Analytical Separation
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
The purpose of separation in analytical chemistry is to _______.
separate different analytes from each other to reduce interference
To deal with Interferences:
❑Using a masking agent to immobilize or chemically bind the
interference species.
❑Converting either the analyte or the interferences into a
separate phase that can be separated mechanically.
Extraction
• Transfer of solute from one phase to another in which is more soluble.
• Phases can be liquid, solid or gas.
Liquid/liquid extraction
Two immiscible layers: organic (S2) and aqueous (S1) in separatory funnel
Immiscible liquids remain in separate phases

Solute “partitions” between two solvents:
❑Solute prefers the solvent that is more like itself
❑Polar and ionic compounds partition into water (s1)
❑Nonpolar compounds partition into organic solvents (s2)
Partition Coefficient (K) works for _______.
molecules that only have one form
K = [S2]/[S1]
![<p>molecules that only have one form</p><ul><li><p>K = [S2]/[S1]</p></li></ul><p></p>](https://assets.knowt.com/user-attachments/bb8a9074-b1f9-499b-9ac5-31b39b8ae92e.png)
Distribution Coefficient (D) works for ______.
species that have multiple forms
ex: acids and bases
Ideally, K is _______.
independent of the total quantity of S
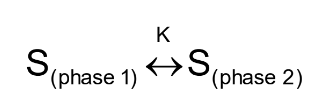
Partition Coefficient (K)
Ratio of solute (S) concentrations in each phase (phase 1 and phase 2) at equilibrium
K = (1-q) V1 / qV2
The larger the partition coefficient, the _______.
smaller amount of solute that remains in phase 1 (q)
q
fraction of S in phase 1 after extraction
q = V1/V1+KV2
pH dependent
V1
volumes of solvent in phase 1
V2
volumes of solvent in phase 2
m
number of moles of S (total)
How to move S to solvent 2 as much as possible?
K should be large and q should be small
The fraction of solute remaining in phase 1 depends on (q):
V1, V2: →larger V2, q smaller ; larger V1, q larger
K → larger K, q smaller
For extraction efficiency, perform _______.
more extractions with small volumes
gives smaller q
To improve extraction efficiency, we want ______.
a small V1, large V2, and small q
Neutral species are more soluble in ______.
organic phase, while charged species are more soluble in water
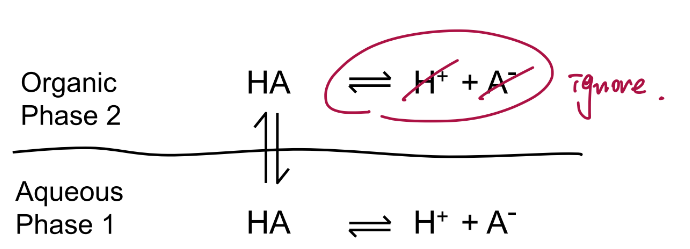
Charge dictates solubility and ______.
pH dictates charge
The analyte present in more than one chemical form, its distribution between two phases is described by:
Distribution Coefficient, D.
❑Takes into account all chemical forms of a compound.
❑Different from partition coefficient (K) for those analytes that can be present in more than one chemical form.
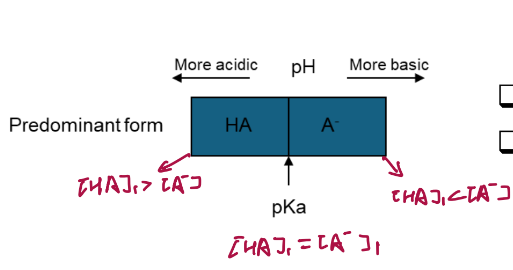
If a solute is an acid or base its charge _______.
changes as the pH changes
Distribution coefficient D is used in place of the ______.
K in the equation for q when dealing with species that has more
than one chemical form
D equation in terms of HA
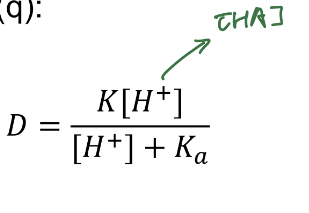
q in terms of D (use for weak acid and base)
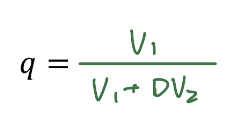
D in terms of a base
αB is the fraction of weak base in the neutral form, B, in the
aqueous phase.
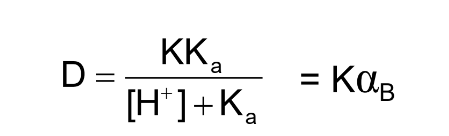
To extract a base into water, use a _______.
pH low enough to convert B into BH+.
To extract an acid HA into water, use a ______.
pH high enough to convert HA into A-.