Chem 202 all units
1/133
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
134 Terms
ketone
R-C=O-R
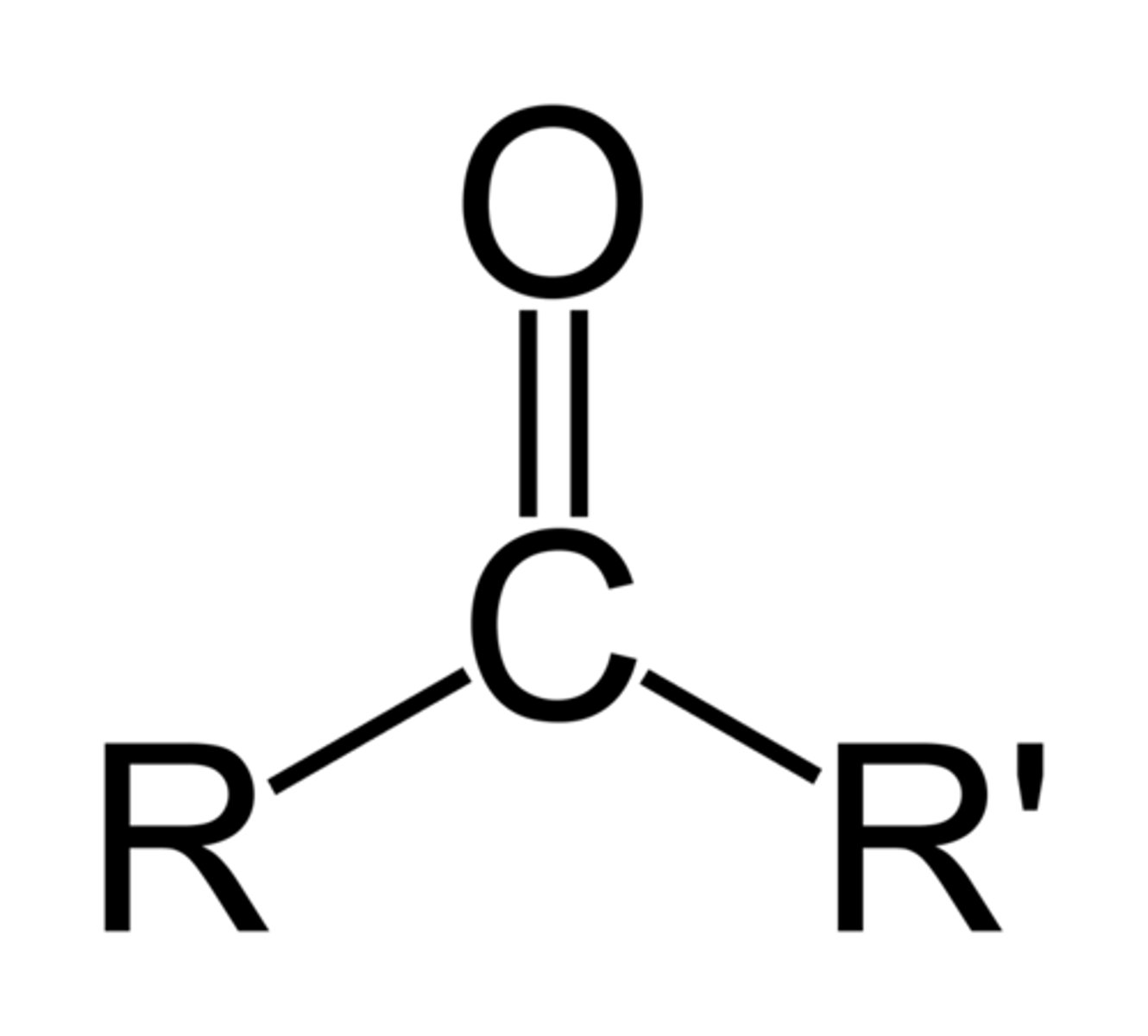
Acid Chloride
R-C=O-Cl
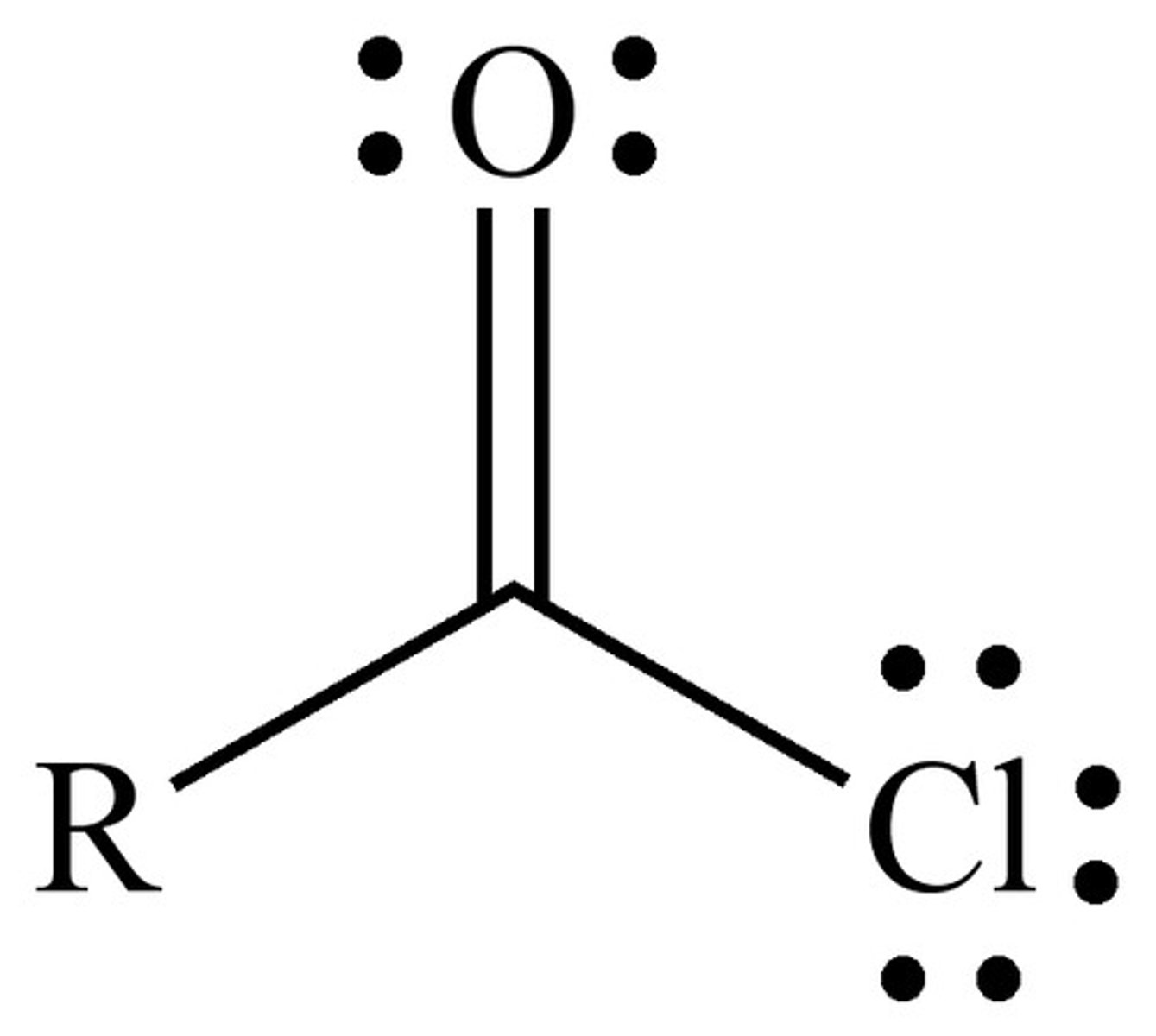
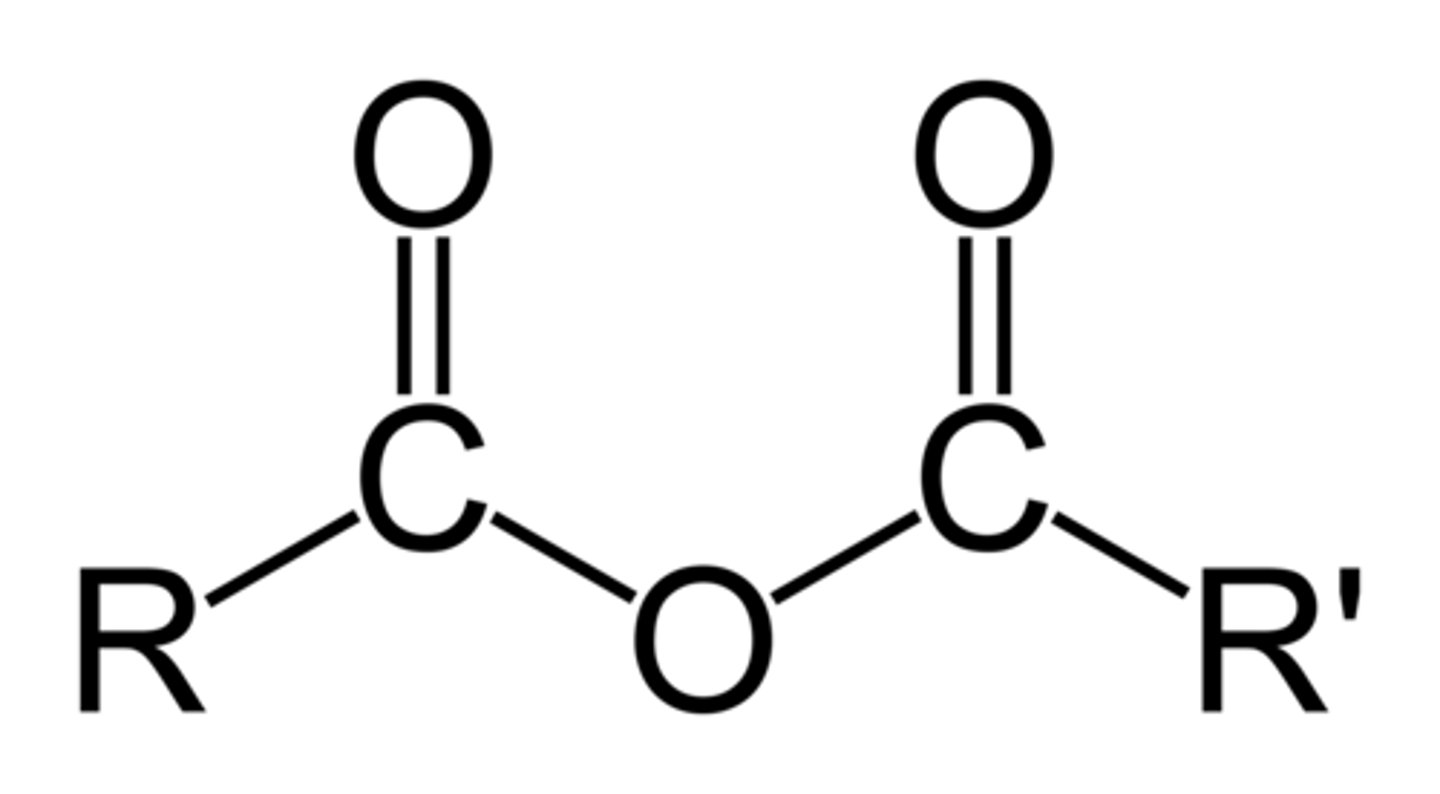
acid anhydride
R-C(=O)-O-C(=O)-R
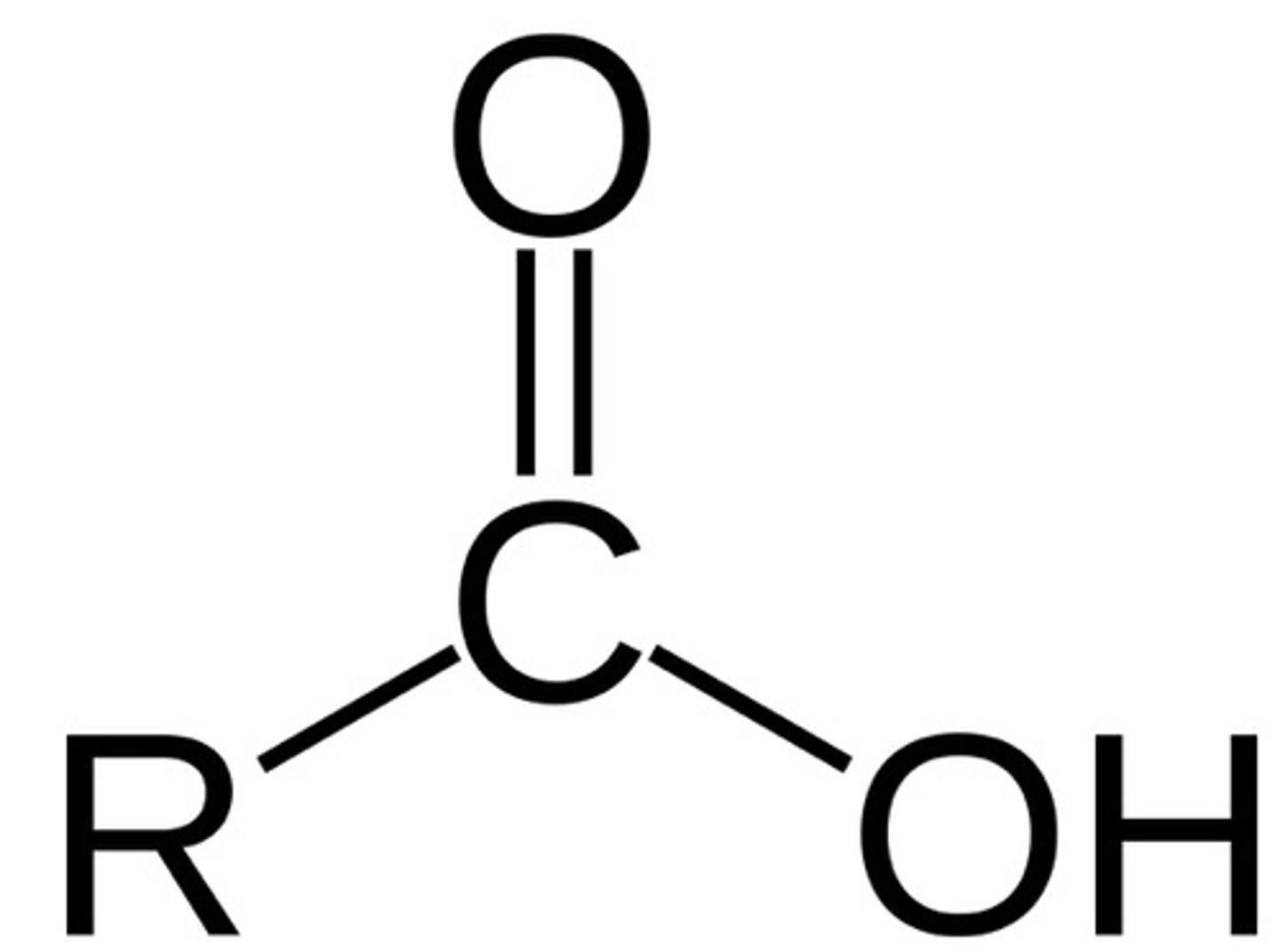
carboxylic acid
R-COOH
Amide
-CONH2, -CONHR, -CONR2
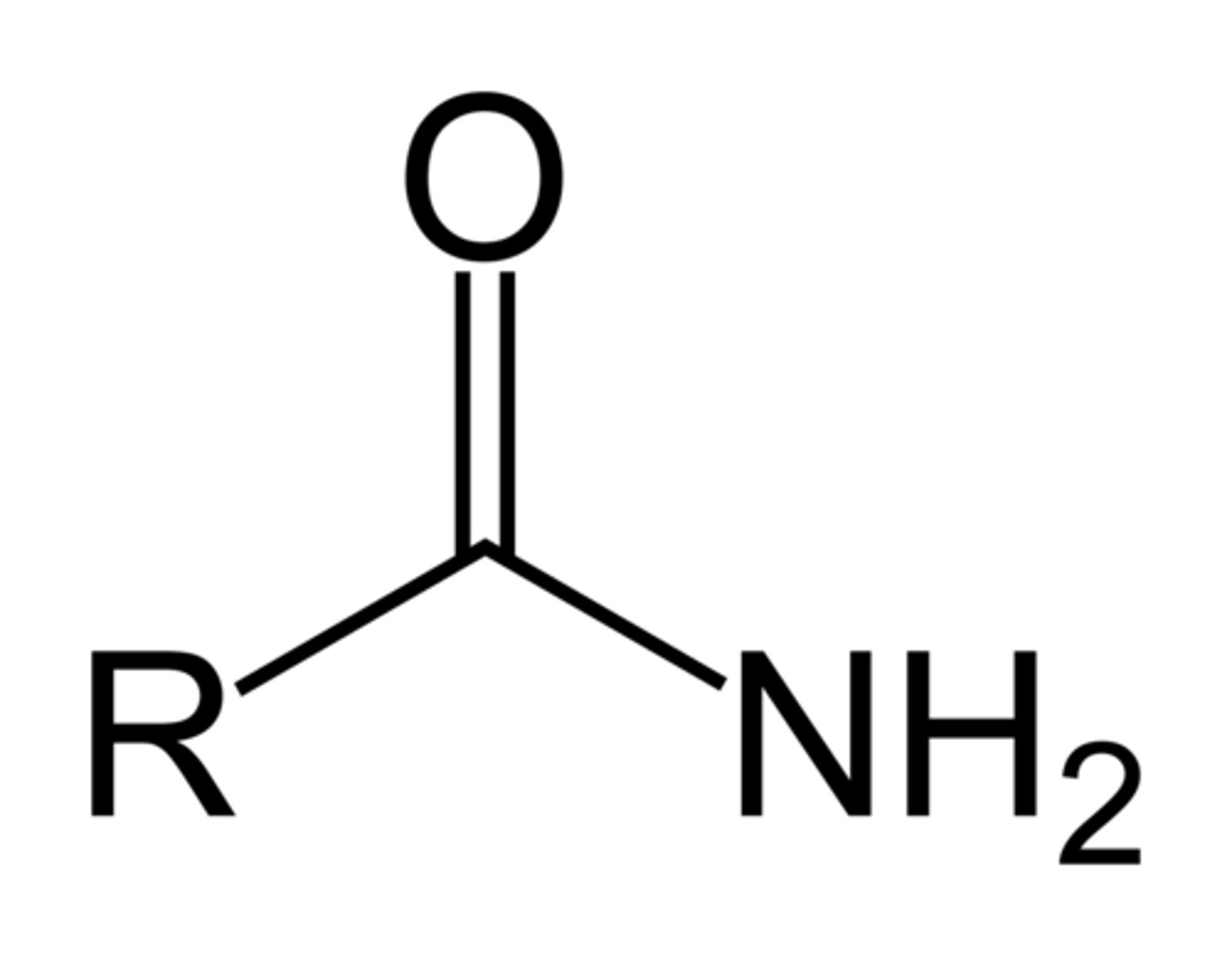
Ester
RCOOR
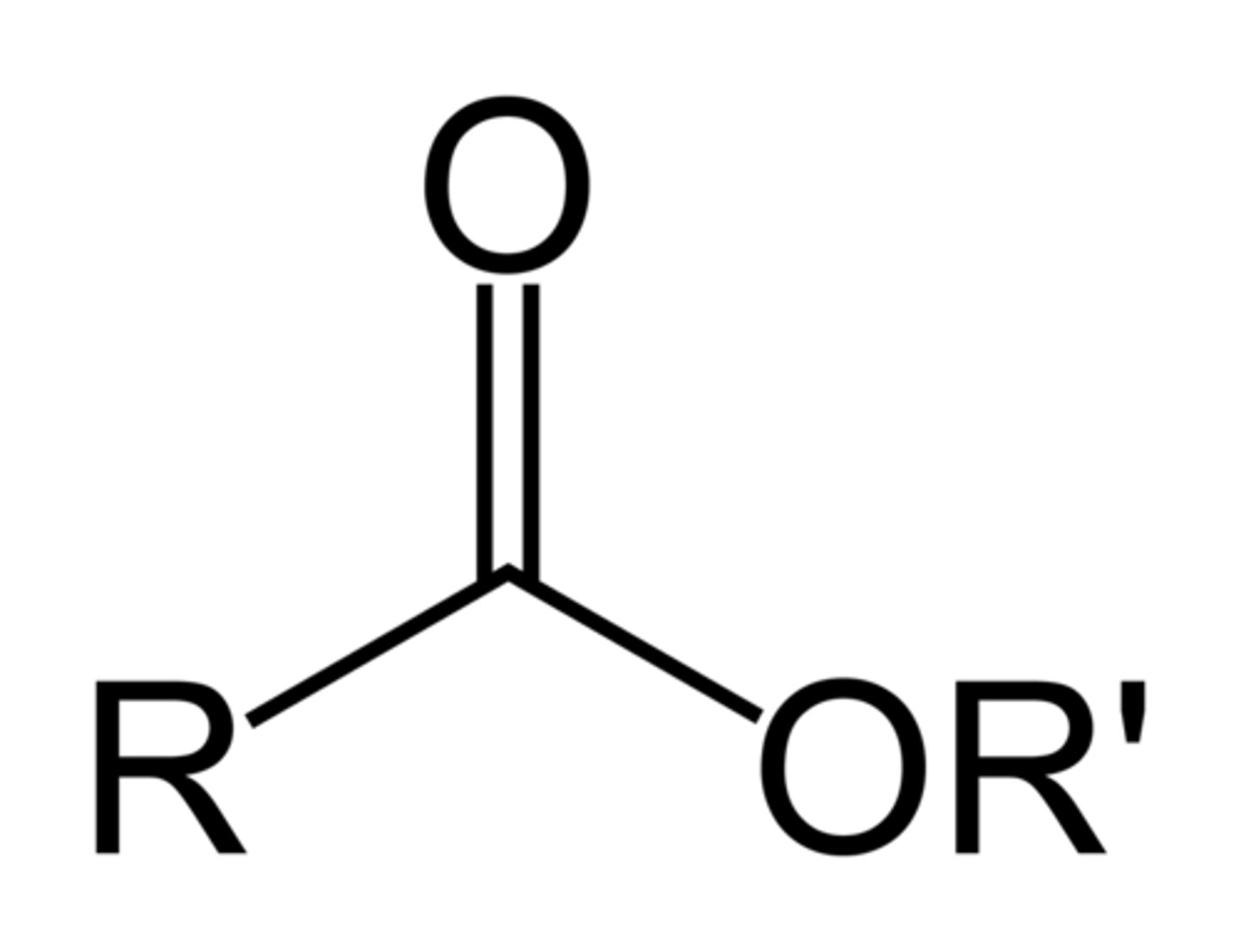
Nucleophilic Addition
a reaction in which an electrophilic π bond reacts with a nucleophile, breaking the π bond and forming 2 new σ bonds.
Ammonia
NH3
Ester Hydrolysis
ester reacts with H2O to produce a carboxylic acid and an alcohol
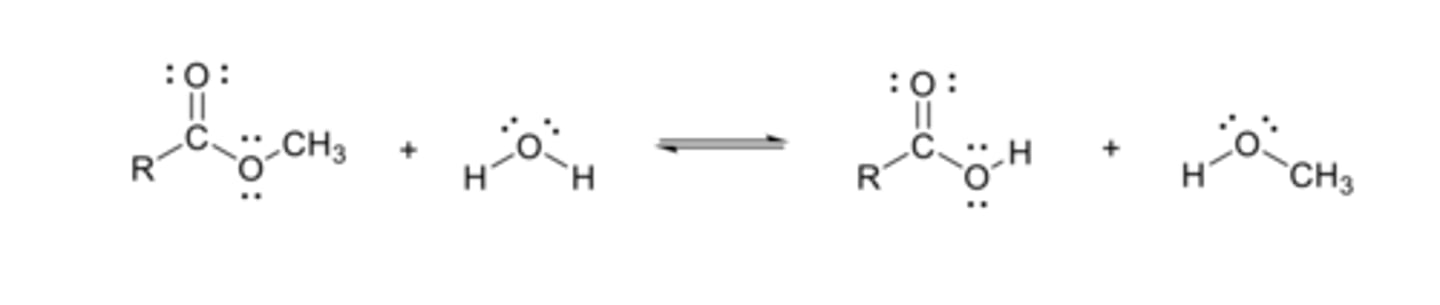
Fischer esterification
The formation of an ester from a carboxylic acid and an alcohol in acidic conditions
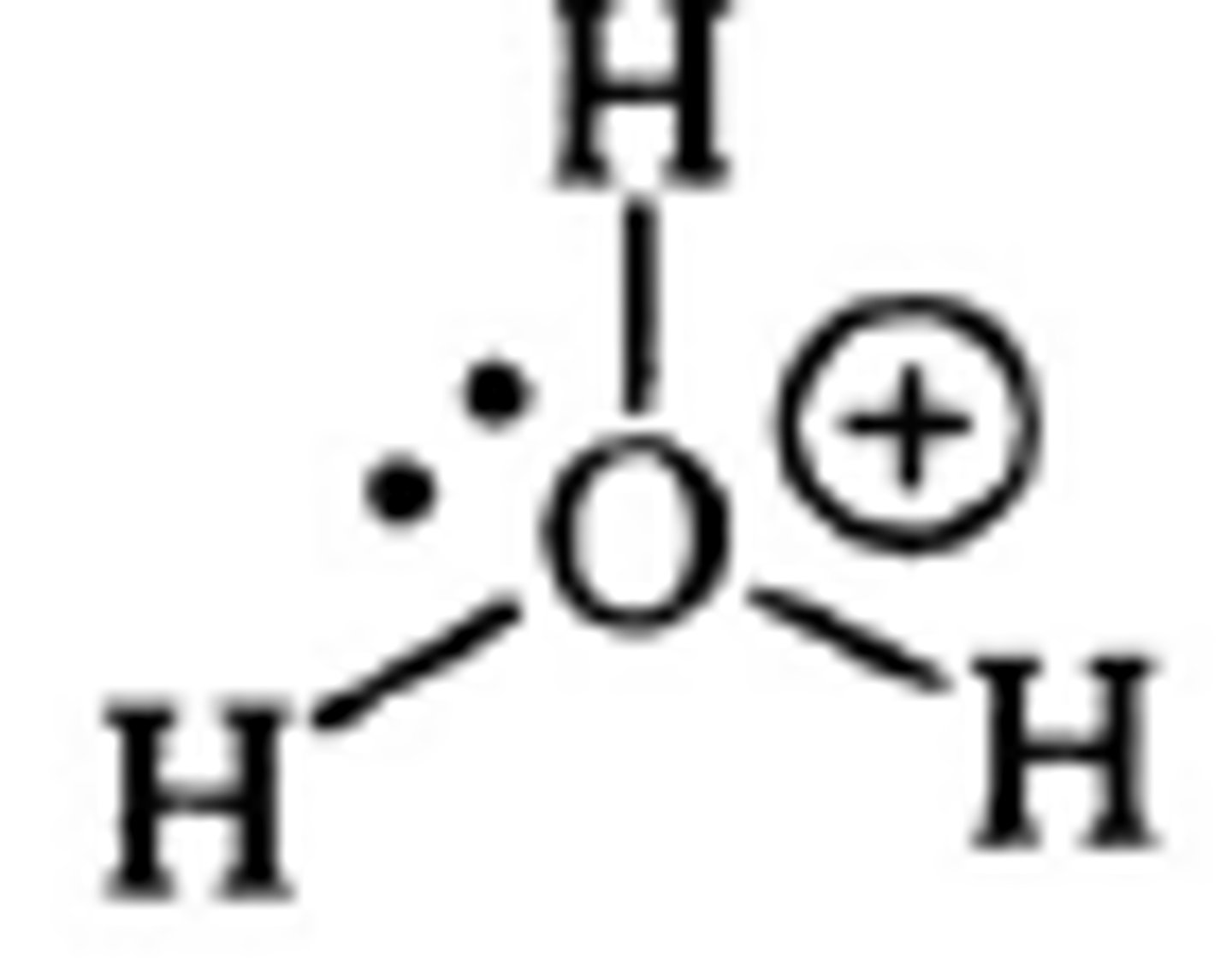
pka of hydronium or ROH2+ (ie.
protonated alcohol)
<0
pka of HCl, HBr, HI
< -8
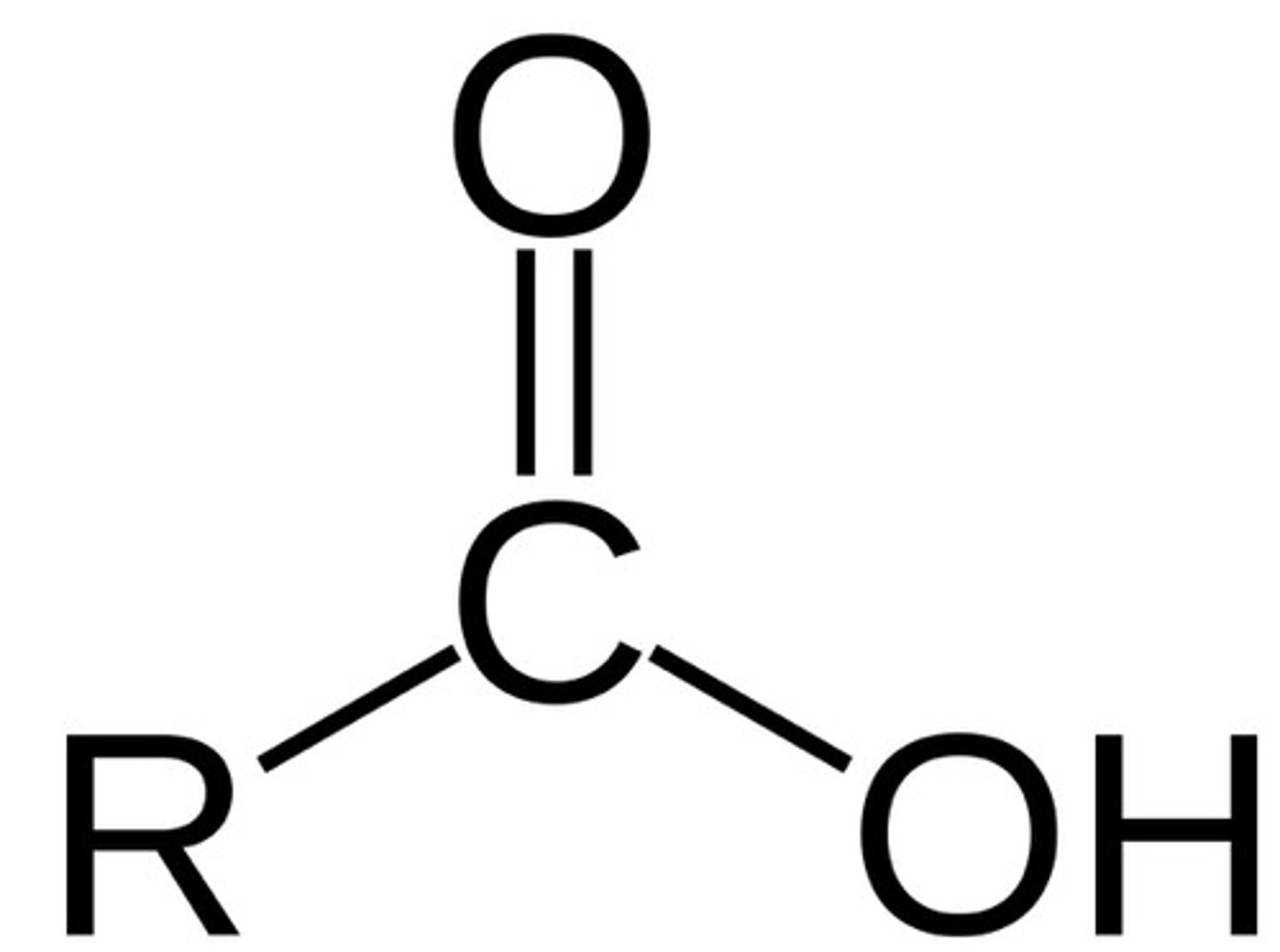
pKa of RCOOH (carboxylic acid)
5
pKa of ammonium ion, protonated
amines
9-11
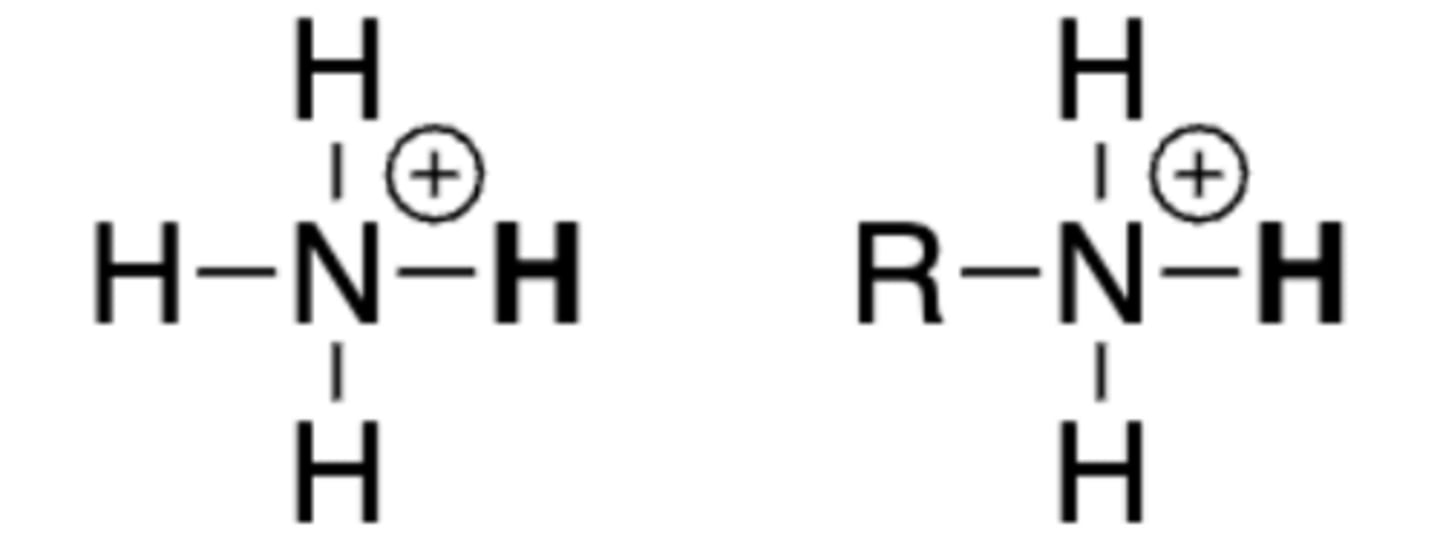
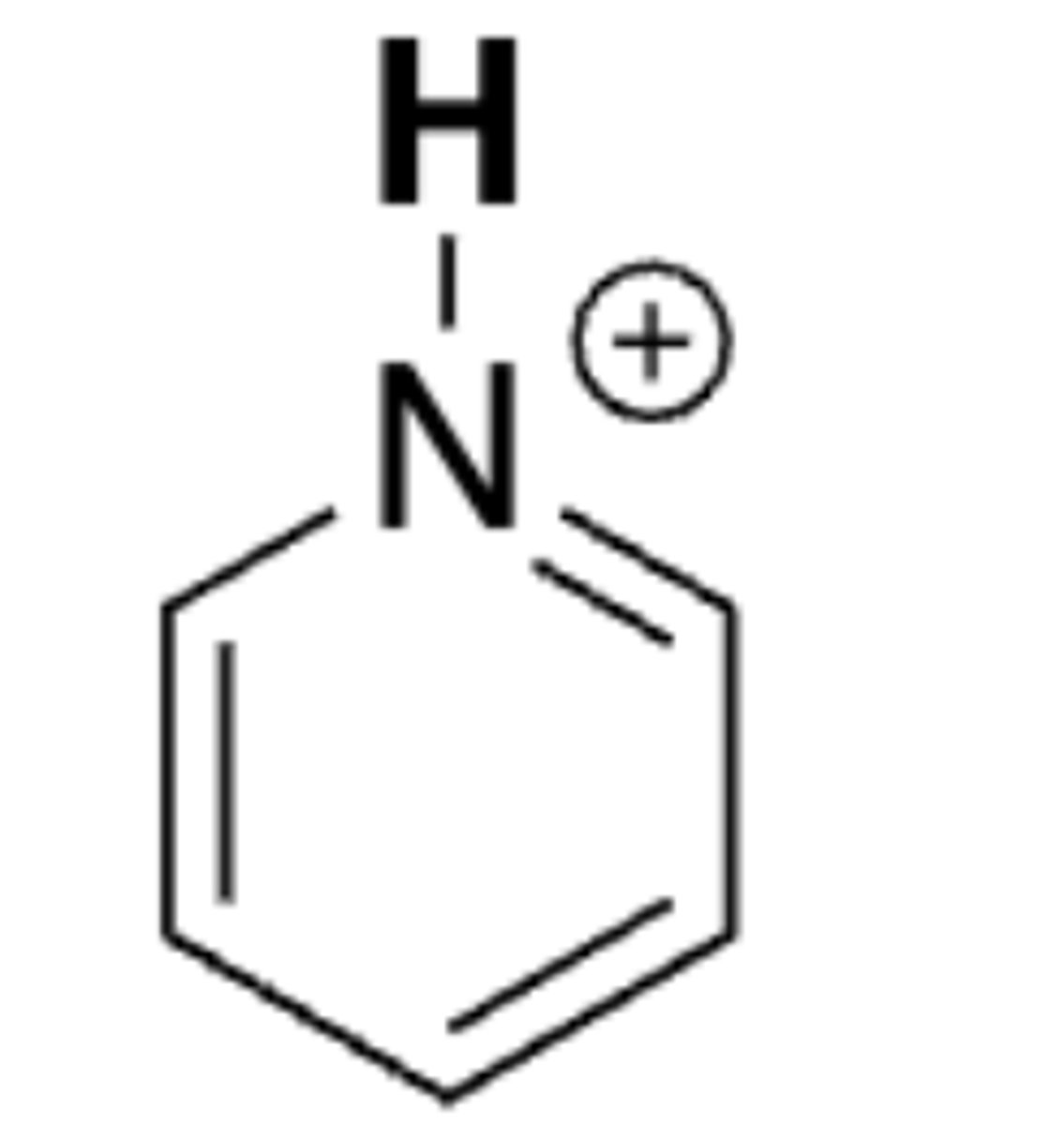
pKa of pyridinium ion
5
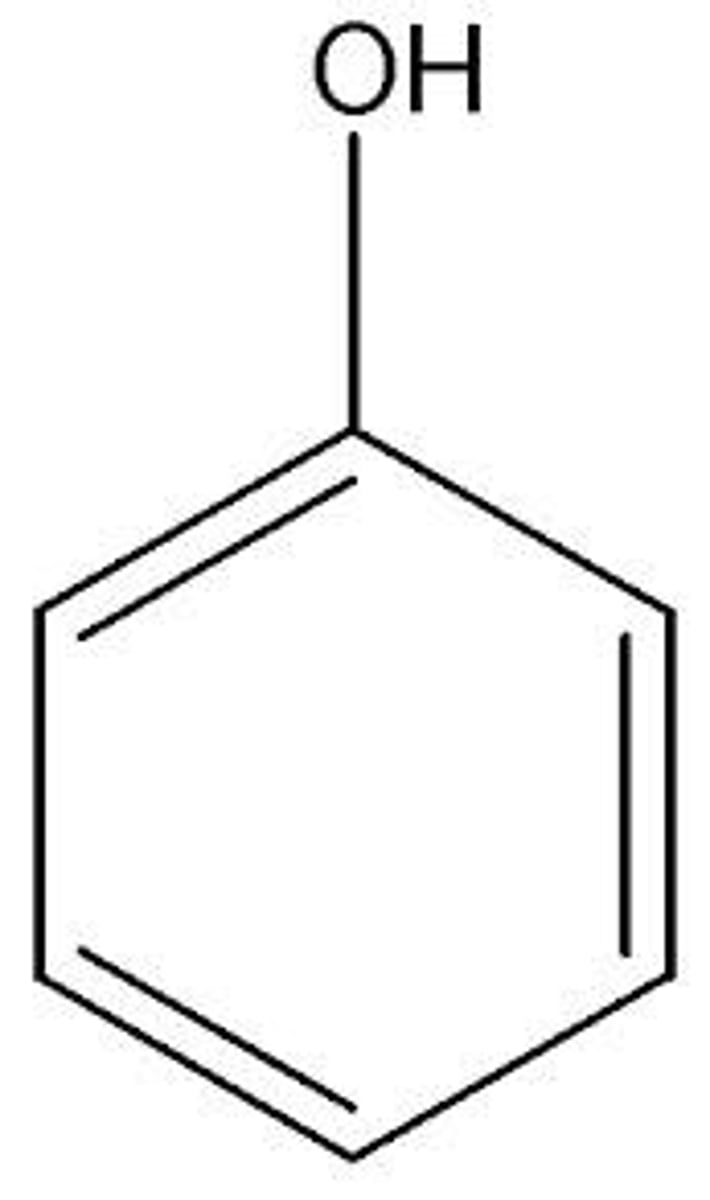
pKa of phenol
10
pKa of ROH or H2O
15

pKa of NH3
38
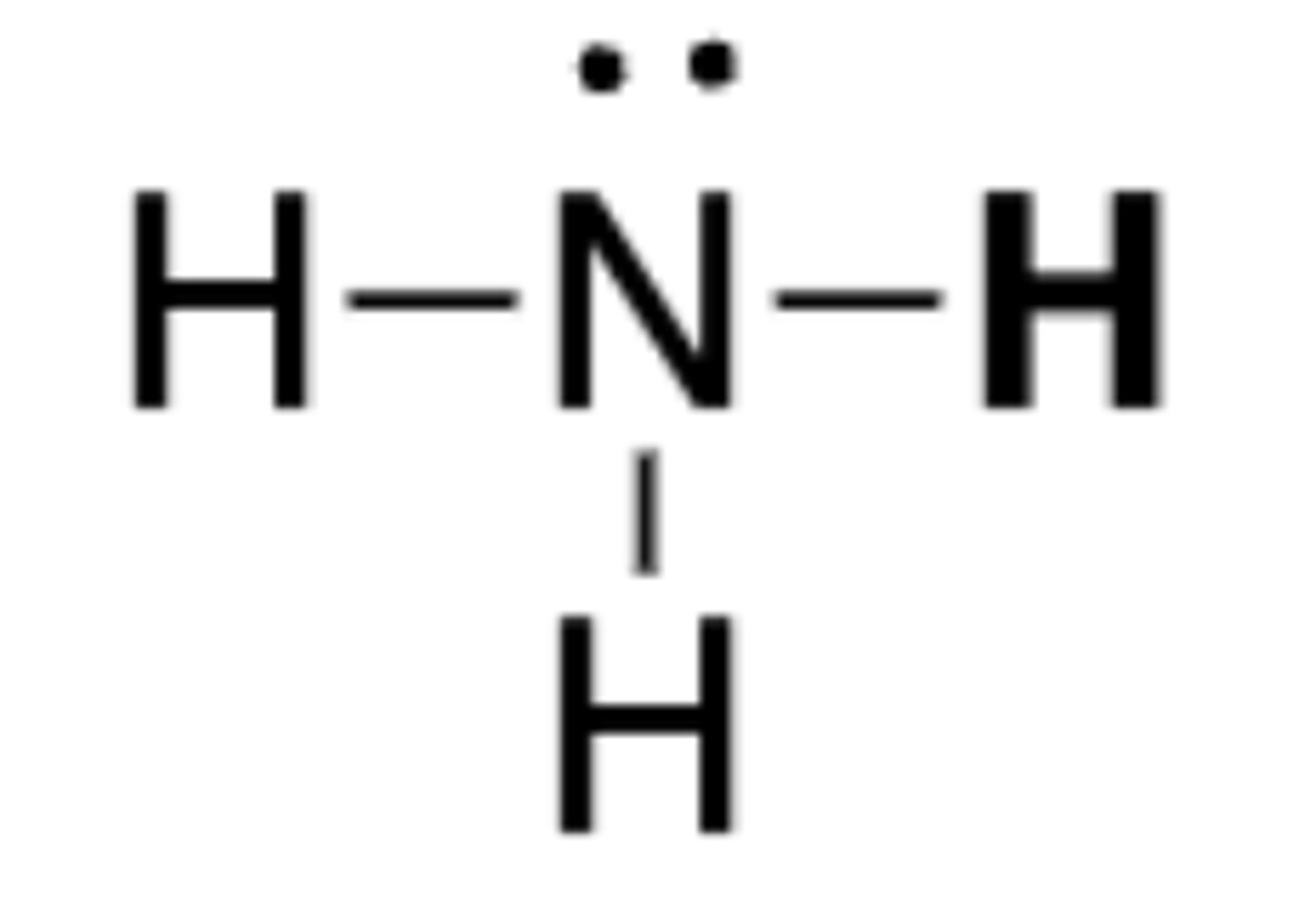
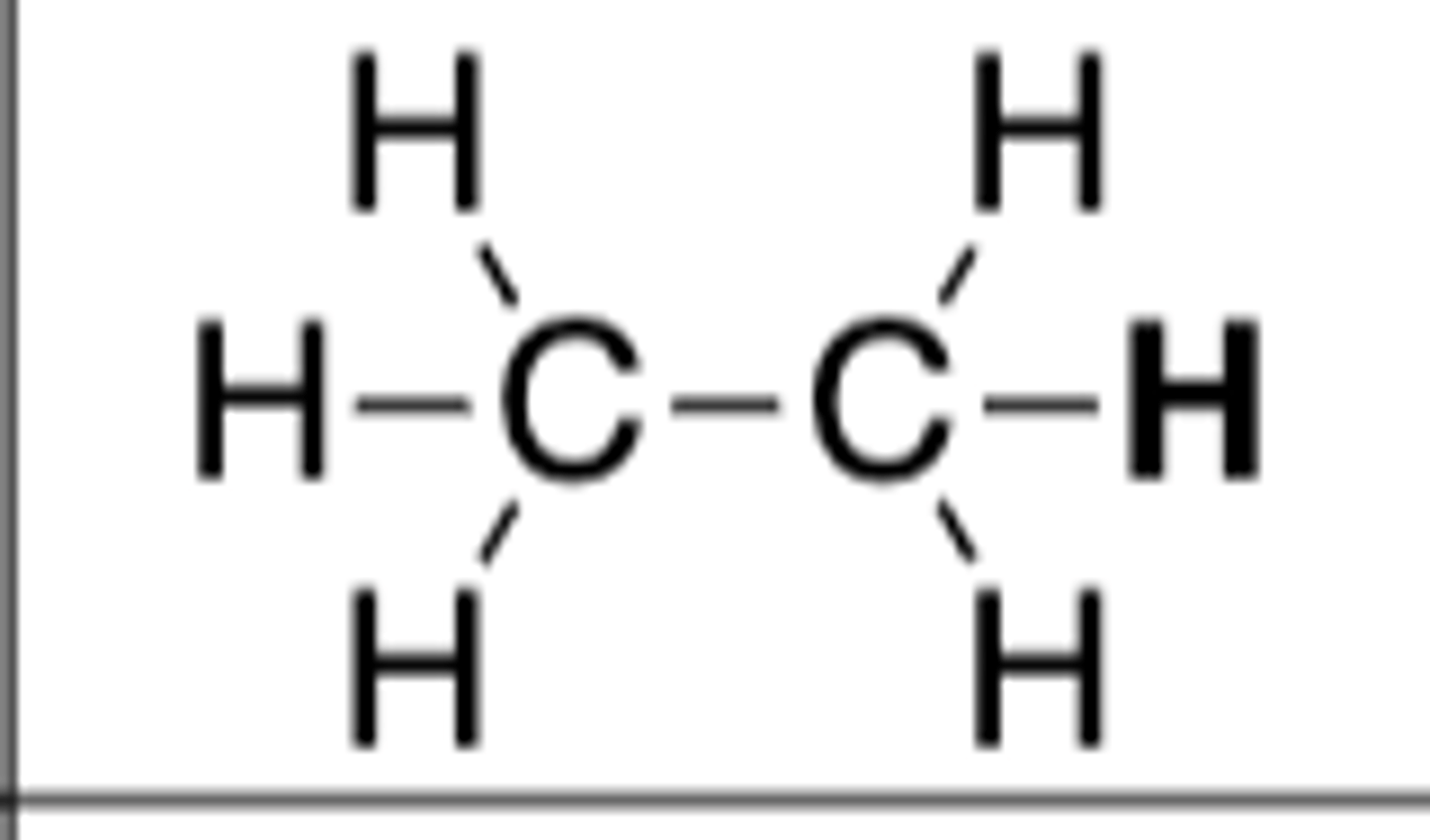
pKa of CH3CH3 (or RCH3)
50
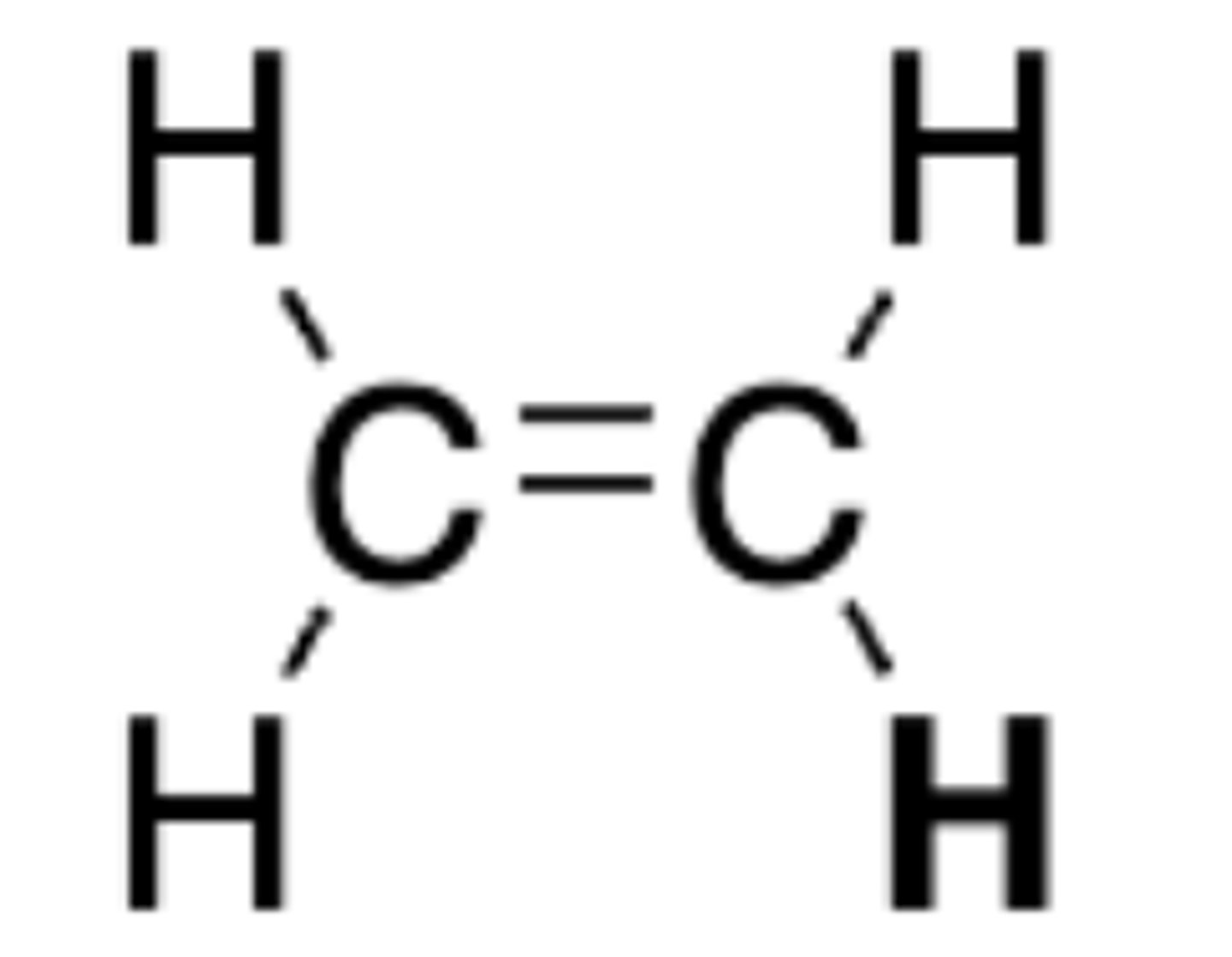
pKa of CH2CH2
44
pKa of HCCH
25
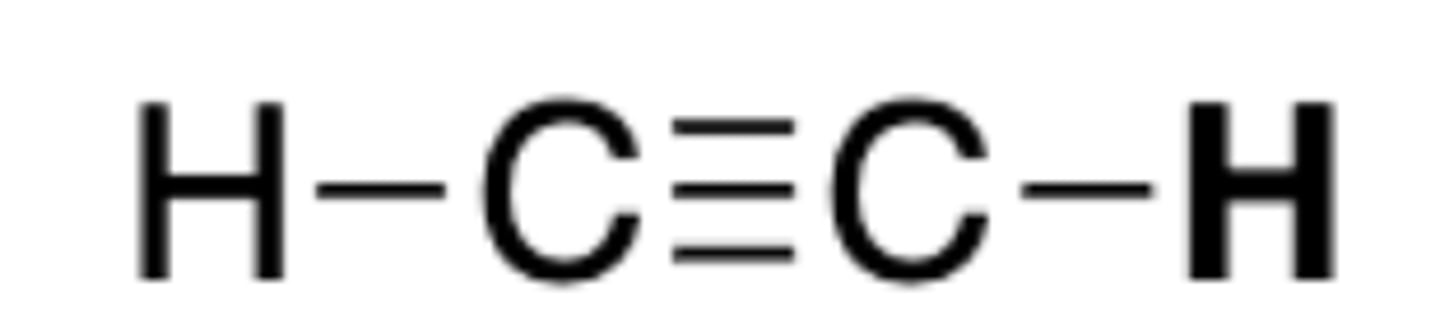
alkyl group
An alkane with a hydrogen atom removed
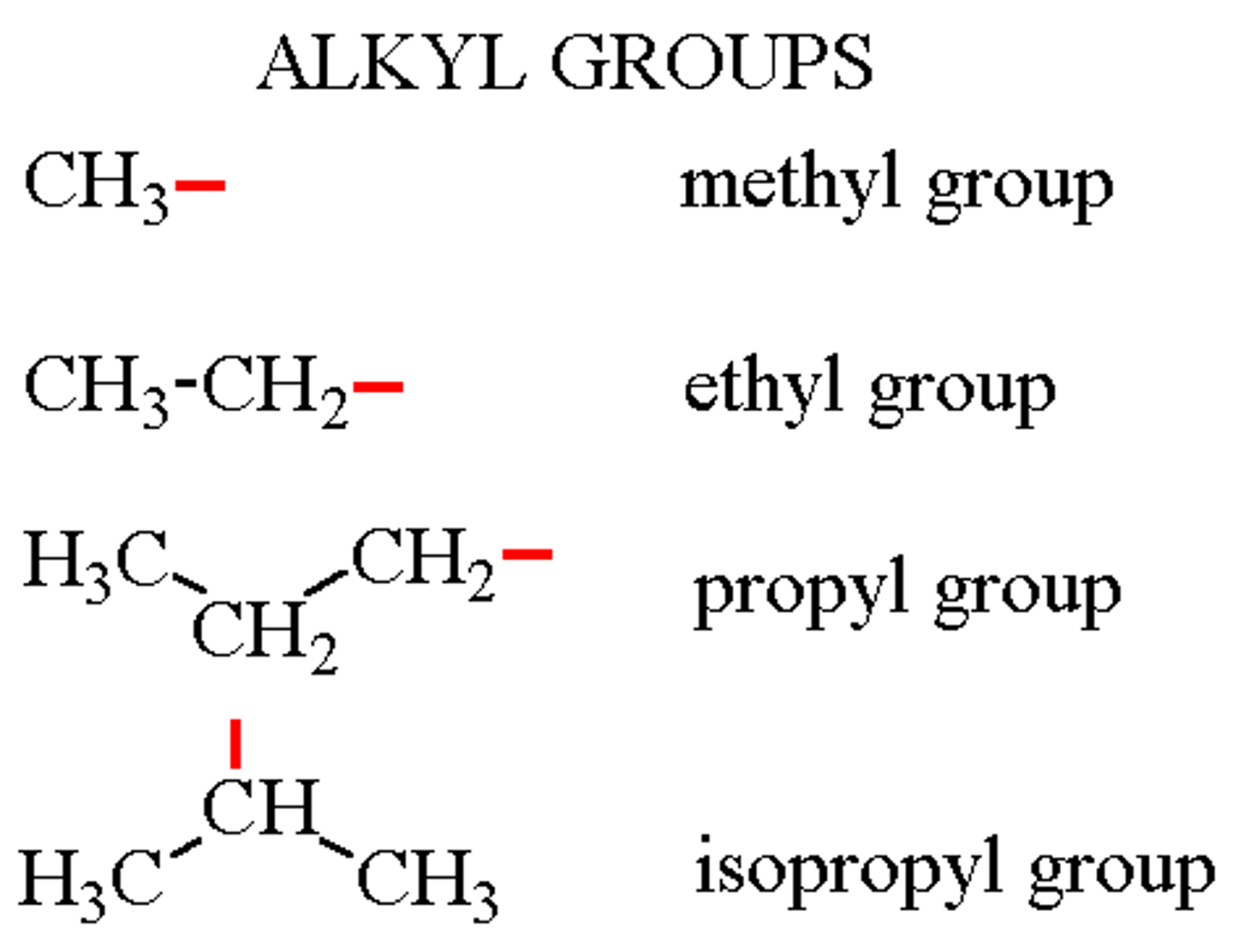
carbonyl compound
any compound that contains a carbonyl group C=O
kinetic product
Faster due to lower activation energy, typically at low temperatures. lower activation energy but higher Gibb's free energy
thermodynamic product
more stable (lower energy) and favored at high temperatures. The product has higher activation energy but lower Gibb's free energy
acid-catalyzed reaction
uses a small amount of acid to speed up a process, with the acid regenerated in the end (e.g., hydration of alkenes)
acid-promoted reaction
requires a stoichiometric amount of acid that is consumed, not regenerated
base-promoted reaction
A base facilitates chemical bond breaking, commonly in ester hydrolysis (saponification) or amide hydrolysis. The base acts as a nucleophile, attacking carbonyl carbons, rather than just a catalyst, as it is consumed in the process.
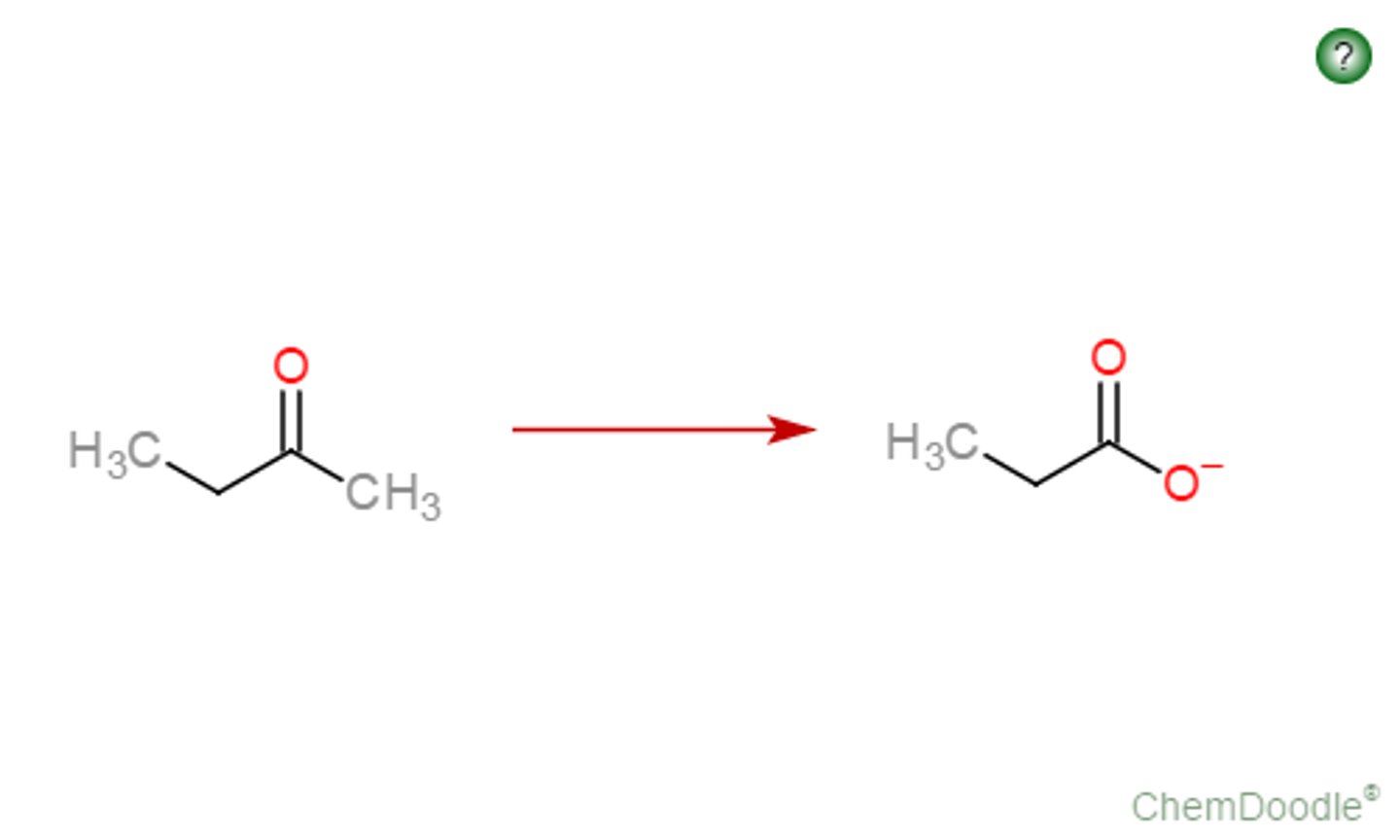
carboxylate anion
conjugate base (RCO2-) of a carboxylic acid, is produced by base-promoted reaction
Leaving Group (LG)
- atom or group of atoms which break away from the molecule
- accepts electrons from C-LG bond
Factors:
1. resonance (increases electron accommodation)
2. atomic radius (larger radius increases electron accommodation)
3. electronegativity (high EN increases electron accommodation)
4. inductive effects
5. formal charge
Excellent:
1. sulfonate ions
2. iodide ion
Moderate:
1. bromide ion
2. chloride ion
3. water
4. alcohols
Rare:
1. fluoride ion
2. amines
3. oxyanions
4. nitranions
Never:
1. alkyl group
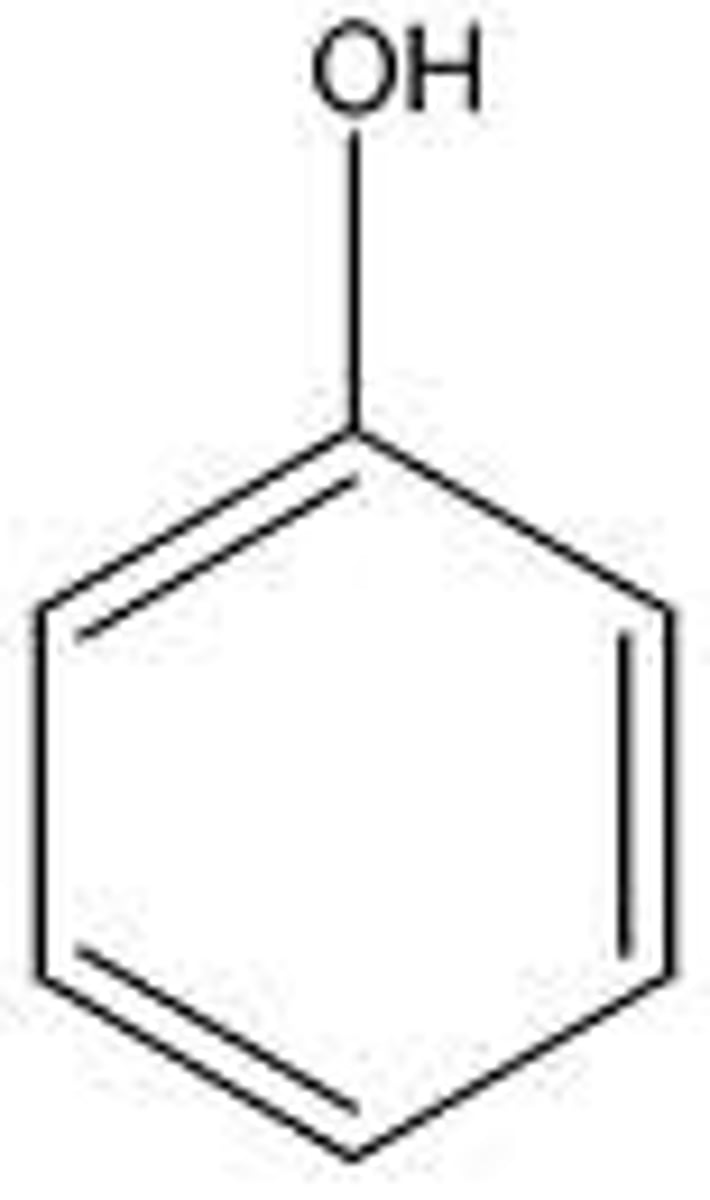
phenol
Benzene ring with -OH
pyridinium ion
5.2/ring w/three double bonds and N-H

Alkyl Halide
An alkane in which one of the hydrogens has been replaced by a halogen.
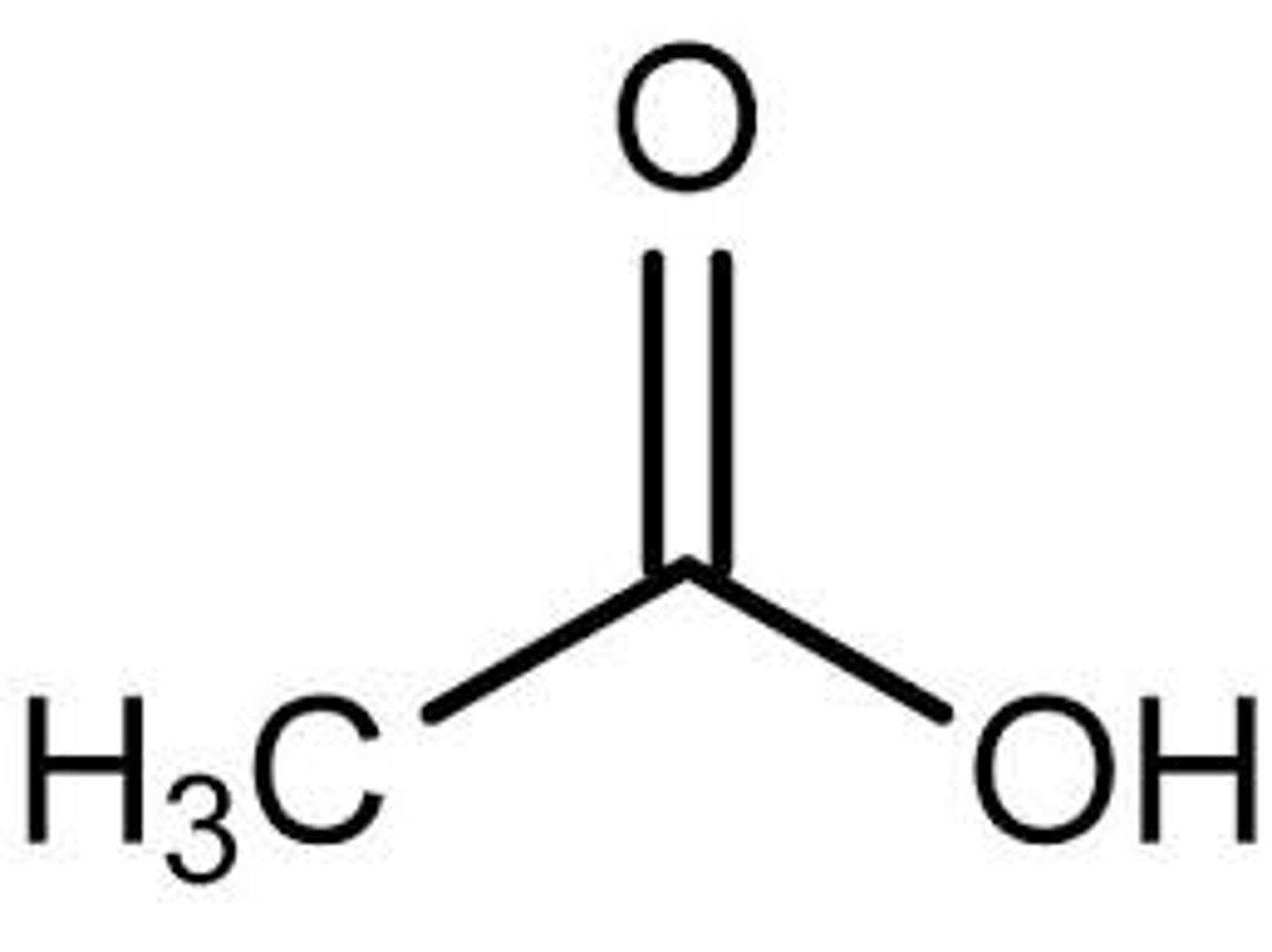
Acetic Acid
CH3COOH
Alkane
a hydrocarbon containing only single covalent bonds
1 carbon
methane
2 carbon chain
Ethane
3 carbon chain
Propane
4 carbon chain
Butane
5 carbon chain
Pentane
6 carbon
Hexane
7 carbon chain
Heptane
8 carbon chain
Octane
9 carbon chain
Nonane
10 carbon chain
Decane
isopropyl
1-methylethyl
CH3CHCH3
First-order units
1/s or s^-1
second-order units
M^-1 s^-1
SN1 rate law
rate = k[alkyl halide]
SN2 rate law
rate = k[alkyl halide][nucleophile]
SN1 activation barrier
stability of C+ intermediate
SN2 activation barrier
accessibility of LUMO simga star
SN1 nucleophile
weak nucleophiles, usually neutral (H2O, ROH, RNH2)
SN2 nucleophile
strong nucleophile (usually charged) -OH,-OR, -NRH
SN1 LUMO for addition
Empty 2p on C+
SN2 LUMO for addition
empty sigma star
SN1 sterochemistry (if RX has an asymmetric carbon)
racemic mixture
SN2 sterochemistry (if RX has an asymmetric carbon)
inversion
asymmetric carbon
A carbon that is attached to four different atoms or groups of atoms.
racemic mixture
a mixture of equal amounts of two enantiomers
SN1 solvent preference
Faster in polar protic (usually the nucleophile)
SN2 solvent preference
Faster in polar aprotic
Polar protic
methanol (CH3OH)
ethanol (CH3CH2OH)
water (H2O)
ammonia (NH3)
polar aprotic
Polar but do not have hydrogens bonded to highly electronegative atoms (DMSO)
SN1 alkyl halide
3>>>2>1
SN2 alkyl halide
1>>>2>3
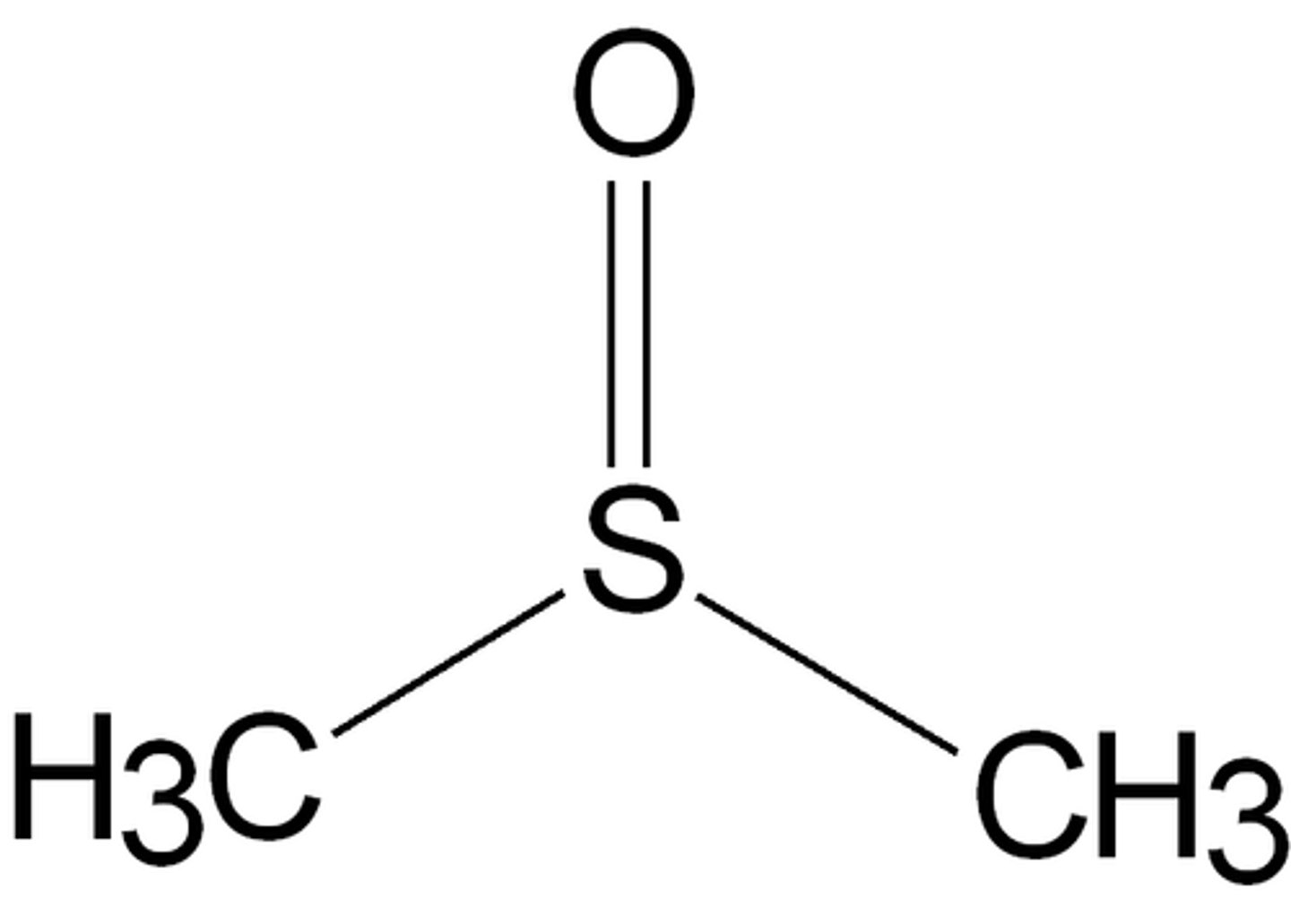
DMSO (dimethyl sulfoxide)
polar aprotic
RX
electrophile
Primary Alkyl Halide (1)
An alkyl halide in which the carbon atom bonded to the halogen is also bonded to 1 other carbon atom.
Secondary Alkyl Halide (2)
The carbon to which the halogen is bonded is attached to two other carbons
Tertiary Alkyl Halide (3)
The carbon to which the halogen is bonded is attached to three other carbons
Zeroth Order Rate Law
Rate = k (change in concentration has no effect on the rate)
First Order Rate Law
Rate = k[A]
Second Order Rate Law
Rate=k[A]^2
Zeroth order integrated rate law
[A]t = [A]0 - kt
First Order Integrated rate law
ln[A] = - kt + ln[A]0
second order integrated rate law
1/[A]t = kt + 1/[A]0
zeroth order units
m/s
Rates
Rates of appearance or disappearance. Dependent on stoichiometric relationships (the balanced equation)
Rate Law
k= rate constant, units vary depending on x and y. x and y can only be determined from experiment.
1. The magnitude of k indicates how fast or slow the reaction is.
2. Rate law is dependent on concentrations, not stoichiometry
3. Must be determined experimentally
4. Rate constant k is not the same as the rate of the reaction
Arrhenius equation
k=Ae^(-Ea/RT) or ln(k2/k1)=-Ea/R((1/T2)-(1/T1))
Half-life (t1/2)
The time it takes for the amount of drug to fall to half of its value; this is a constant in first-order kinetics. This is independent of initial concentrations.
Steps for a ester hydrolysis
PADPED
Kinetics
"How fast?"
- Collisons
- mechinsim/pathway
- catalysis
- path dependent
Thermodynamics
"How far"
- Equilibrium (delta G naut = -RTlnK)
- Path independent
Free Energy (delta G)
Spontaneity
Enthalpy (delta H)
The property that quantifies thermal energy transfer in or out of a system at constant pressure
Entropy
How is energy spread out in a system? Higher entropy correlates with more possible microstates, representing greater matter dispersal
1st Law of Thermodynamics
Energy cannot be created or destroyed, only exchanged
2nd Law of Thermodynamics
The entropy of the Universe increases in all spontaneous processes.
Gibbs Free Energy Equation
ΔG = ΔH - TΔS
hydrophobic effect
The observed tendency of nonpolar substances to aggregate in aqueous solution and exclude water molecules. This increases the water entropy as they no longer have to make a rigid structure around non-polar substances.
Le Chatelier Equation
Delta G= RTln(Q/K)
Lewis Acid
A compound that accepts an electron pair
Lewis Base
A compound that donates an electron pair
Bronsted-Lowry acid
A compound that donates a proton (H+) in a reaction
Bronsted-Lowry base
A compound that accepts a proton (H+) in a reaction
Arrhenius Acid
A compound that, when dissolved in water, increases the hydronium ion concentration
Arrhenius Base
A compound that, when dissolved in water, the hydroxide ion concentration increases
Lewis acid-base adduct
A compound that contains a coordinate covalent bond formed between a Lewis acid and a Lewis base.