Principles of Chemistry - Paper 1
1/48
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
49 Terms
Structure and Movement of Solids
particles are close together, touching, in regular arrangement, vibrate around fixed positions
Structure and Movement of Liquids
particles are touching, irregular arrangement, move randomly
Structure and Movement of Gases
particles are far apart, irregular arrangement, move quickly and randomly
Interconversions between the States of Matter
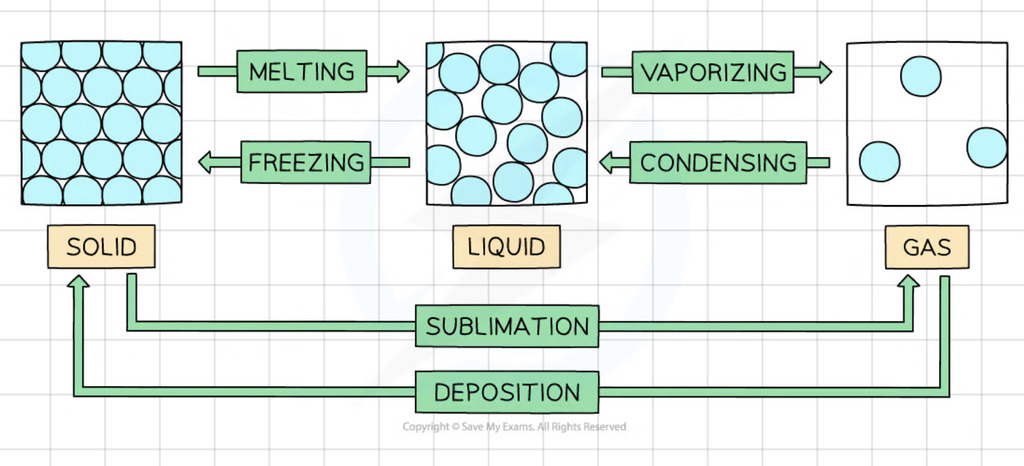
Melting
Particles gain kinetic energy, move faster, further apart
Boiling
Particles gain kinetic energy, move faster, more particles have enough kinetic energy to overcome intermolecular forces and move further apart.
Condensing
Particles lose kinetic energy, move close together and more slowly.
Freezing
Particles lose kinetic energy, move more slowly and become regularly arranged.
Dilution
process of reducing the concentration of a given solute in its solution, using water
Diffusion
spreading out of particles from a high to low concentration until there is an equal concentration
Solute
substance that dissolves in solvent
Solvent
liquid solute dissolves in
Solution
liquid formed when solute dissolves in solvent
Saturated Solution
solution which contains as much dissolved solid as possible at a particular temperature
Element
substance that only contains one type of atom, cannot be split into anything simpler by chemical means
Compound
two or more elements chemically combined in fixed proportion
Mixture
various substances physiclly combined in any proportion with no chemical reaction
Pure Vs. Impure Substance
Pure: fixed melting and boiling point
Mixture: melt and boil over range of temperatures
Separation Techniques
filtration
crystallisation
simple distillation
fractional distillation
chromatography
Filtration
separate insoluble solid from liquid
Crystallisation
separate solute from solution
Simple Distillation
separate components of solution
Fractional Distilation
separate mixture of liquids
Paper Chromatography
separate components of mixtures (coloured inks: mixture of dyes)
same type of paper, same solvent
Rf Value
distance moved by spot/ distance moved by solvent front
Investigation using Paper Chromatography
put spots of each ink on pencil line
pour some solvent into bottom of beaker
place paper in beaker so spots are above level of solvent
leave until solvent has risen up paper
Atom
smallest piece of element that can still be recognised as that element
Molecule
two or more atoms chemically bonded
Atomic Number
number of protons in atom’s nucleus
Mass Number
total number of protons and neutrons in atom’s nucleus
Isotope
atoms of same element/ same number of protons
dufferent number of neutrons/ different mass number
Relative Atomic Mass
average mass of atom compared to mass of carbon-12
Electrical Conductivity and Acid-Base Character - Metals
conduct electricity
have oxides which are basic, reacting with acids to give a salt and water
Electrical Conductivity and Acid-Base Character - Non-Metals
do not conduct electricity (except for graphite)
have oxides which are acidic or neutral
Noble Gases
do not react
full outer shell of electrons
Mole
unit for amount of substance
Determining Formula of Metal Oxide by Combustion
weight 2.4g magnesium ribbon
heat strongly in crucible untill all forms magnesium oxide
keep lid on to stop MgO escaping but keep small gap so air can enter
reweigh crucible + lid + MgO
reheat and reweigh to constant mass
Determining Formula of Metal Oxide by Reduction
measure mass of empty boiling tube
place metal oxide in and measure mass
pass methane over copper (II) oxide and burn off excess gas
heat copper (II) oxide strongly using bunsen burner
heat until metal oxide completely changes colour
measure mass of tube remaining metal powder
Ionic Bonding in terms of Electrostatic Attraction
strong electrostatic forces of attraction between oppositely charged ions
Why do GIL have high b.p.?
strong ionic bonds
between oppsositely charged ions
require a lot of energy to break bonds
Electrical Conductivity of Ionic Compounds
solid → strong ionic bonds between many ions in fixed position, ions not free to move
molten/ dissolved in water → ions are free to move
What is a Covalent Bond?
strong electrostatic forces of attraction between nuclei of atoms that make up bond and shared pairs of electrons
Diamond Properties
hard
high m.p.
does not conduct electricity
Why is diamond hard?
has tetrahedral lattice structure/ each atom is bonded to 4 other atoms
not arranged in layers
Why does diamond not conduct electricity
no delocalised eletrons
all electrons held firmly in covalent bonds
Properties of Graphite
soft
high m.p.
conducts electricity
Why is graphite soft?
structure is in layers
weak forces of attraction between layers
slide over one another easily
each atom is bonded to 3 other atoms
Why can graphite conduct electricity?
one delocalsied electron per carbon atom
free to move
Why do GCL have high m.p.?
strong covalent bonds
many
require a lot of energy to break bonds