c4 - predicting chemical reactions
1/37
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
what metals are known as the alkali metals
group 1
how many electrons do alkali metals have in their outer shell and what does this mean
1 electron so they all have similar chemical properties
what are the 3 physical properties of alkali metals
low mp and bp
low density
very soft - can be cut with knife
why dont alkali metals form covalent bonds
they lose their 1 electron on their outer shell so easily that sharing it is out of the question
why does the reactivity of alkali metals increase down the group
the atoms get bigger down the group
the outer electron is further sway from the nucleus
the further the electron is from the positive attraction of the nucleus , the easier it can be lost
what properties do alkali metals and normal metals share
good conductors of heat + electricity
shiny - only seen in alkali metals when freshly cut but then turn dull with oxidation
how does lithium (alkaline metal) react with water
it moves around the surface fizzing vigorously
whats the word equation for the reaction of lithium and water
2Li + 2H2O → 2LiOH+ H2
how does sodium (alkaline metal) react with water
move over top of water with vigorous fizzing and may catch fire with an orange flame
how does potassium (alkaline metal) react with water
moves over top of water with vigorous fizzing and may catch fire with lilac flame
how does rubidium react with water
they explode
where are halogens found
group 7
how many electrons do halogens have on their outer shell
7 electrons so they all have similar chemical properties
whats the trend of the size of each element and the number of full electron shells in group 7
increase down the group
means the outer shell is farther away from the nucleus and the positive protons are shielded by more electron shells
the further the outer shell is from the positive nucleus, the harder it is to attract another electron
whats the trend of reactivity in group 7
reactivity decreases with the size of the atom (down the group)
how many more electrons do halogens need to become stable
need 1 more atom
achieves this when a halogen atom can receive an electron and forms a single covalent bond
whats the trend of the melting and boiling points in halogens and why
they increase down the group as down the group the molecules become larger and the intermollecular forces become stronger
more energy is needed to overcome these forces
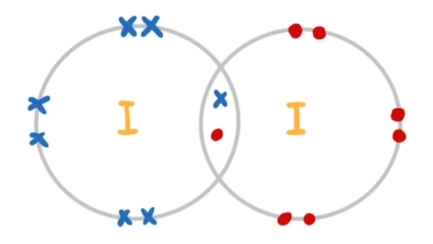
whats a diatomic molecule
two atoms covalently bonded together in a molecule
are halogens diatomic molecules
yes because they share one pair of electrons in a covalent bond (e.g. Cl 2 , Br 2, I2 )
whats chlorine characteristics (halogen)
fairly reactive poisonous green gas (has low boiling point)
whats bromine’s characteristics (halogen)
posinous reddy brown liquid which gives off orange vapour at room temp
whats iodine’s characteristics (halogen)
dark grey crystalline solid which gives off purple vapour when heated
what are properties all halogens possess
non metals - dont conduct electricity
brittle and crumbly when solid
poisonous and smelly
Halogens react with _______ to form salts.
alkili metals
what does the reactivity of an atom mean
how easy it is to lose or gain electrons - the easier it is the more reactive the atom
whats a displacement reaction
a reaction that occurs when a more reactive element replaces a less reactive element in a compound
whats the method for the experiment to compare the reactivity of halogens
place 2 drops of potassium chloride solution in each 3 dimples on your dimple tile
repeat with potassium bromide in the next row and potassium iodine in the final row
place 2 drops of chlorine water in each 3 dimples down left hand column of dimple tile
repeat with bromine water and iodine water in the next columns
observe if there is a colour change with either chlorine, bromine or iodine
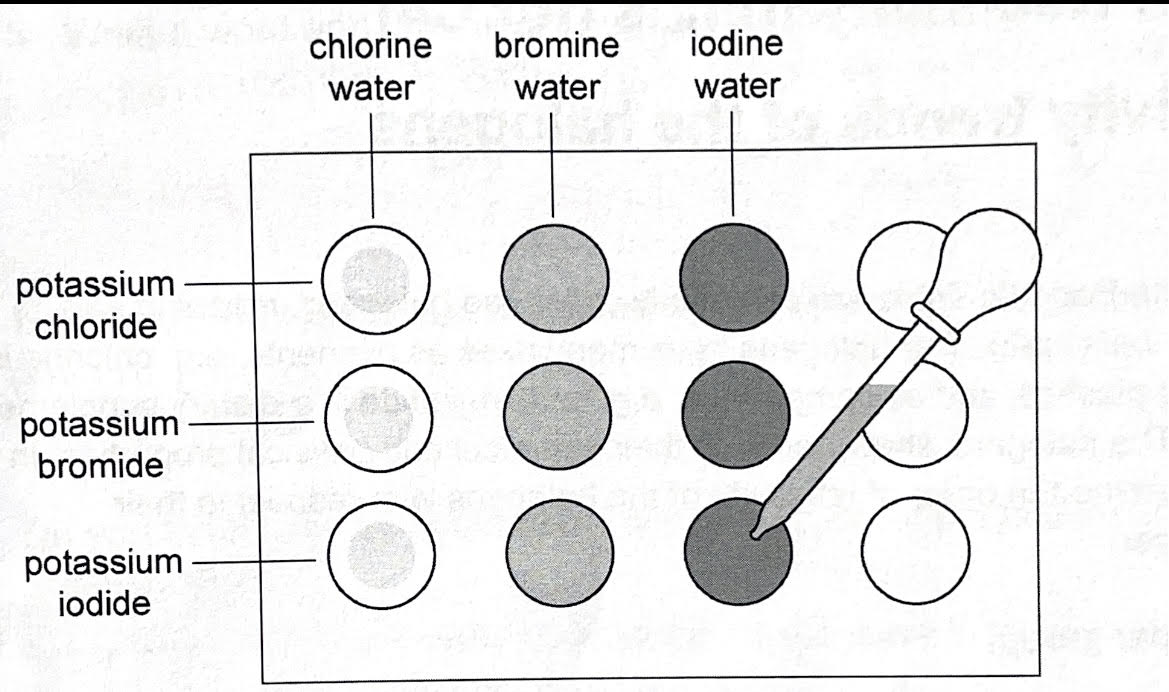
how do you tell in the experiment for reactivity trends of halogens which is most reactive
the order of reactivity is: chlorine > bromine > iodine.
because chlorine could displace bromine and iodine, bromine could only displace iodine, but iodine could not displace chlorine or bromine.
how do you tell by observing is a halogen has been displaced in an experiment
if you see a colour change then a reaction has heppaned ad the halogen has replaced teh halide ions in the salt
what are the colours of halide salts, bromide, chlorine and iodine
Halide salts (e.g. KCl, KBr, KI) are colourless
Bromine water is orange
Chlorine water is colourless
Iodine water is brown
what is the name for all the gases in group 0
noble gases
what can be said about all the noble gases
all colourless gasses at room temp
all made up if single atoms
all inert
non flammable
what does inert mean
they are not generally reactive so they usually dont react with anything
why are all the noble gases inert
they have a full outer shell of electrons - dont easily give up / gain electrons
what are the properties of group zero
the boiling point , melting point ans density all increase as you go down the group
how metals react with _____ tells you about their reactivity
acids
whats the reactivity series
potassium
sodium
calcium
magnesium
aluminium
zinc
iron
copper
silver
what happens when you put a more reactive metal into a solution of a less reactive metal salt
the reactive metal will replace the less reactive metal in the salt