Cancer Chemotherapy
1/102
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
103 Terms
Cancer Treatment Modalities
Surgery and Radiation _ % cancer patients will be cured by; the rest are cured by chemo
25
Chemotherapy
will result in a cure or prolonged remission in only 10% of patients in selective tumor types
Chemo allows for _ regression of disease systems
temporary
antineoplastic therapy
ant cancer
Cytotoxic drugs
any drugs that has a toxic effect on cells; also kills host cells
Cytostatic
stops proliferation
Therapeutic Index (TI)
tells us how safe a drug is: TI = TD50/ED50
TD50
the toxic dose of a drug for 50% of the population
ED50
he effective dose for 50% of the population
High TI means theres a big
gap between a helpful dose and a harmful
one — the drug is safer
A low TI means the effective and toxic doses
are close — the drug requires careful monitoring
Single agent drug therapy
in the earlier days of chemotherapy development
Combination chemotherapy:
long-term remissions, more effective prevention of resistance, and tolerable side effects with maximal doses
Rationale
use of multiple agents, each with cytotoxic
activity against the targeted tumor type but with different
mechanisms of action, allows maximum cell killing by each
agent
Cells resistant to one agent might still be sensitive to the
other drugs in the regimen: patterns of cross-resistance must
be taken into consideration in
formulating drug combination
High-dose chemotherapy with stem-cell rescue (autologous)
and bone marrow transplant from a compatible donor led to
cures for some patients with acute myelocytic leukemia and chronic myelocytic leukemia
Combined modality chemotherapy
drugs with other cancer treatments (e.g., radiation therapy)
Combination chemotherapy
combining agents with different mechanisms of action, different resistance mechanisms, and different dose limiting toxicities
Neoadjuvant chemotherapy
initial therapy- preoperative: Treatment given as a first step to shrink tumor before the main treatment is given
Reduces the bulk (size) of primary tumors prior to
surgical resection, to make resection possible or less difficult
Often used in patients with __ advanced tumor types
locally
may _ local recurrence
decrease
The initial clinical response of the tumor mass can serve as an
indication of tumor sensitivity to the drugs used; therefore an
indication to
continue/ not continue this chemotherapy after
surgery
Adjuvant chemotherapy
post-operative: Additional cancer treatment
given after the primary treatment to prevent the risk that cancer will
come back
based on
cell kinetic principles
Surgery shifts tumor cell growth from
“plateau phase” into “exponential phase”
Best time to use chemotherapy is early (right after the
surgery) when tumor burden is small and tumor cells that
are present are actively growing in the “exponential phase”
and therefore susceptible to chemotherapy
Palliative therapy
without curative intent, increase life expectancy &
life quality
Supportive therapy
to reduce toxicity, and side affects
Barriers to chemo
Toxicity to normal cells
• Tumor heterogeneity (intrinsic resistance)
• Drug resistance (acquired or endogenous)
• Not all cells in the tumor are replicating at the same time
complications of chemo
Anticancer drugs =
usually selectively toxic to dividing cells- including normal host cells
The epithelium of the gastrointestinal (GI) tract
nausea and vomiting, directly damage the proliferating mucosa of
the GI tract
Bone Marrow
caused by destruction of proliferating
hematopoietic stem cells or progenitor cells; results in a
decrease in all blood elements
GI
Nausea, vomiting, mucositis, GI infections- almost all drugs
hematological
Anemia, leukopenia, thrombocytopenia- almost all drugs
skin
Alopecia, darkening of the skin, nail changes
endocrine
Infertility, irregular periods, amenorrhea- almost all drugs
neurological complications
Peripheral neuropathy, fatigue, loss of interest, confusion-
common with the plant alkaloid vincristine
chemotherapeutic drugs
alkylating agents
drugs that work by directly modifying/damaging DNA. Cell-cycle nonspecific action
Antimetabolites
inhibit DNA synthesis - most effective during S-phase
Vinca alkaloids (inhibitors of mitosis)
prevent cell division by binding to tubulin & preventing formation of mitotic spindles –most effective during M-phase
Antitumor antibiotics
bind DNA to prevent replication or
RNA synthesis
Action sites of chemo drugs
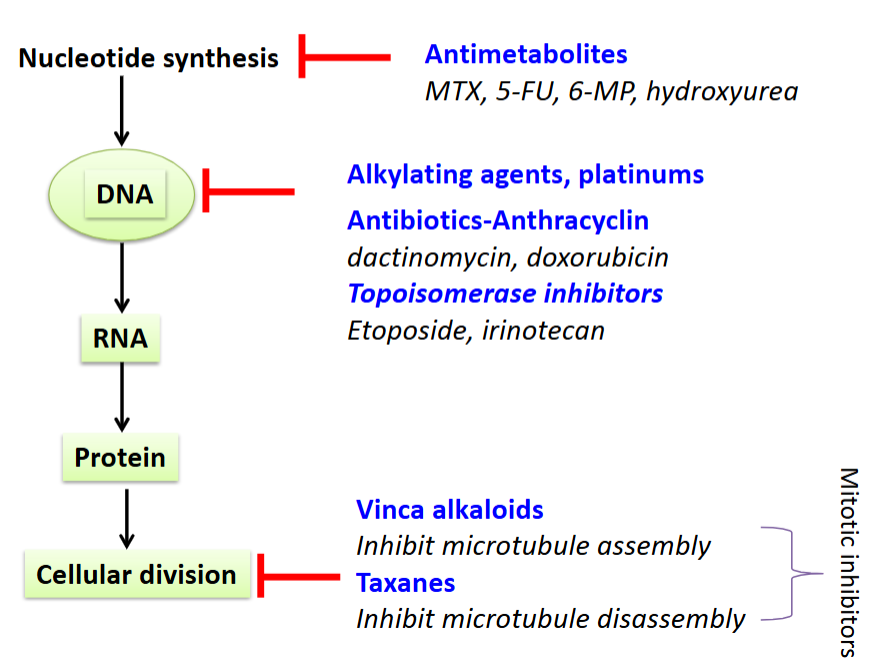
cancer drugs and cell cycle
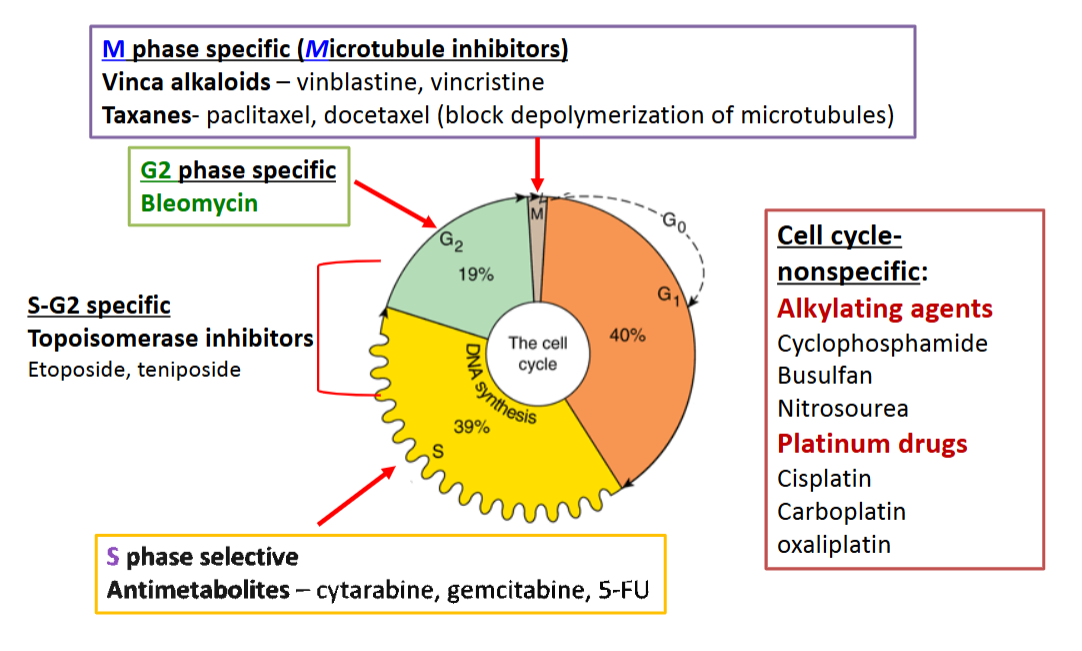
Alkylating Agents
General Indications
Broad spectrum activity against a range of solid and hematological
malignancies. Typically used in combination regimens
mechanisms of action
covalent DNA binding drugs
adding an _ group to DNA
alkyl
Transfers alkyl group to
nucleophilic sites on DNA, leading to DNA crosslinking or modification — disrupts DNA replication and transcription - Cell cycle non- specific
mechanisms of resistance
increase dna repair; decrease uptake
increase in GST attaches a
protective molecule glutathione. Prevents from damaging DNA.
ex of agents
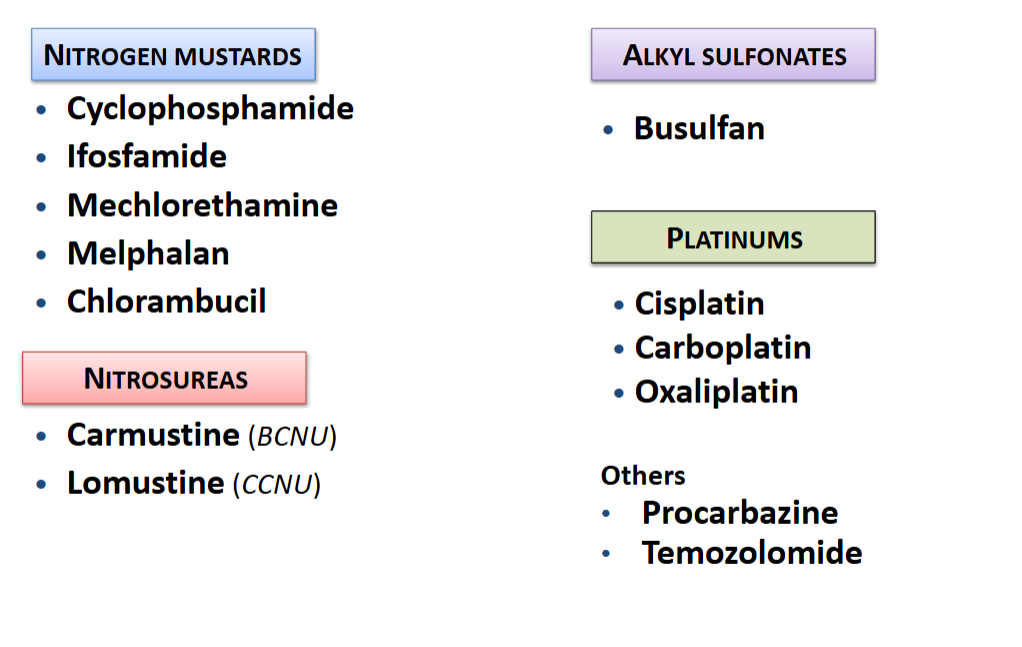
Platinums
indications
Cancers of testes, ovary, bladder, head and neck, lung
MOA
Covalently binds to DNA bases and disrupts DNA function
• Crosslinks N7 guanine on DNA
MOR
DNA excision repair mechanisms
Platinums
Cisplatin
Broad range activity against solid tumors
• Toxicity– Nephrotoxicity, Ototoxicity
• Synergistic with other classes and used in combination
Carboplatin
Similar spectrum of activity as cisplatin, but with less nephrotoxicity
• Toxicity: Myelosuppression (bone marrow toxicity)
Oxaliplatin
Approved as a 2nd line treatment for metastatic colorectal cancer
• Tumors resistant to cisplatin or carboplatin are not cross-resistant to
oxaliplatin
• Dose limiting toxicity – Neurotoxicity
Antimetabolites
indications
Broad spectrum activity against a range of solid and
hematological malignancies. Typically used in combination
regimens.
dose limiting toxicity
Bone marrow suppression & GI toxicity
MOA
Inhibit nucleotide synthesis
Purine Analogs
Pyrimidine Analogs
Folic Acid Analogs
• Cells in S phase most susceptible
Purine Analogs
MOA
Inhibits synthesis of nucleotides
from purine bases (adenine, guanine)
• Incorporation into DNA and RNA
Inhibits DNA and RNA synthesis
toxicity
Bone marrow suppression
• GI & liver dysfunction
MOR
decreased activity of HGPRT
(hypoxanthine-guanine
phosphoribosyltransferase)
required for metabolic activation
Pyrimidine analogs
MOA
Inhibition of DNA synthesis by
interfering with enzymes involved in
pyrimidine synthesis
5-FU: blocks thymidylate synthase,
gemcitabine: ribonucleotide reductase
Toxicity
5-FU: myelosuppression, skin toxicity, GI
toxicity
MOR
Enzyme (e.g., thymidylate synthase or
ribonucleotide reductase) induction
Thymidylate synthase converts dUMP to dTMP in the process of ___
DNA synthesis
Folic Acid Analogs
MOA of MTX
Competitively inhibits dihydrofolate reductase (DHFR), a key
enzyme in intracellular folate homeostasis, to inhibit the synthesis
of tetrahydrofolate (THF) from folic acid (FA) [FA THF]
MOR
Induction of DHFR through gene amplification/other mechanisms
• Decreased drug transport
• Decreased formation of cytotoxic MTX polyglutamate (key metabolite)
Toxicities
Myelosuppression (bone marrow suppression): DLT
• GI effects (mucositis)
• Hepatic: acute and chronic liver toxicity
• Nephrotoxicity: MTX crystals in acidic urine (to prevent: aggressive
alkalized hydration)
• Dermatologic: skin reactions (dermatitis; photosensitivity)
Leucovorin rescue (antidote for MTX in cancer)
Leucovorin is given to decrease the damage done to normal cells by MTX.
• Usually begins 24 to 36 hrs following MTX infusion
MITOTIC SPINDLE INHIBITORS
AND
TOPOISOMERASE INHIBITORS
Vinca Alkaloids
indications
hematological malignancies, breast &
pediatric cancers
MOA
Binds to tubulin —> Inhibit polymerization
into microtubules, disruption of mitotic
spindle formation
M phase cell cycle arrest
toxicity
Vinblastine-bone marrow suppression
• Vincristine- neurotoxicity
MOR
increased efflux via MDR P-gylcoprotein
(Pgp)
Taxanes
indications
Paclitaxel –
broad spectrum activity
against solid tumors (primarily breast)
Docetaxel
similar activity as paclitaxel
MOA
Mitotic inhibitors
• Prevent microtubule disassembly into tubulin monomers: “frozen Mitosis”
• Stablizes GDP-bound tubulin, inhibition of mitosis and cell division (M phase block).
toxicity
Bone marrow suppression & GI toxicity, neutropenia, peripheral neuropathy
MOR
MDR P-glycoprotein mediated drug efflux
• Tubulin mutations
Targeted therapy for Cancer
cytotoxic chemo
Although these drugs can be effective, side effects limit
their use.
• Lack of selectivity – kill tumor cells, but cause toxicity in
normal (non-tumor) cells as well
Targeted EGFR
EGFR
a transmembrane receptor involved in a cell growth, survival, migration and invasion
EGFR activity and/or expression is
deregulated in human
epithelial tumors
– Gene amplification or
mutations: Head & neck,
lung, breast cancers