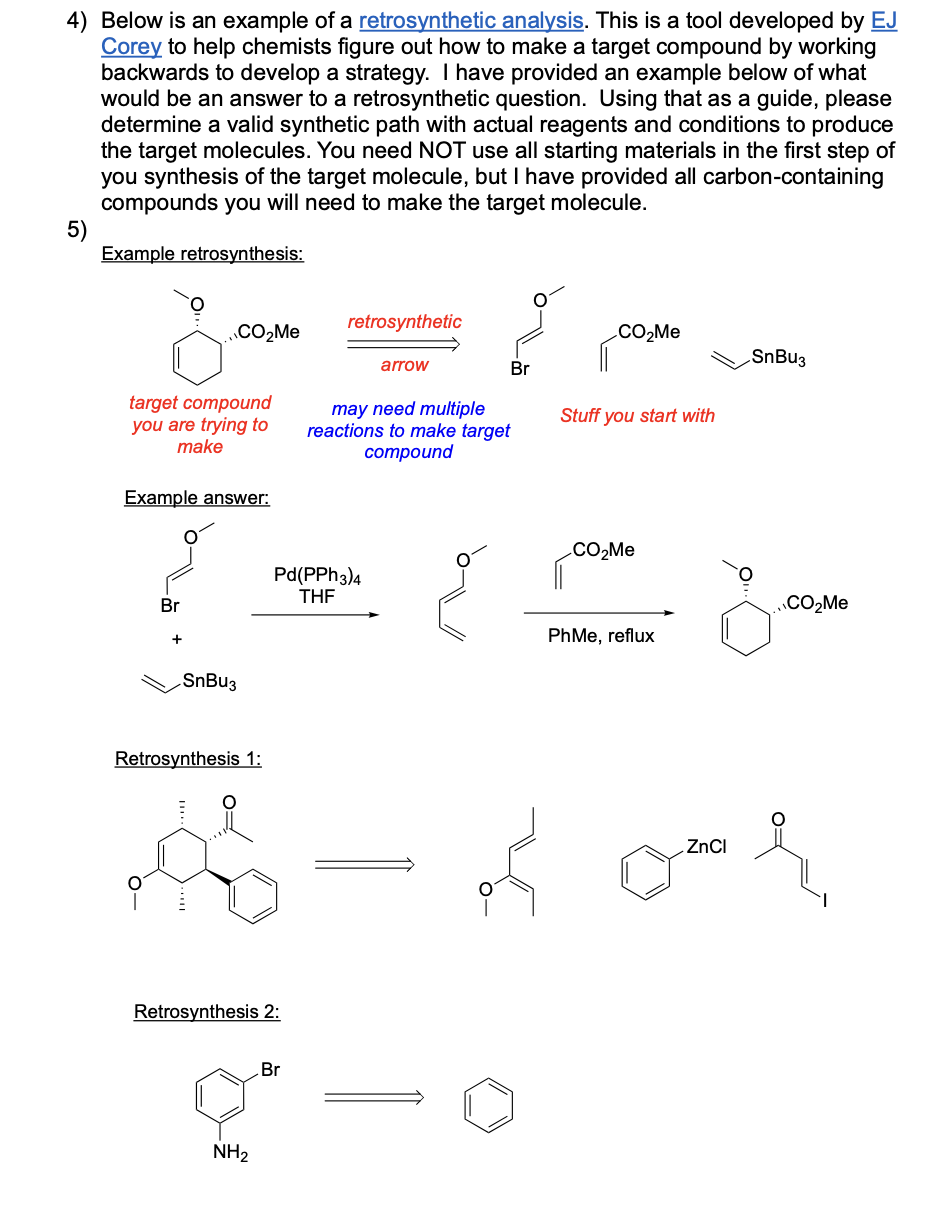
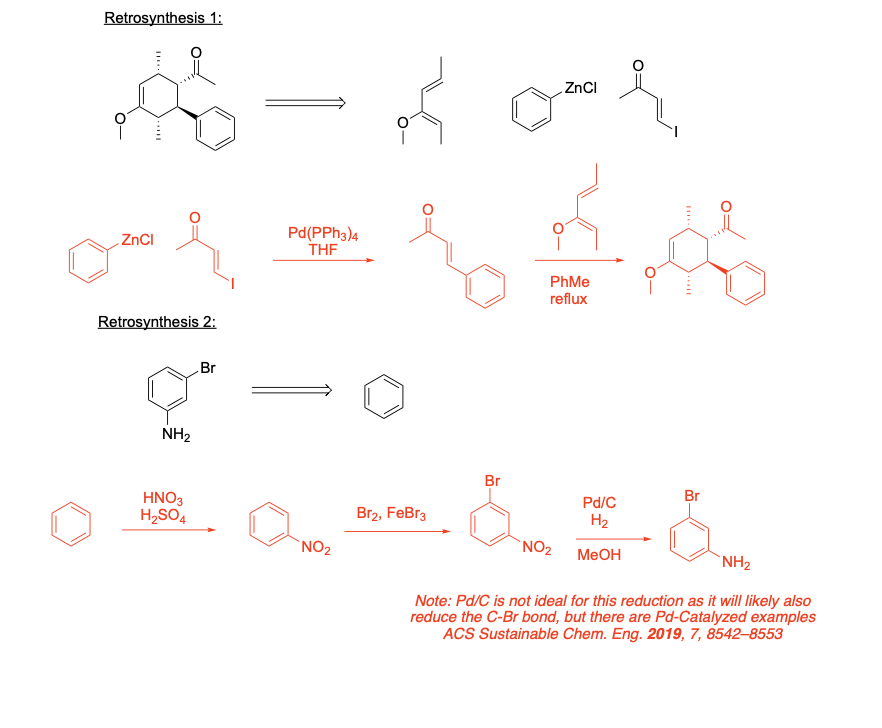
Ochem Midterm 1+2, SI Worksheets
1/99
Earn XP
Description and Tags
You already know broh
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
100 Terms
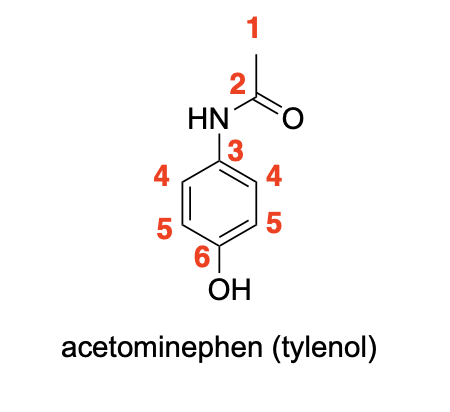
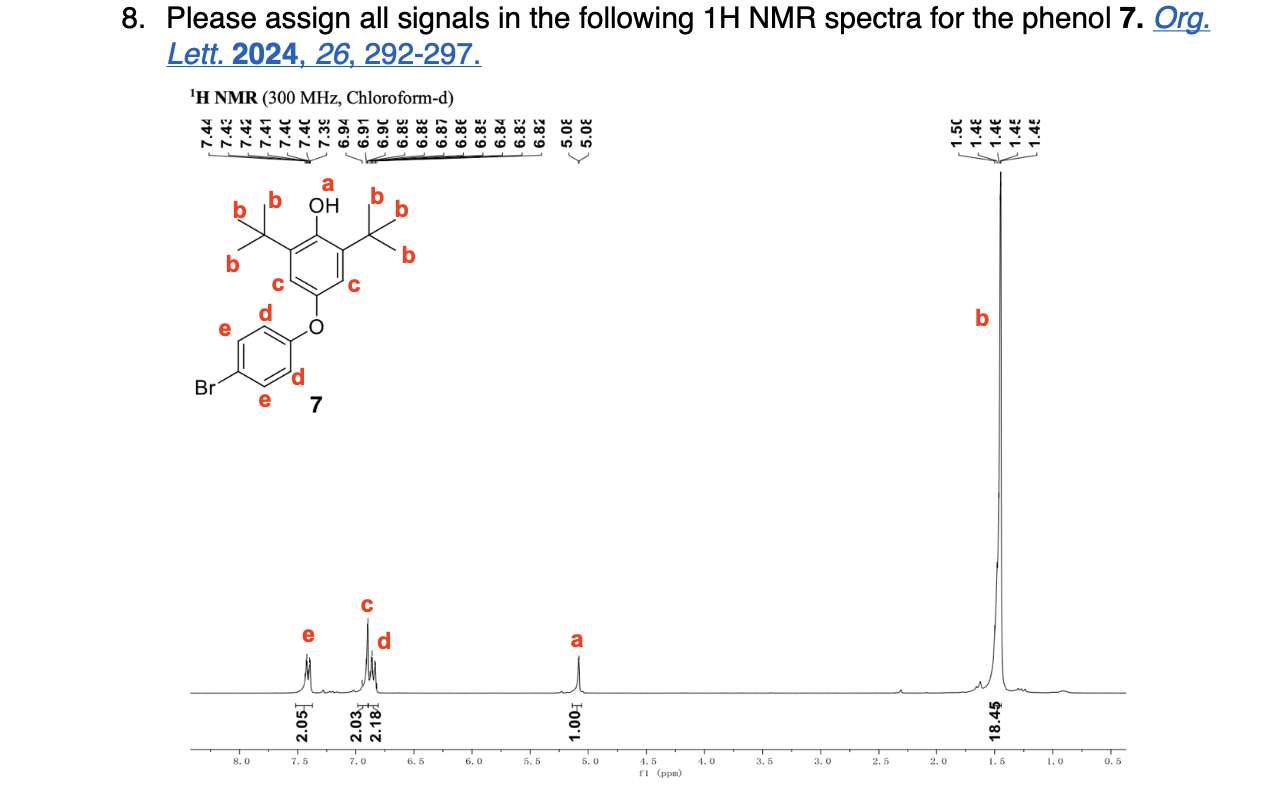
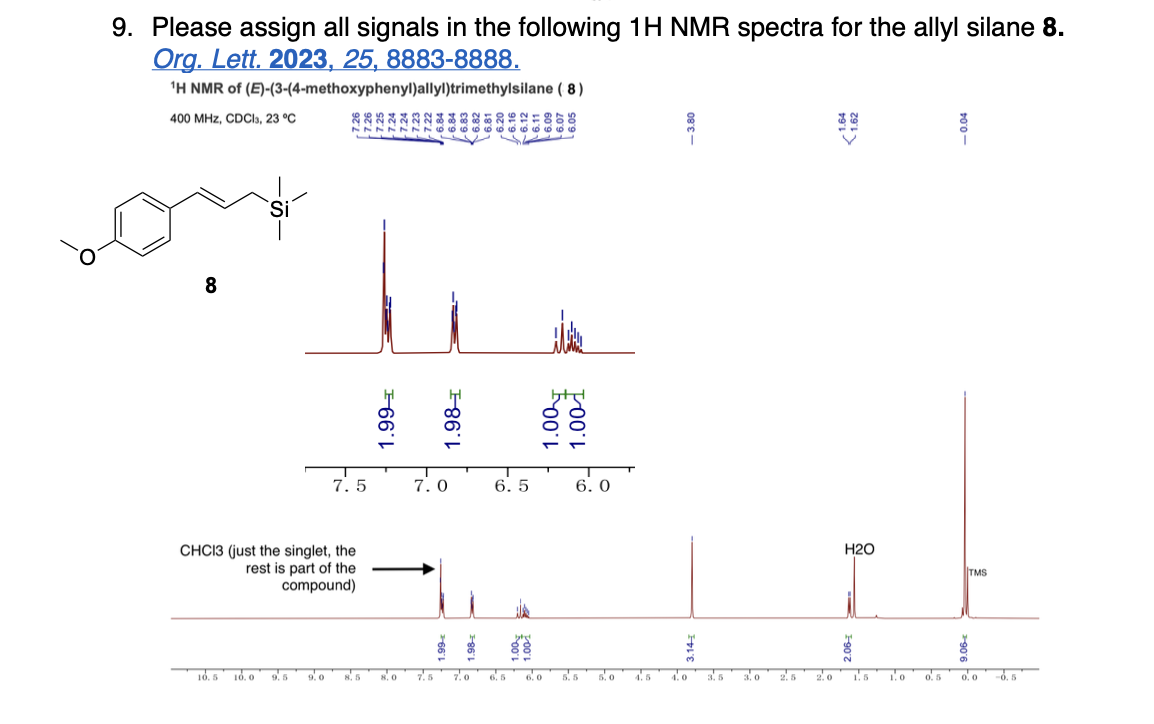
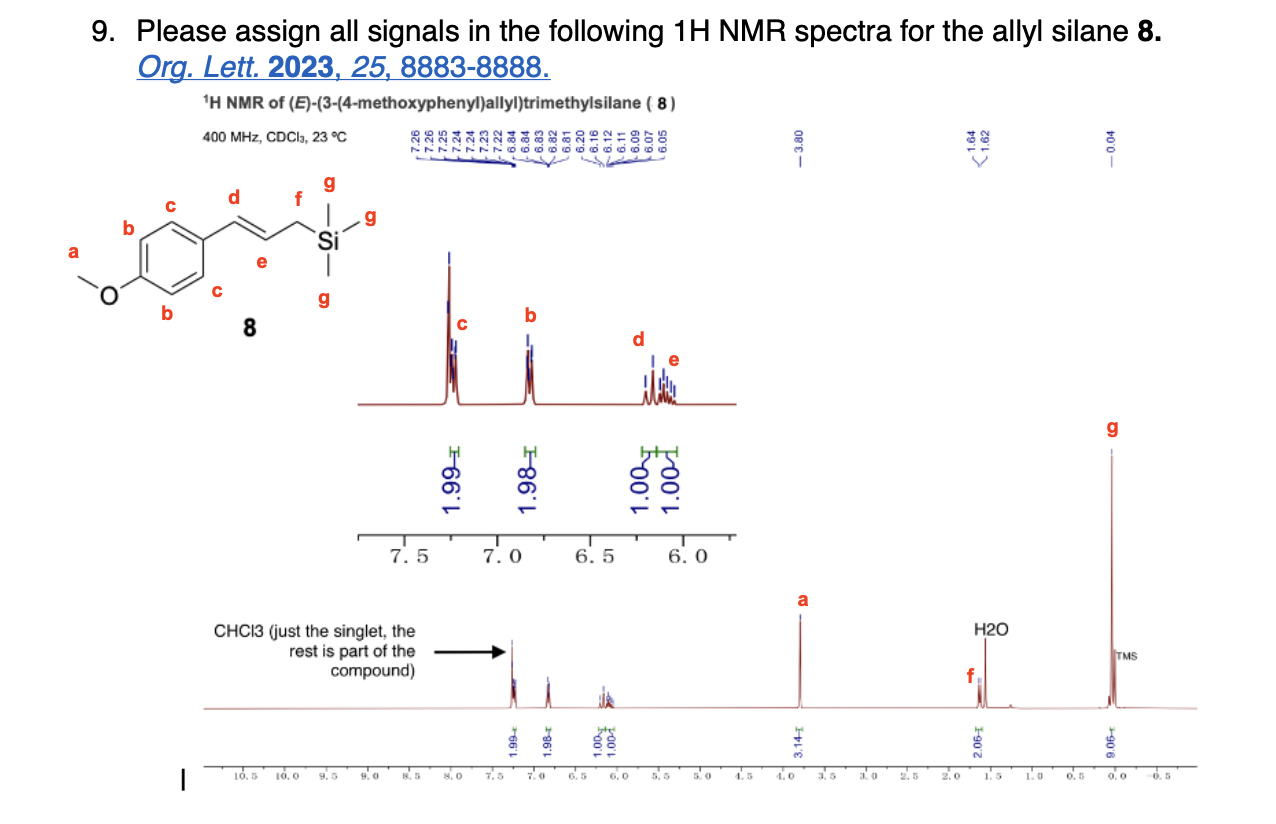
Label 1H NMR Signals
Label 1H NMR Signals
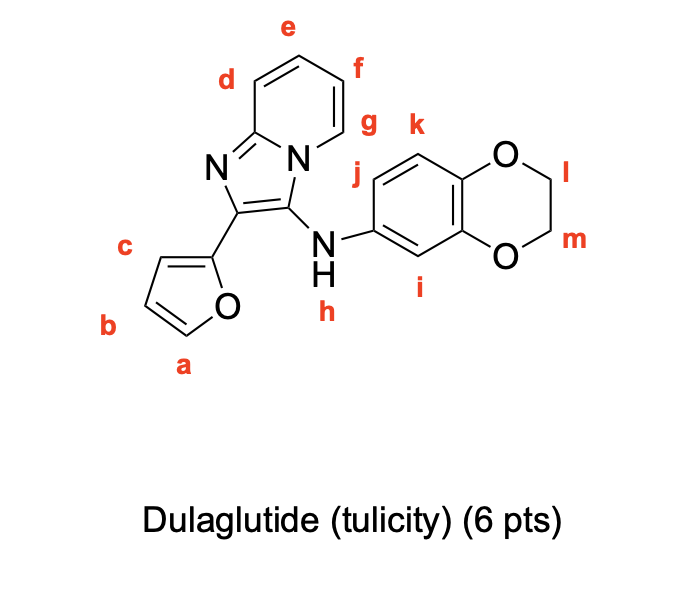
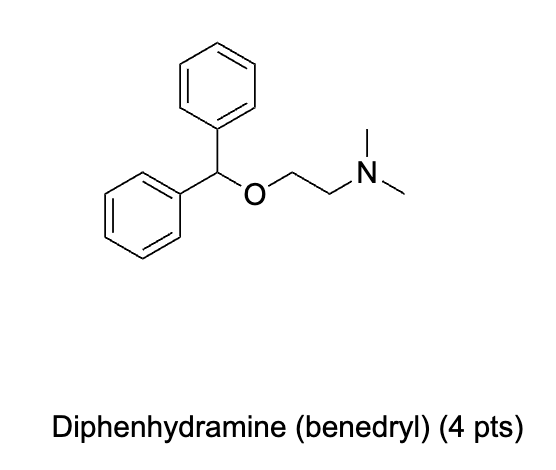
Label 1H NMR Signals
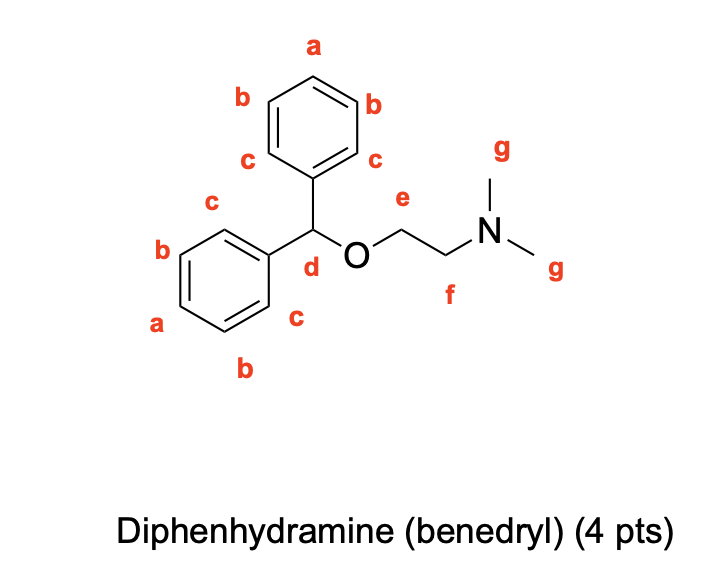
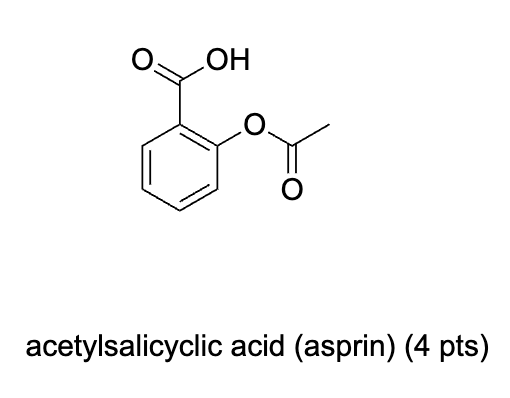
Label 1H NMR Signals
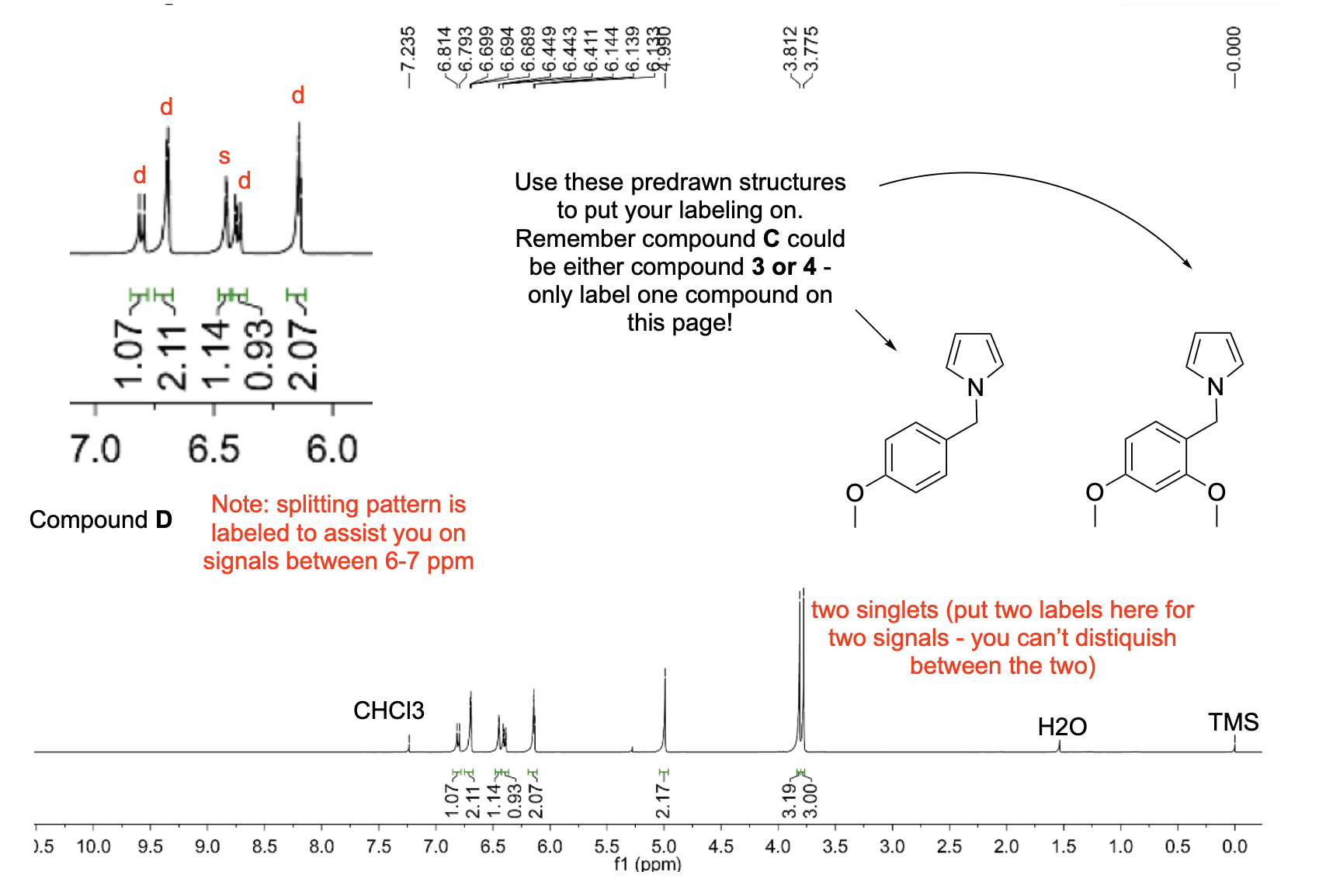
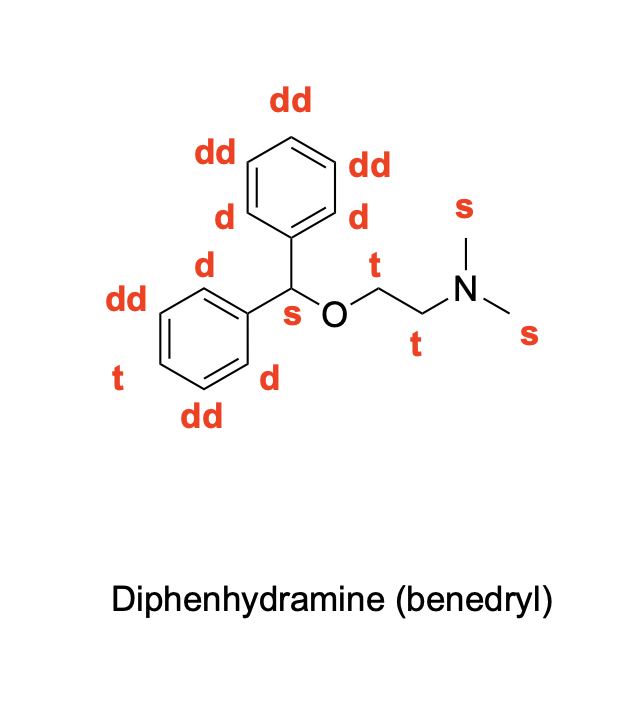
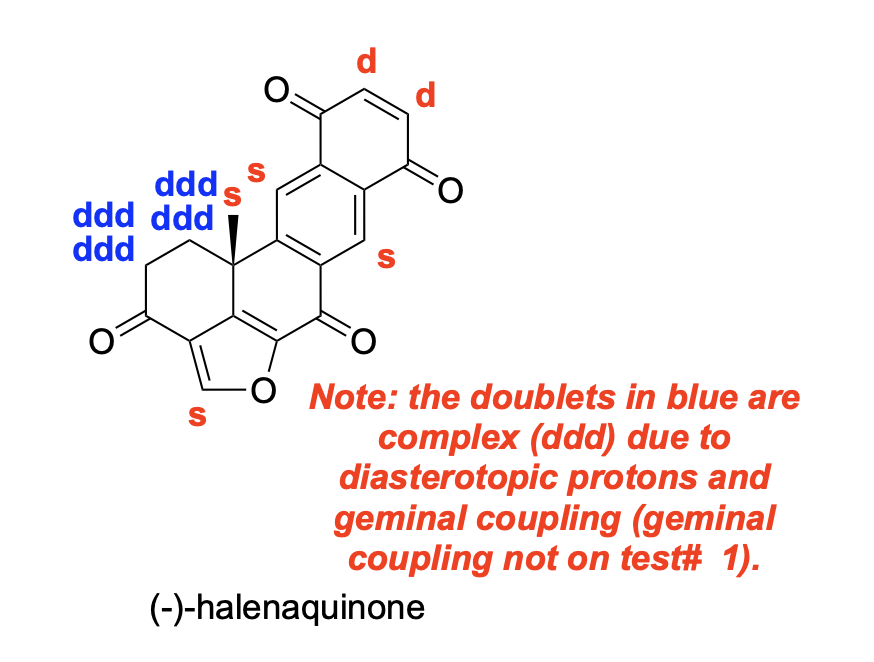
denote the expected splitting pattern with abbreviated label in a 1H NMR spectra for each indicated position in the following structure
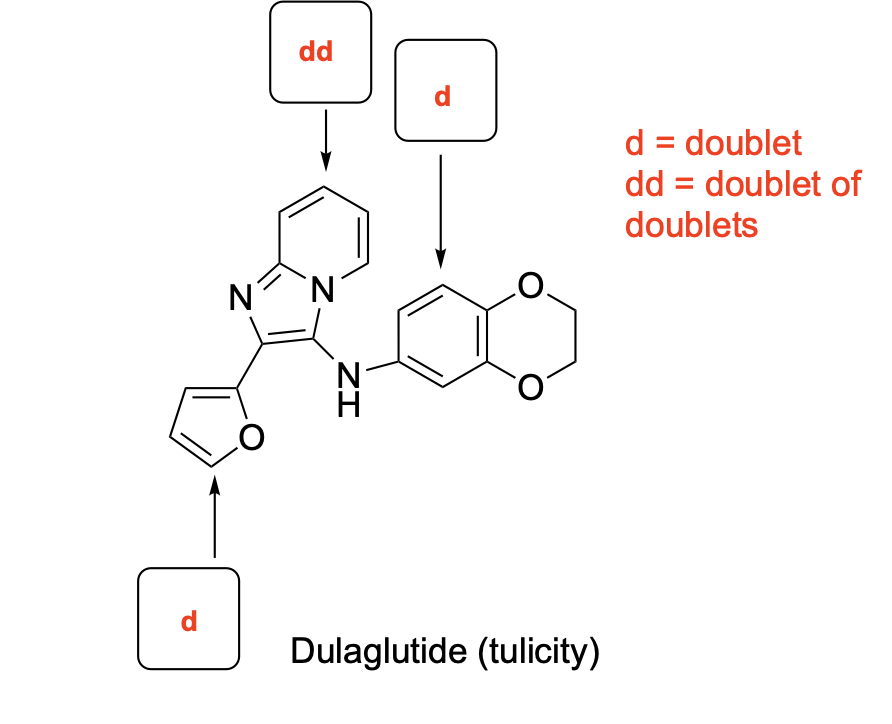
denote the expected splitting pattern with abbreviated label in a 1H NMR spectra for each indicated position in the following structures.
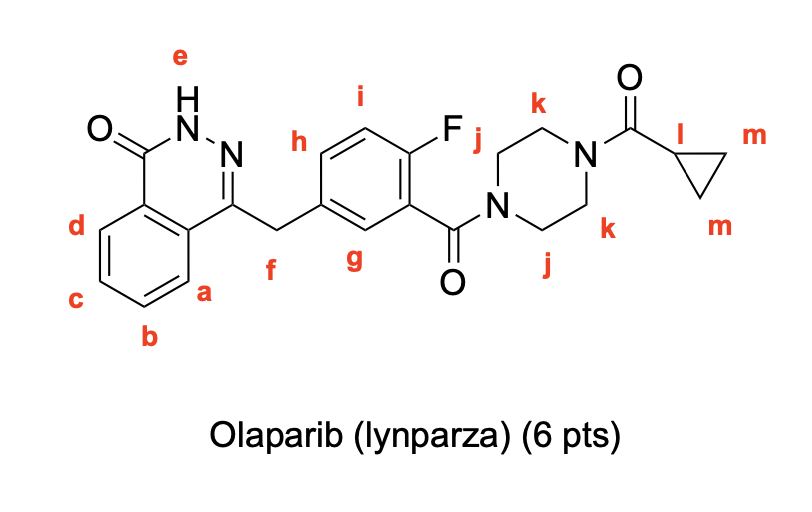
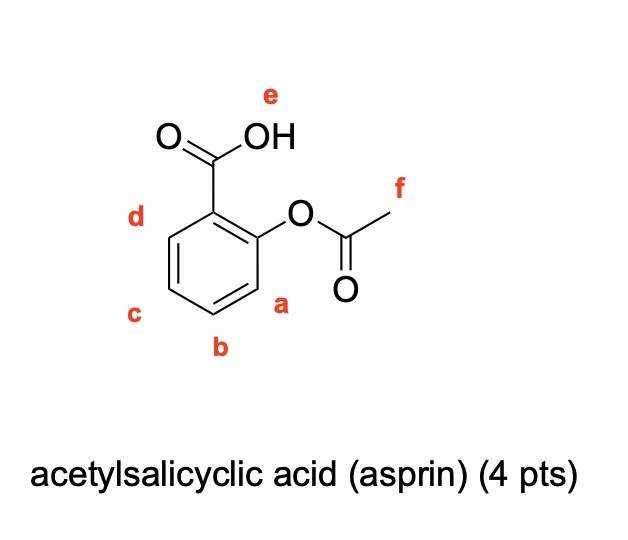
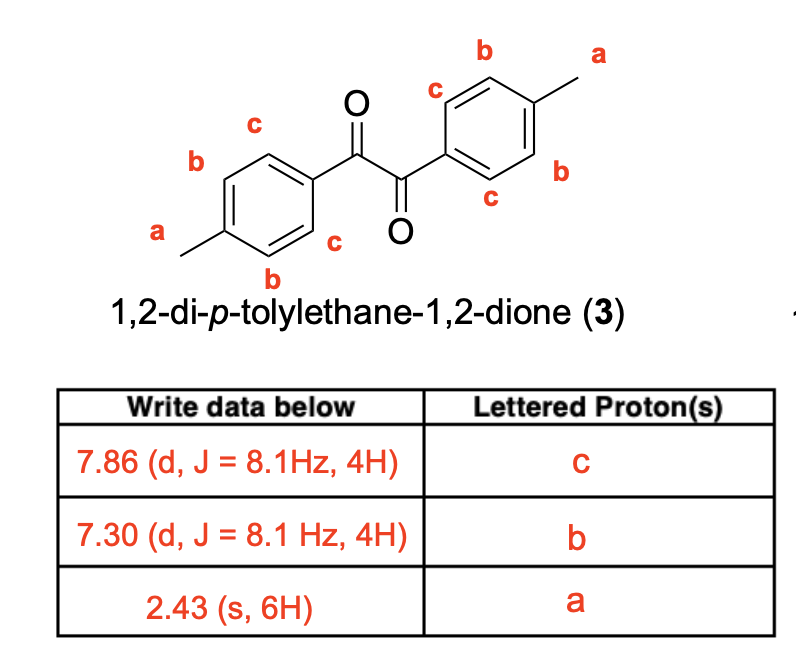
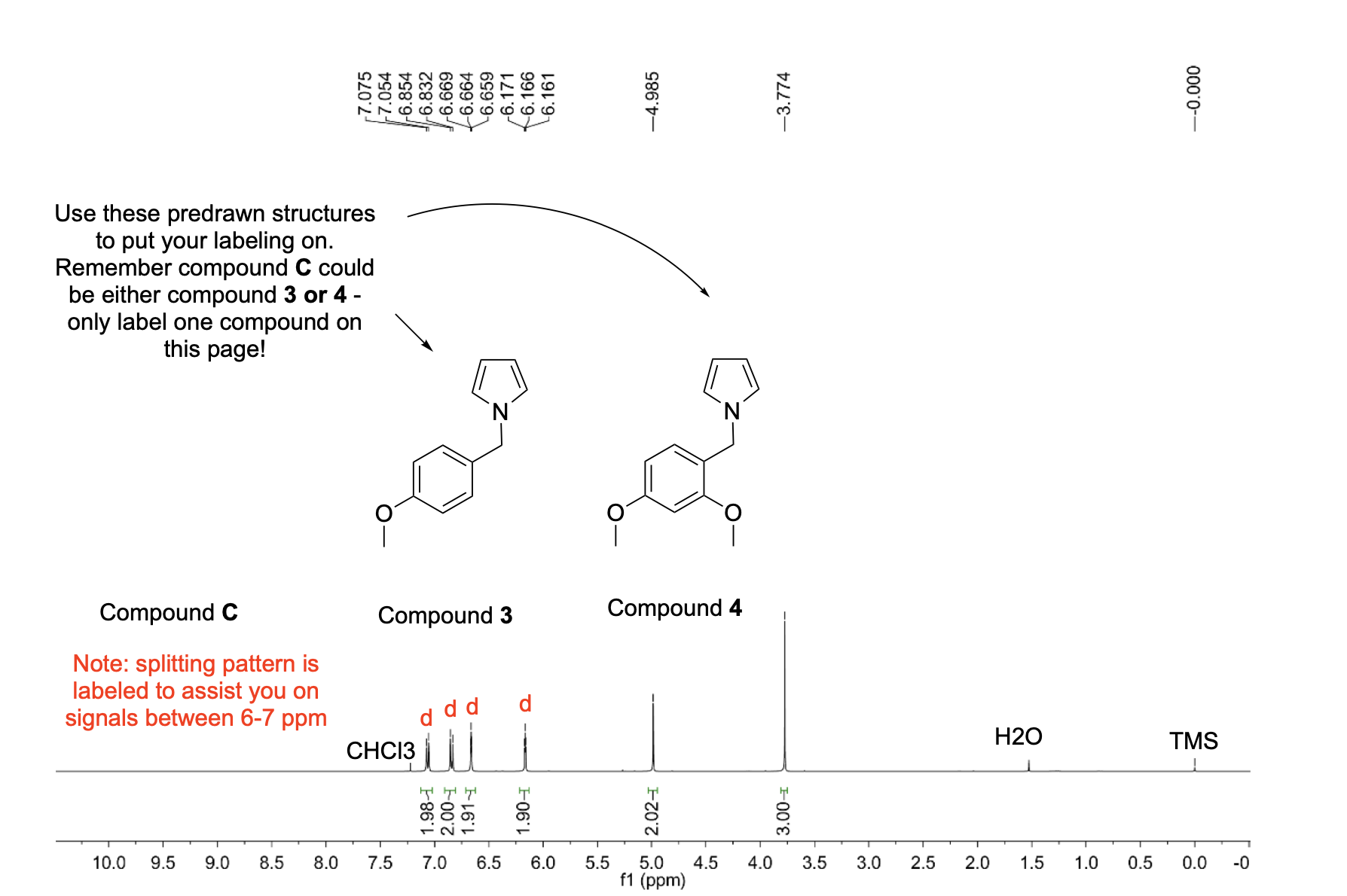
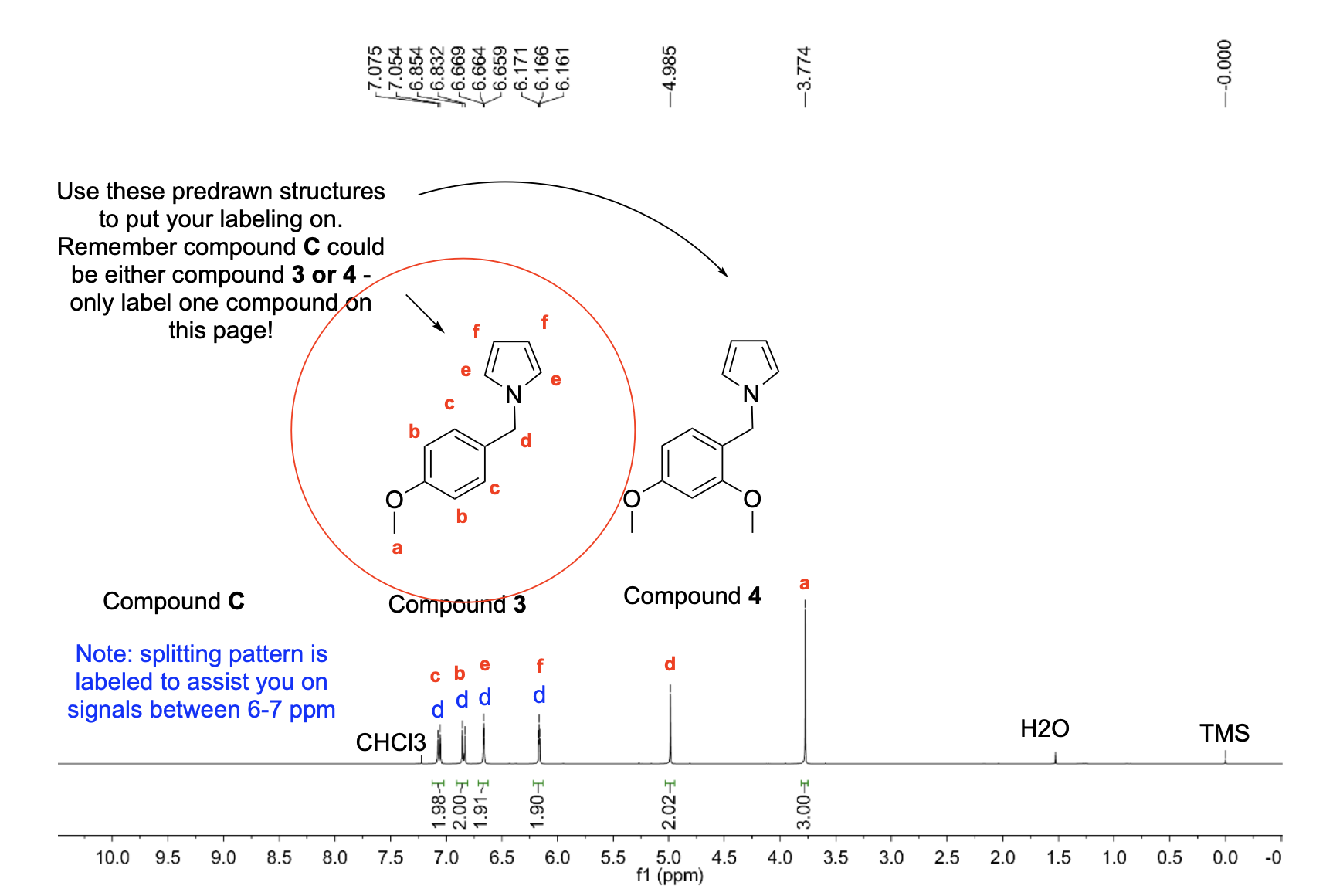
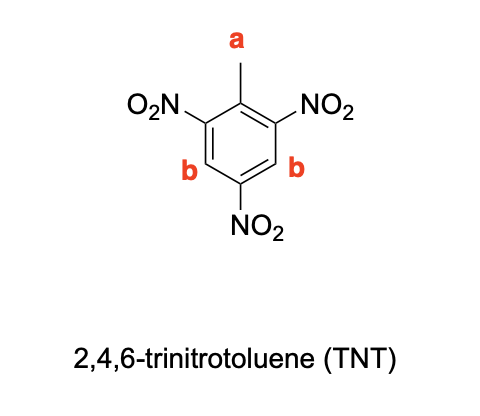
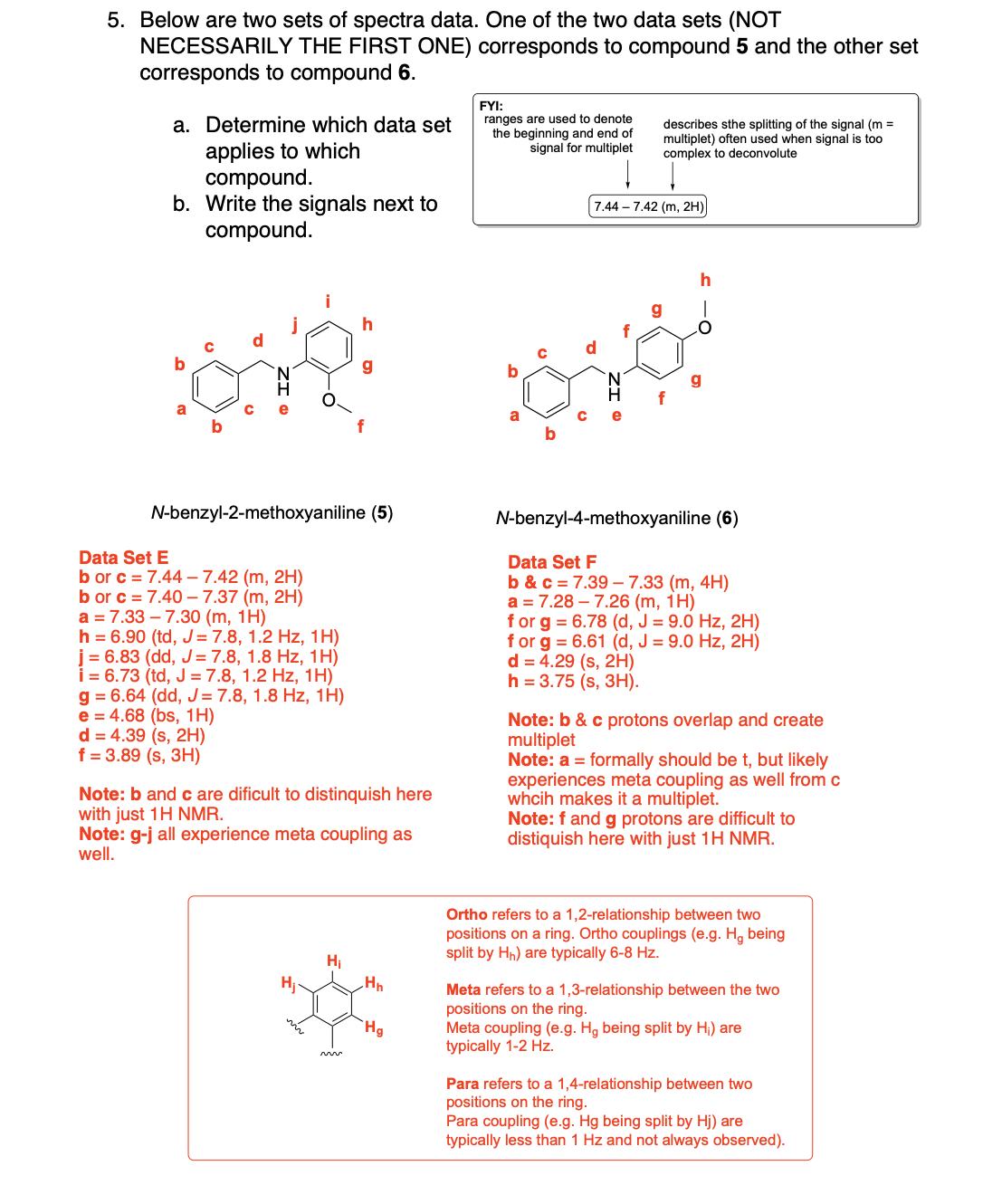
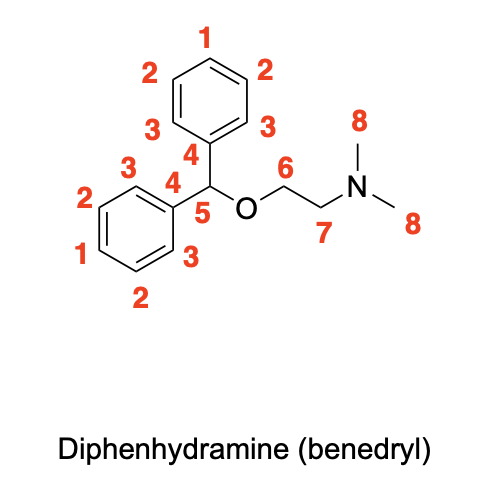
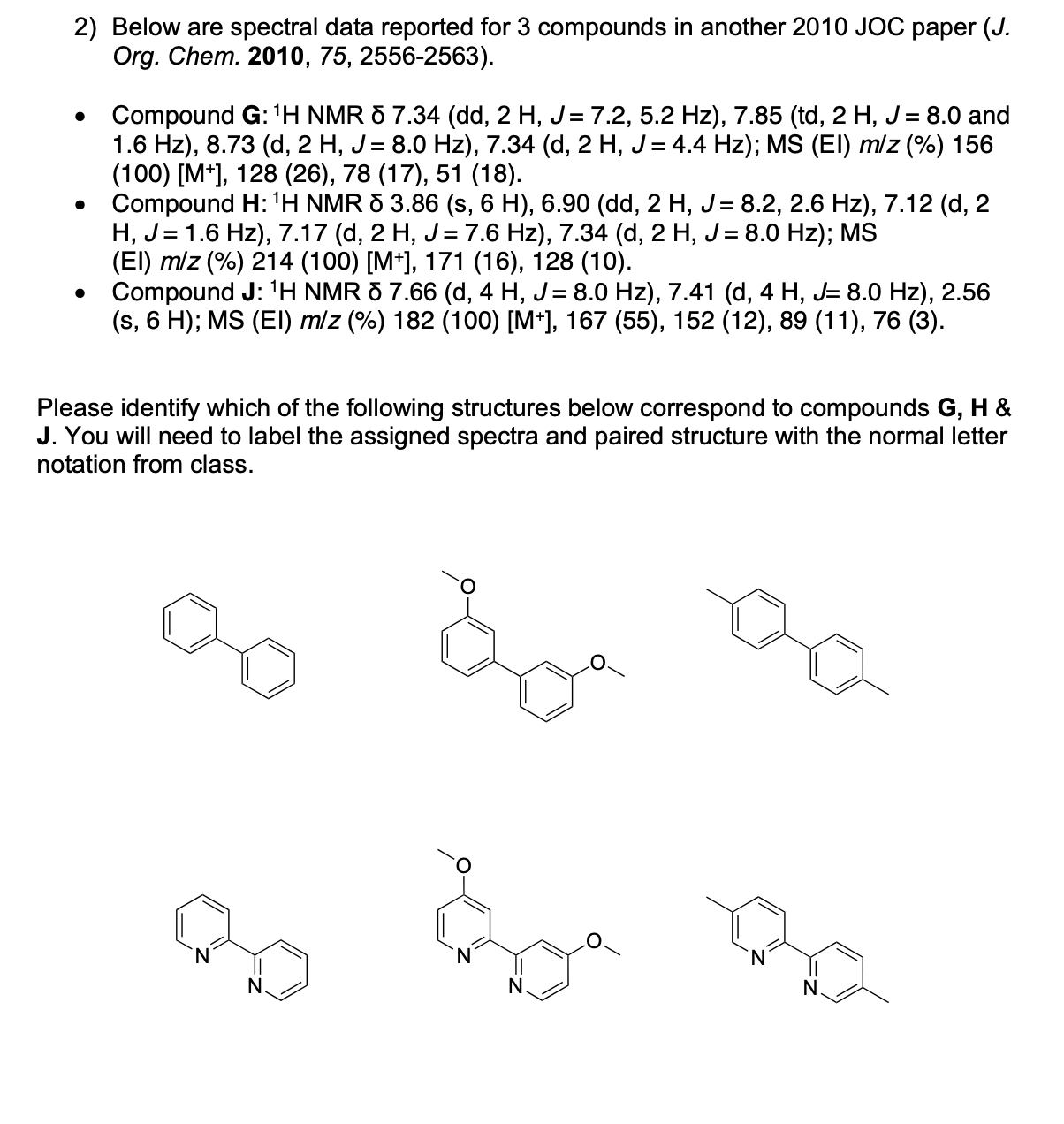
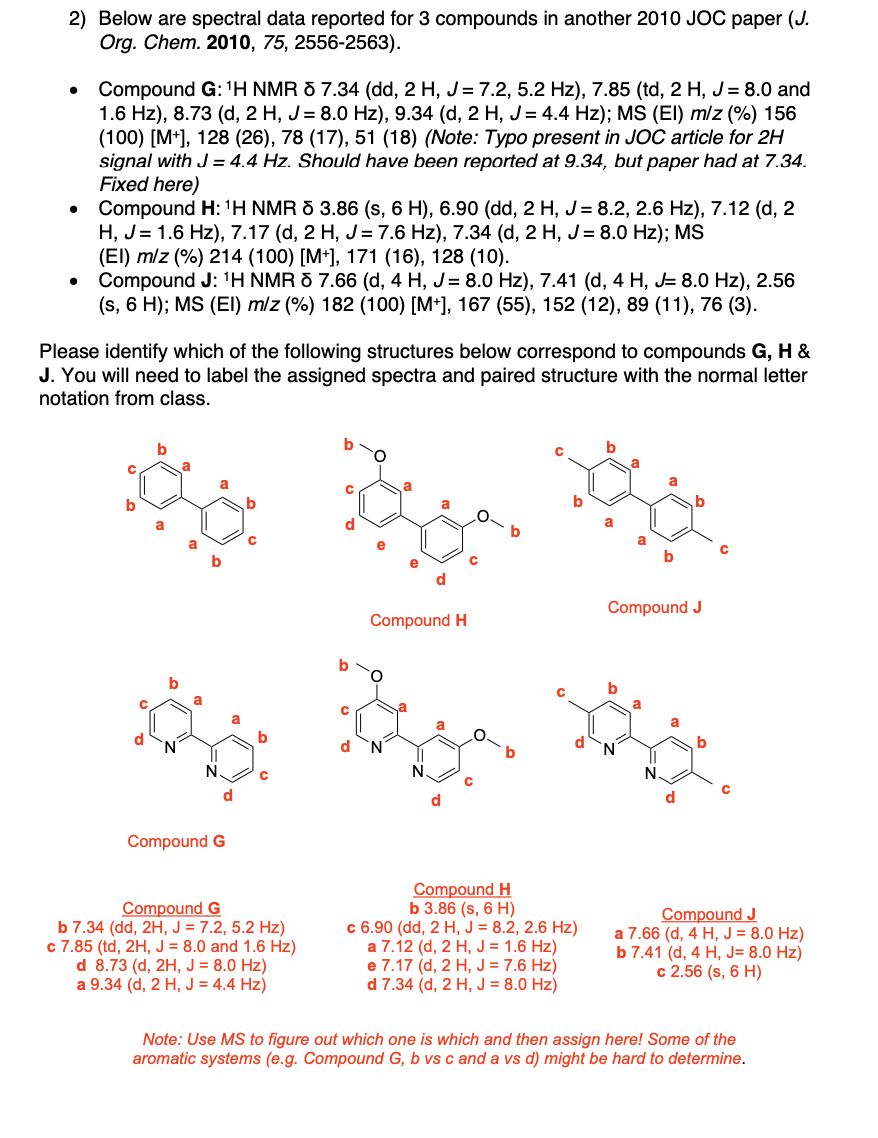
Label the following compounds for unique 1H NMR signals using normal lettering notation
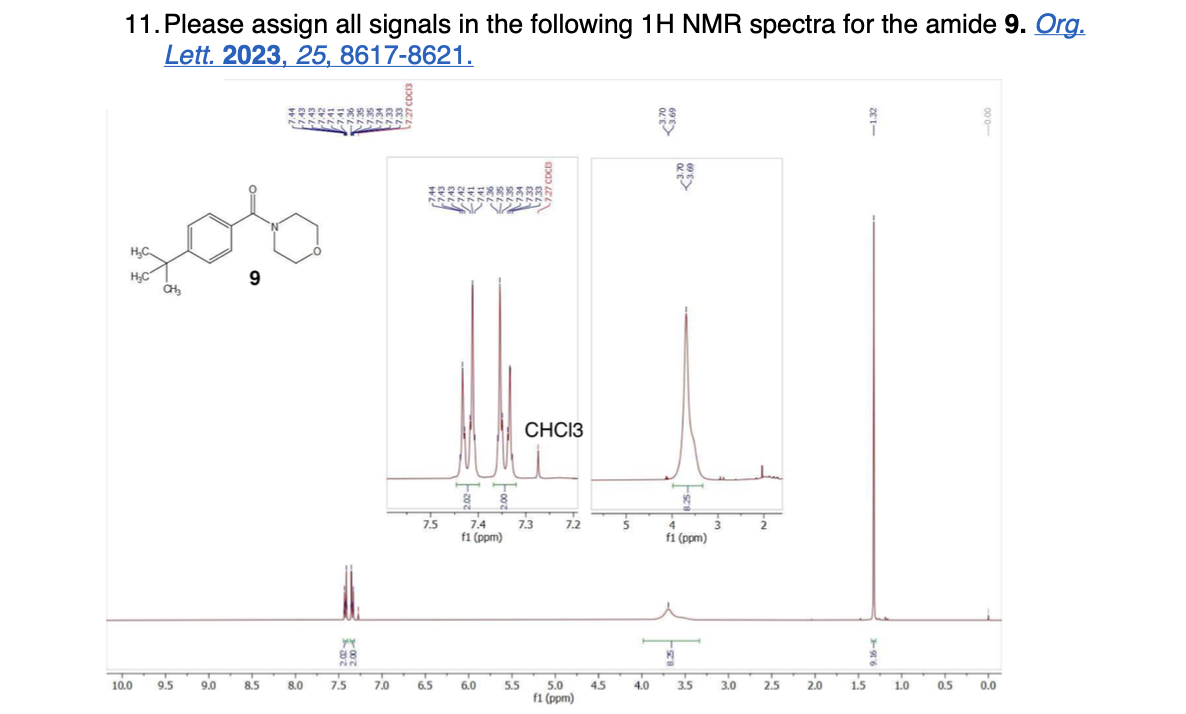
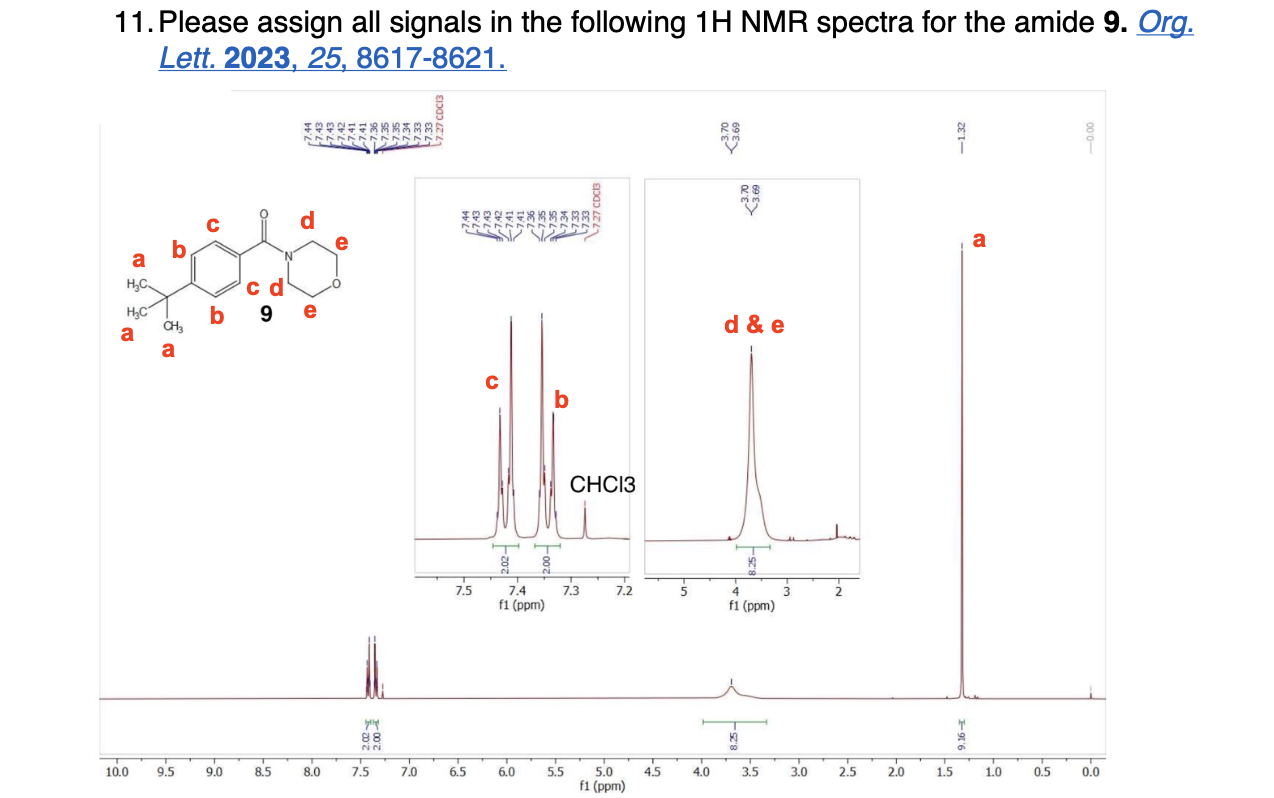
Label the following compounds for unique 1H NMR signals using normal lettering notation
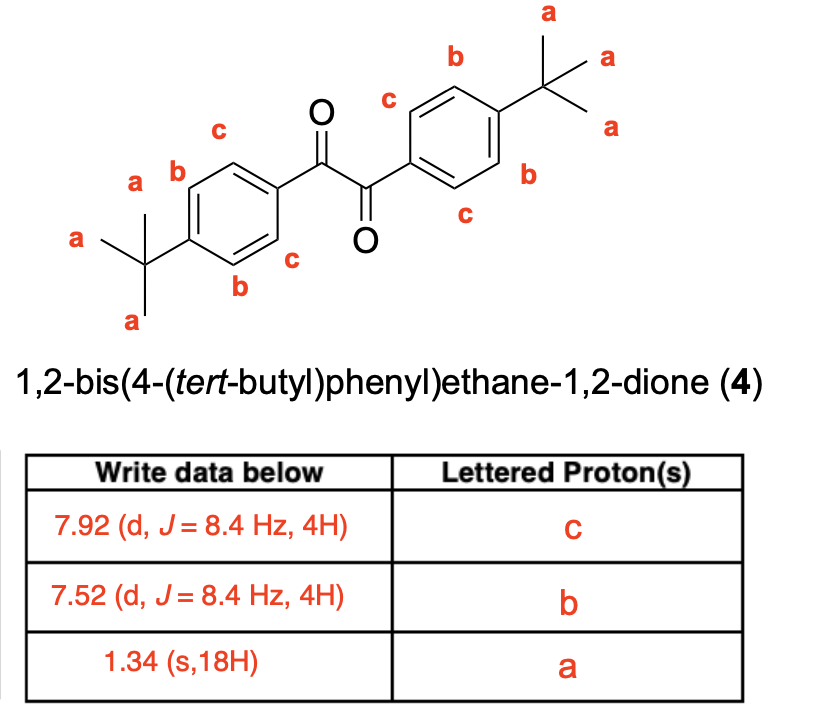
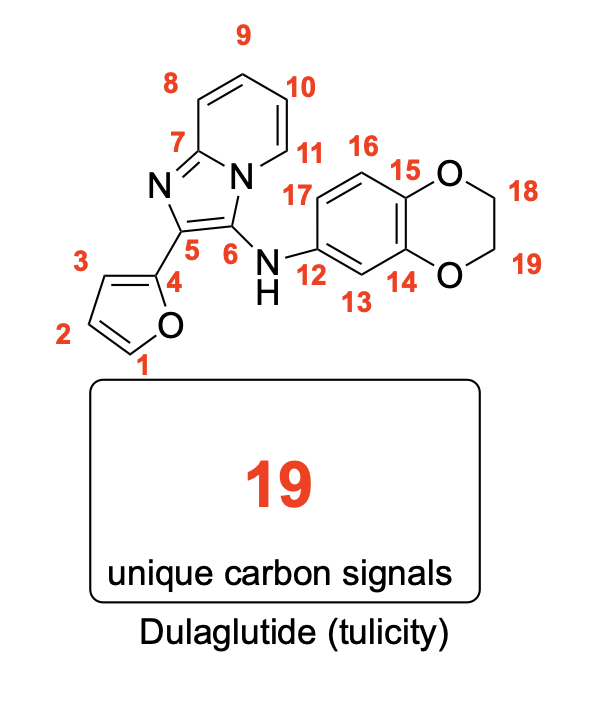
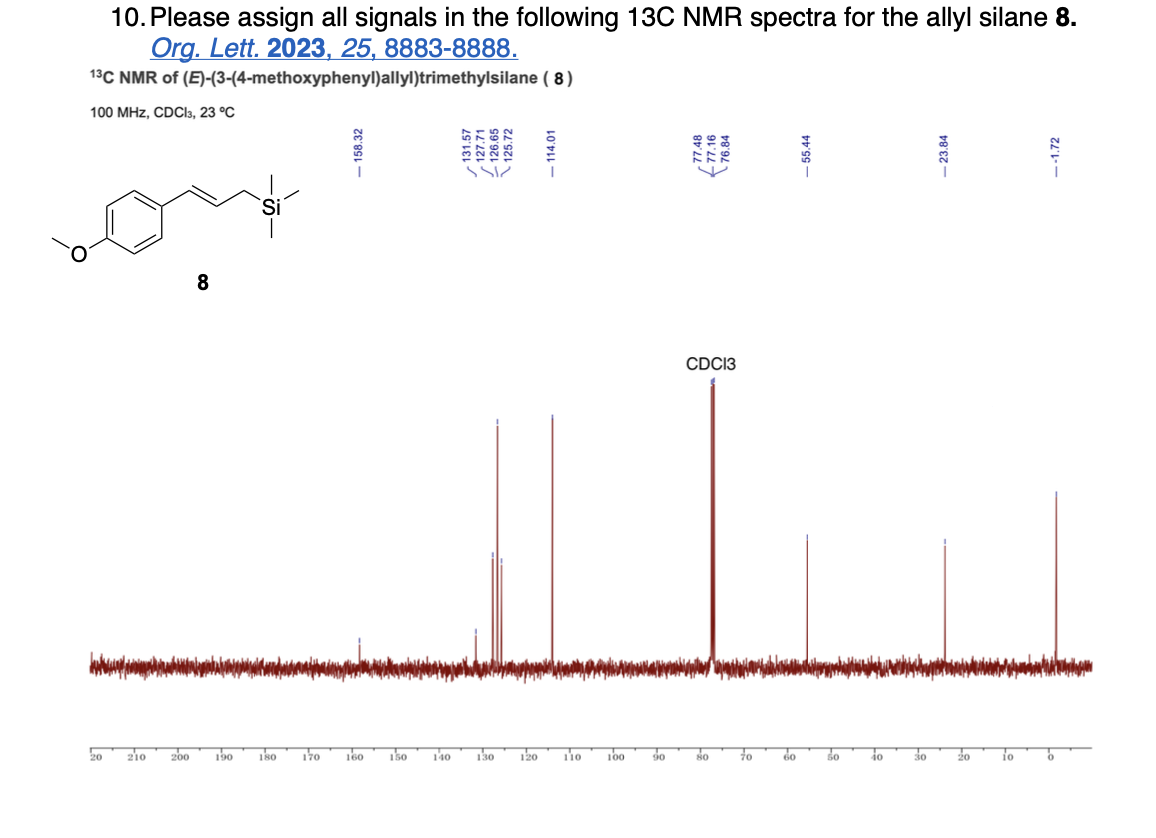
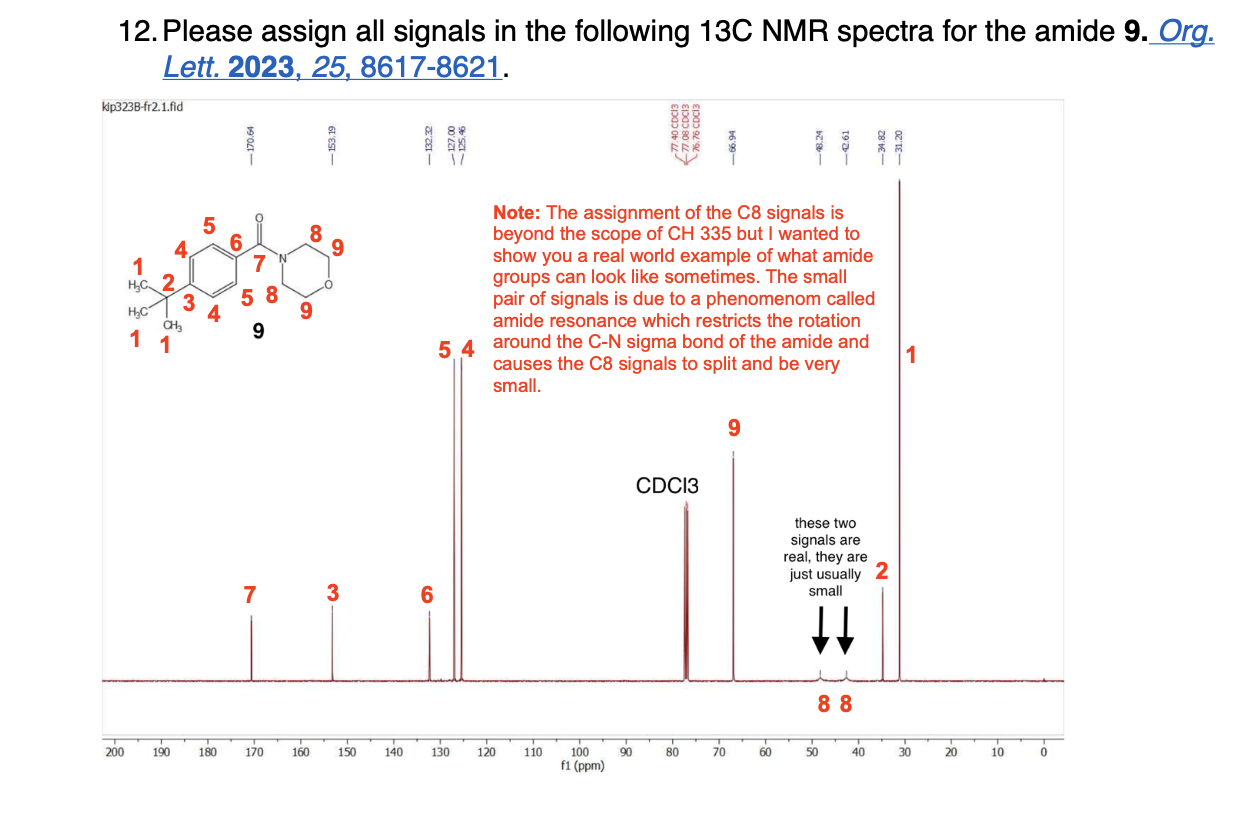
How many unique 13C NMR signals will be observed
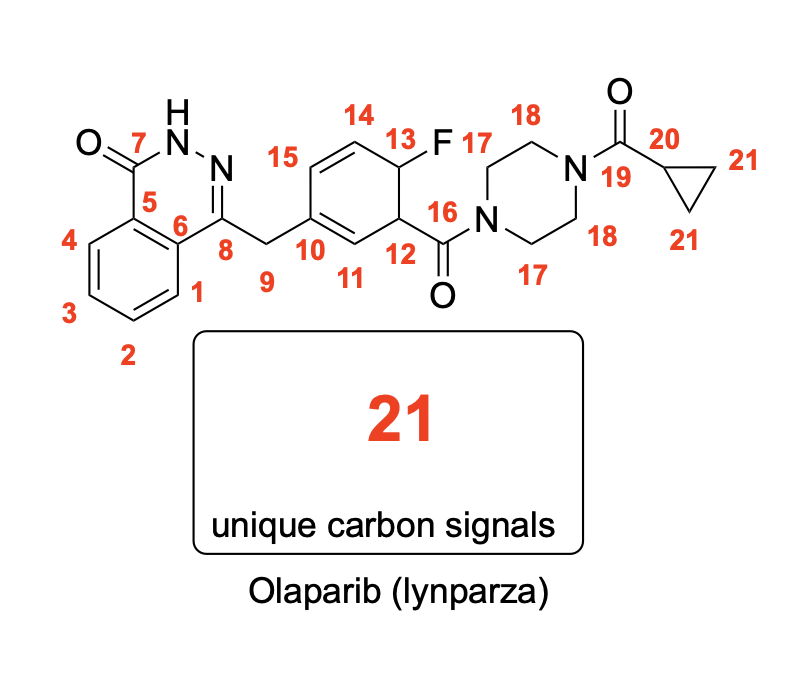
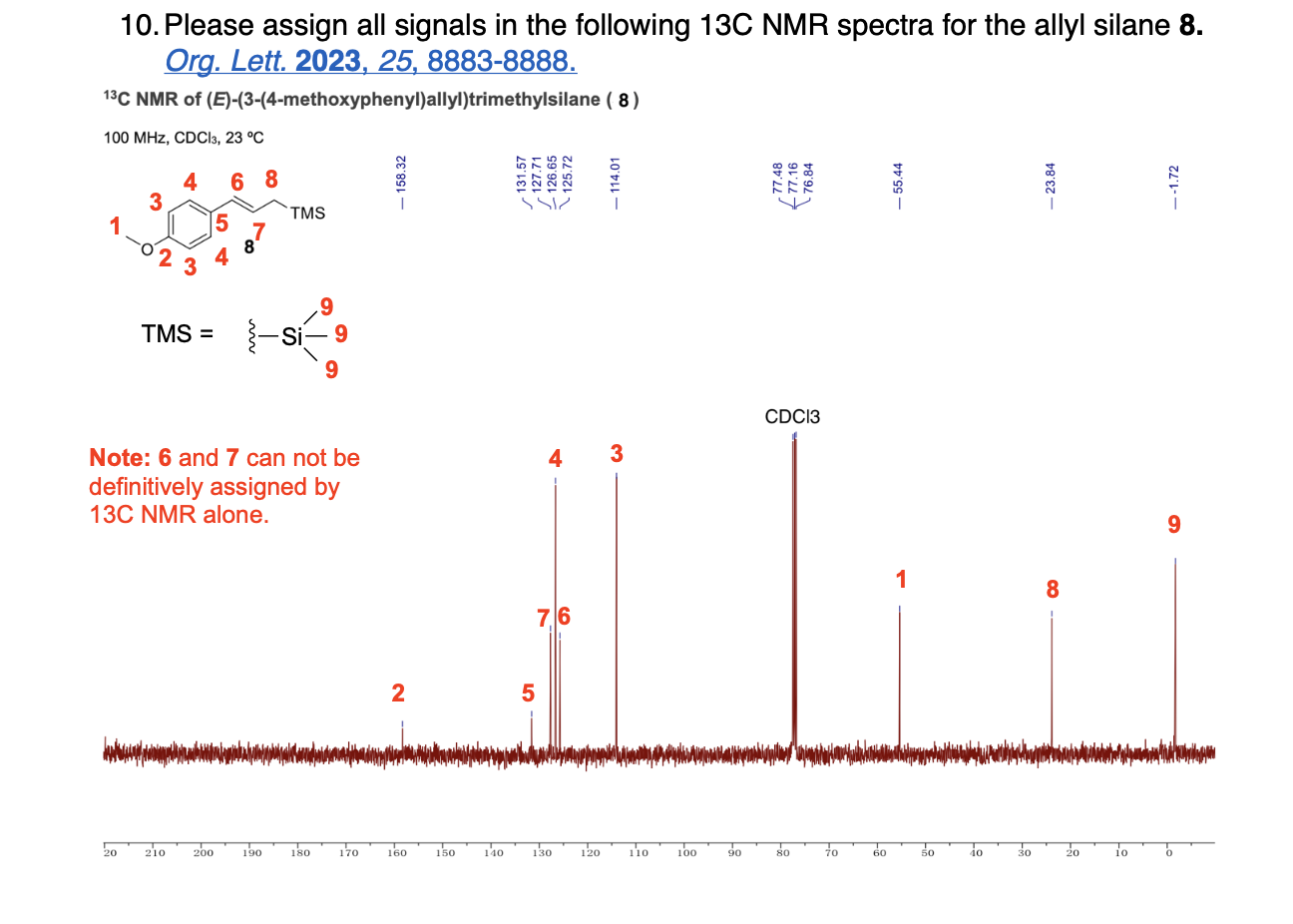
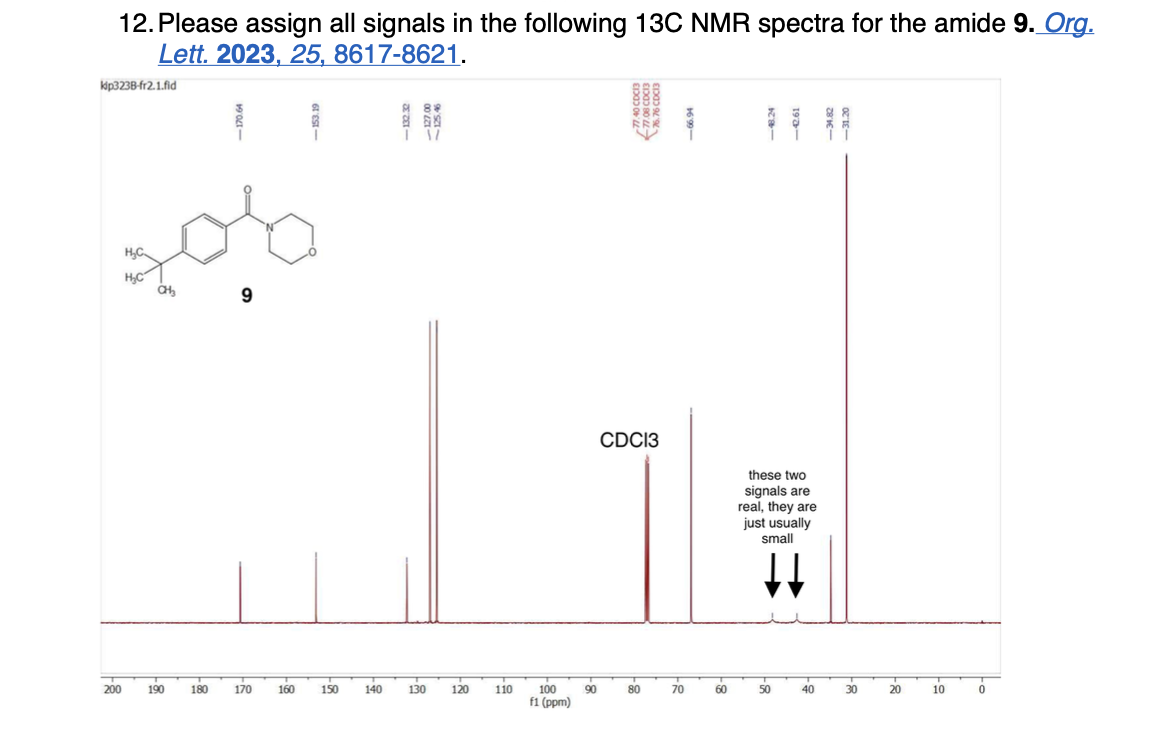
How many unique 13C NMR signals will be observed
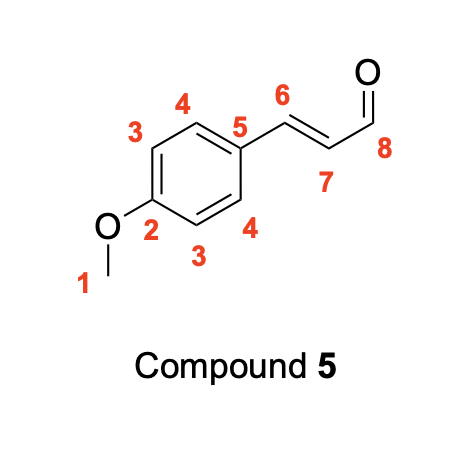
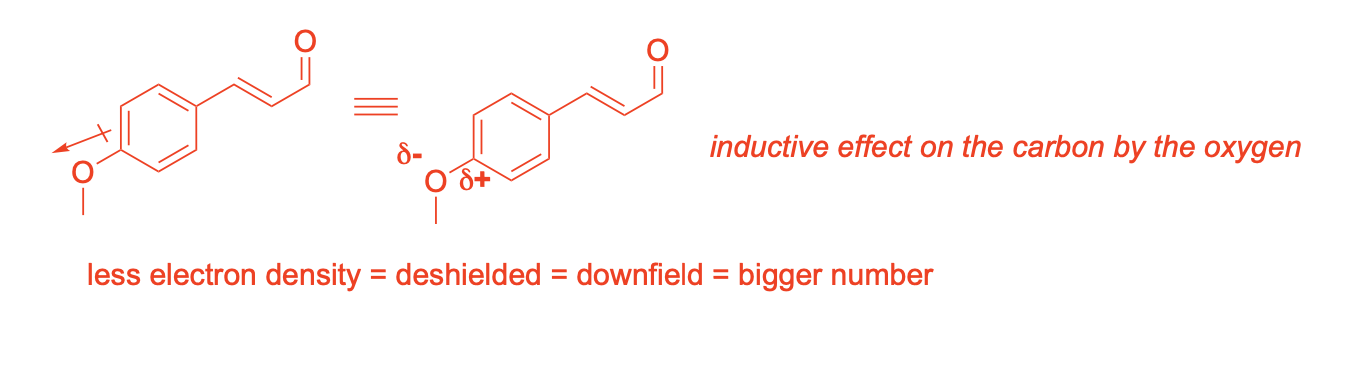
Using resonance drawings, explain why the chemical shift of C3 is further upfield (shielded) than C4
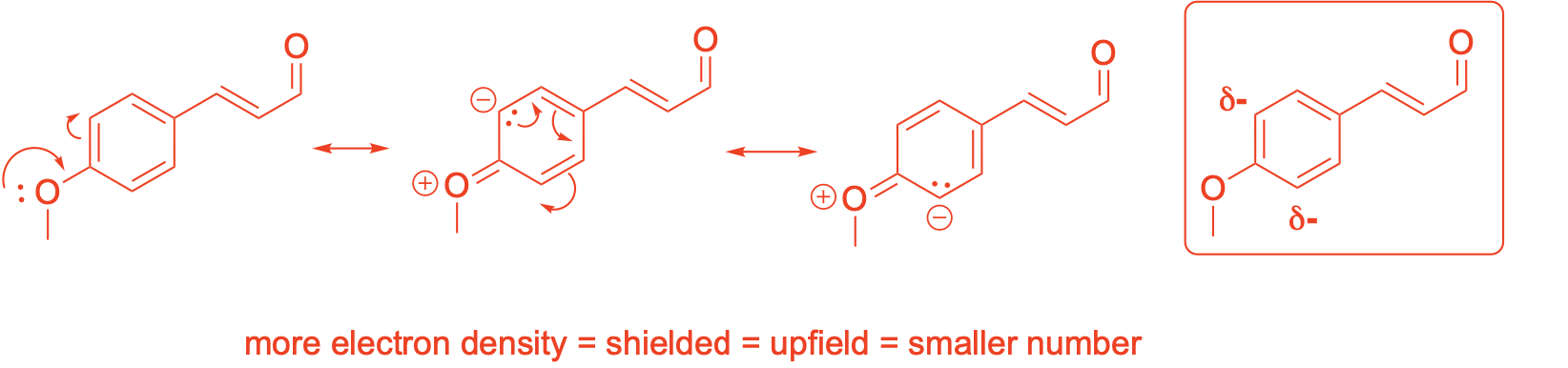
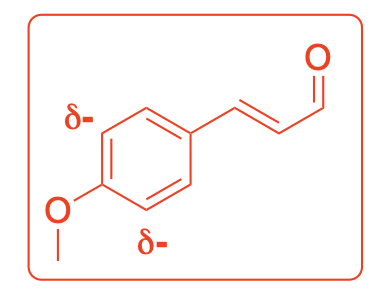
Using resonance drawings, explain why the chemical shift for C6 is further downfield (deshielded) than C7
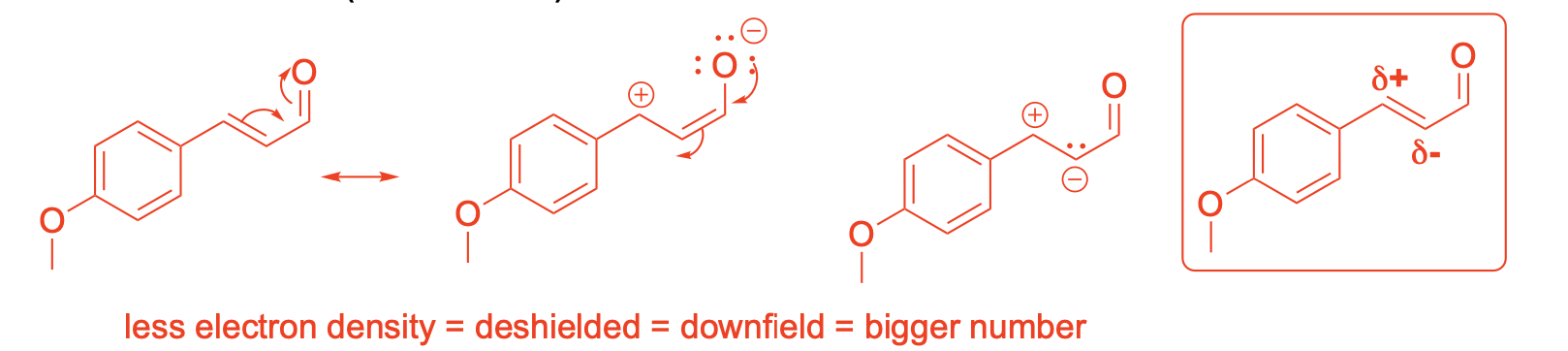
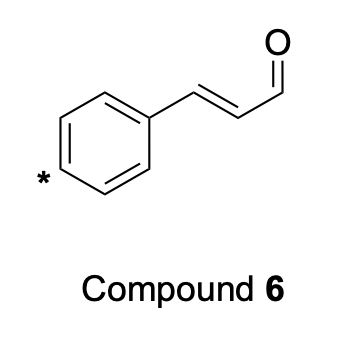
If you collect the 13C NMR spectra of the desmethoxy (without methoxy) compound 6, the chemical shift for the starred (*) carbon is dramatically more upfield (127.9 ppm) than denoted carbon 2 in compound 5 (159.8 ppm). How do you explain that chemical shift difference?
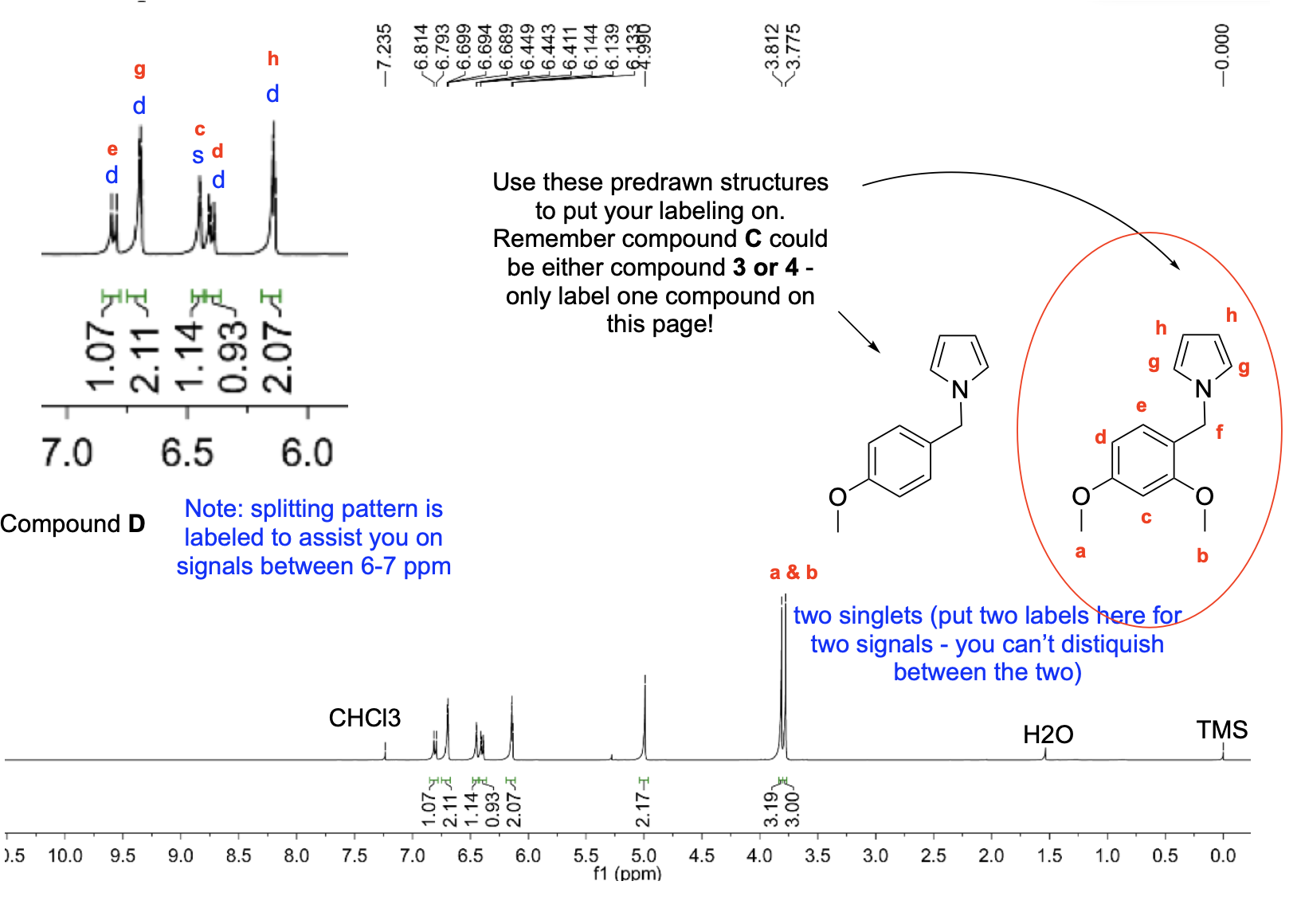
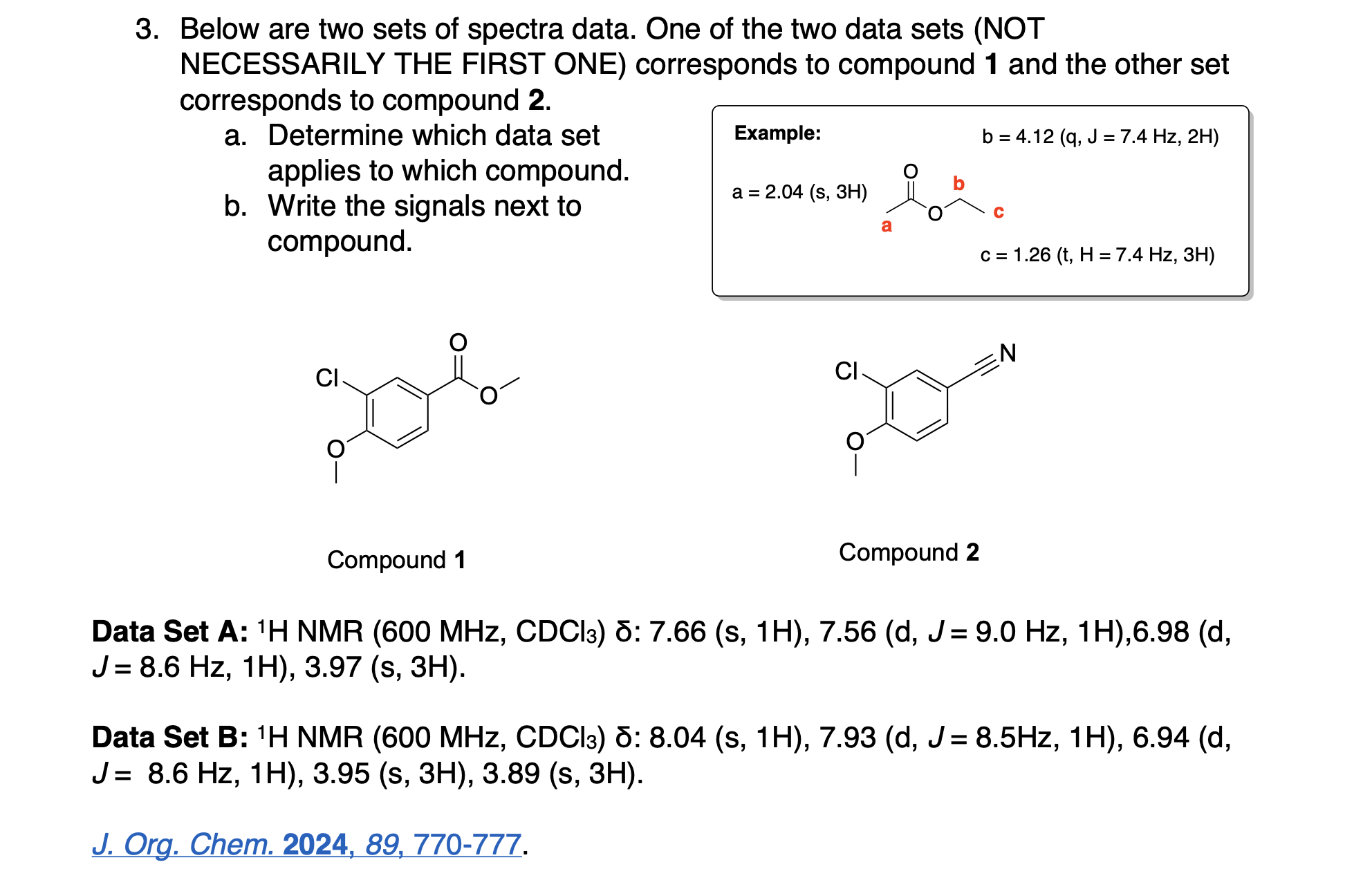
Please label the following compounds for unique 1 H NMR signals using are normal lettering notation:
Please label the following compounds for unique 1 H NMR signals using are normal lettering notation:
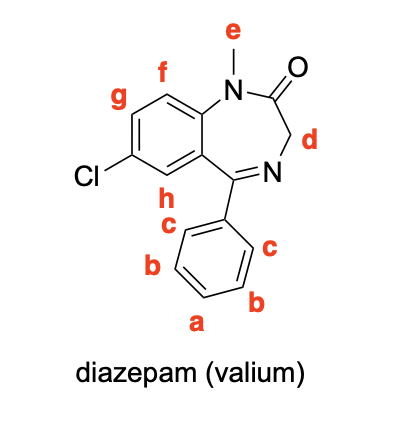
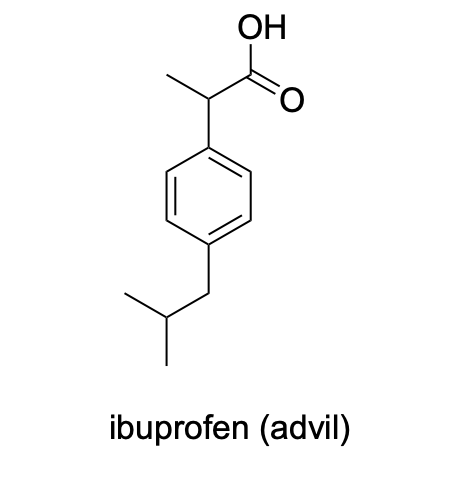
Please label the following compounds for unique 1 H NMR signals using are normal lettering notation:
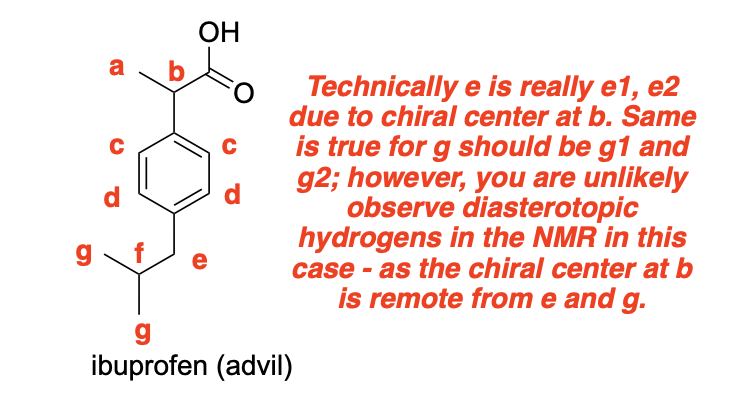
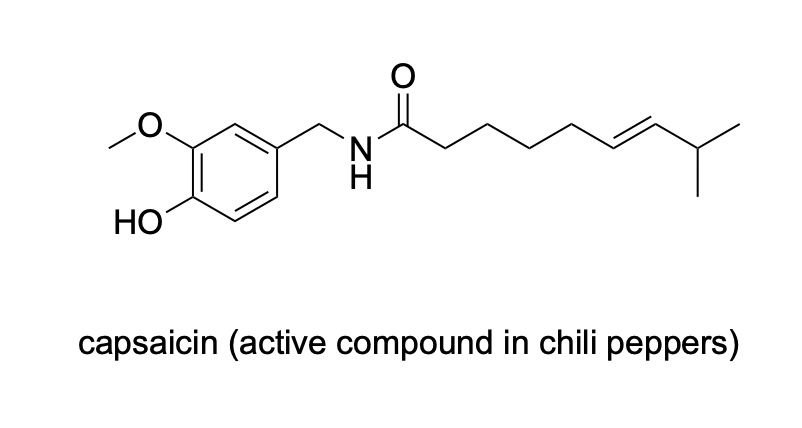
Please label the following compounds for unique 1 H NMR signals using are normal lettering notation:
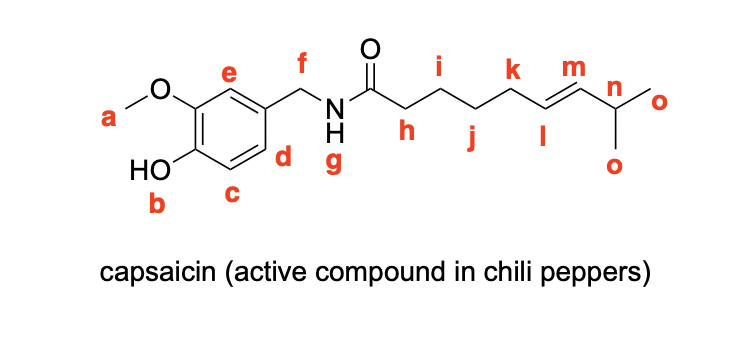
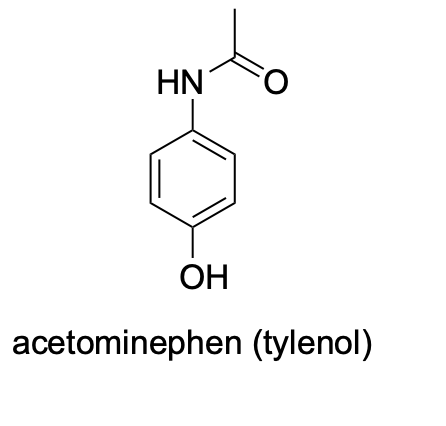
Please label the following compounds for unique 1 H NMR signals using are normal lettering notation:
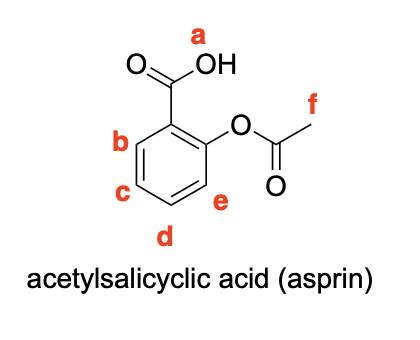
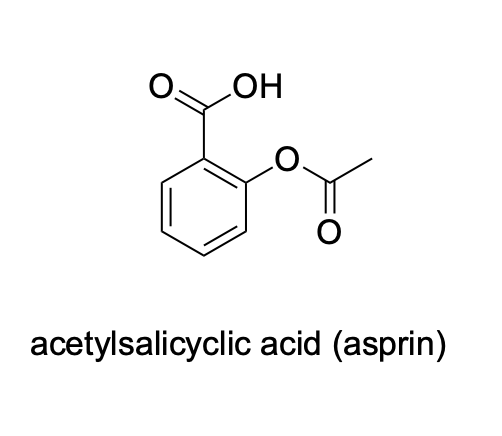
Please label the following compounds for unique 1 H NMR signals using are normal lettering notation:
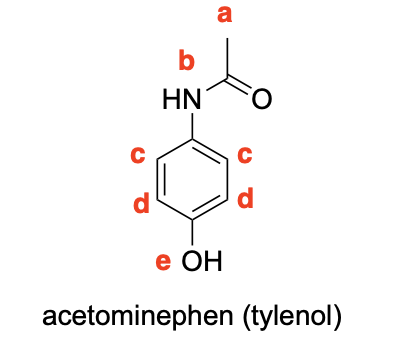
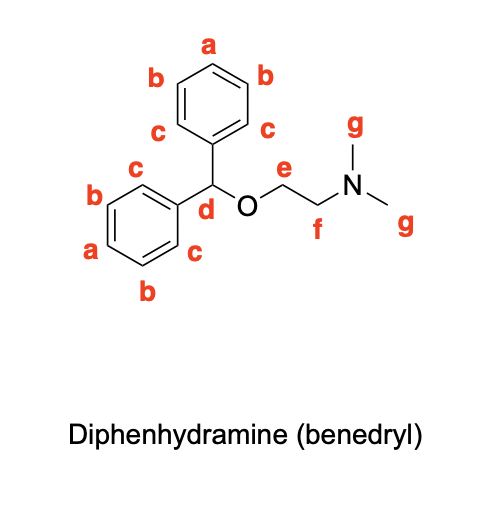
Please label the following compounds for unique 1 H NMR signals using are normal lettering notation:
Please label the following compounds for unique 1 H NMR signals using are normal lettering notation:
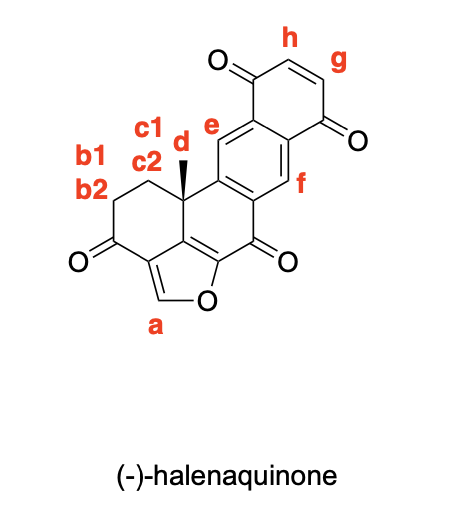
Please denote the expected splitting pattern in a 1H NMR spectra for each signal in the following structure:
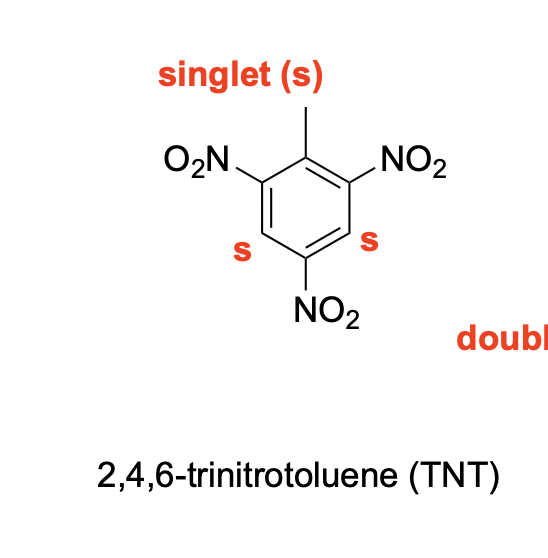
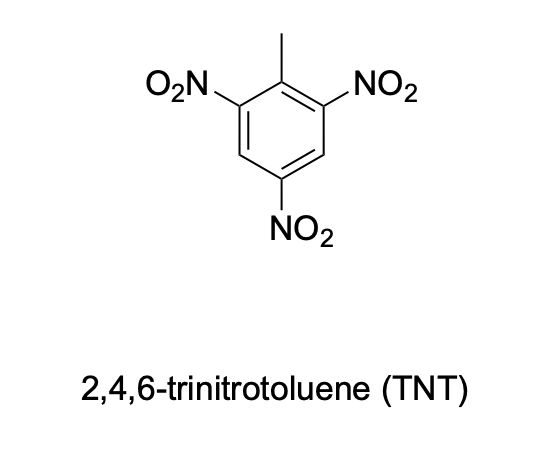
Please denote the expected splitting pattern in a 1H NMR spectra for each signal in the following structure:
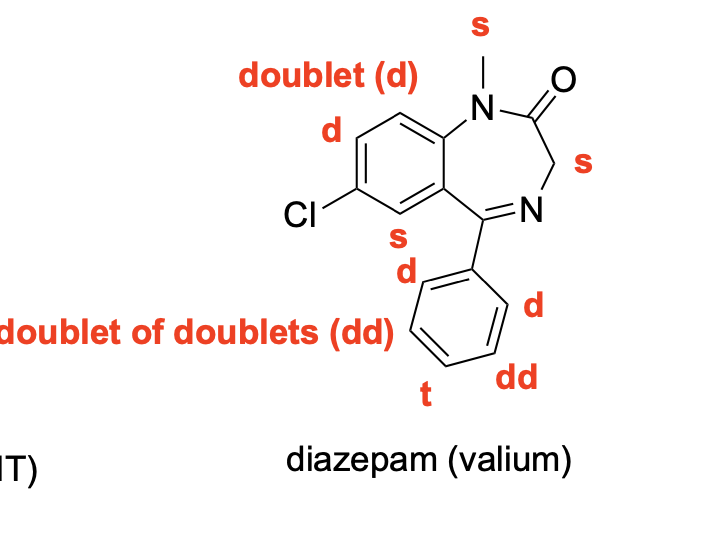
Please denote the expected splitting pattern in a 1H NMR spectra for each signal in the following structure:
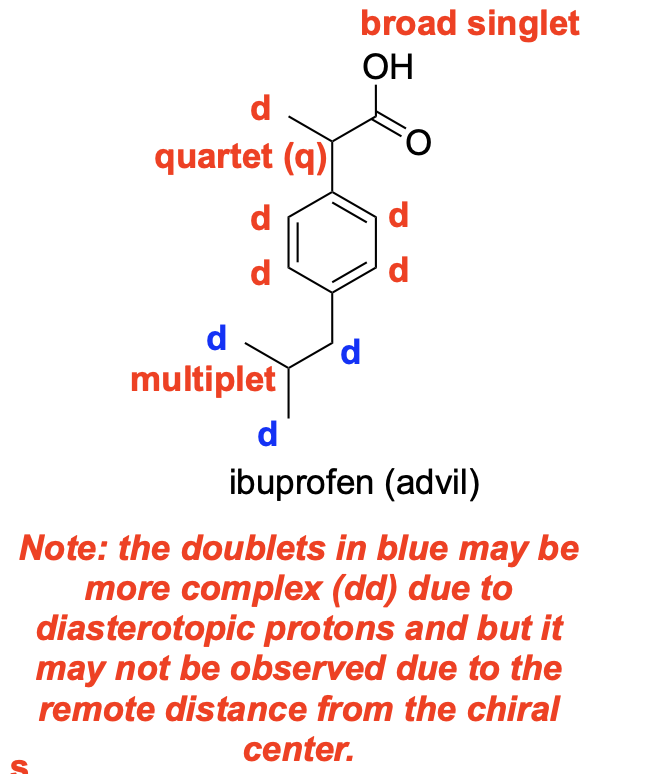
Please denote the expected splitting pattern in a 1H NMR spectra for each signal in the following structure:
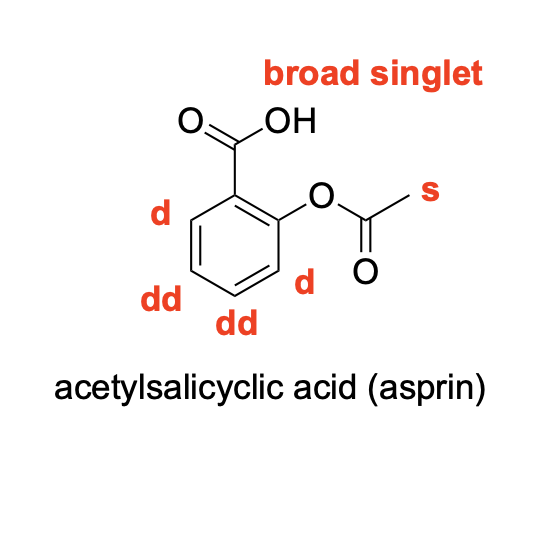
Please denote the expected splitting pattern in a 1H NMR spectra for each signal in the following structure:
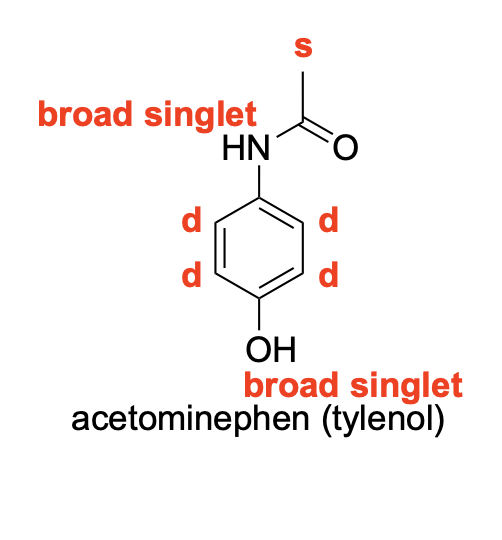
Please denote the expected splitting pattern in a 1H NMR spectra for each signal in the following structure:
Please denote the expected splitting pattern in a 1H NMR spectra for each signal in the following structure:
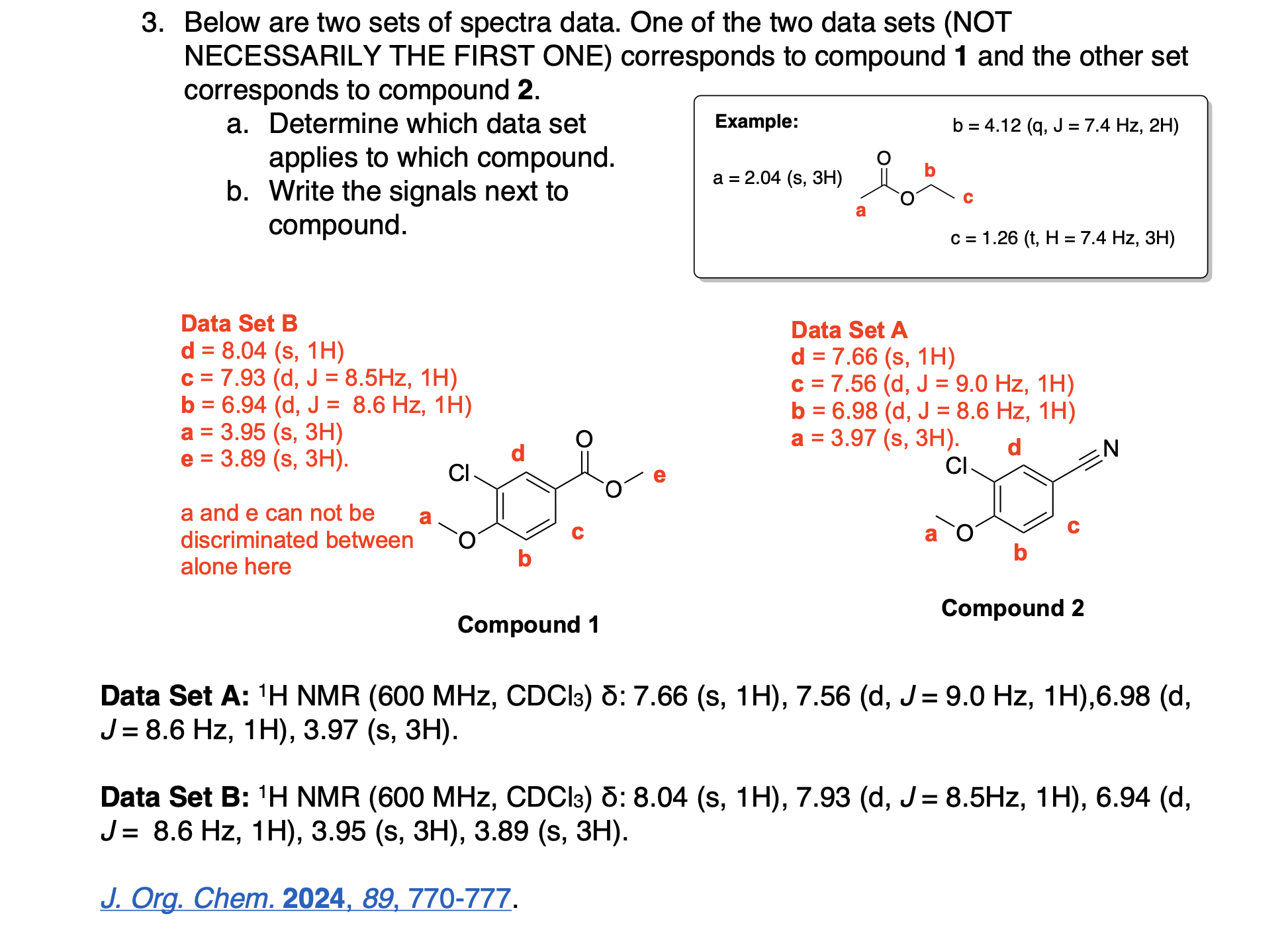
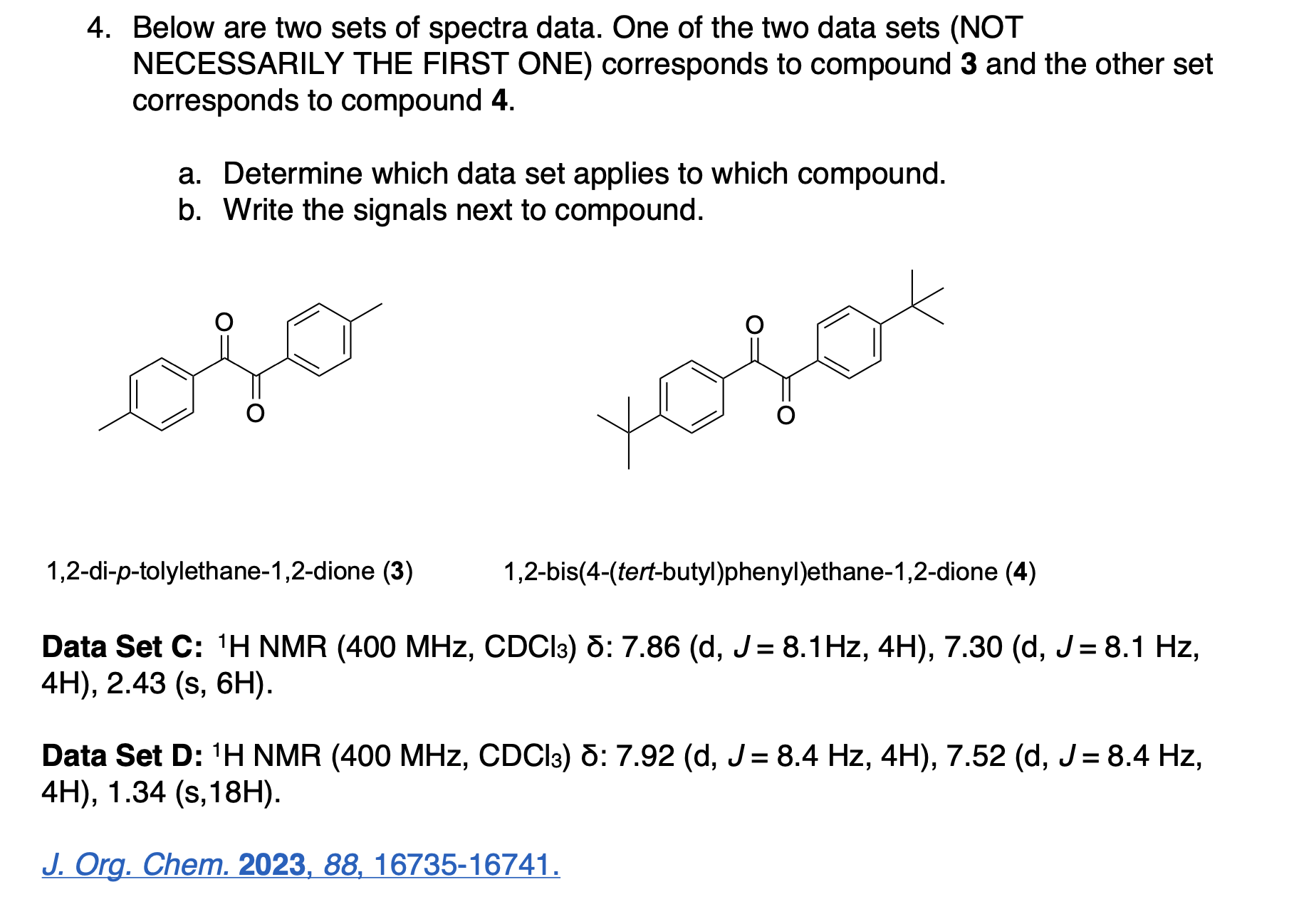
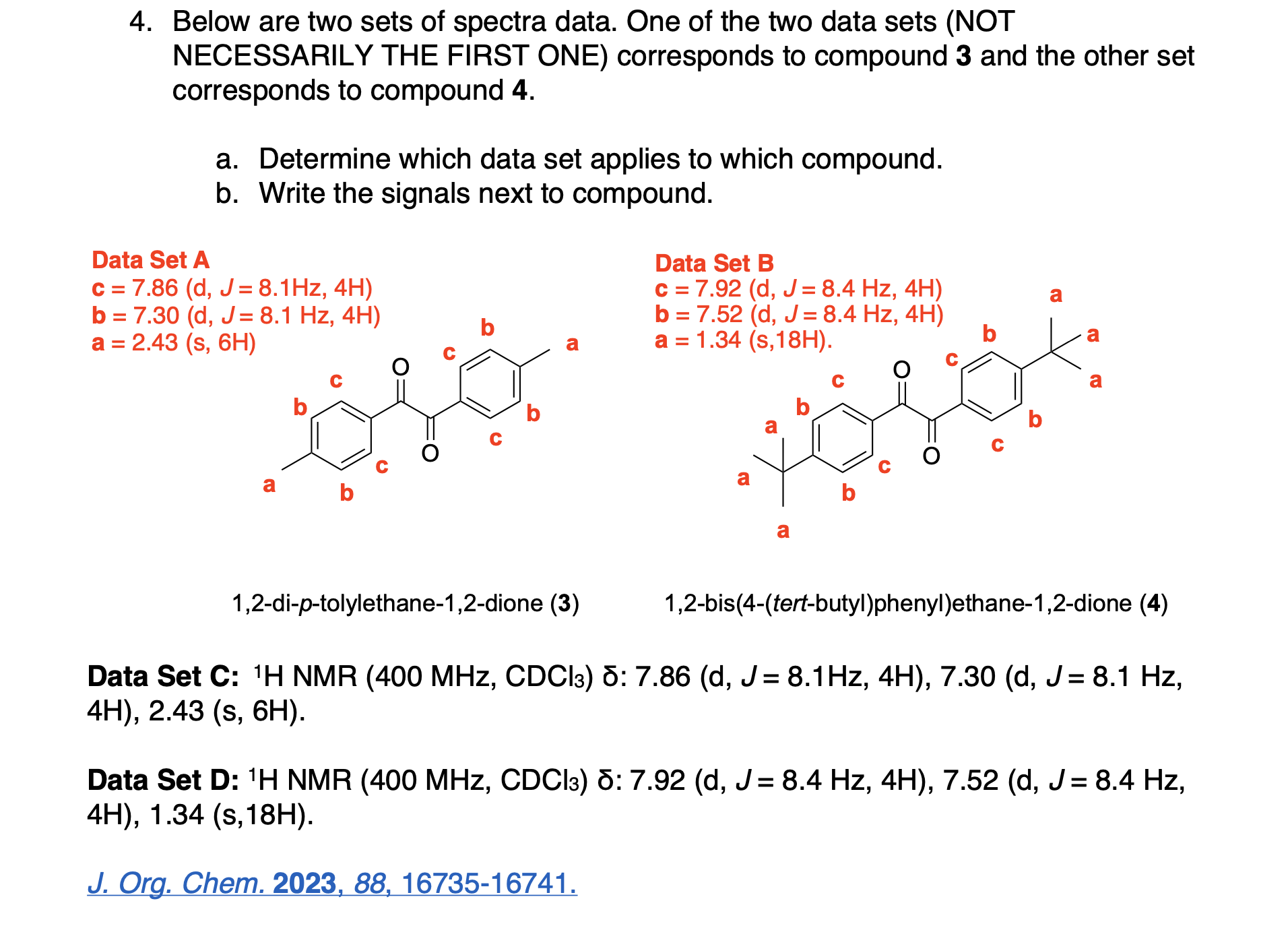
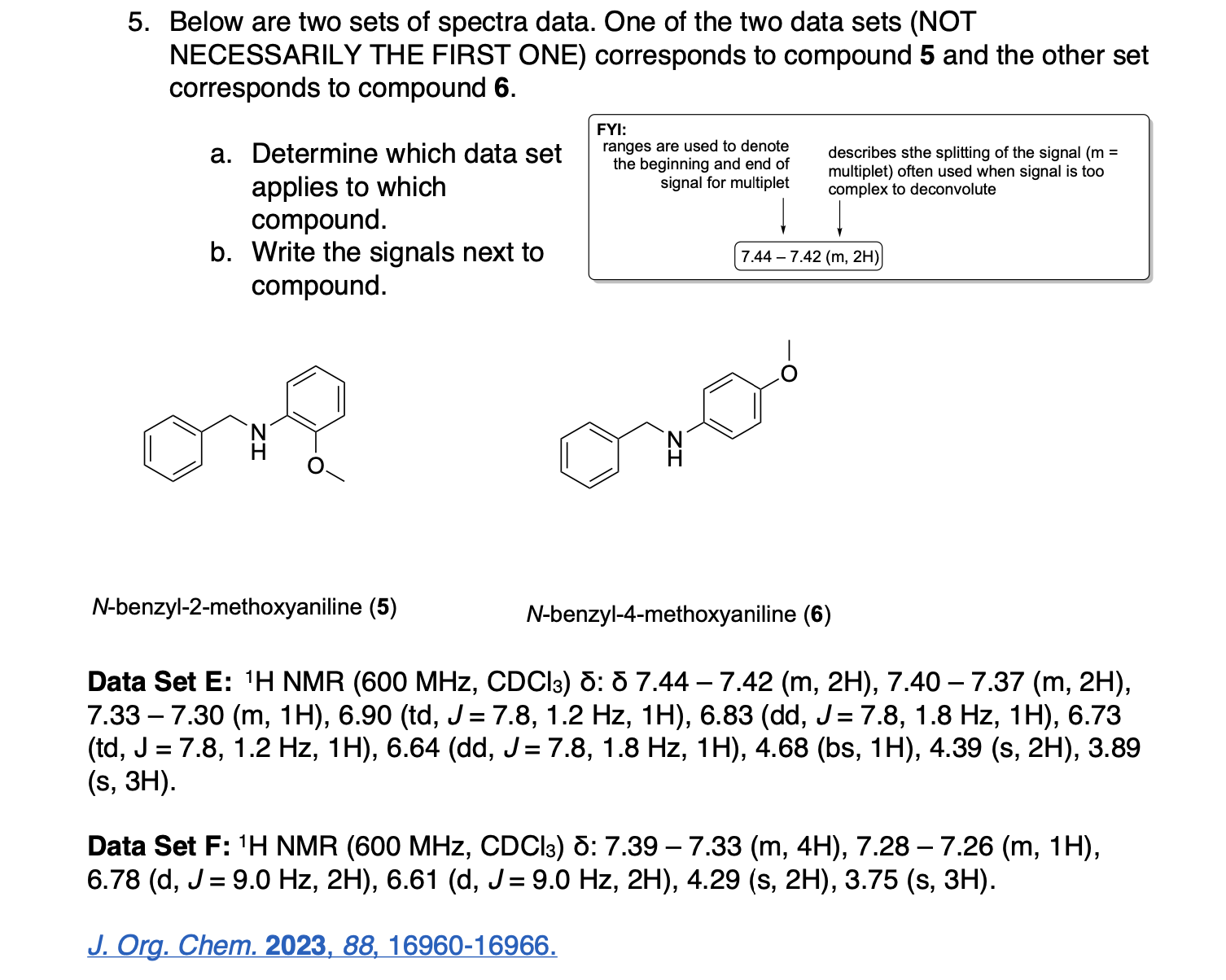
Please label the following compound for unique 13C NMR signals using our normal numbering notation:
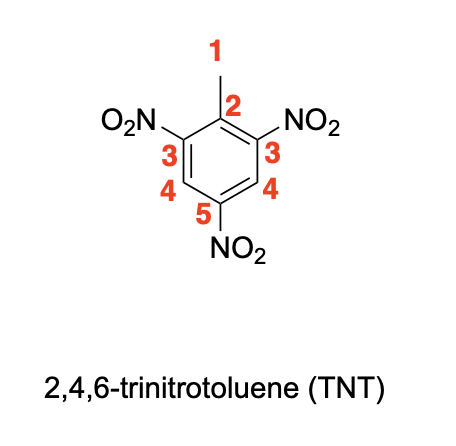
Please label the following compound for unique 13C NMR signals using our normal numbering notation:
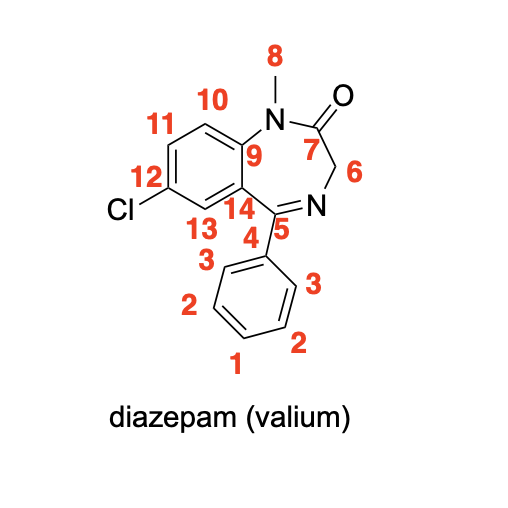
Please label the following compound for unique 13C NMR signals using our normal numbering notation:
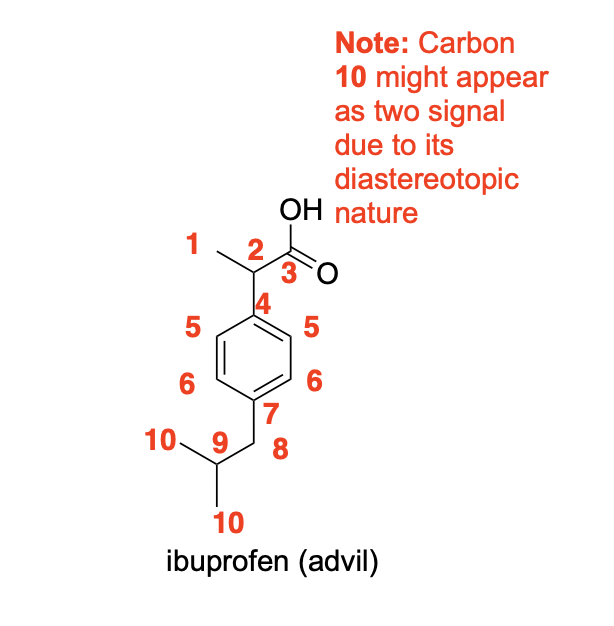
Please label the following compound for unique 13C NMR signals using our normal numbering notation:
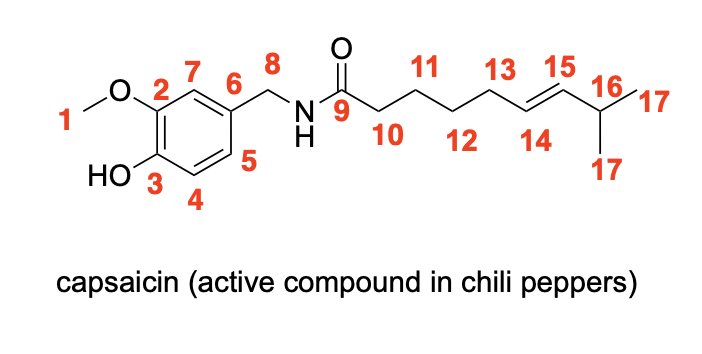
Please label the following compound for unique 13C NMR signals using our normal numbering notation:
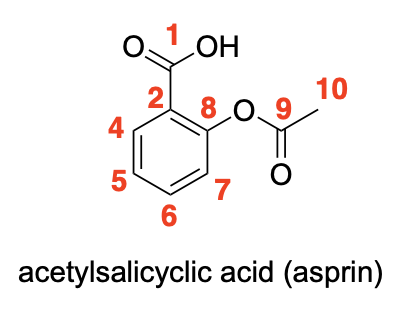
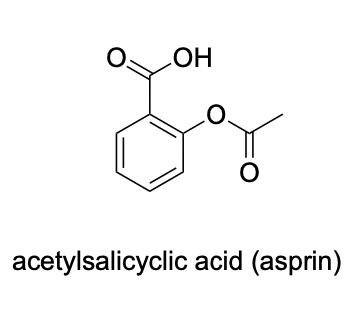
Please label the following compound for unique 13C NMR signals using our normal numbering notation:
Please label the following compound for unique 13C NMR signals using our normal numbering notation:
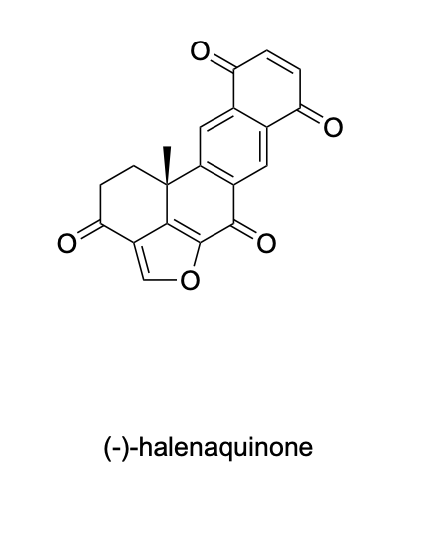
Please label the following compound for unique 13C NMR signals using our normal numbering notation:
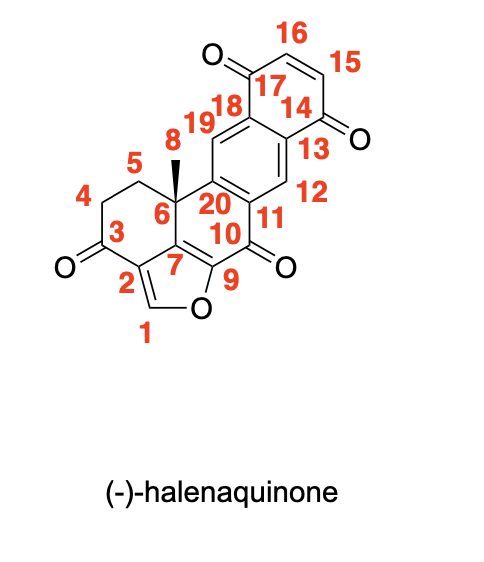
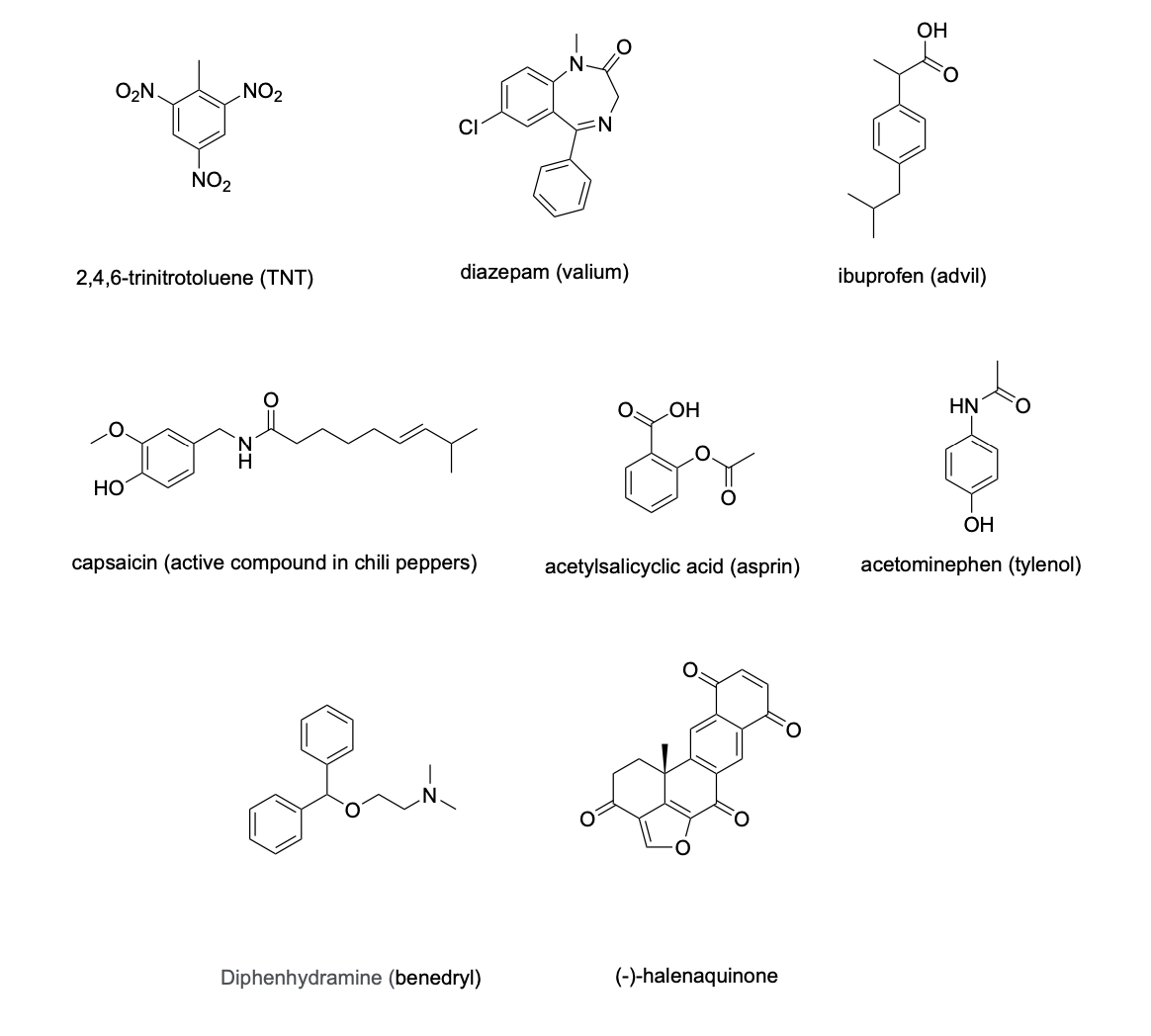
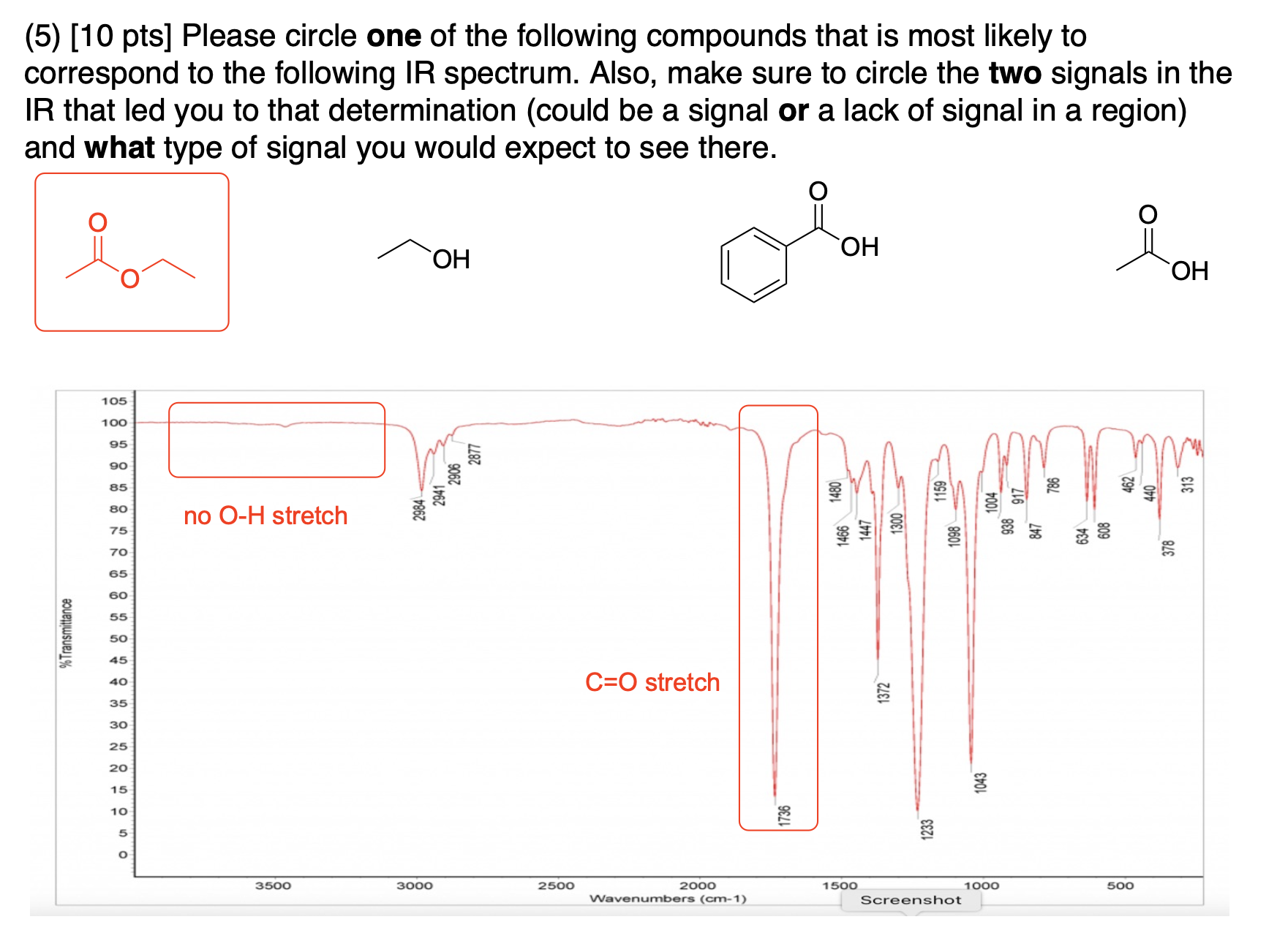
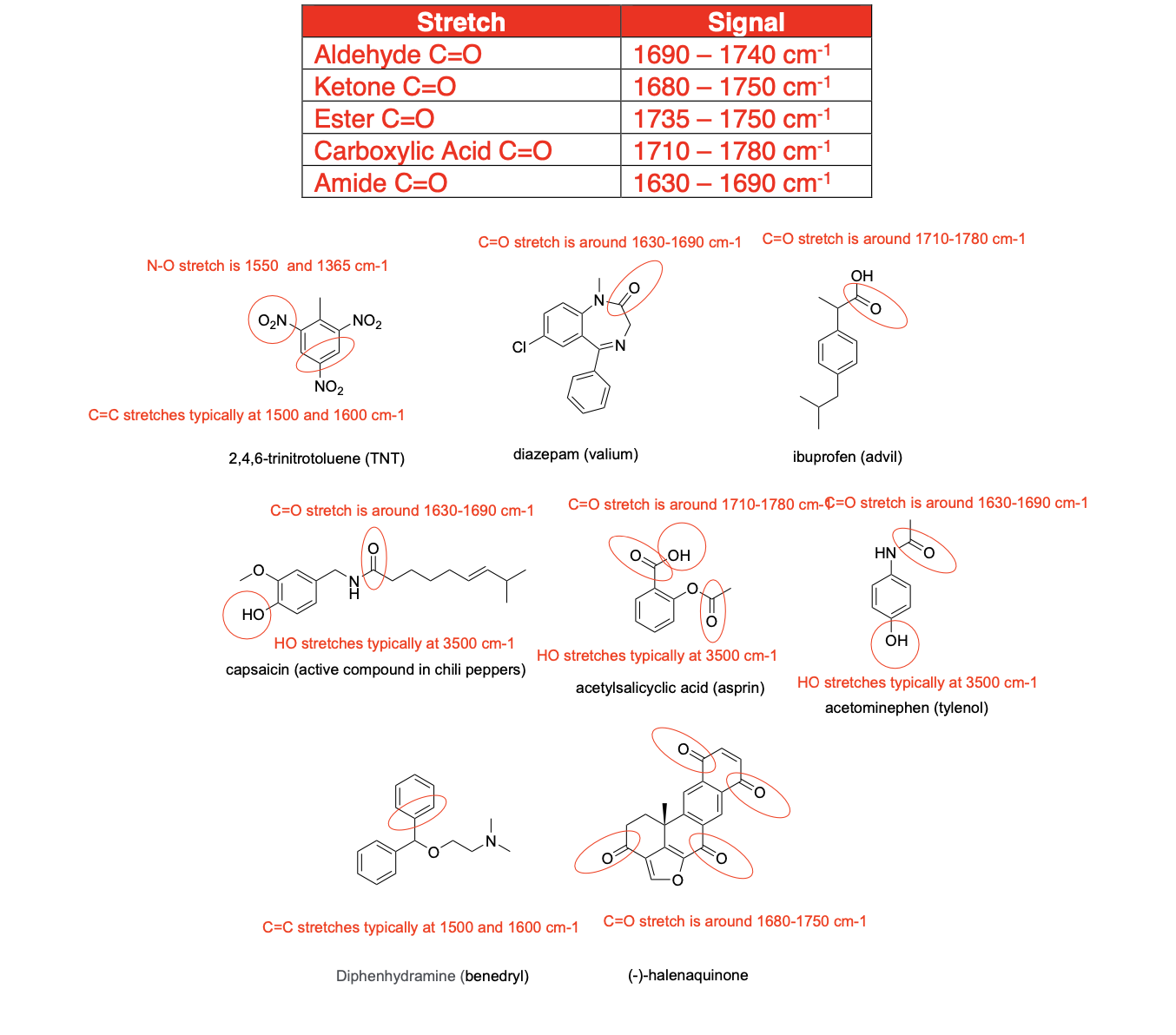
circle at least one appropriate functional group that we learned in class would give a signal in an IR spectrum (excluding C-H stretches) and provide an approximate location for the signal in cm-1 for the following compounds:
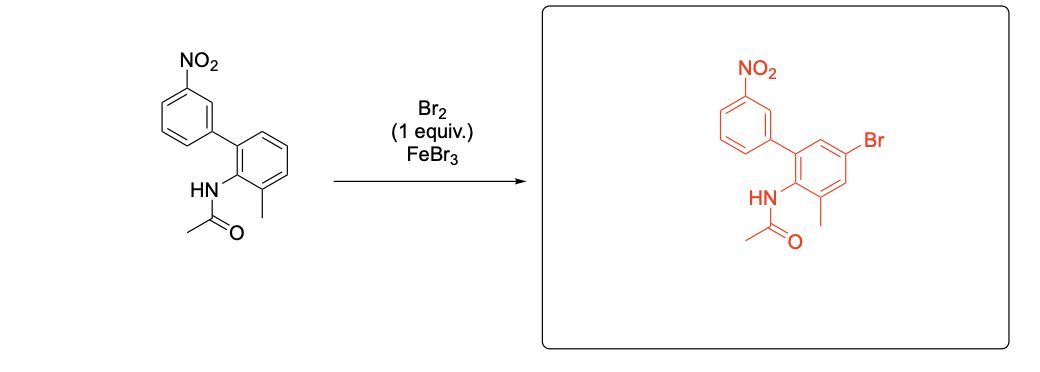
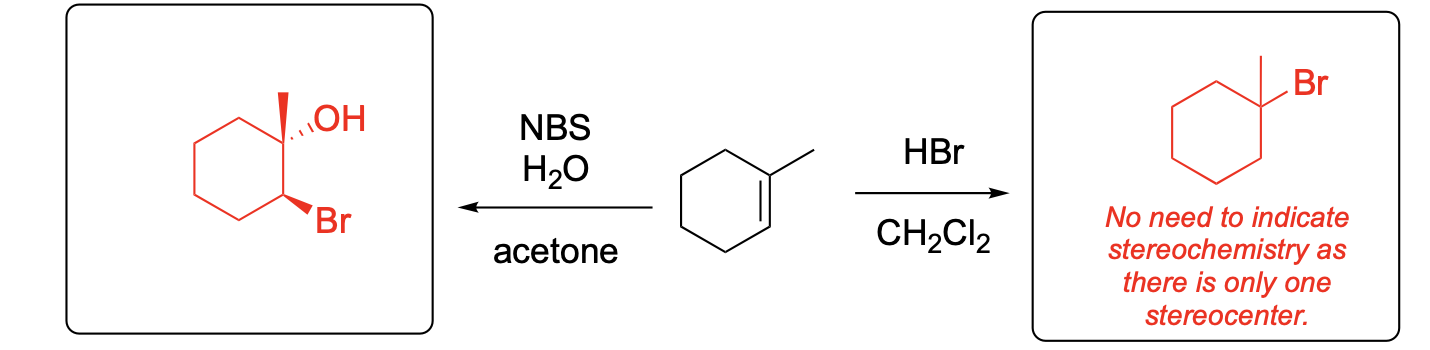
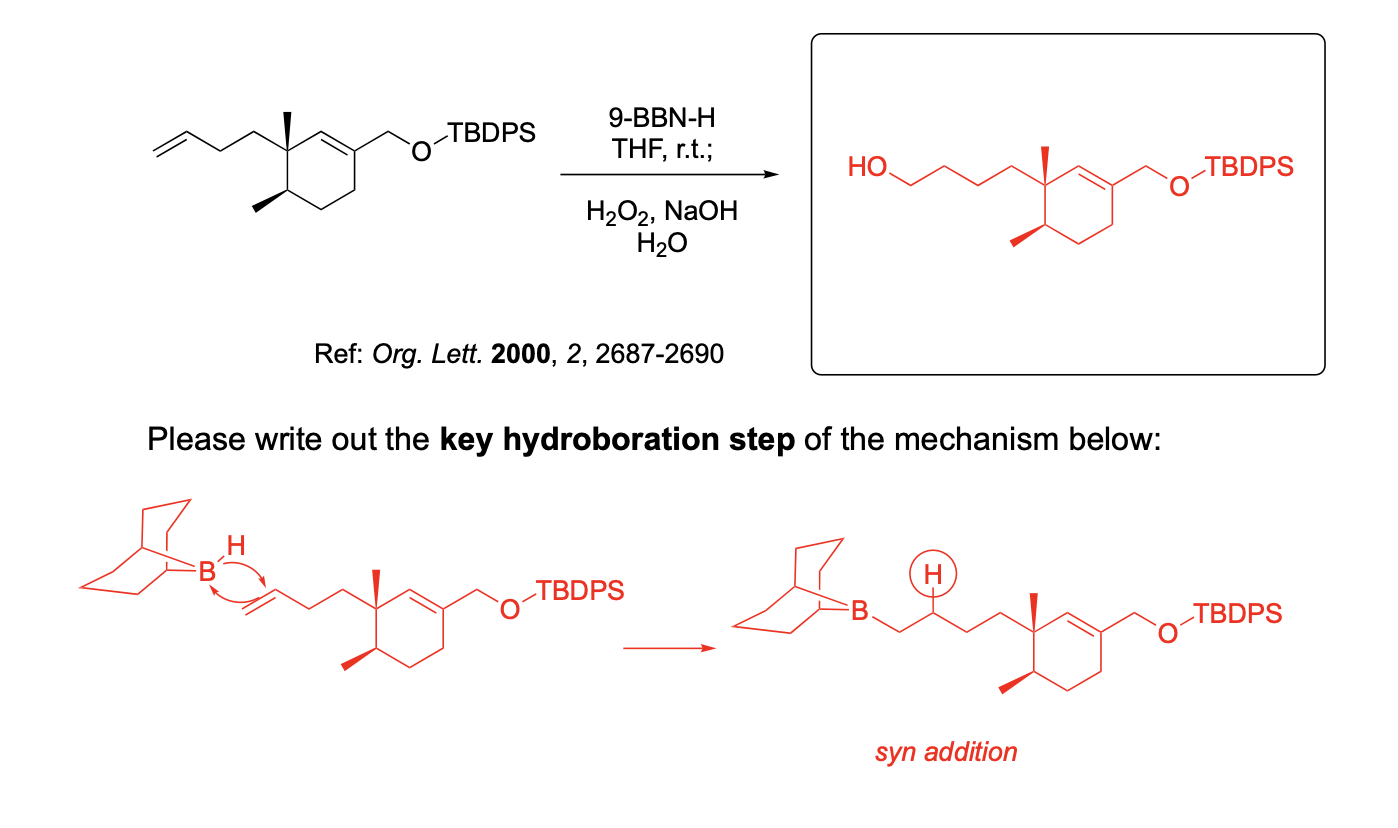
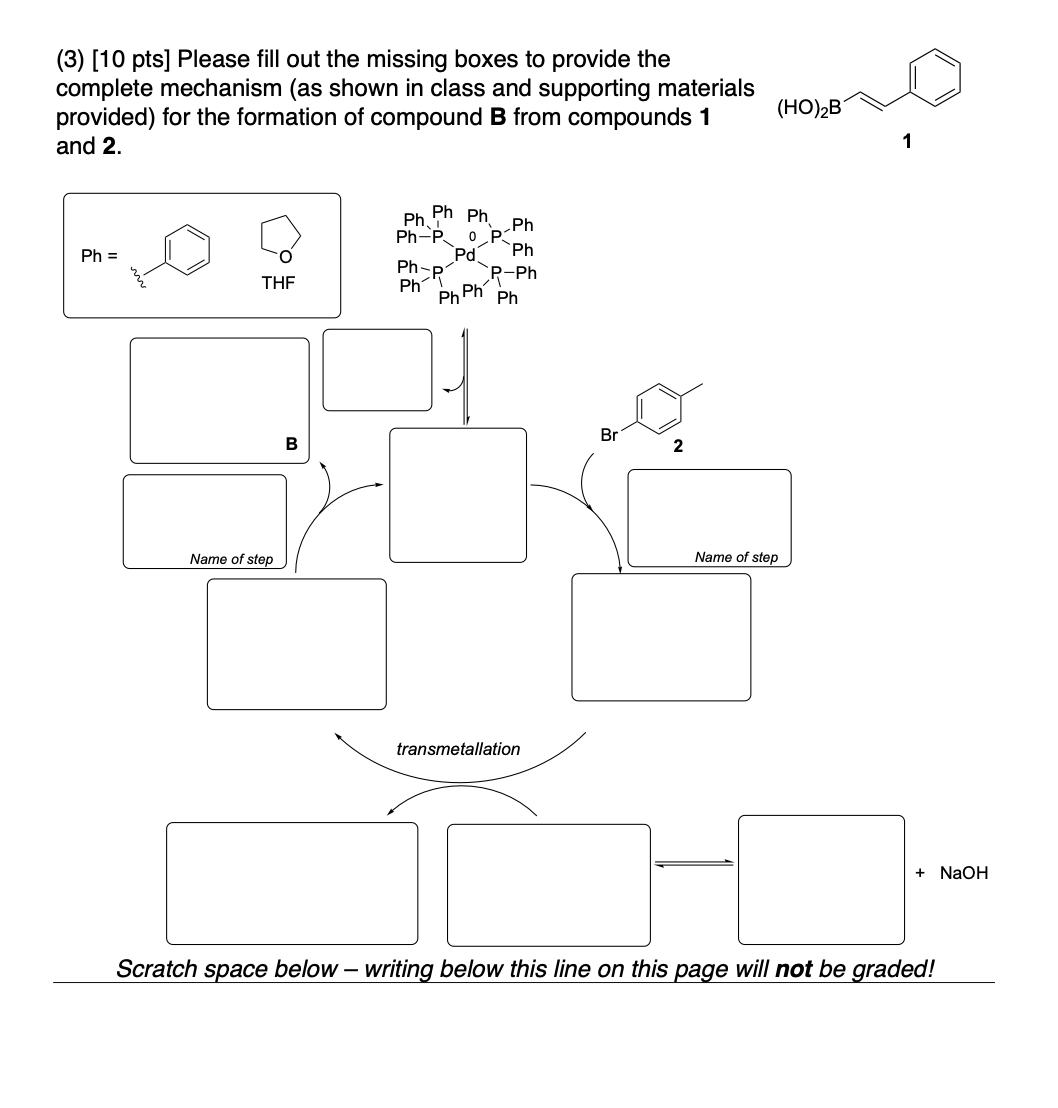
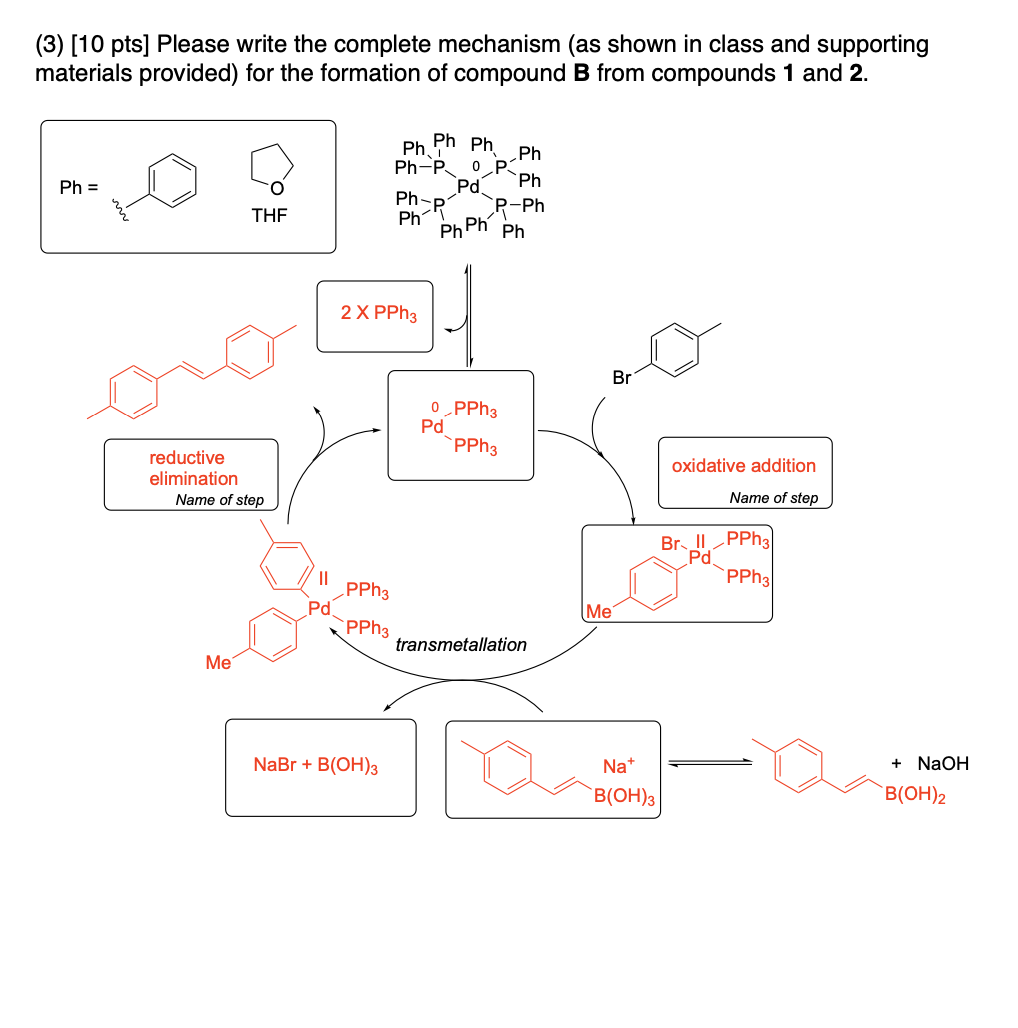
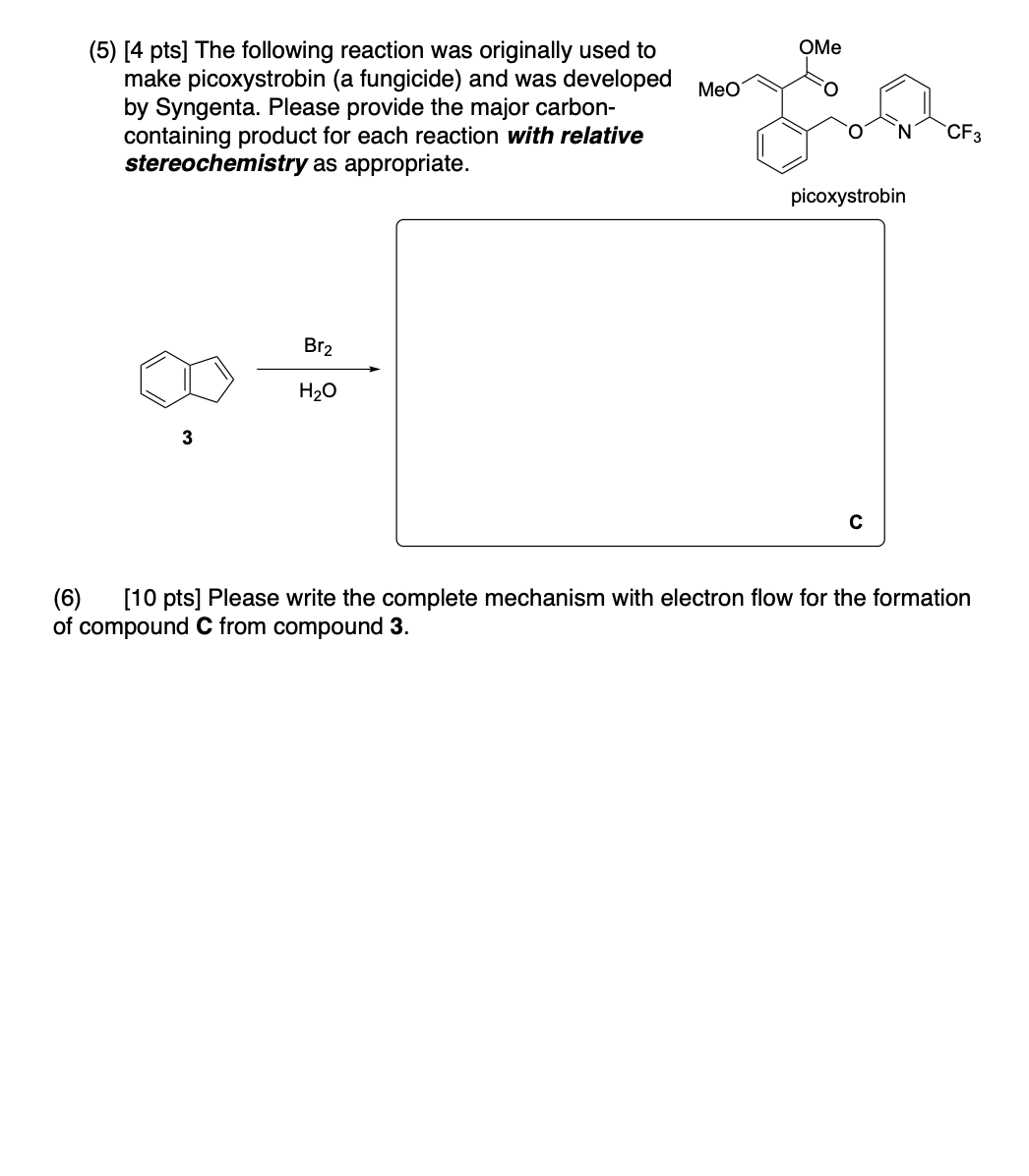
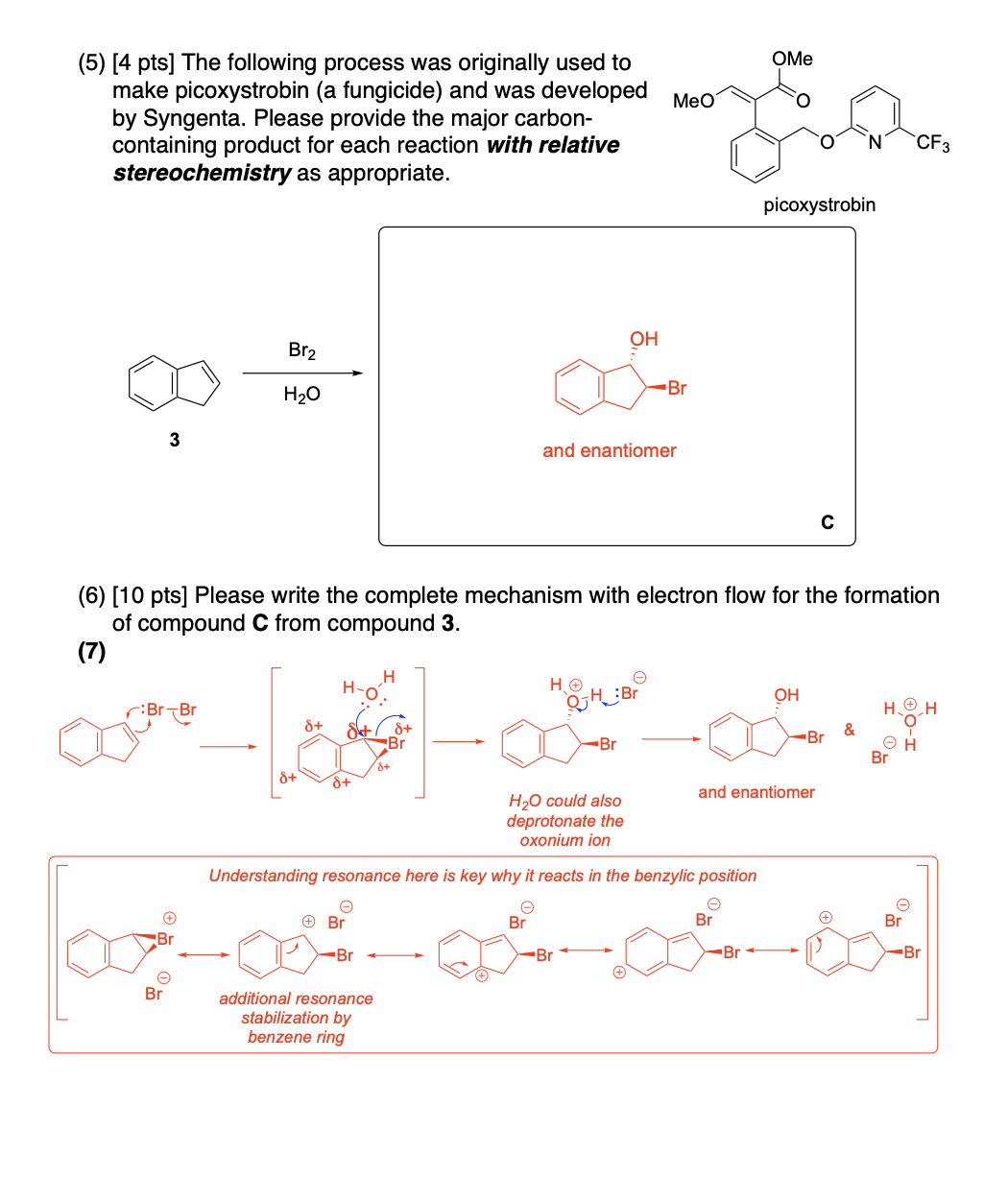
Please provide the major carbon-containing product for each reaction with relative stereochemistry as appropriate.
Please provide the major carbon-containing product for each reaction with relative stereochemistry as appropriate.
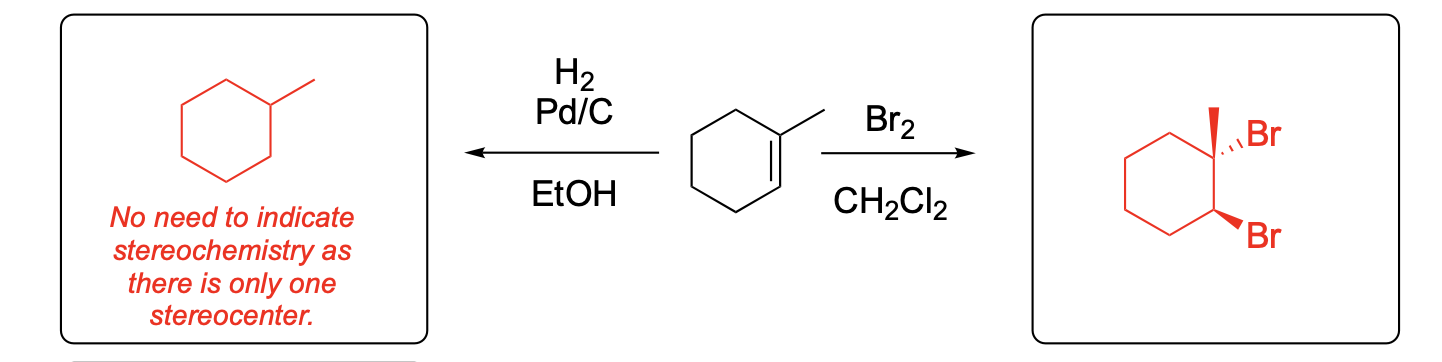
Please provide the major carbon-containing product for each reaction with relative stereochemistry as appropriate.
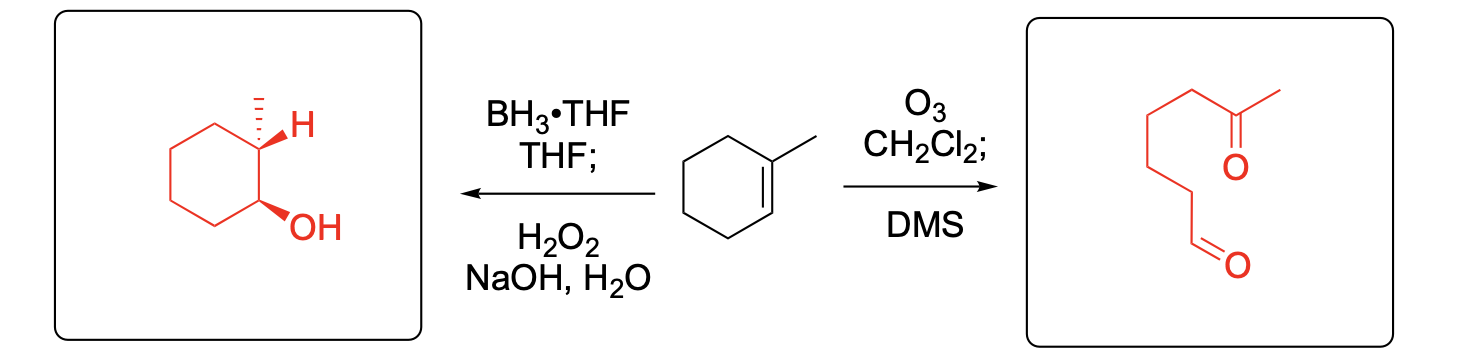
Please provide the major carbon-containing product for each reaction with relative stereochemistry as appropriate.
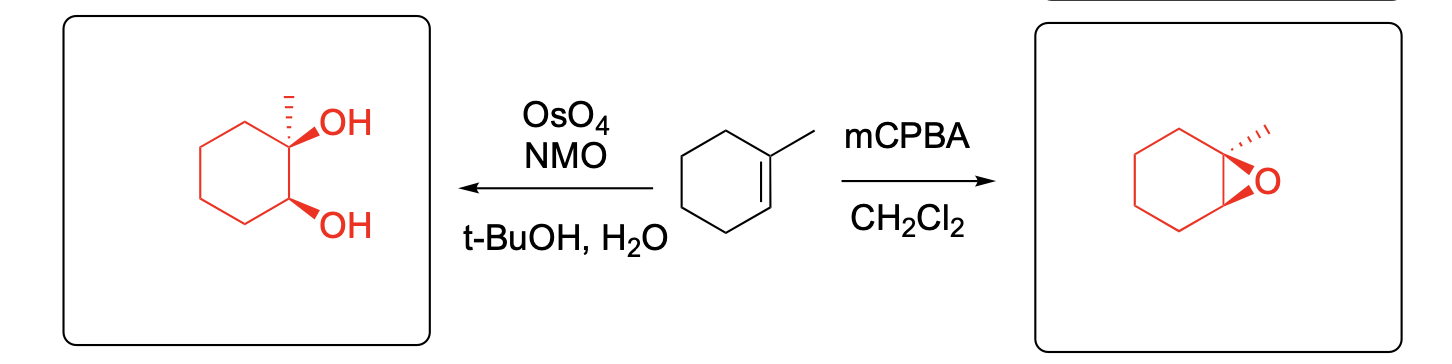
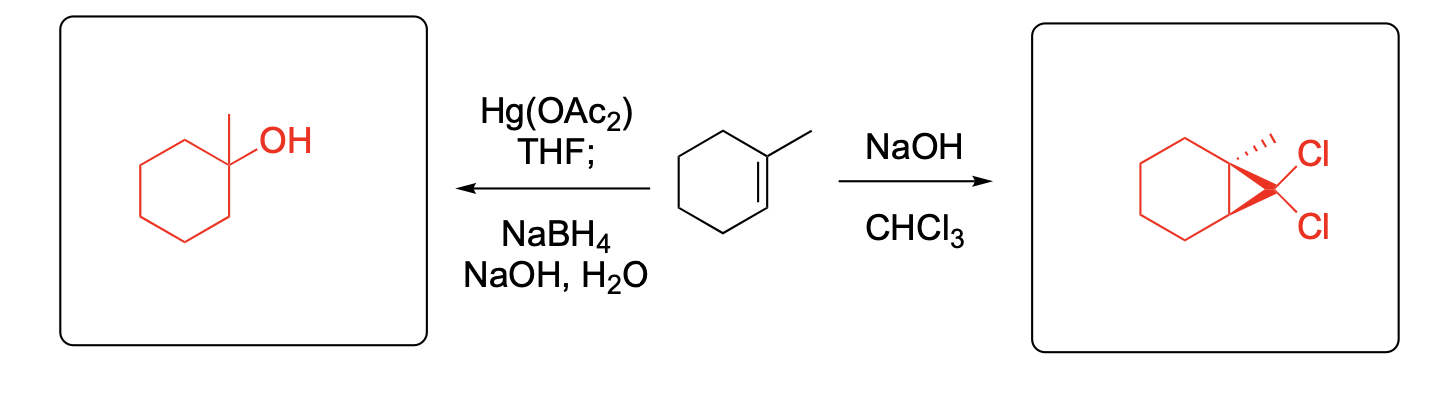
Please provide the major carbon-containing product for each reaction with relative stereochemistry as appropriate.
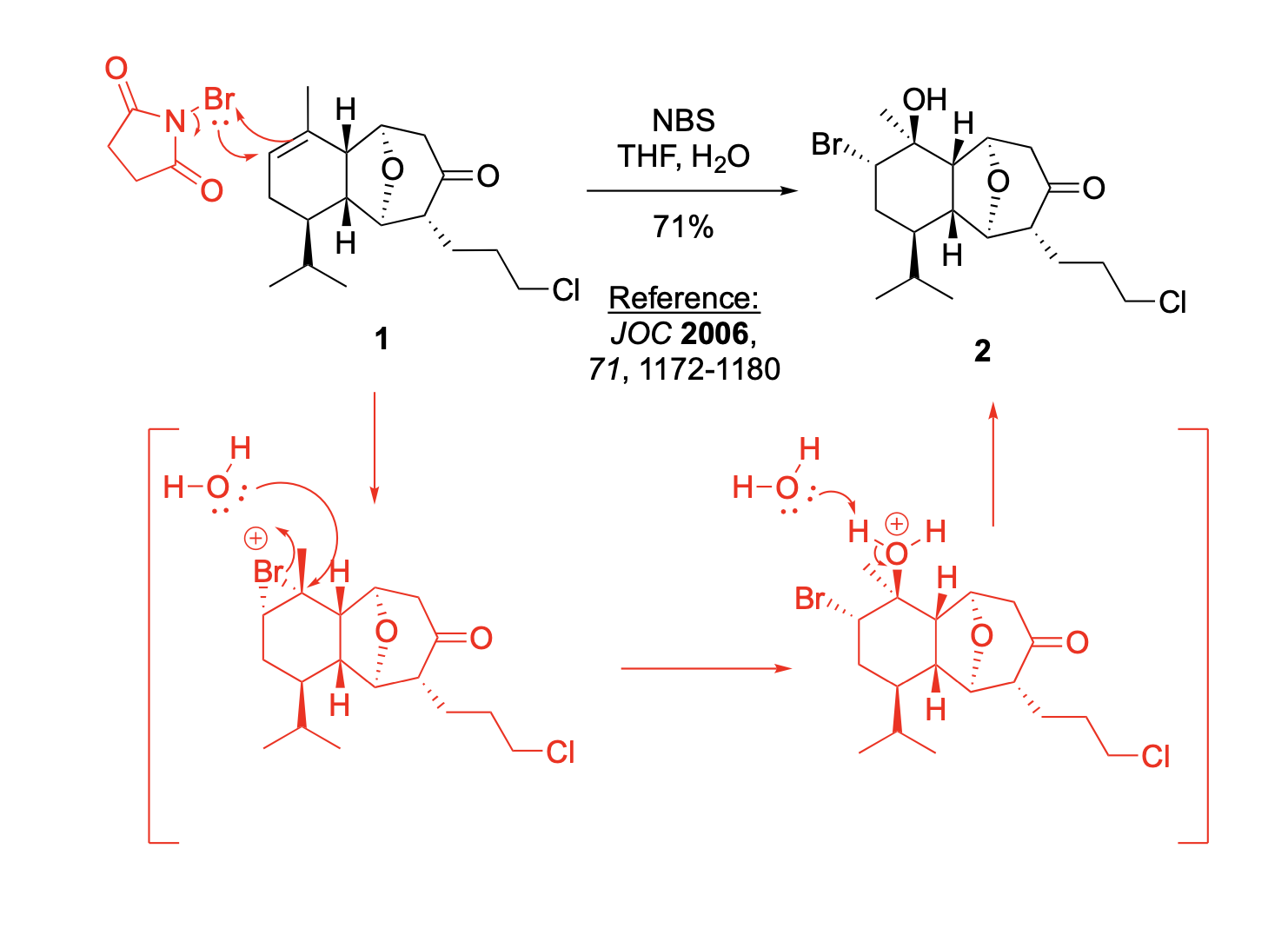
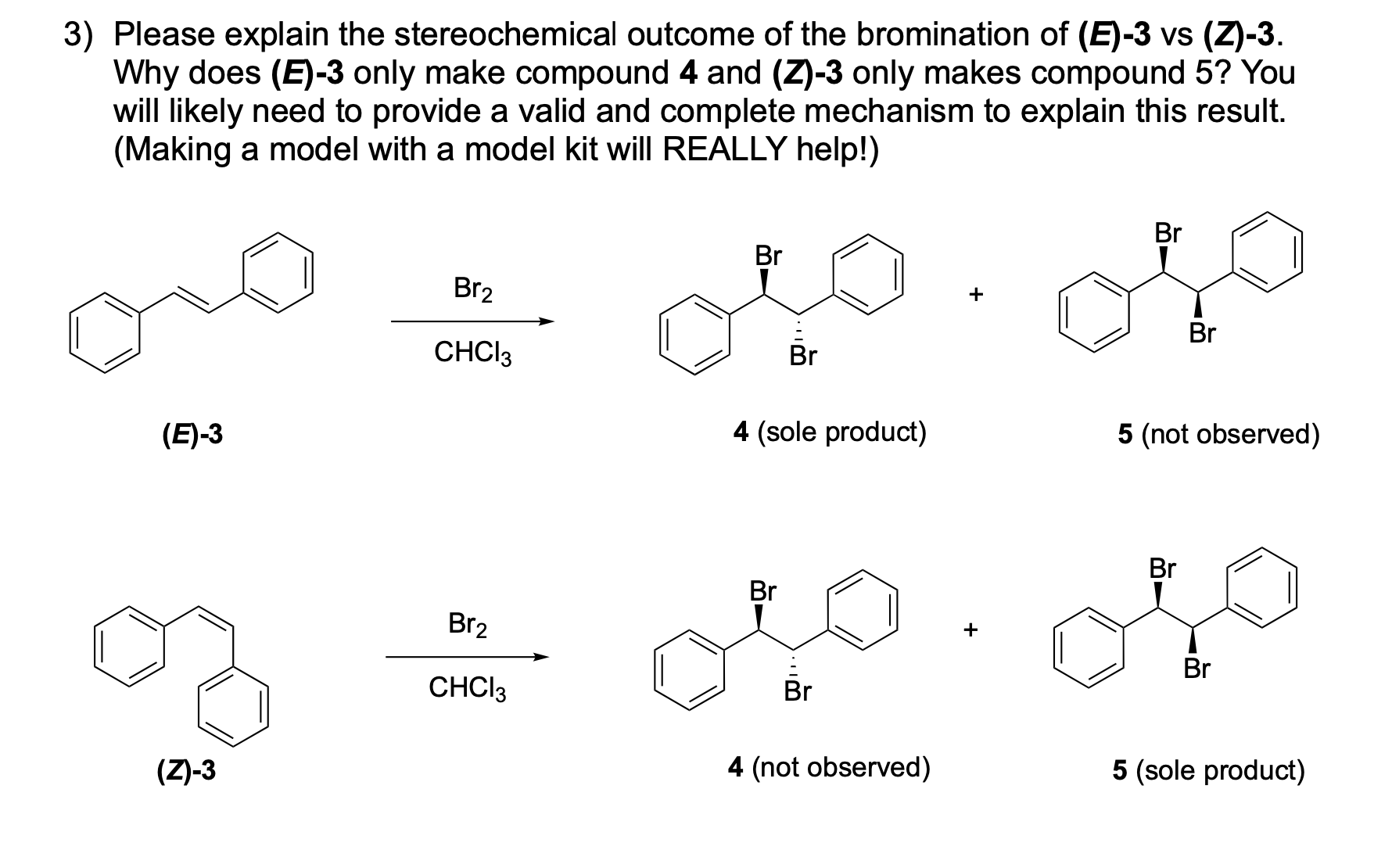
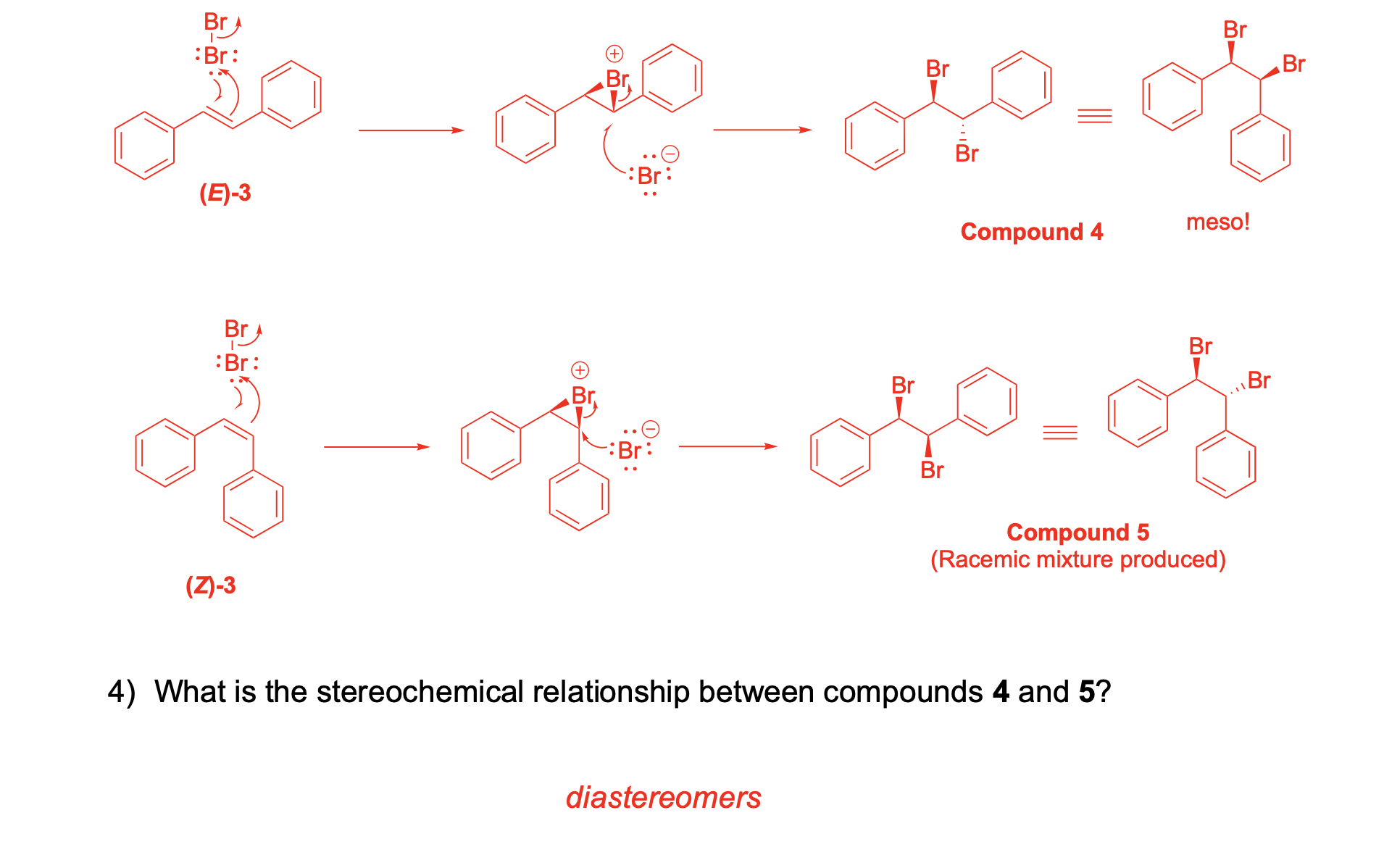
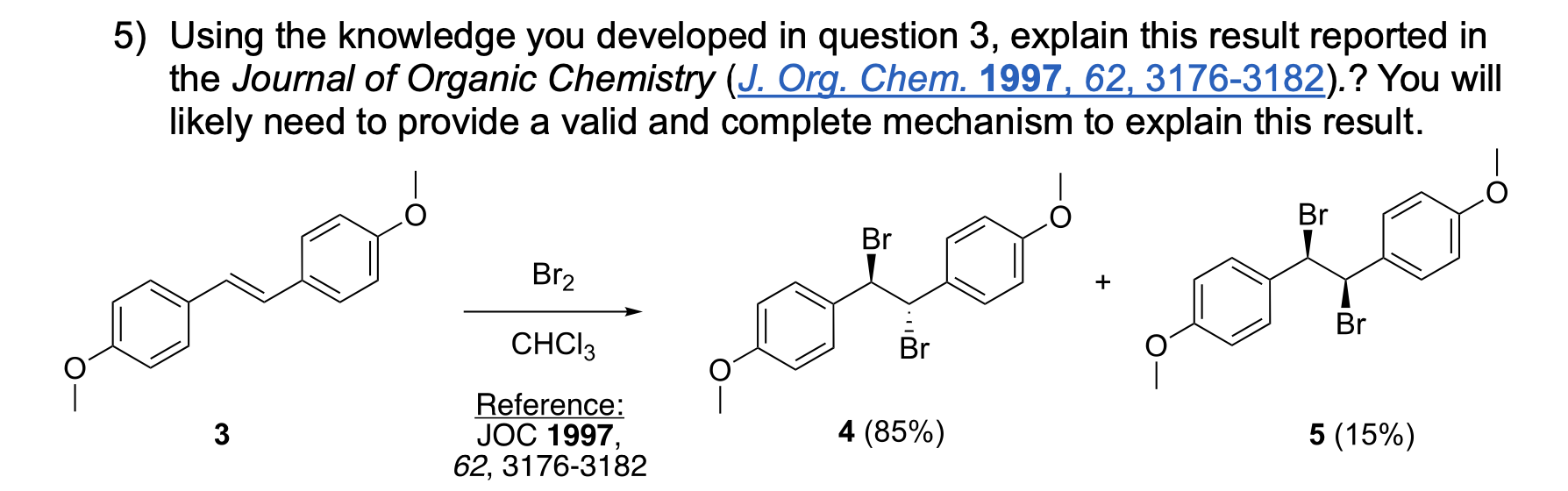
AND What is the stereochemical relationship between compounds 4 and 5?


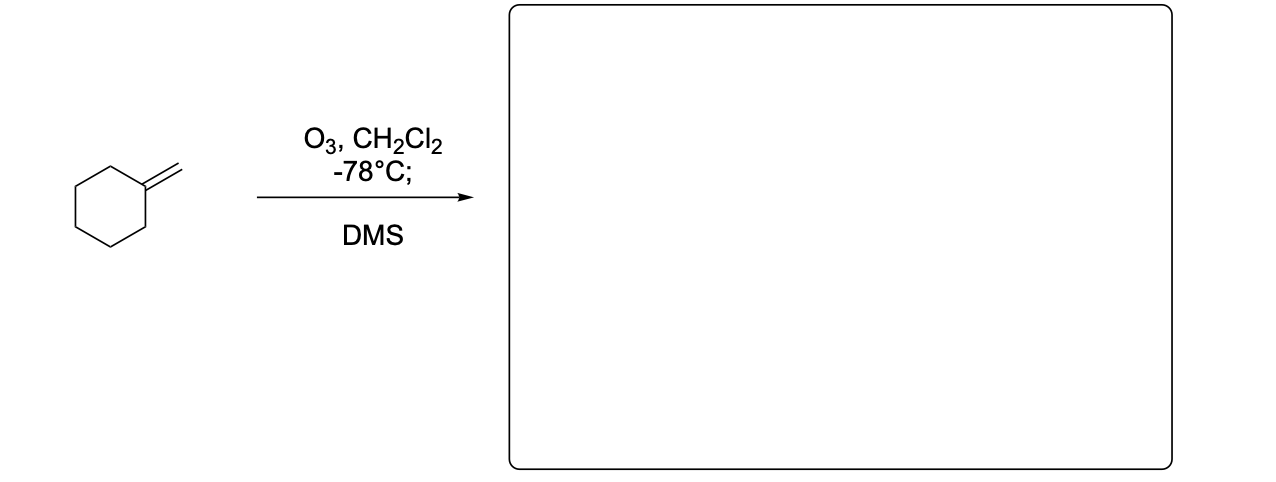
Please write the major product(s) from each reaction. Note that as this is an ozonolysis, there can often be TWO products formed equally.
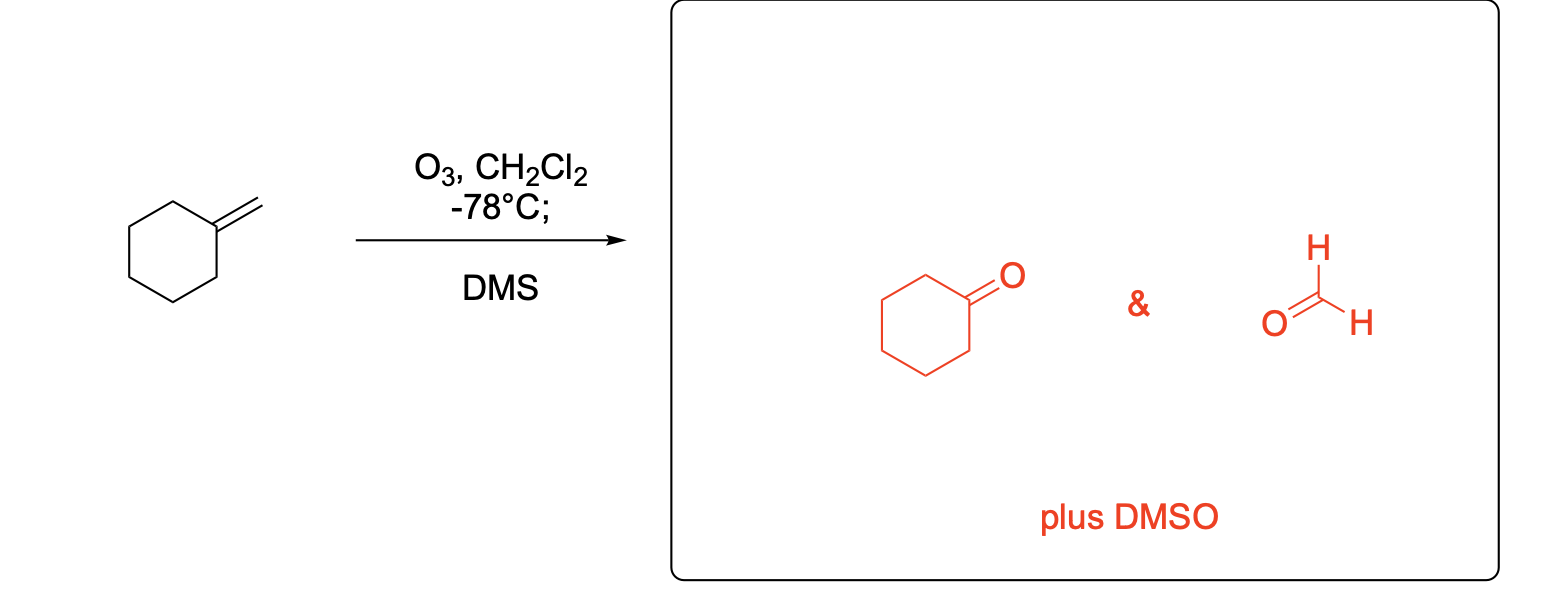
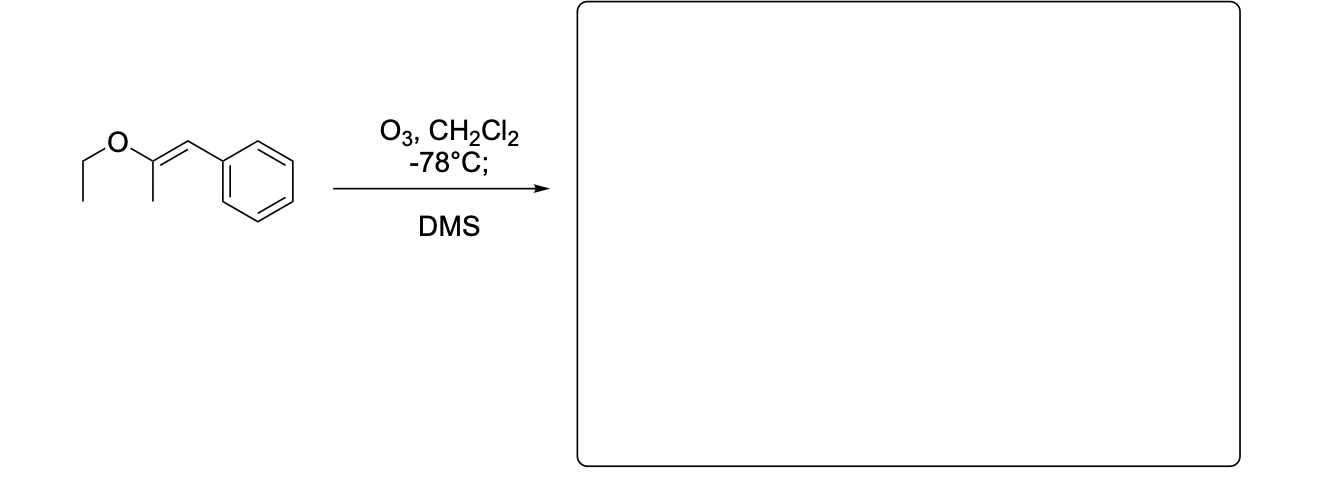
Please write the major product(s) from each reaction. Note that as this is an ozonolysis, there can often be TWO products formed equally.
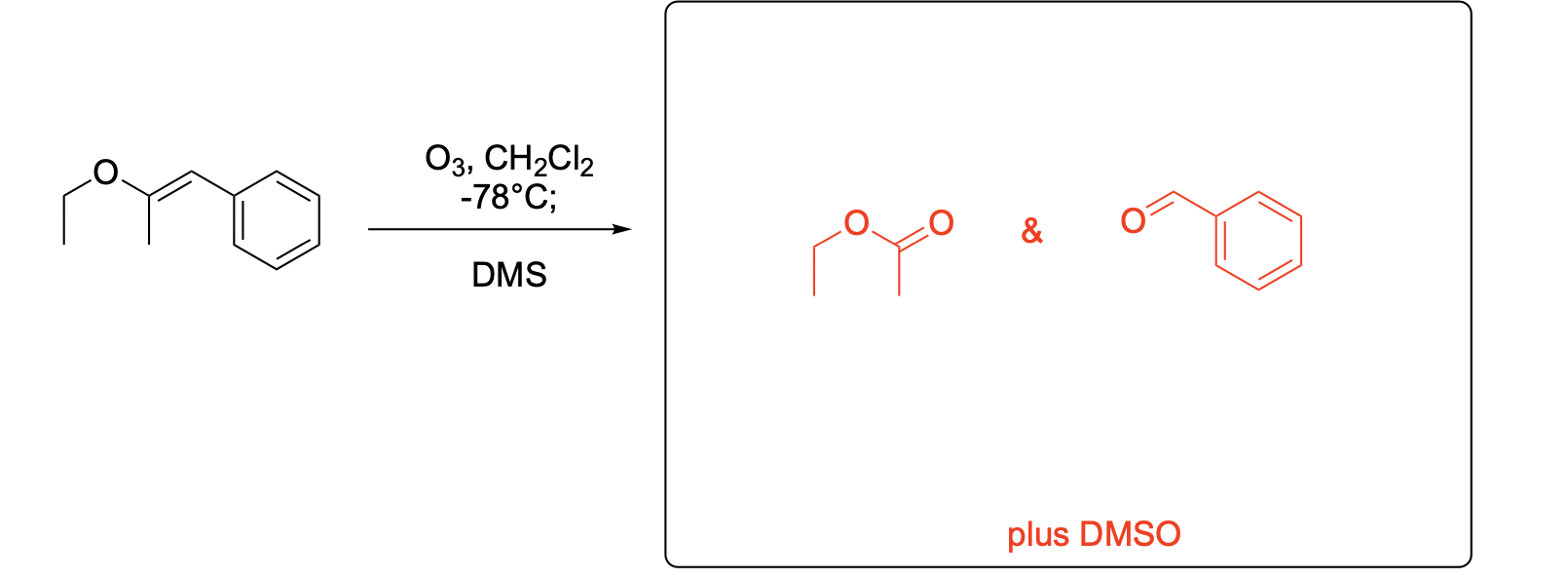
Please write the major product(s) from each reaction. Note that as this is an ozonolysis, there can often be TWO products formed equally.
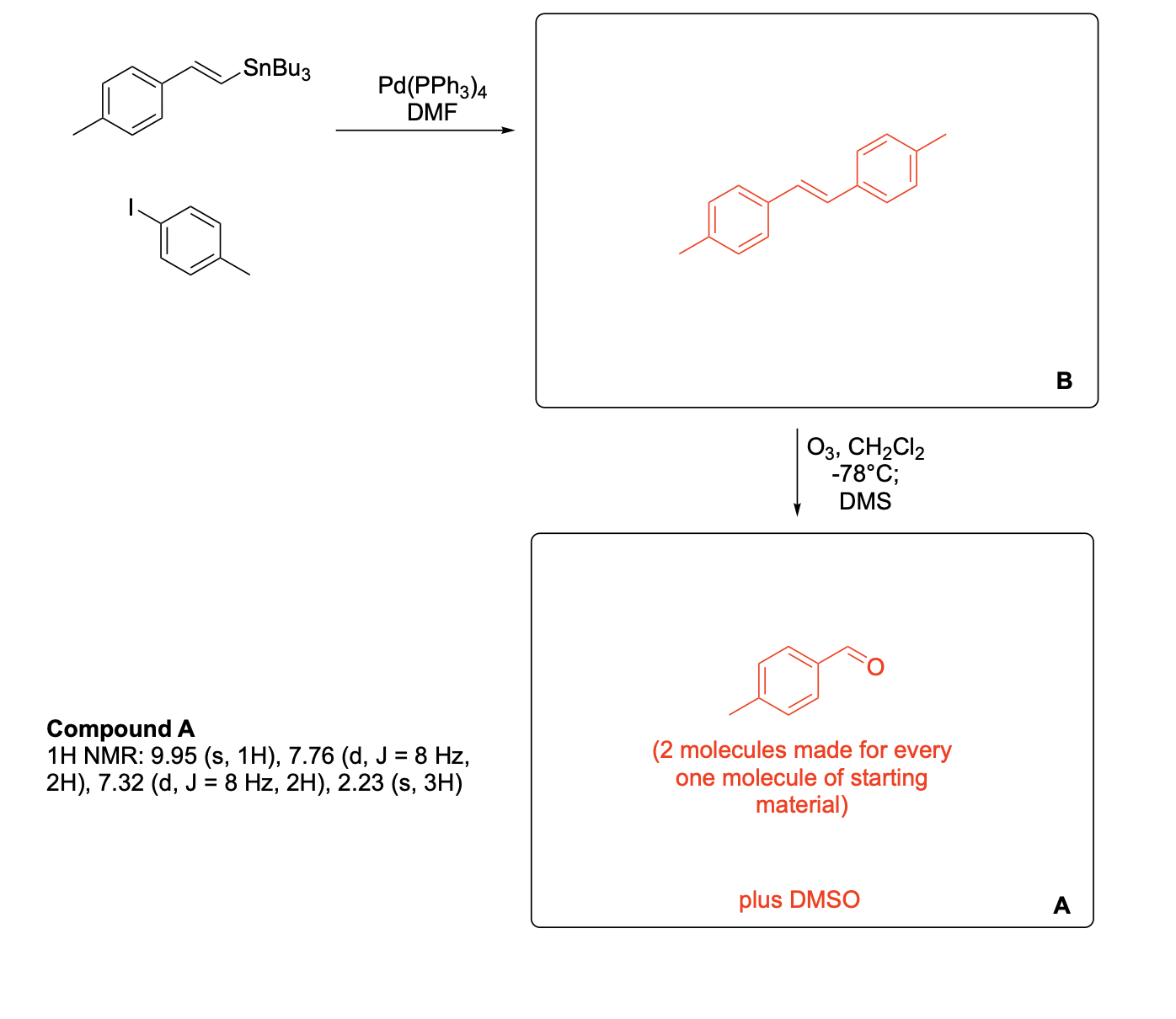
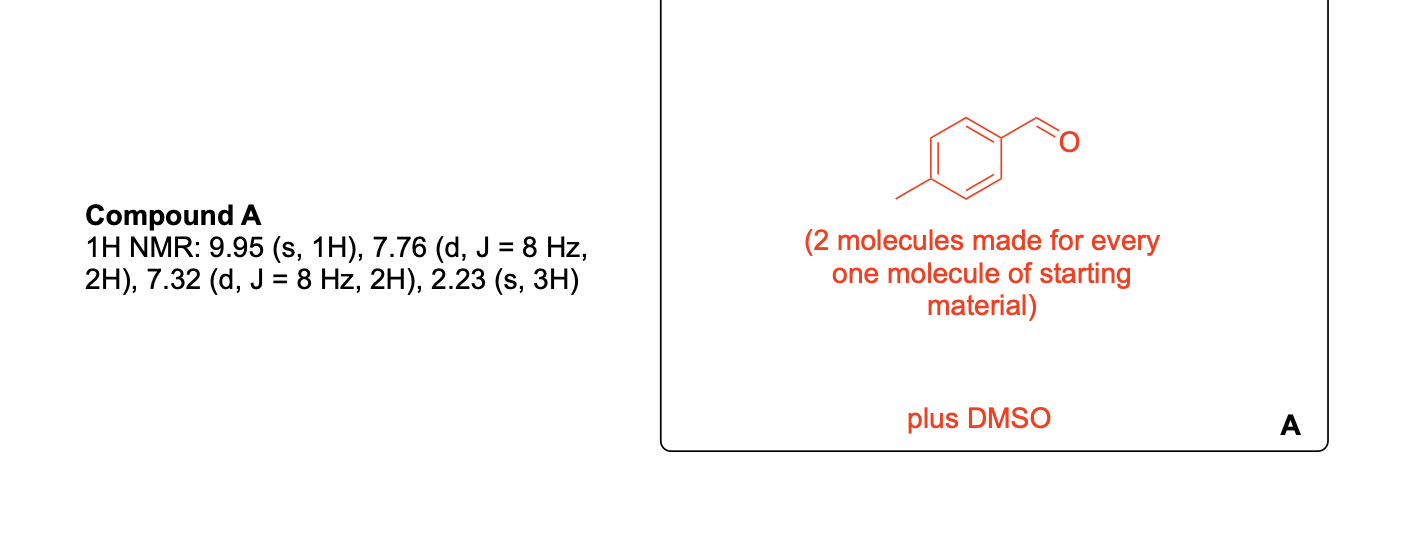
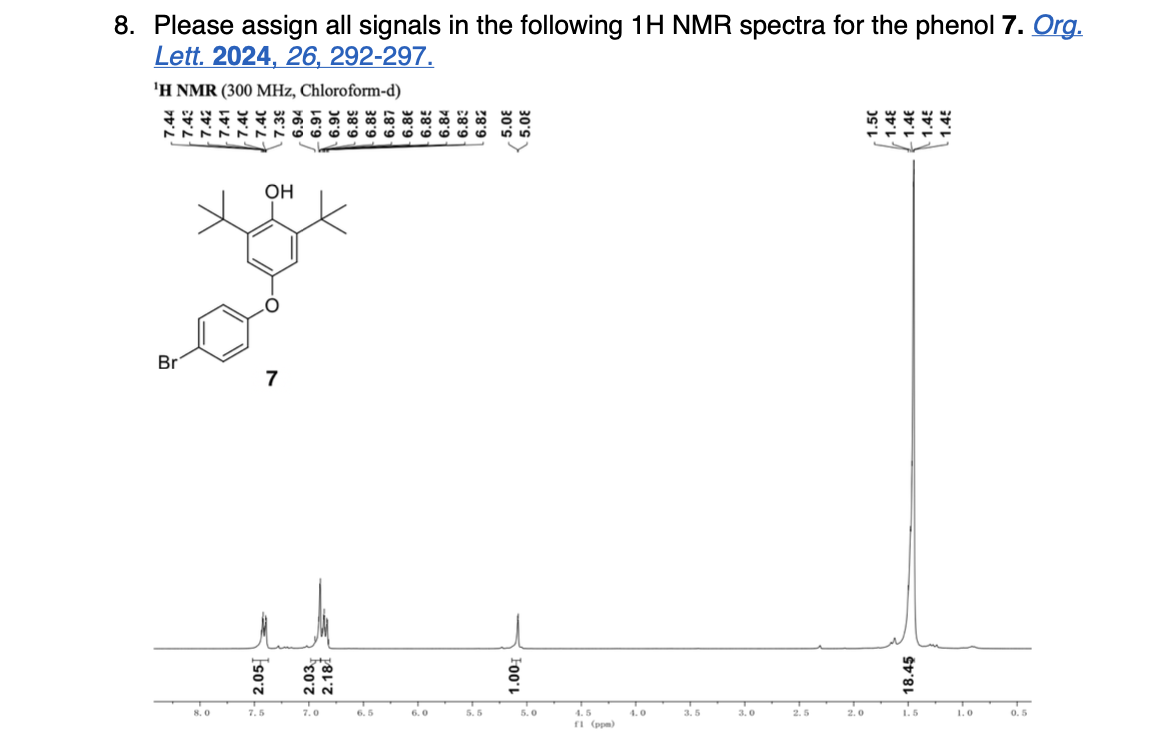
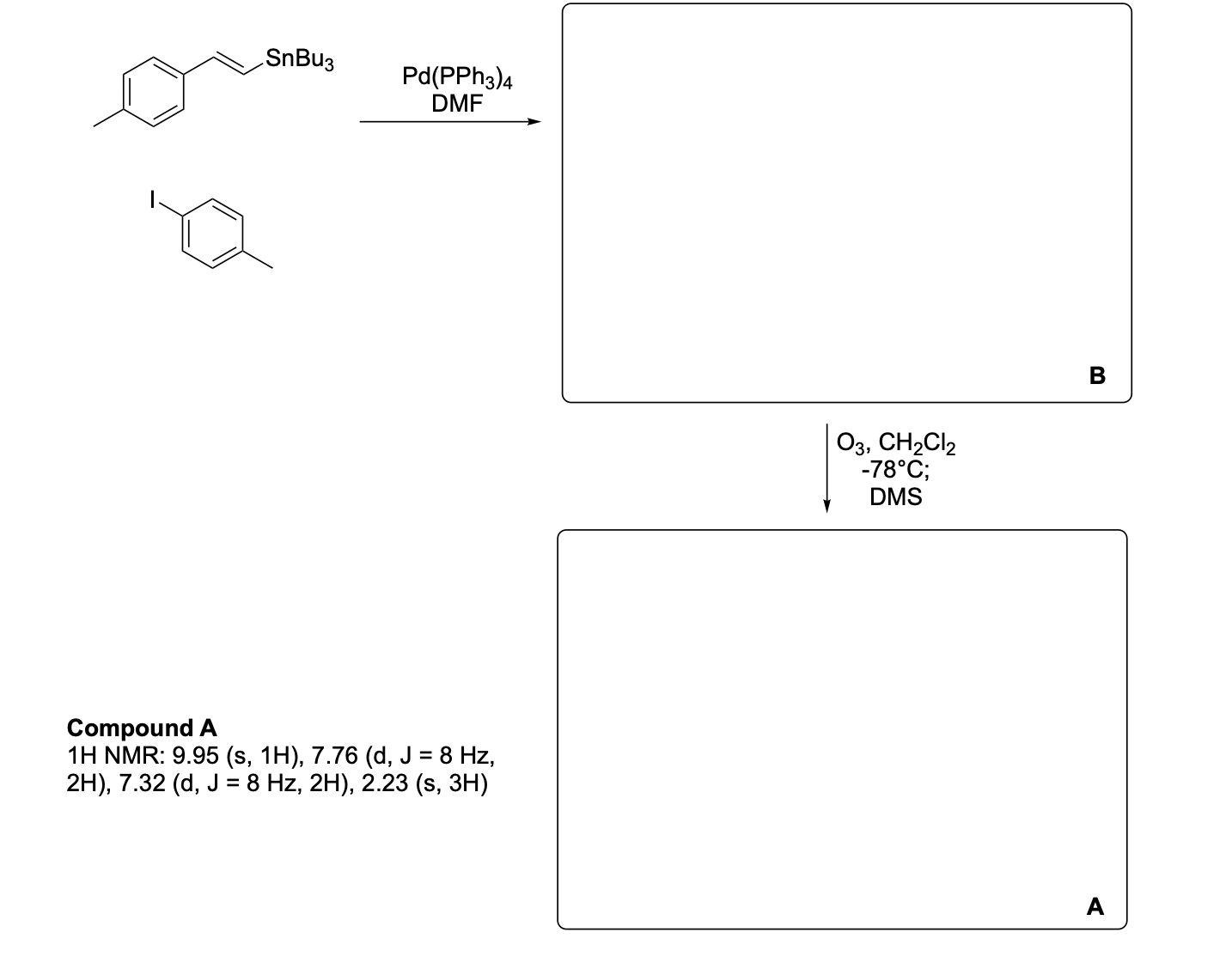
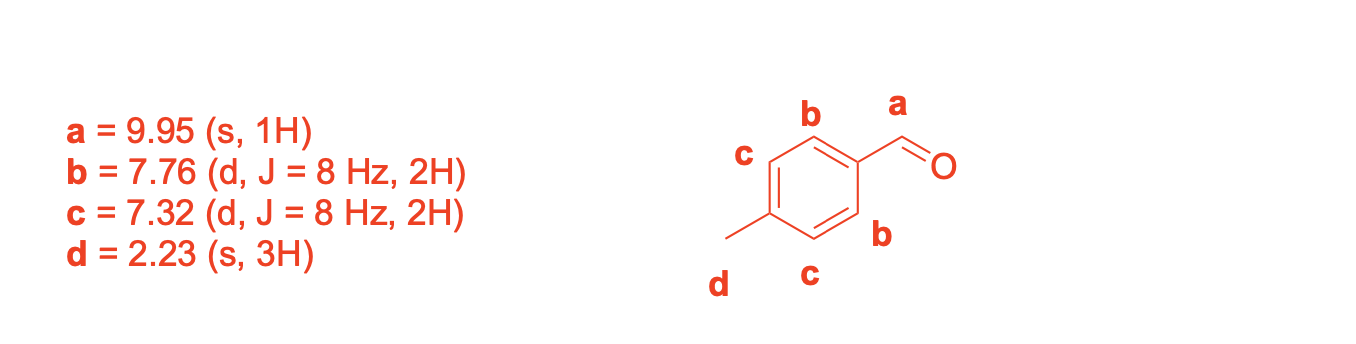
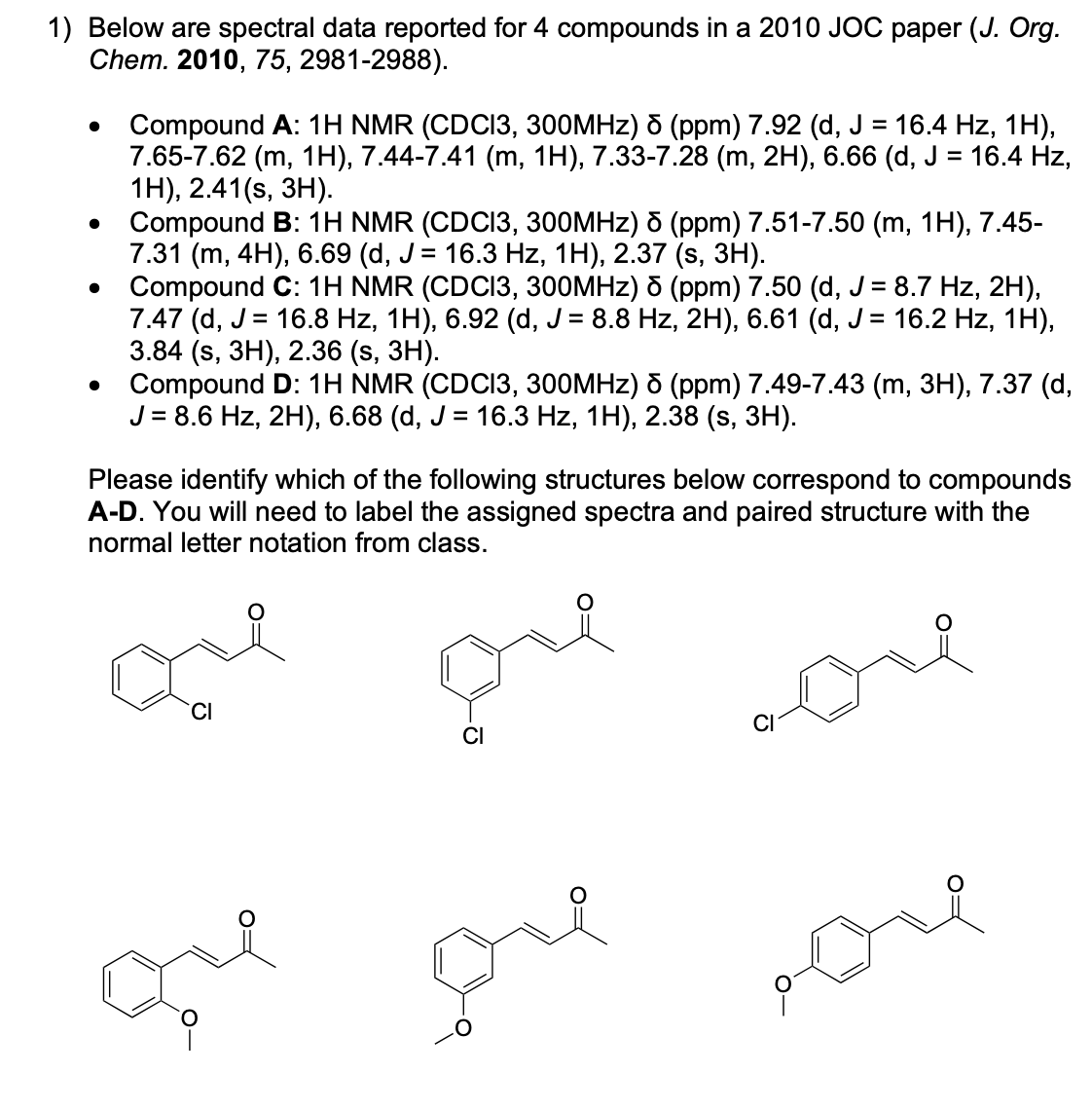
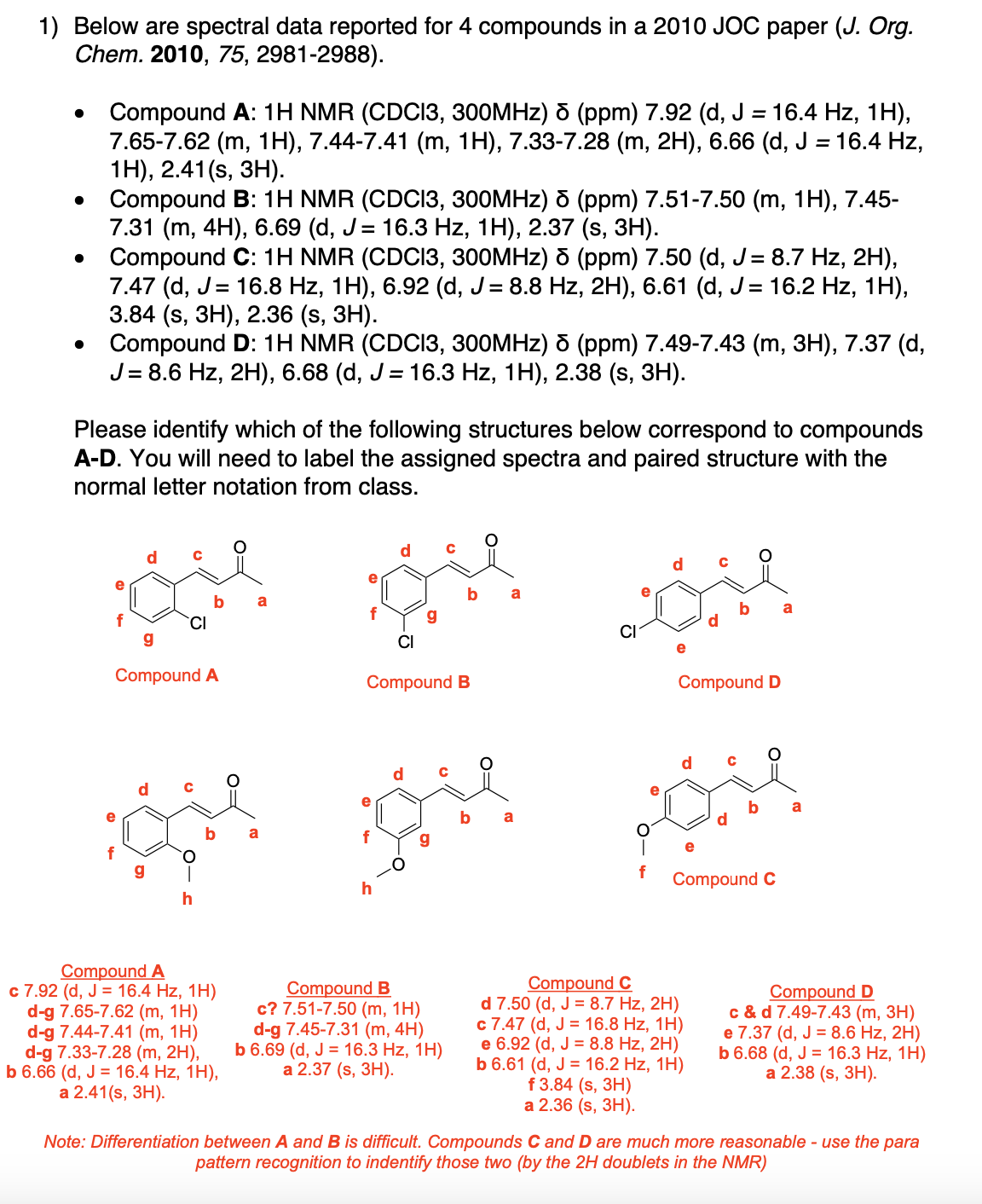
Please label and assign 1H NMR spectra data provided for compound A.
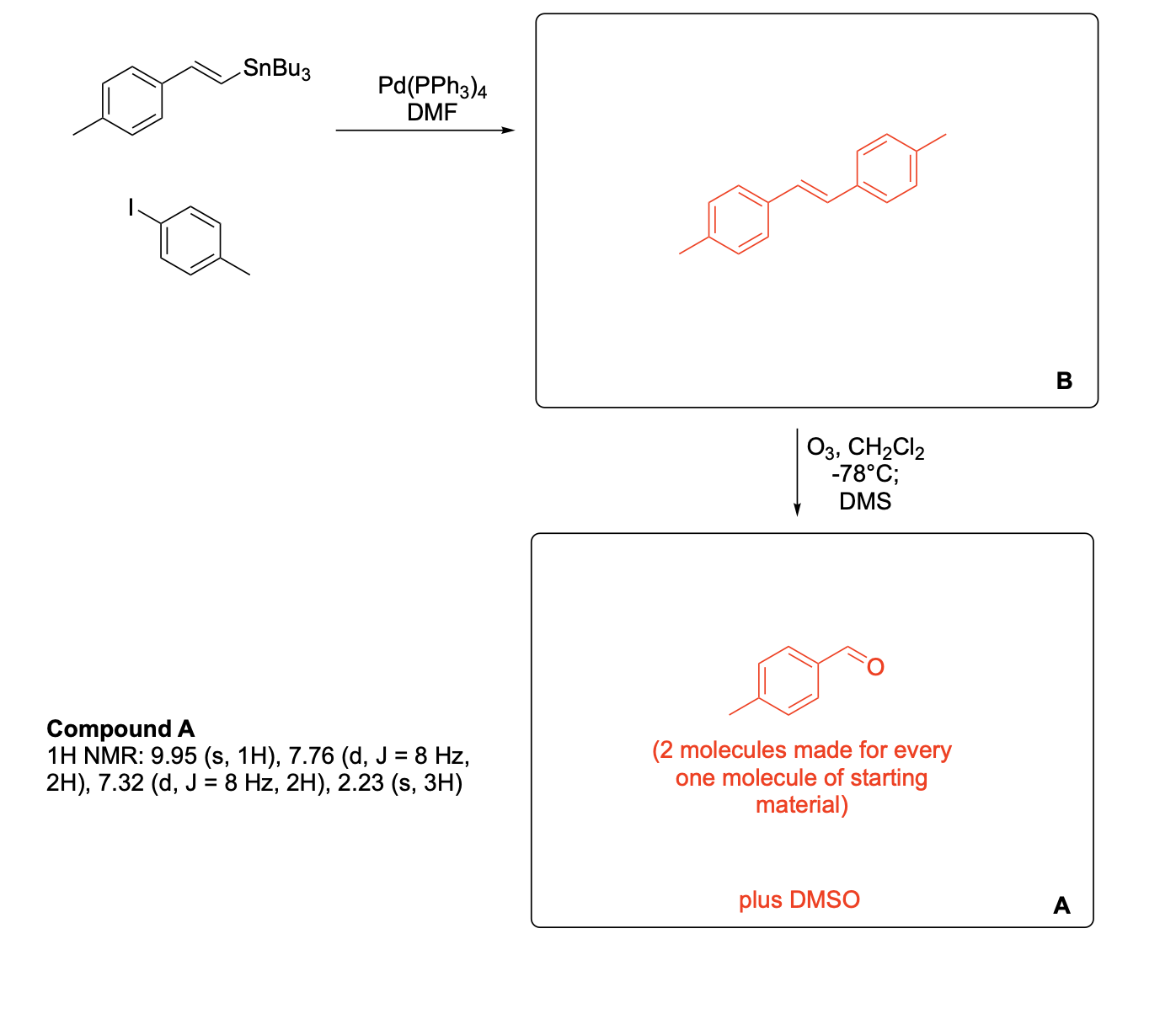
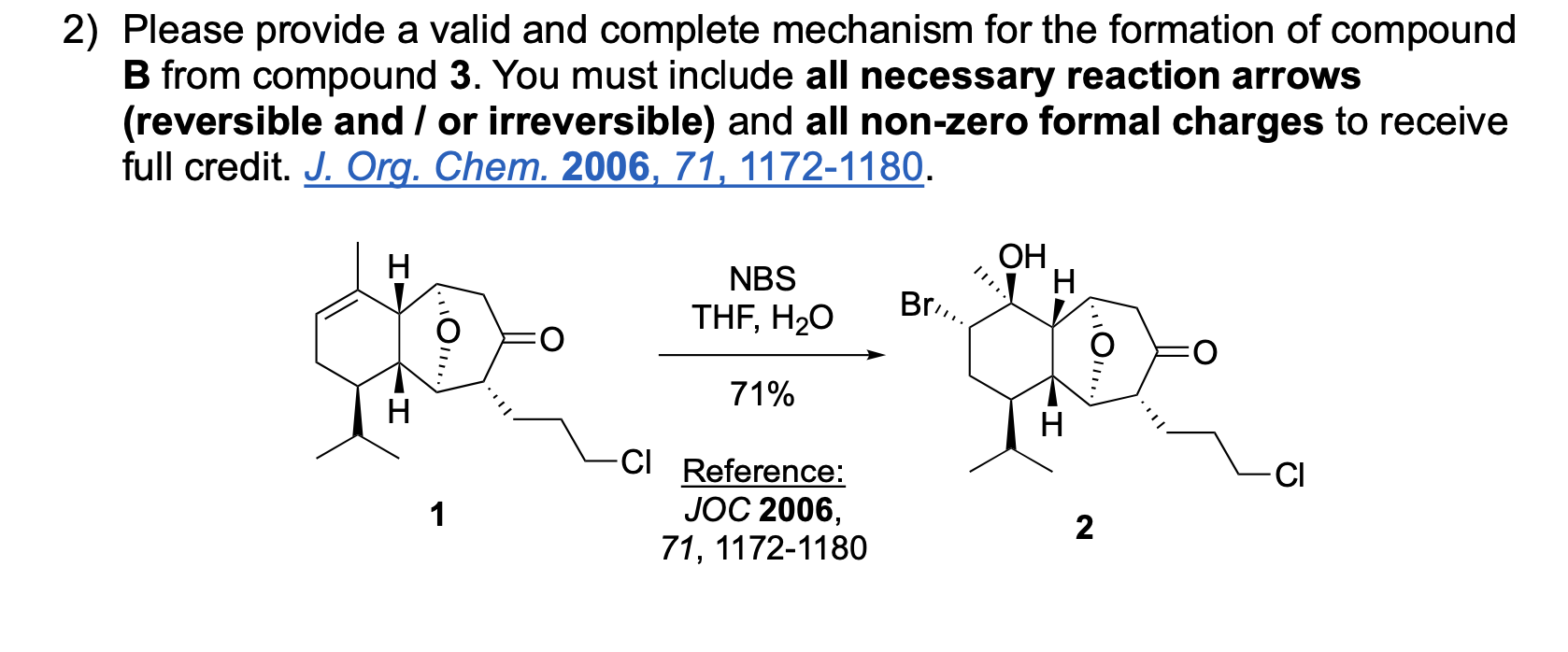
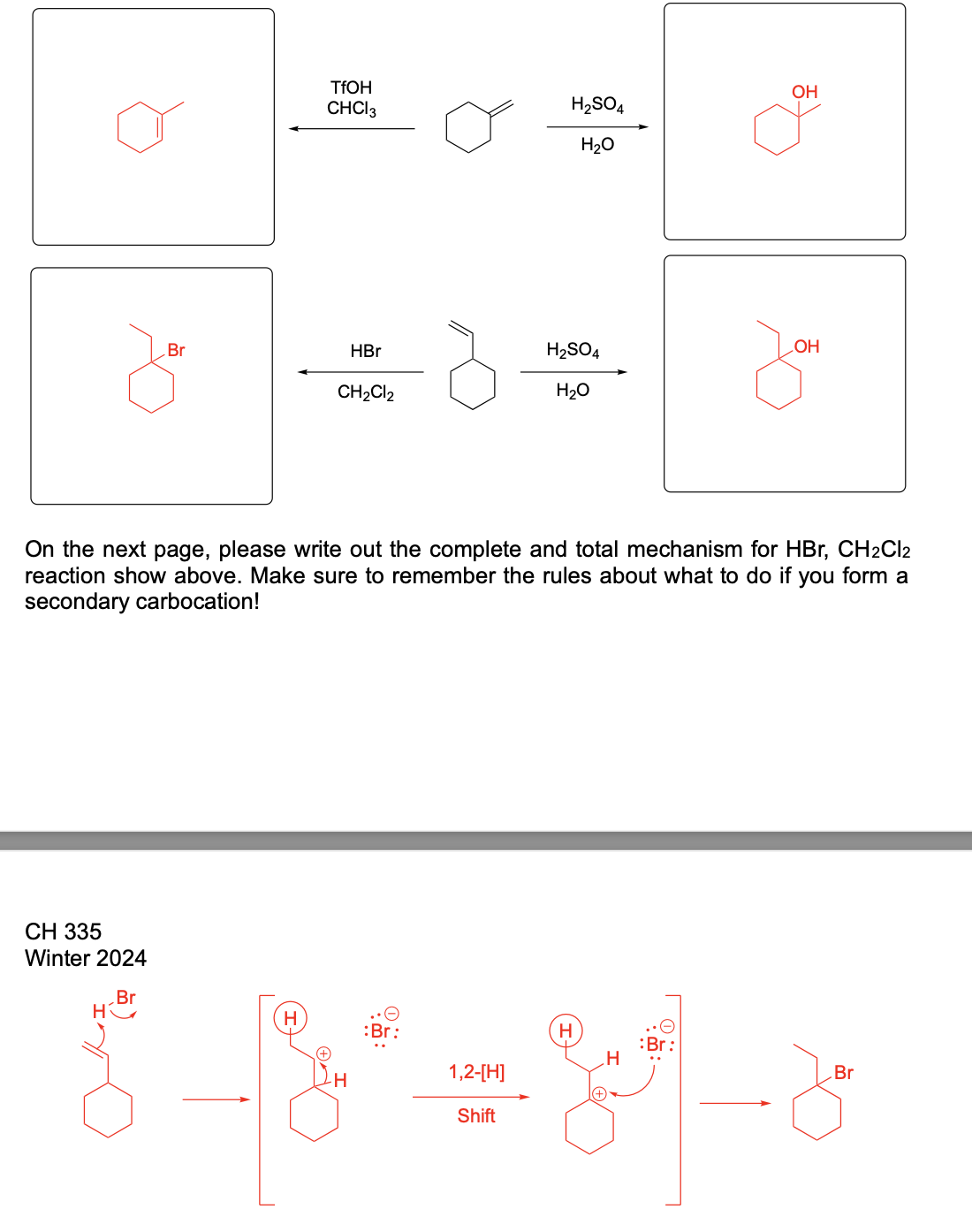
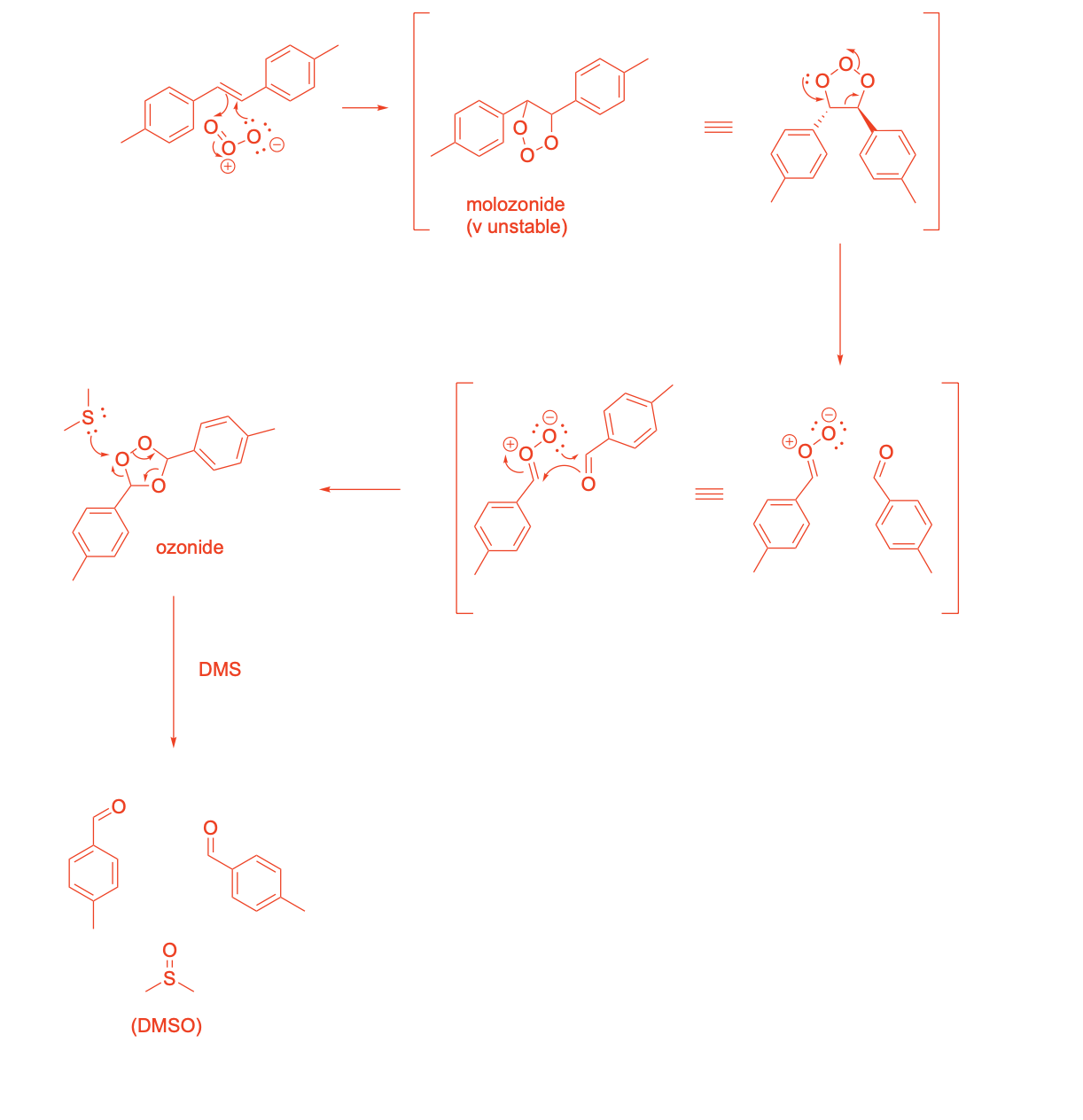
Please write the complete mechanism for the formation of compound A from intermediate B.
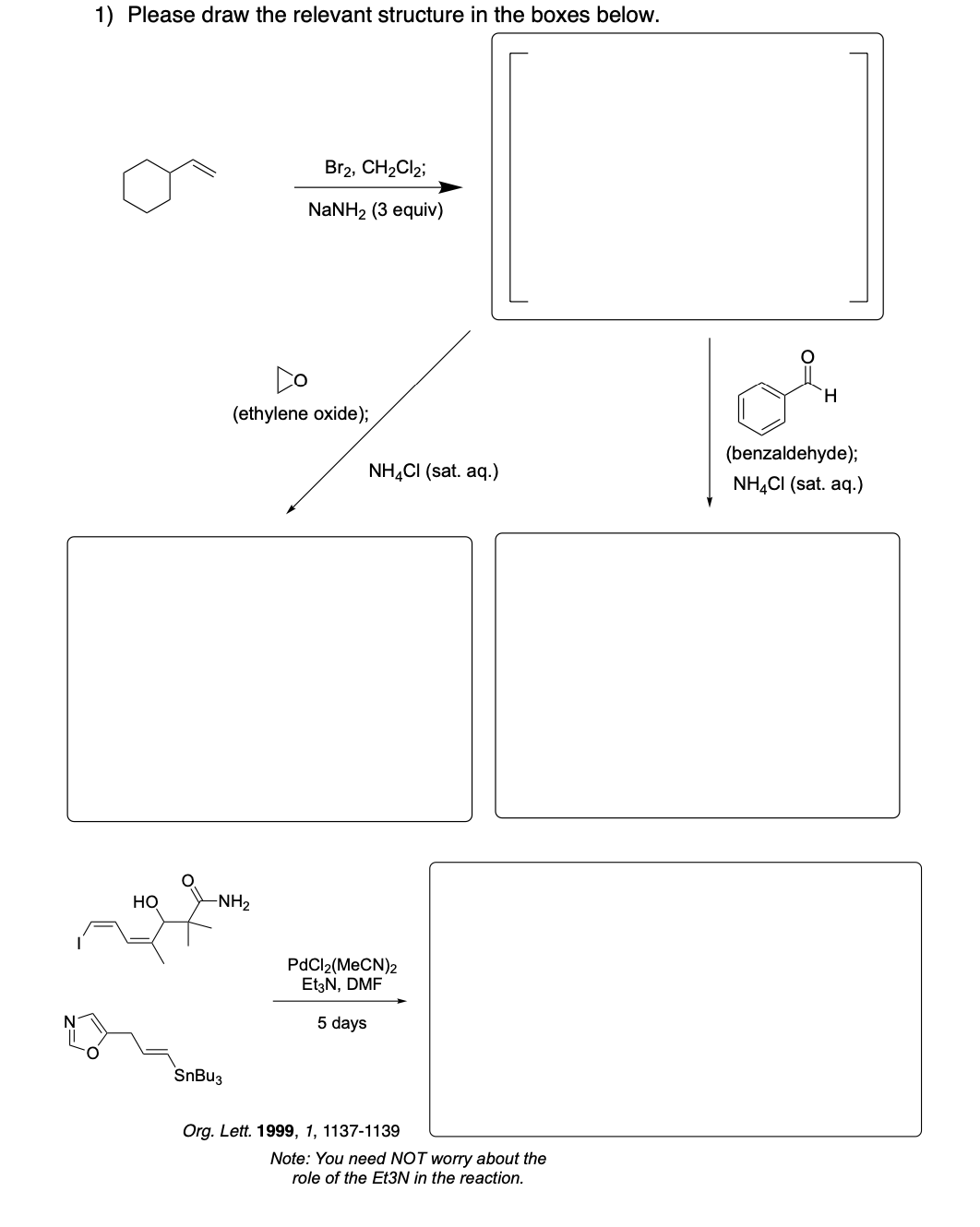
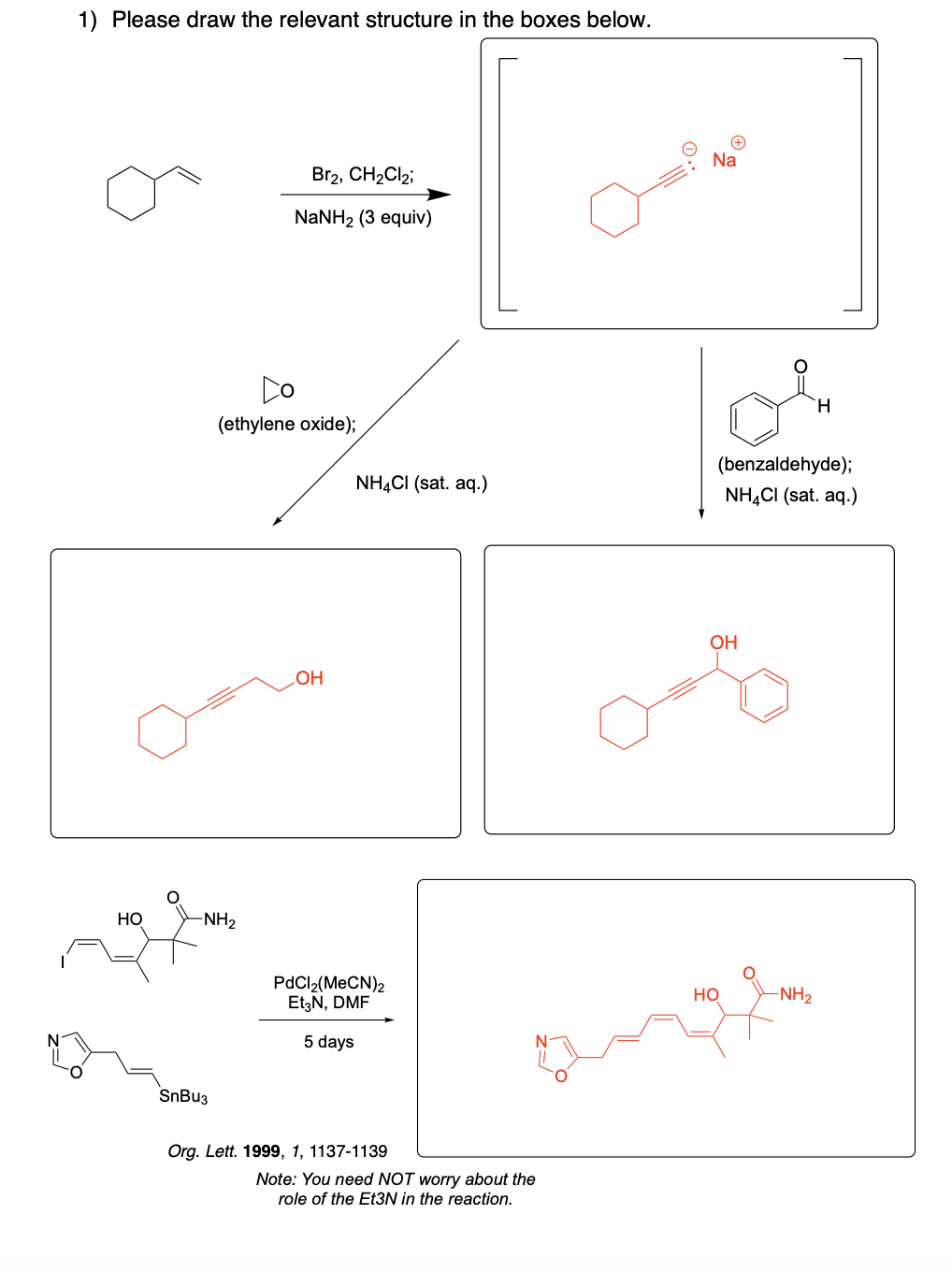
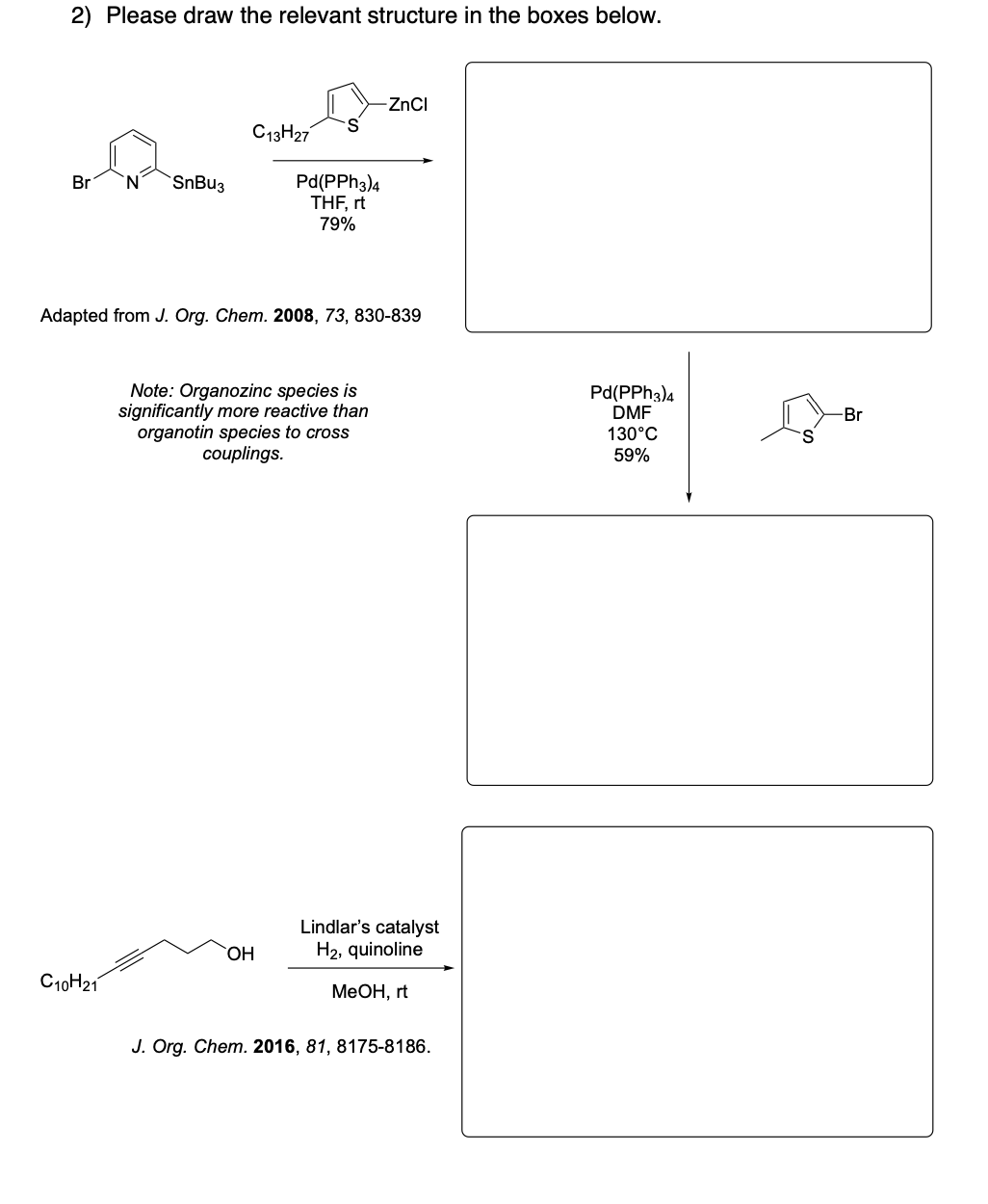
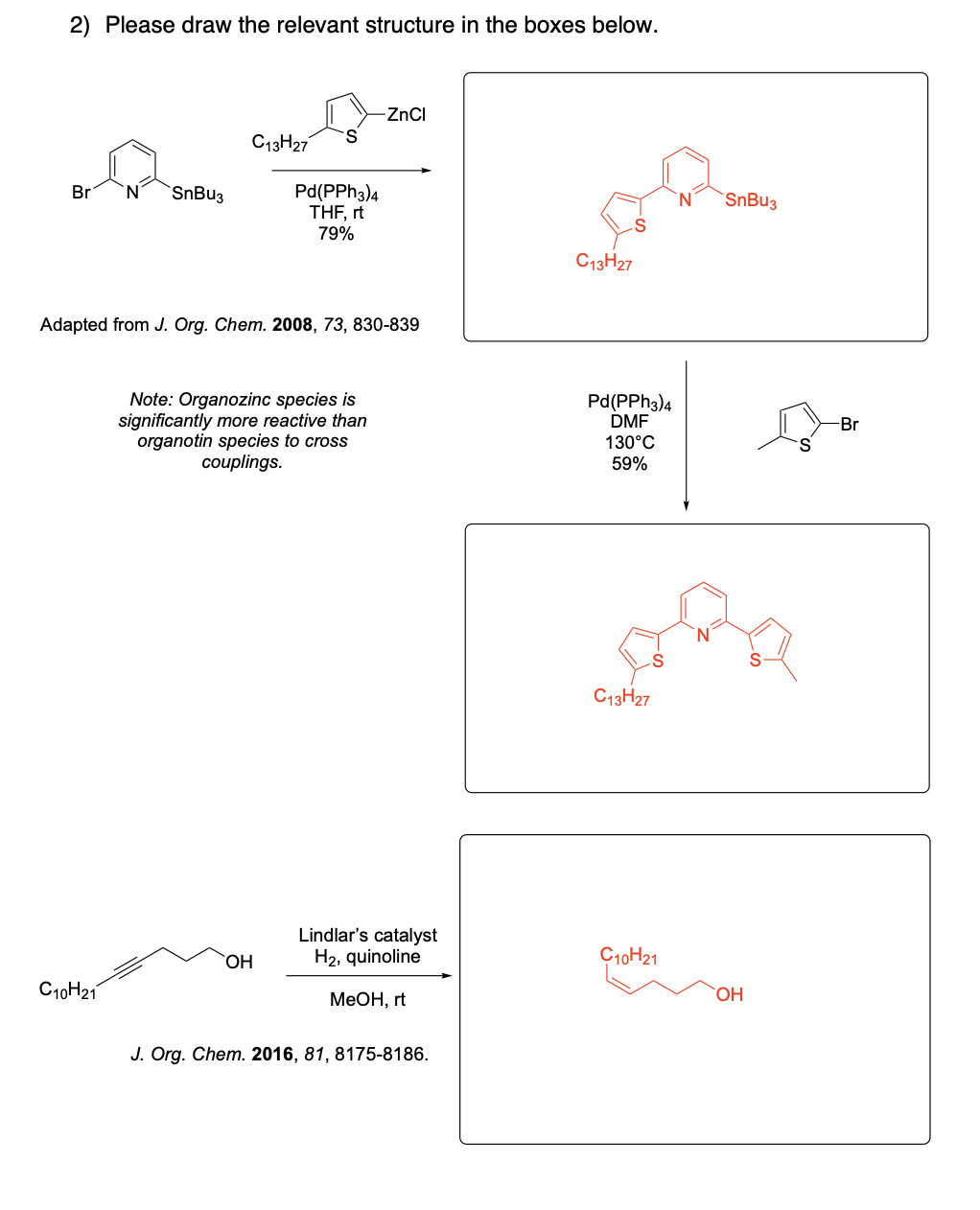
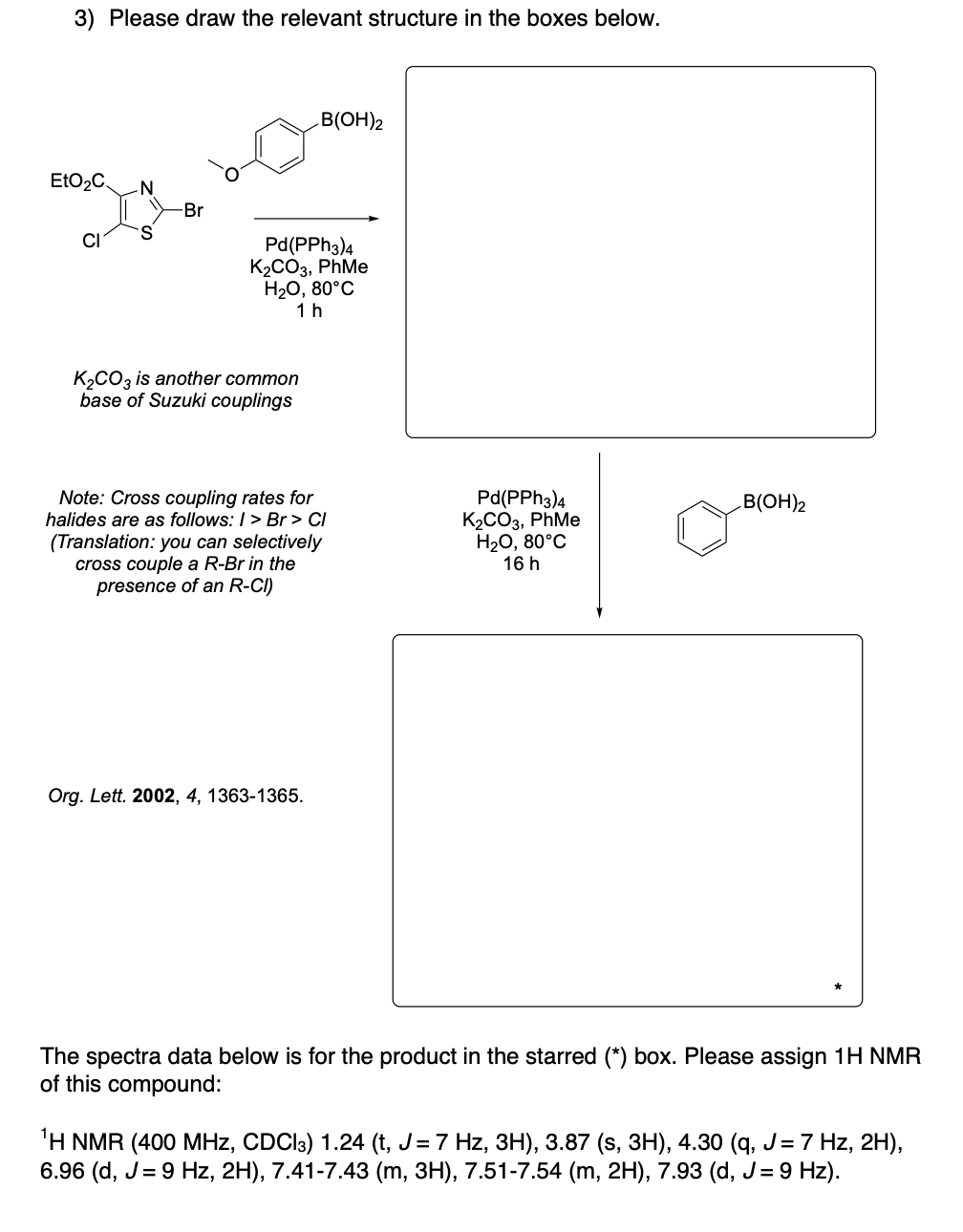
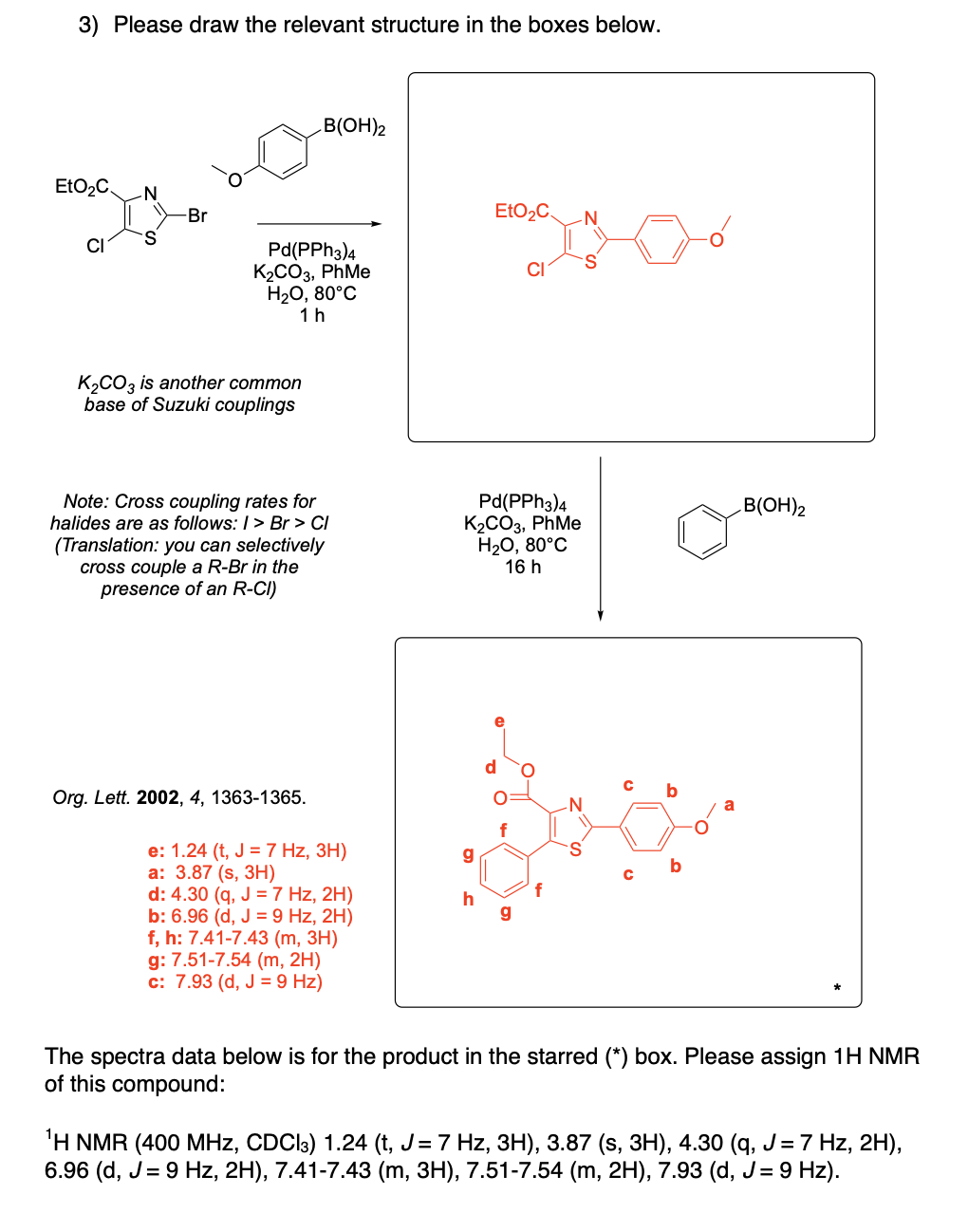
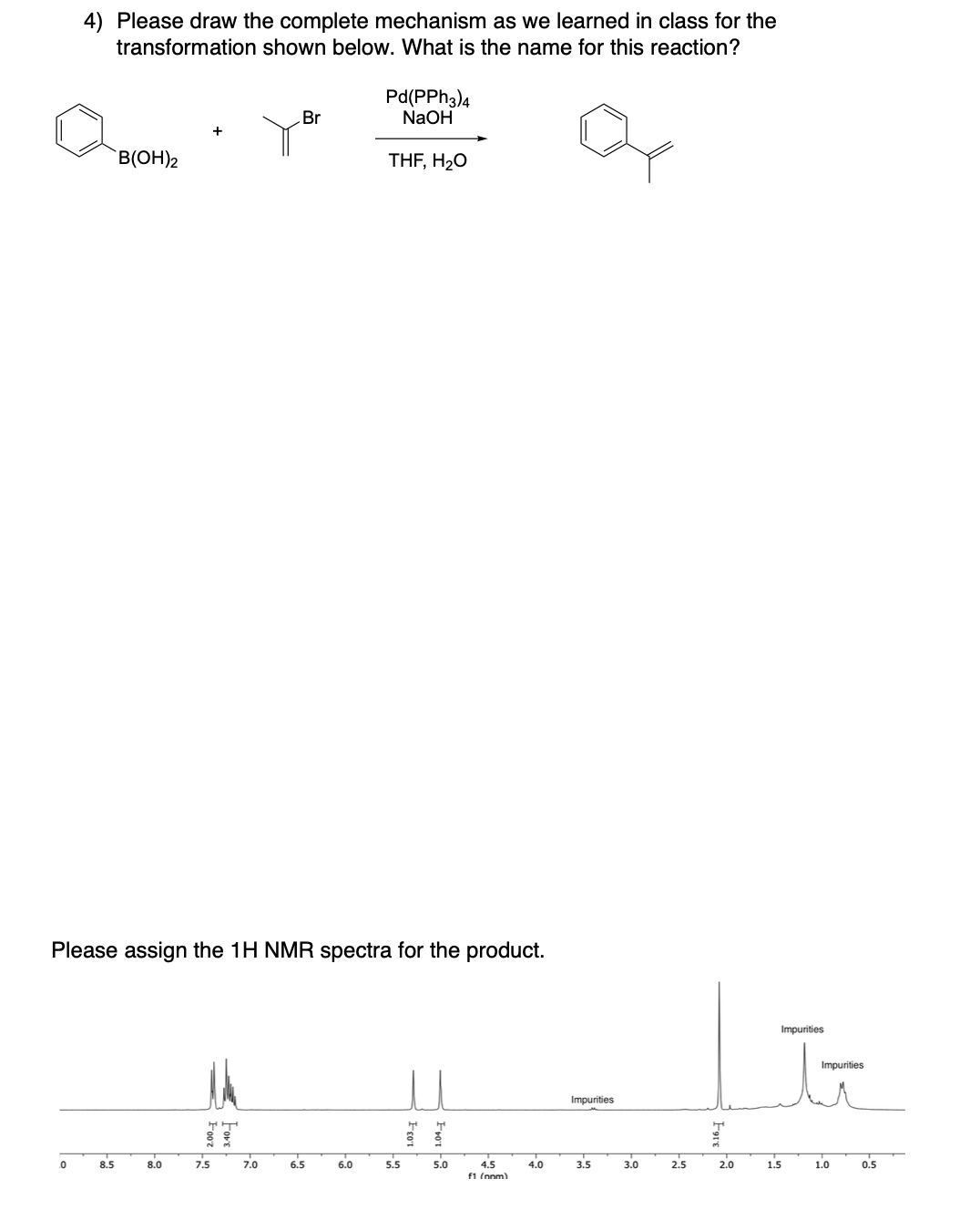
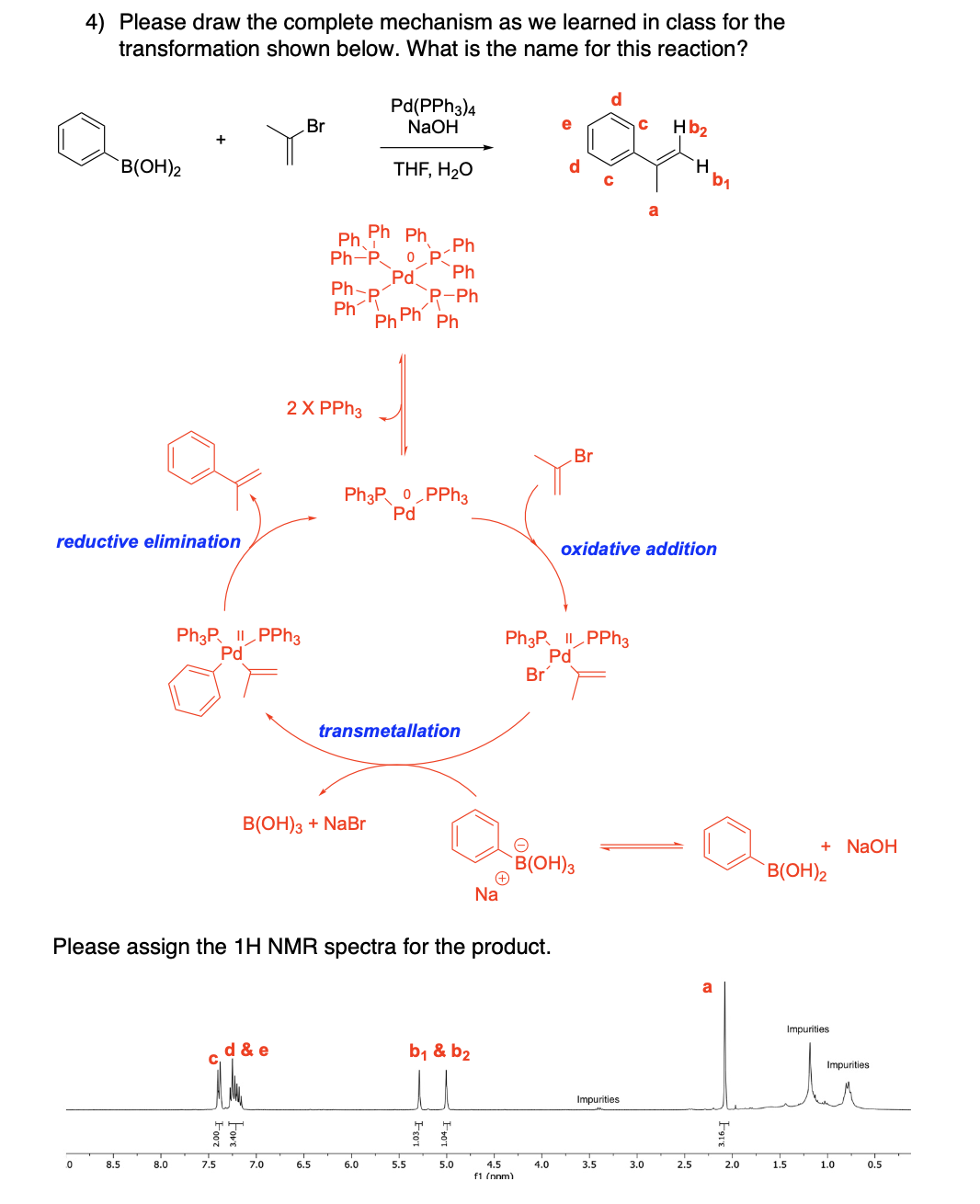

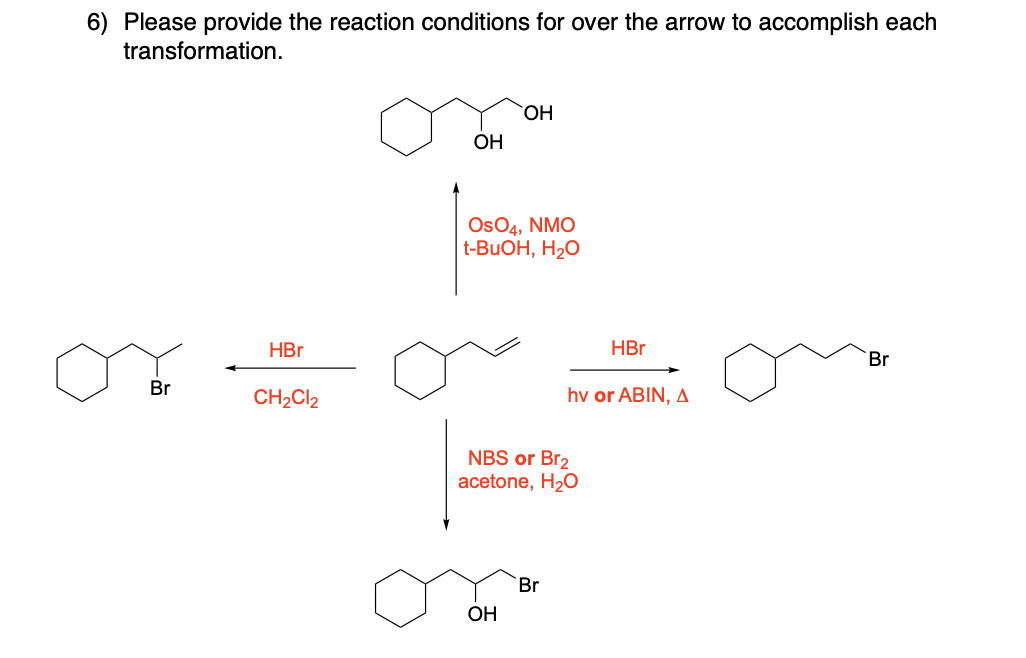
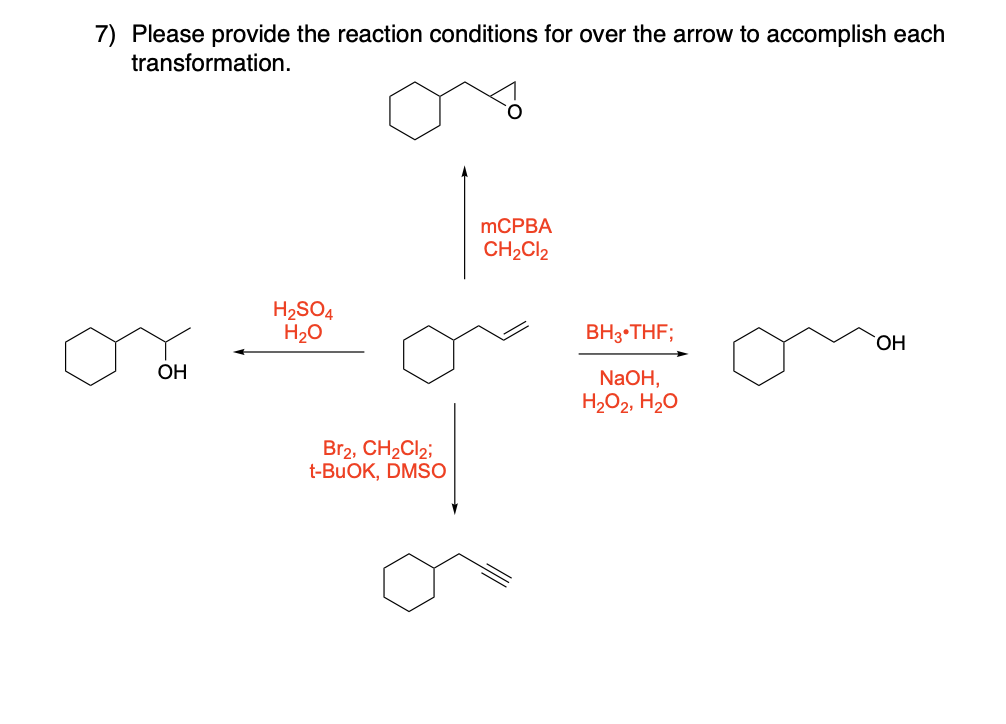
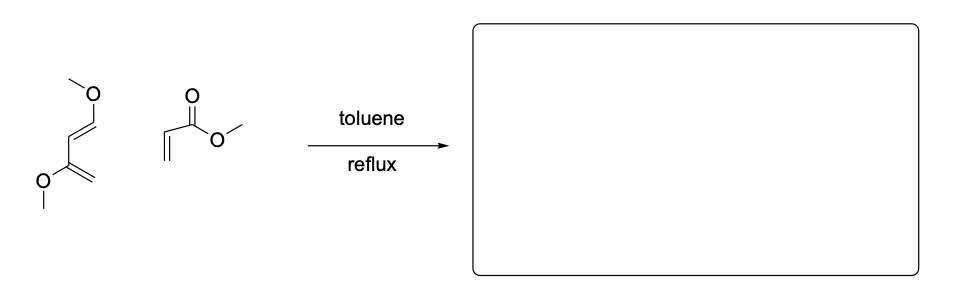
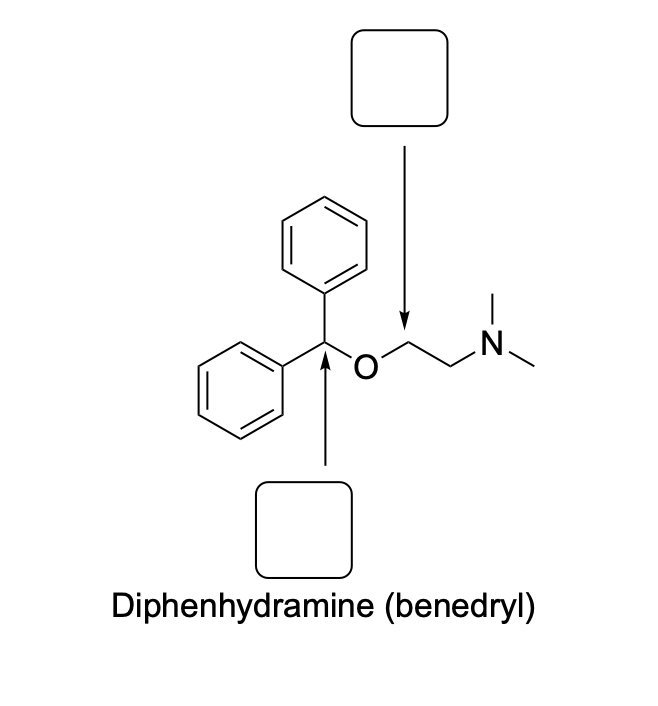
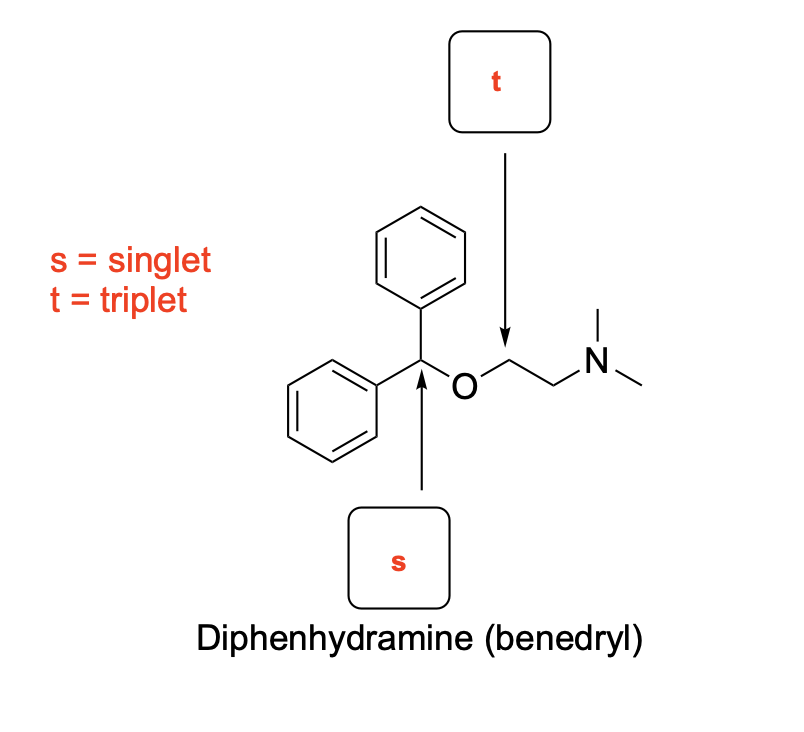
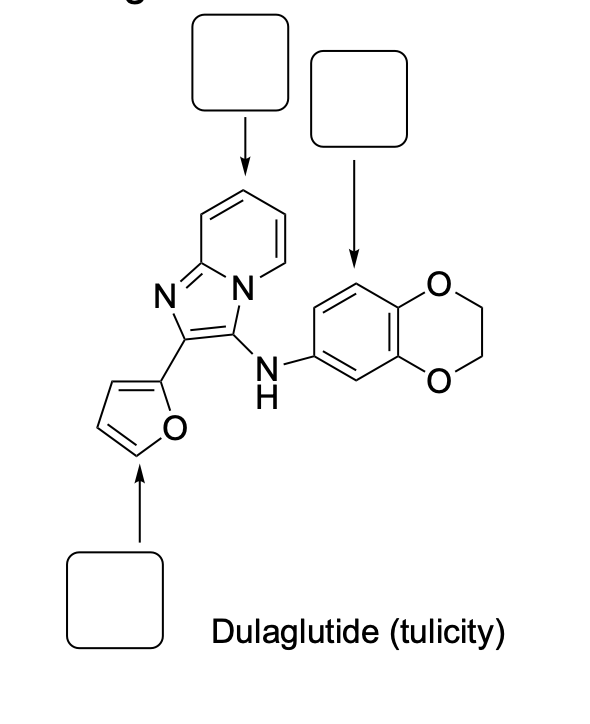
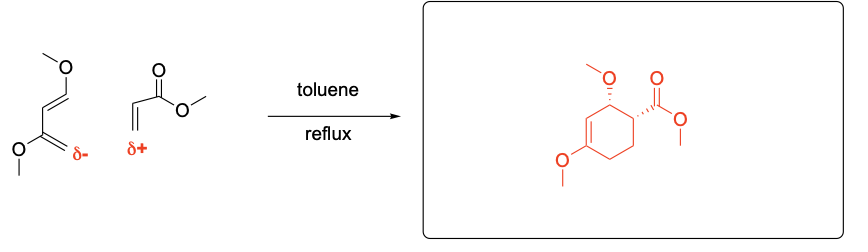
Please draw the relevant structure with stereochemistry (as appropriate) in the boxes below.
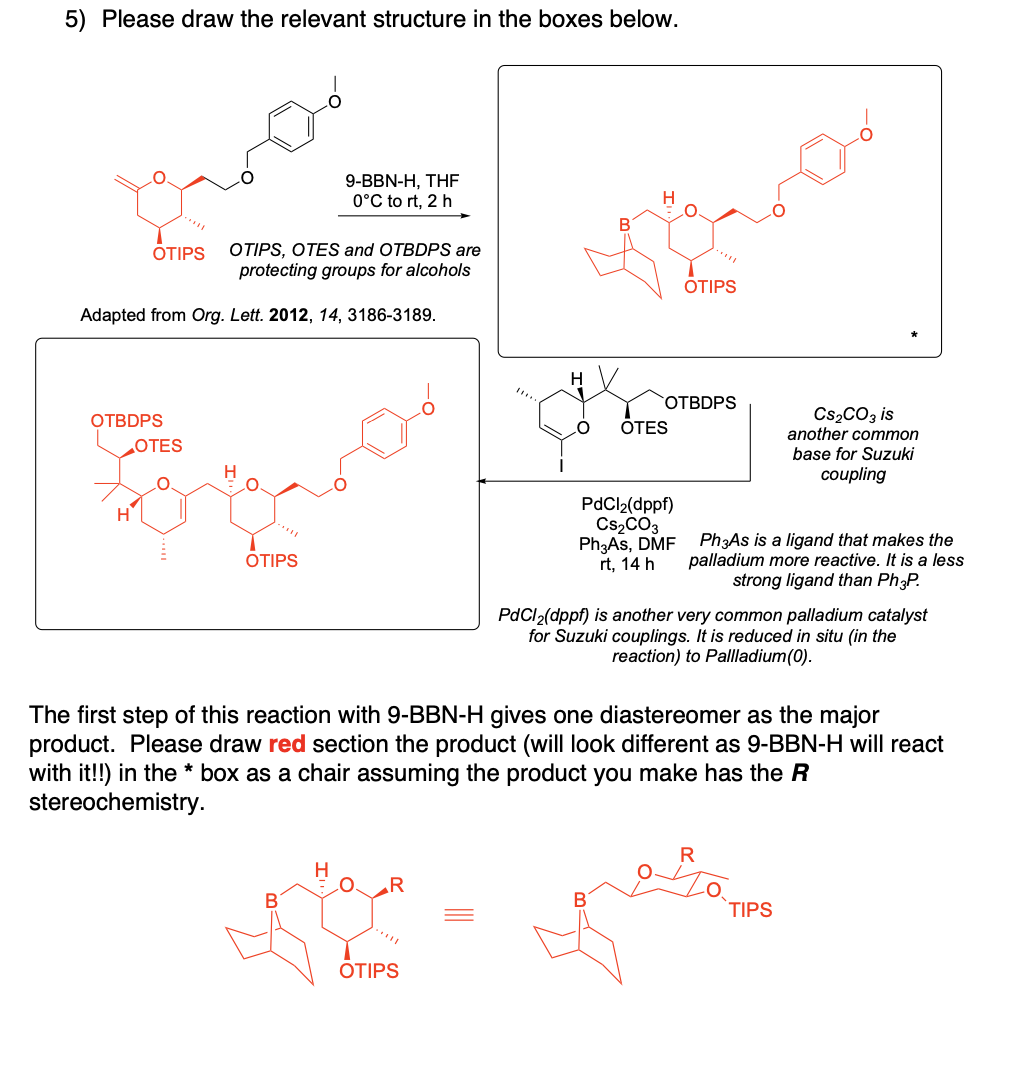
Please draw the relevant structure with stereochemistry (as appropriate) in the boxes below.
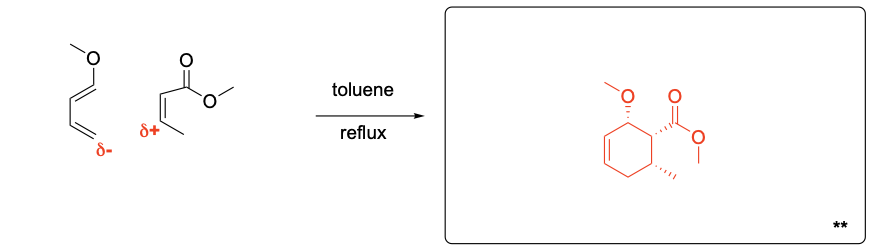
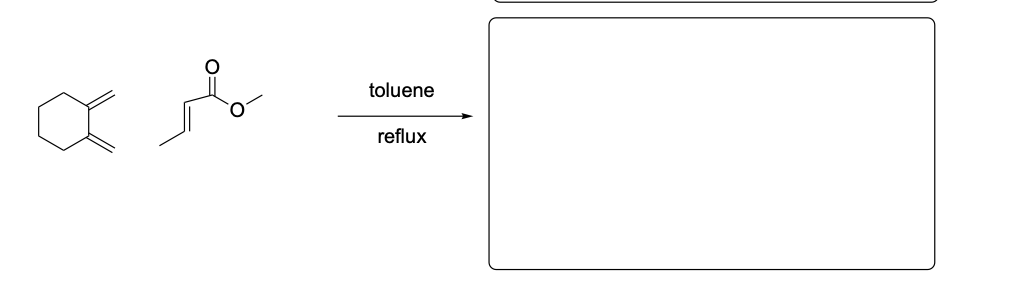
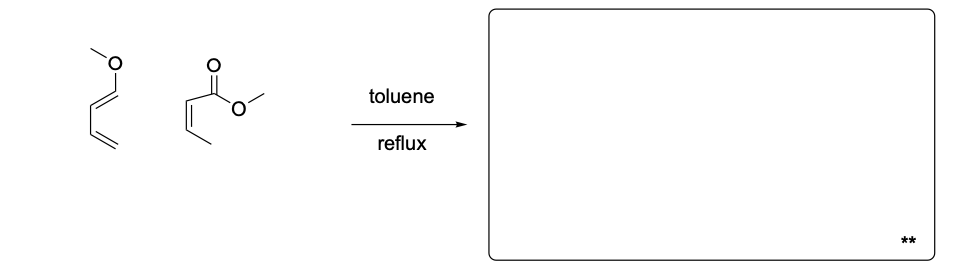
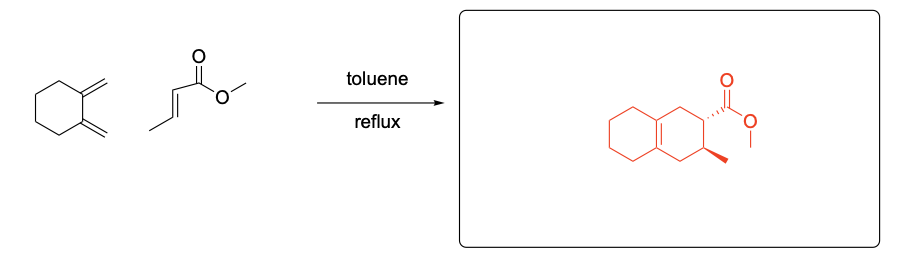
Please draw the relevant structure with stereochemistry (as appropriate) in the boxes below.
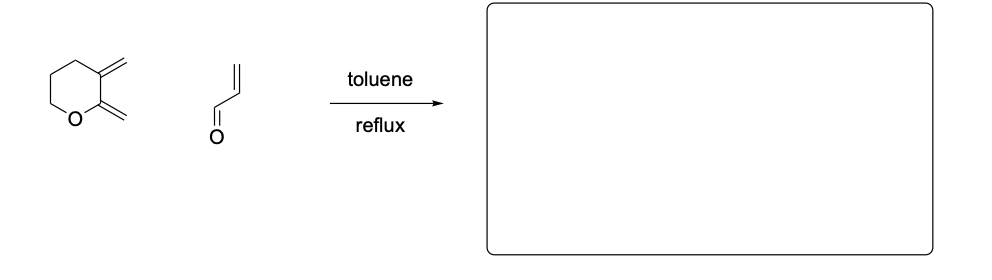
Please draw the relevant structure with stereochemistry (as appropriate) in the boxes below.
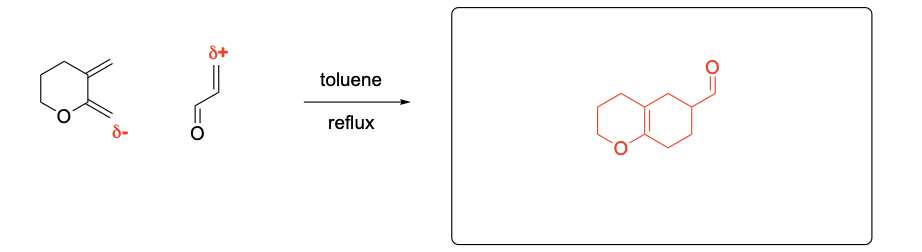
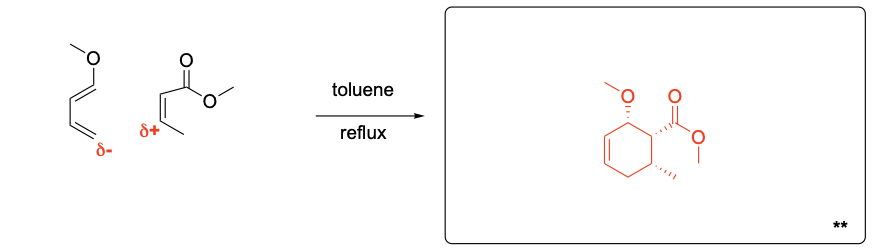
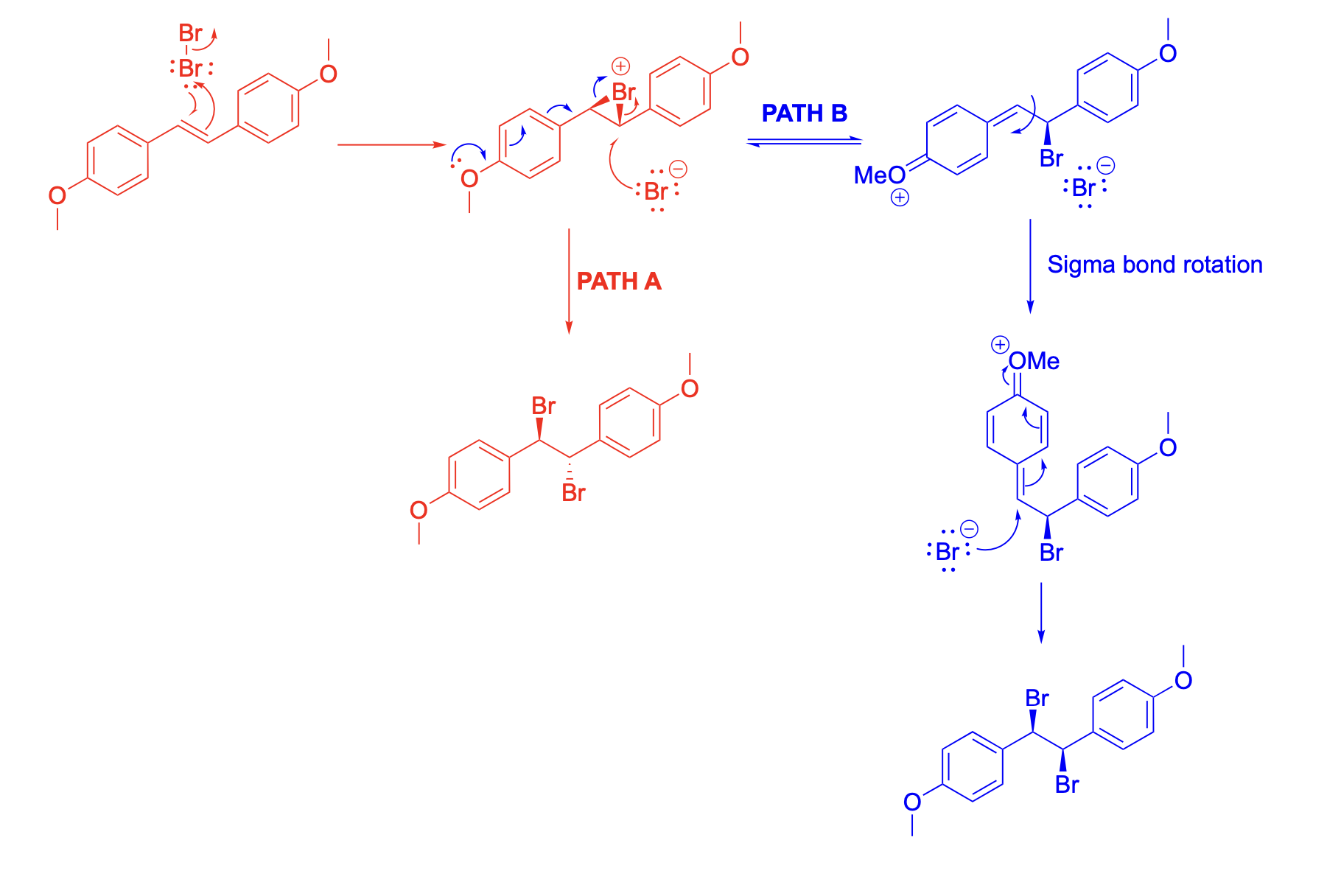
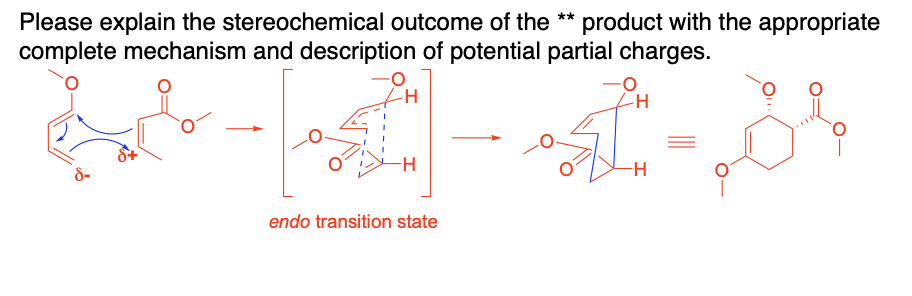
Please explain the stereochemical outcome of the ** product with the appropriate complete mechanism and description of potential partial charges.
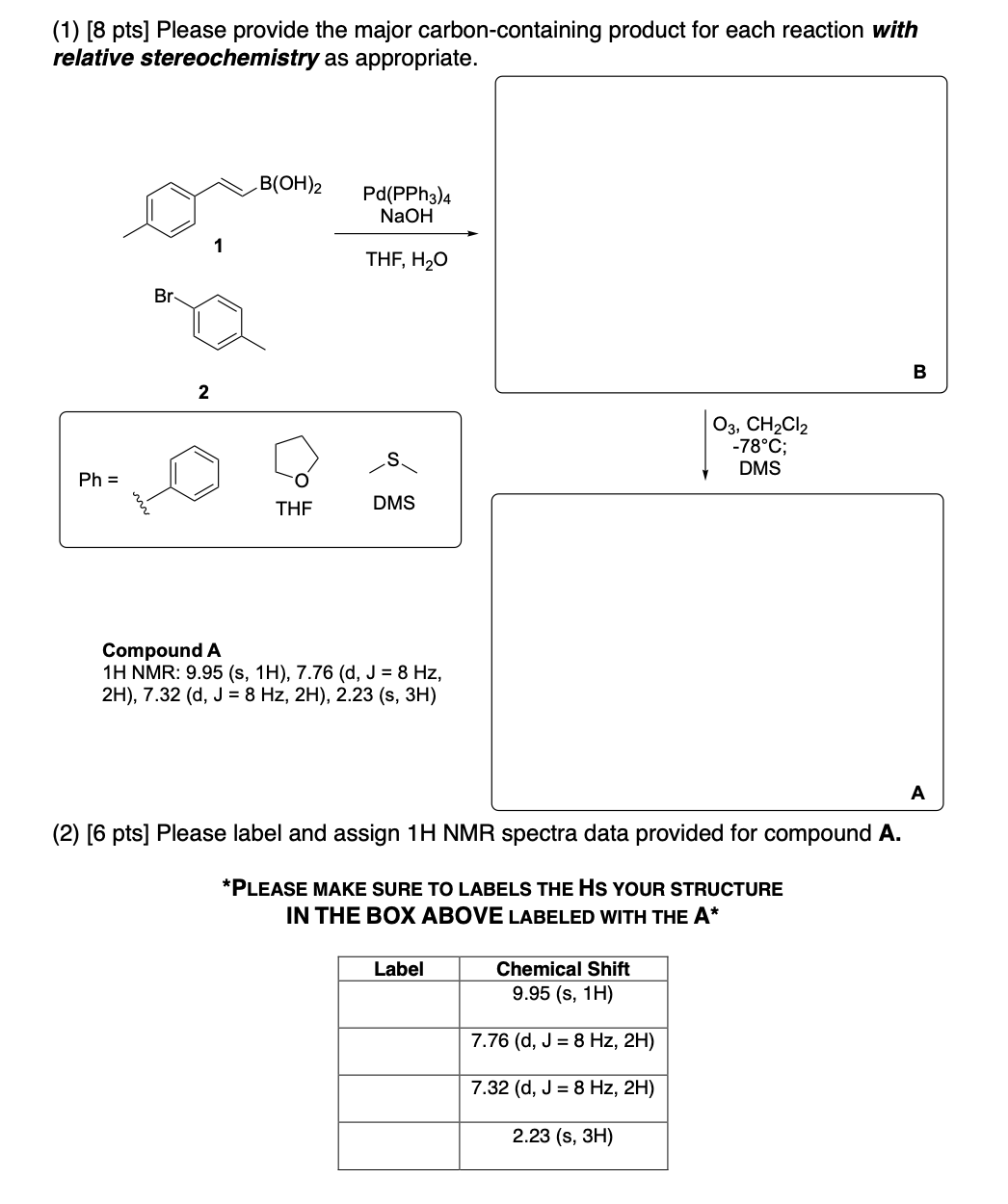
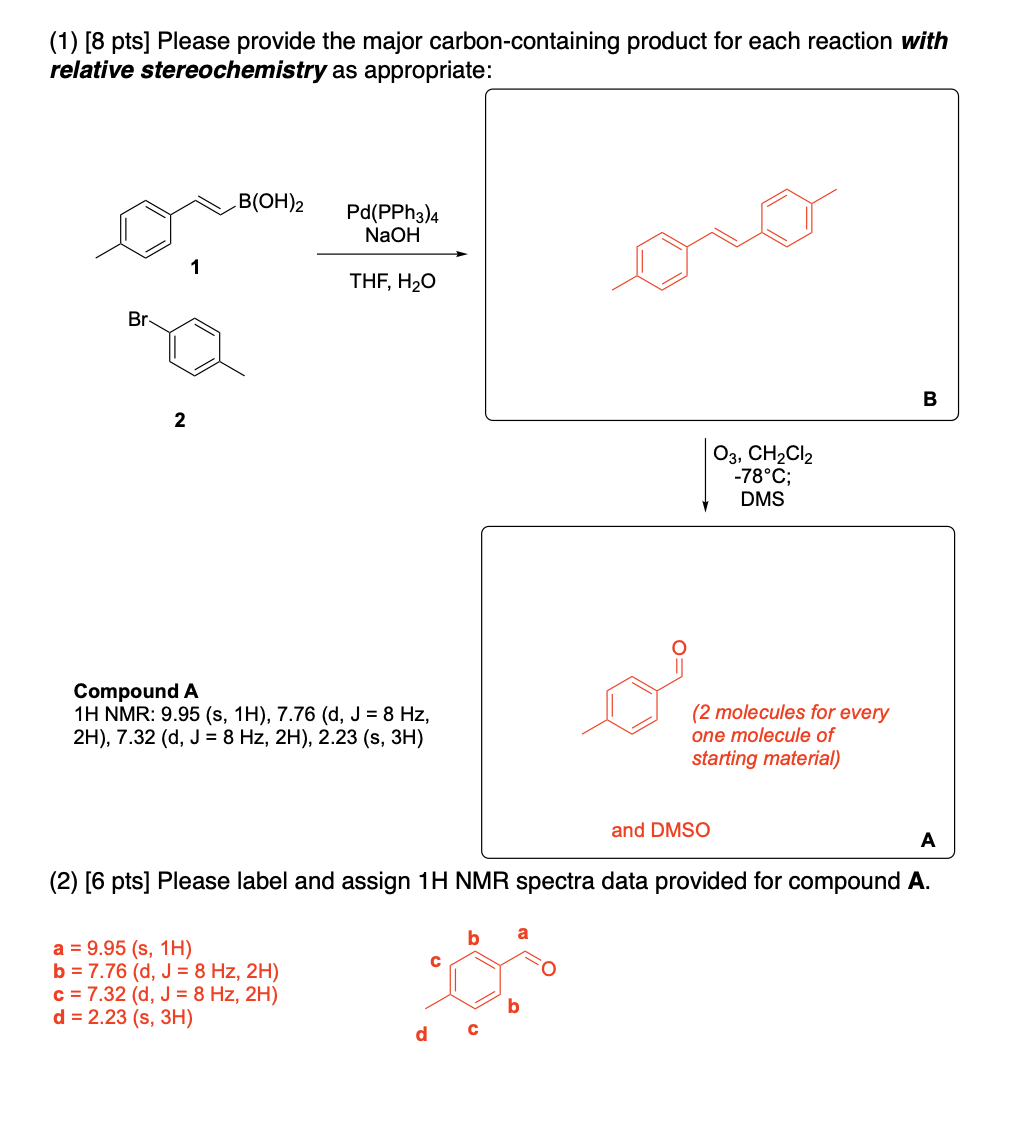
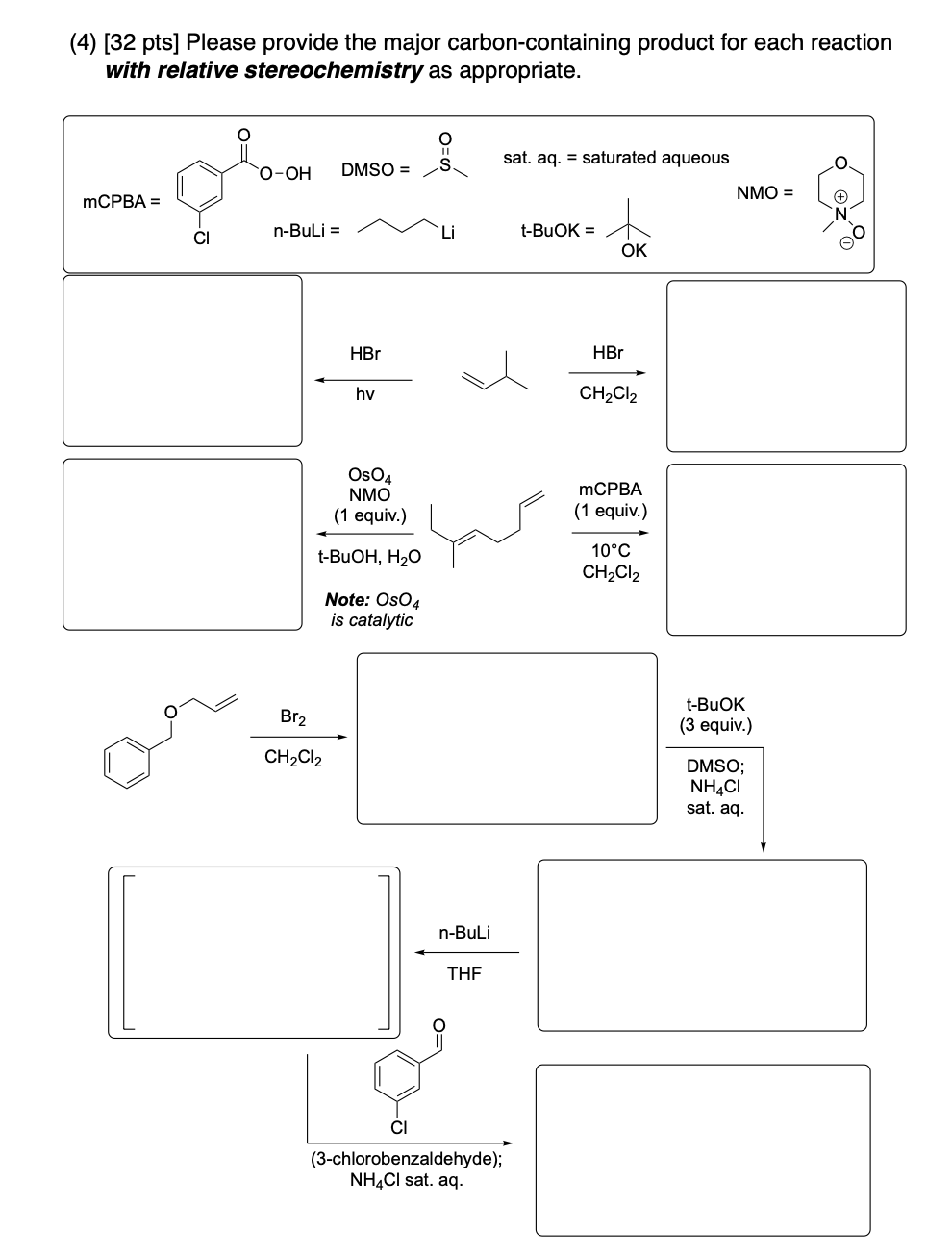
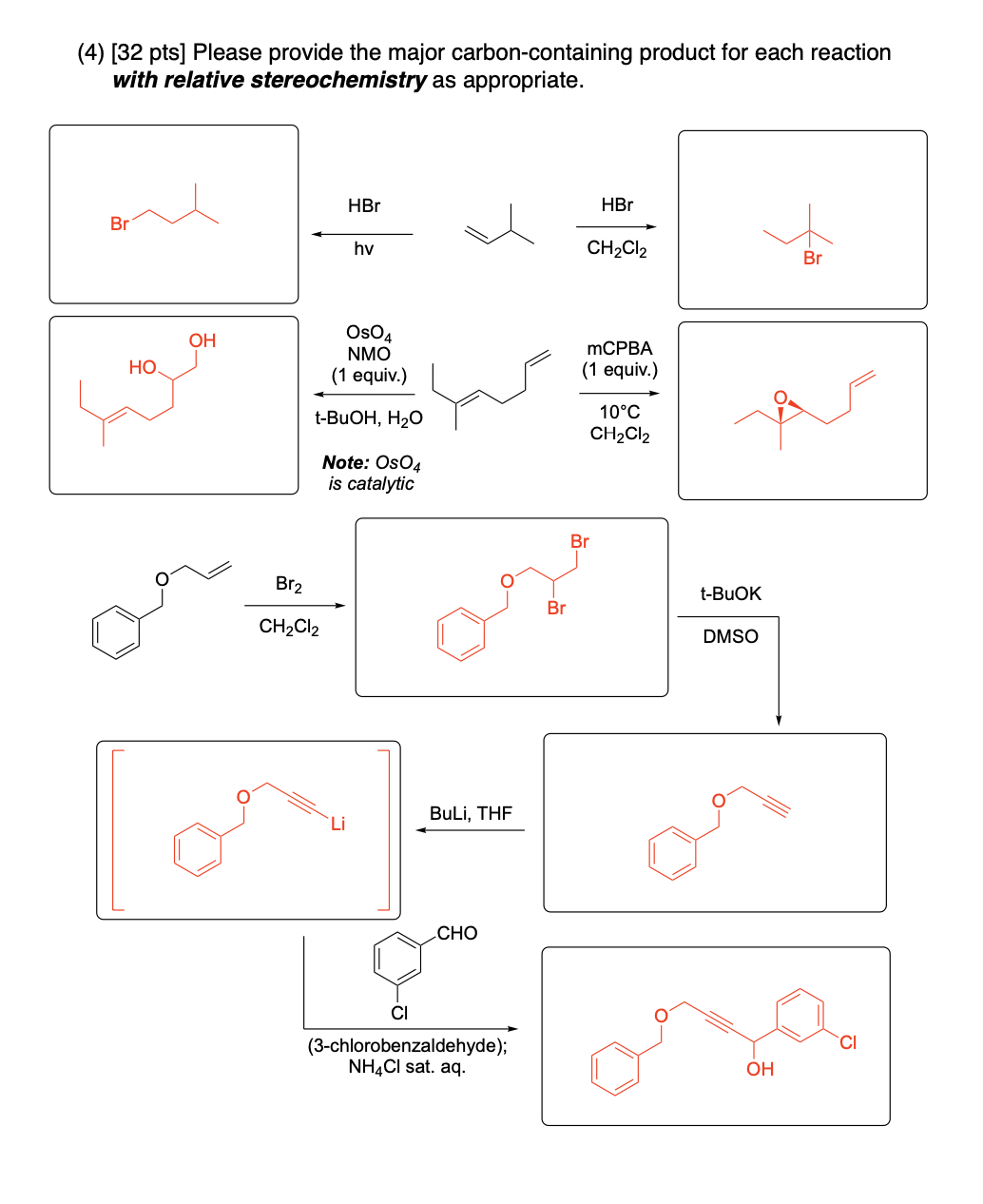
Please provide the major carbon-containing product for each reaction with relative stereochemistry as appropriate.
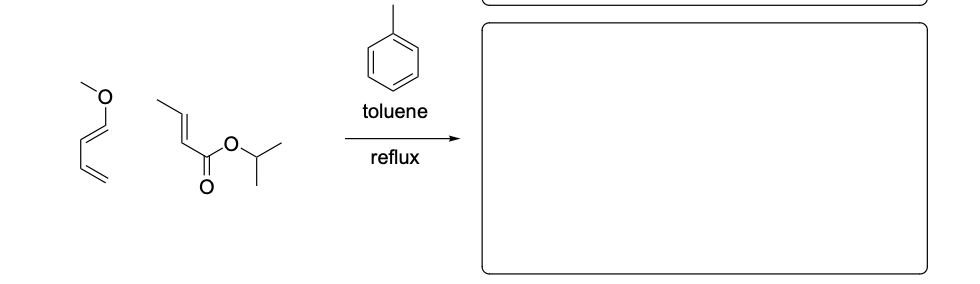
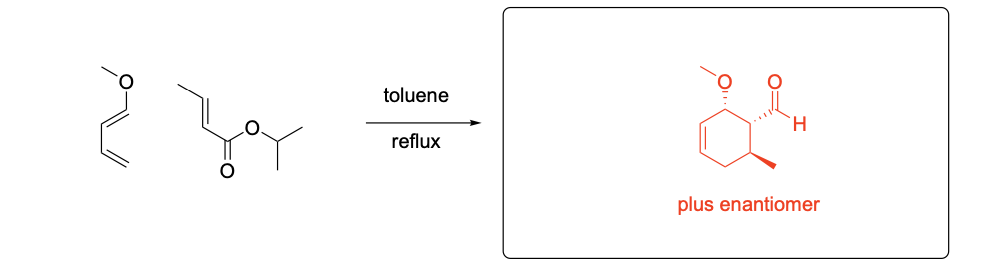
Please provide the major carbon-containing product for each reaction with relative stereochemistry as appropriate.
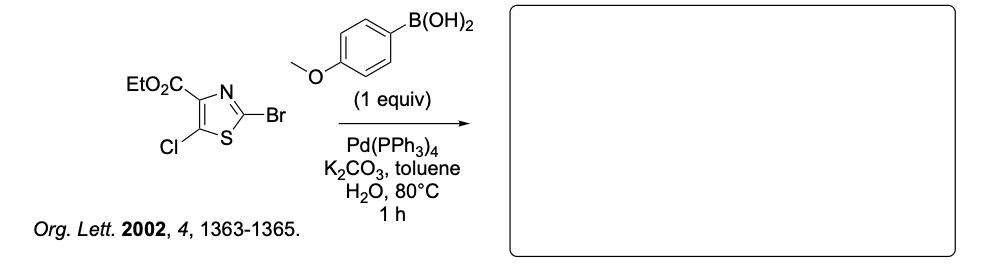
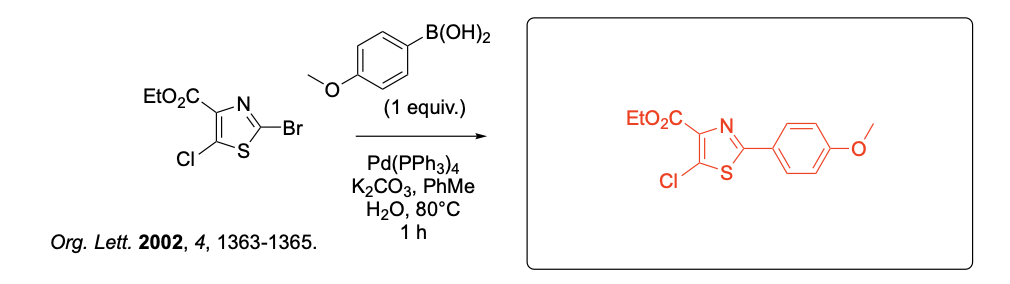
Please provide the major carbon-containing product for each reaction with relative stereochemistry as appropriate.
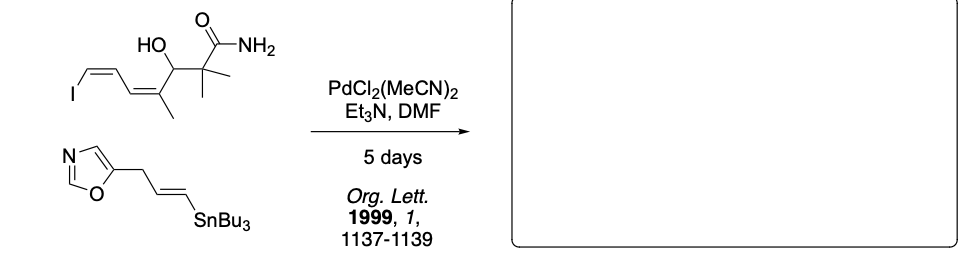
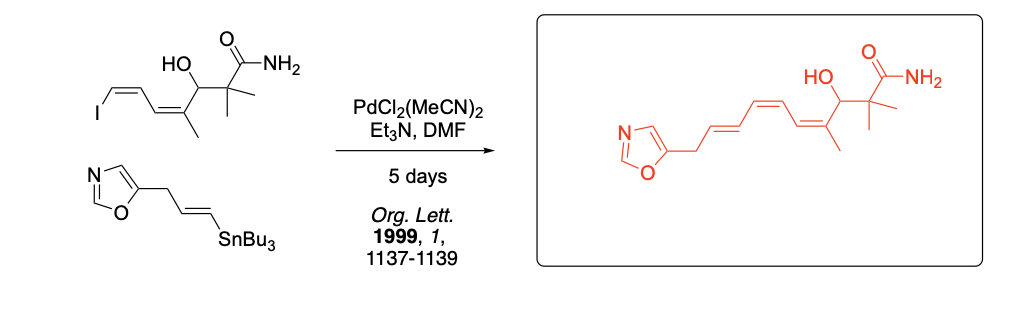
Please provide the major carbon-containing product for each reaction with relative stereochemistry as appropriate.
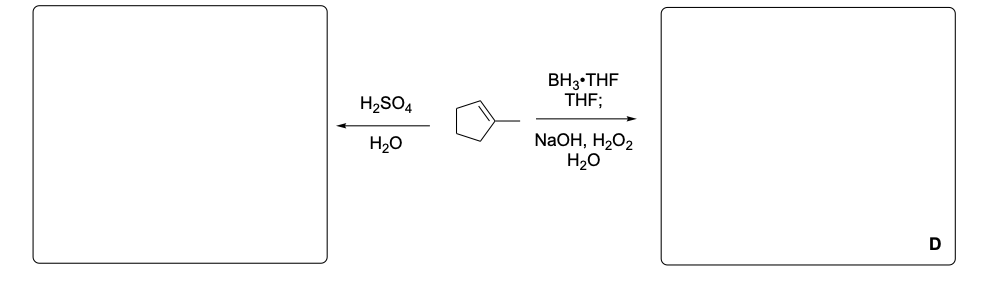
Please provide the major carbon-containing product for each reaction with relative stereochemistry as appropriate.
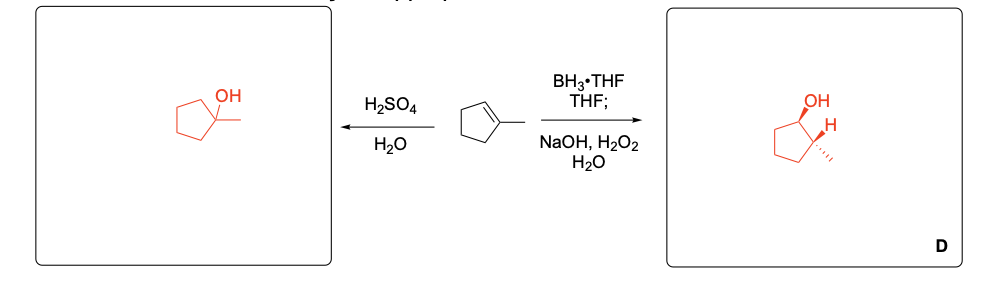
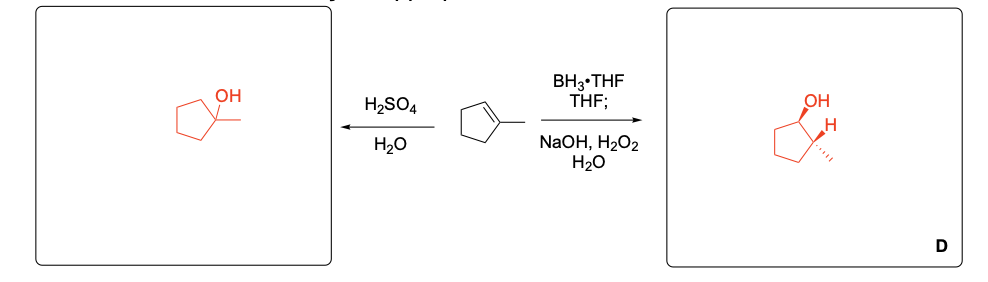
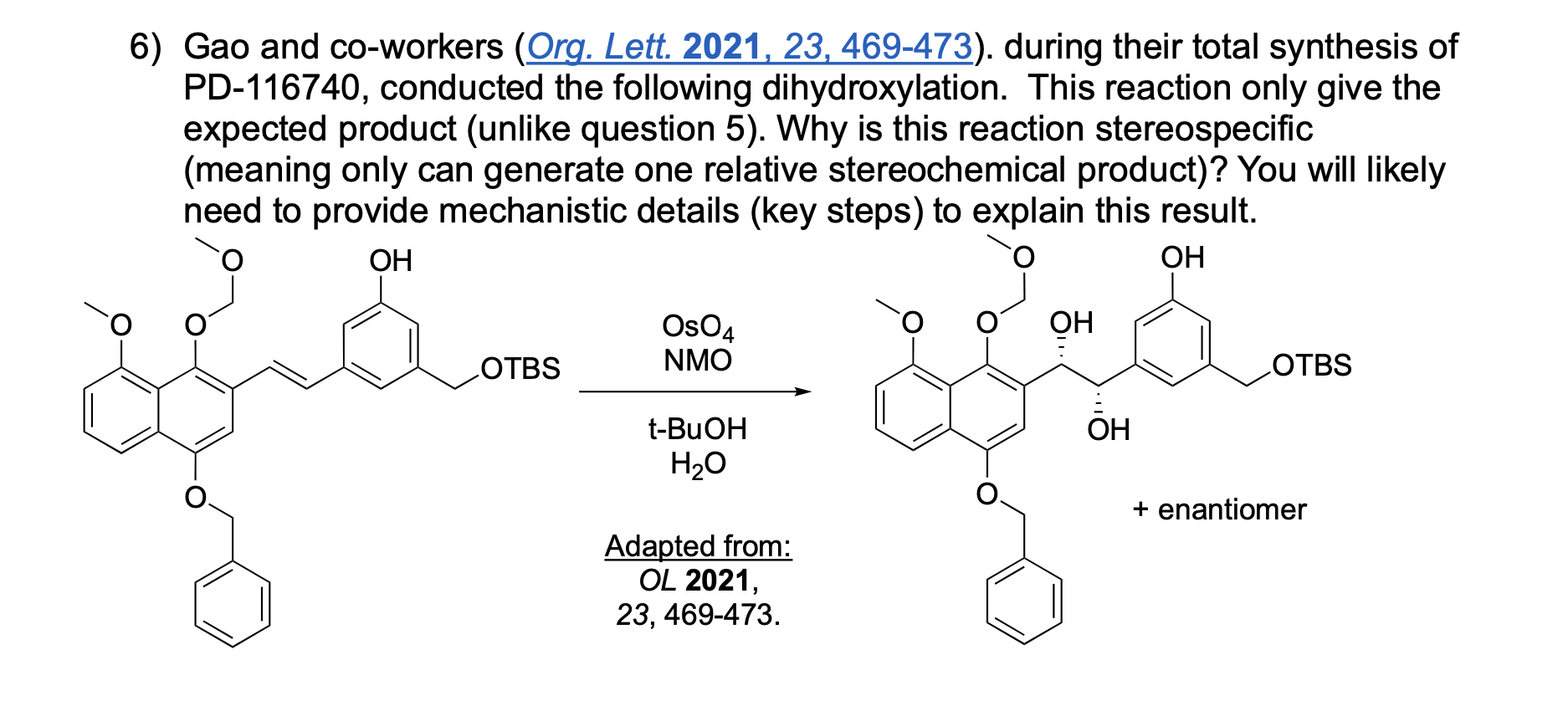
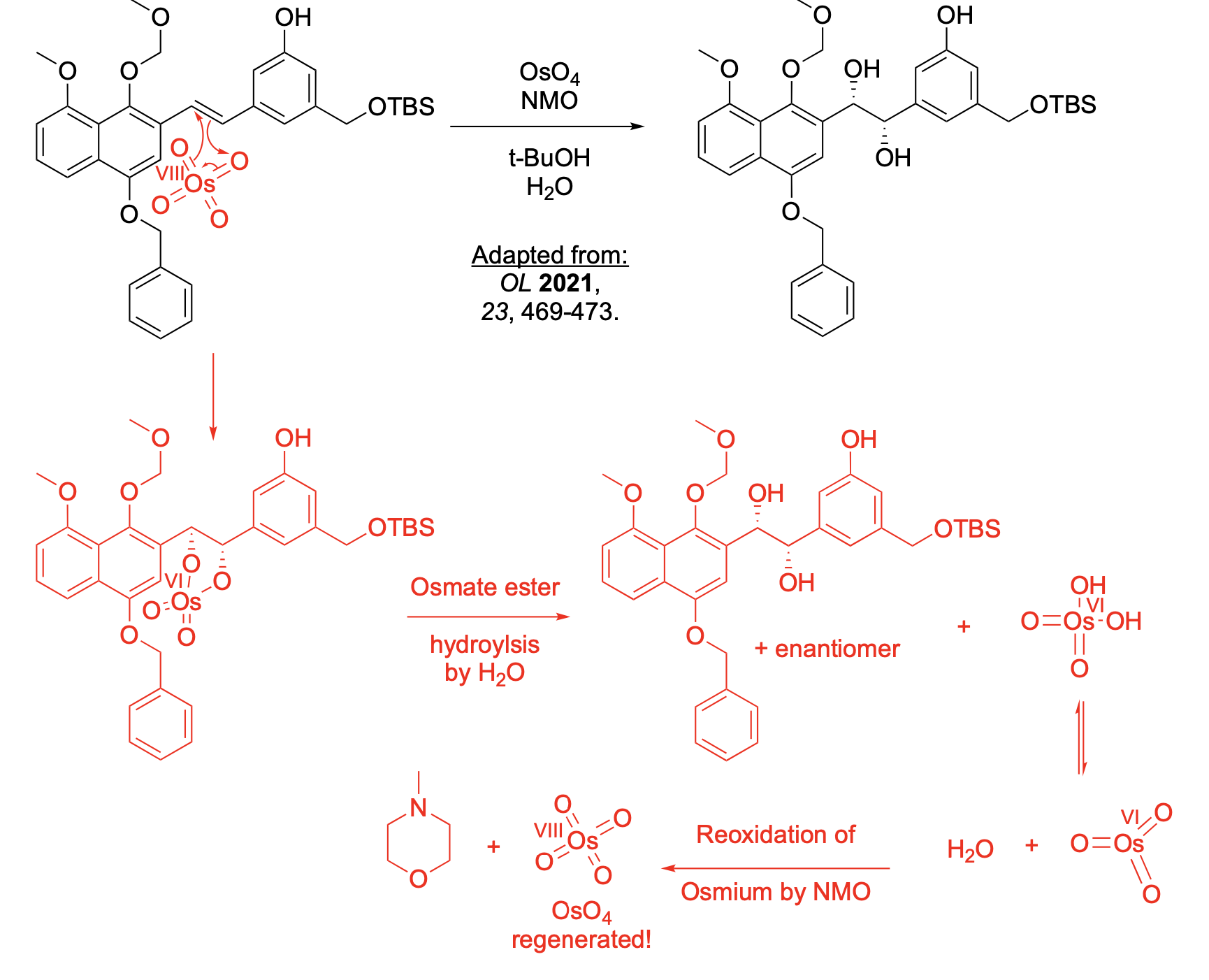
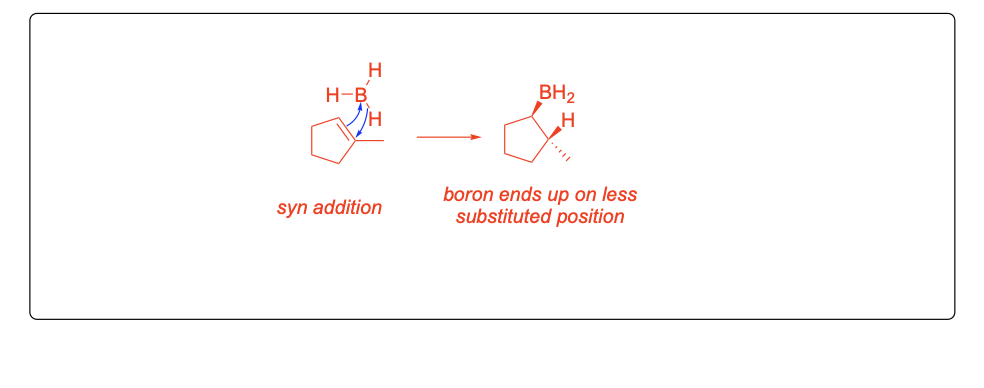
Please explain the observed stereochemistry in compound D. You DO NOT need to provide a complete mechanism – only the initial first step of the reaction to explain the stereochemical outcome.
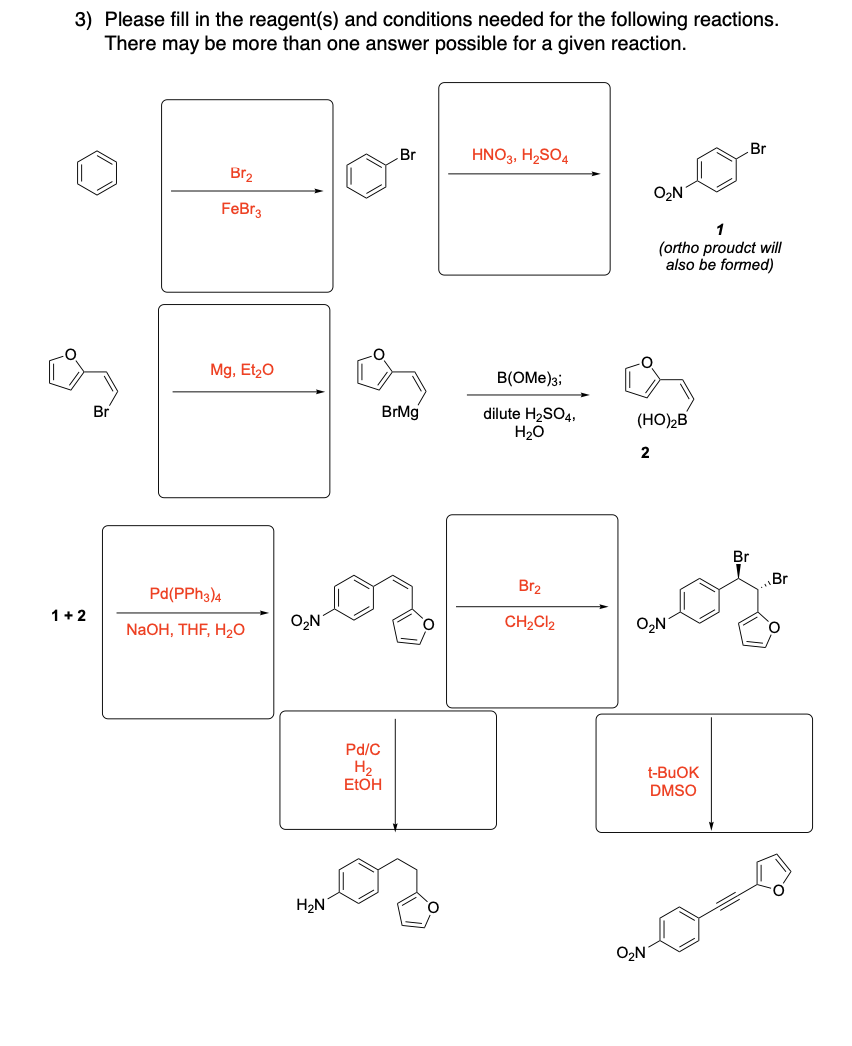
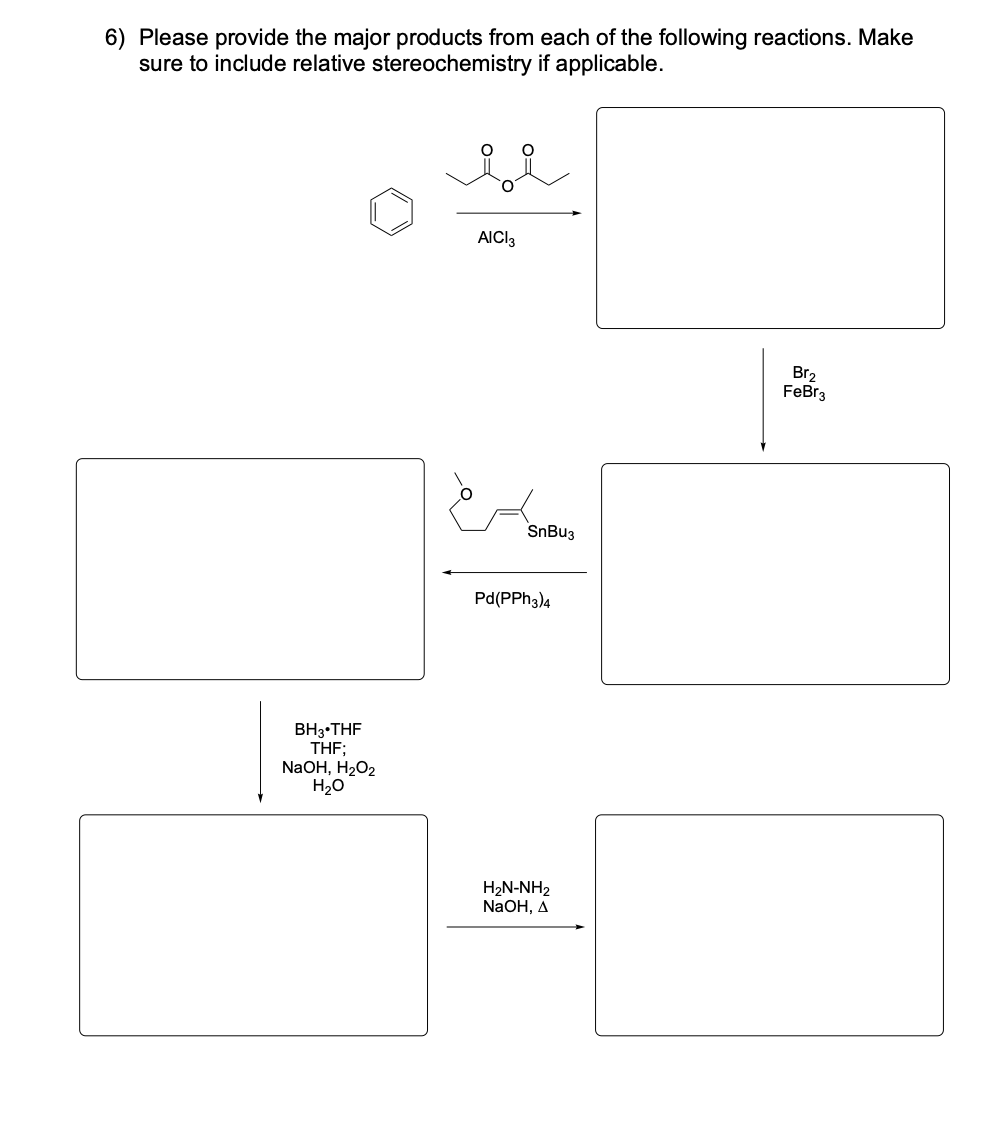
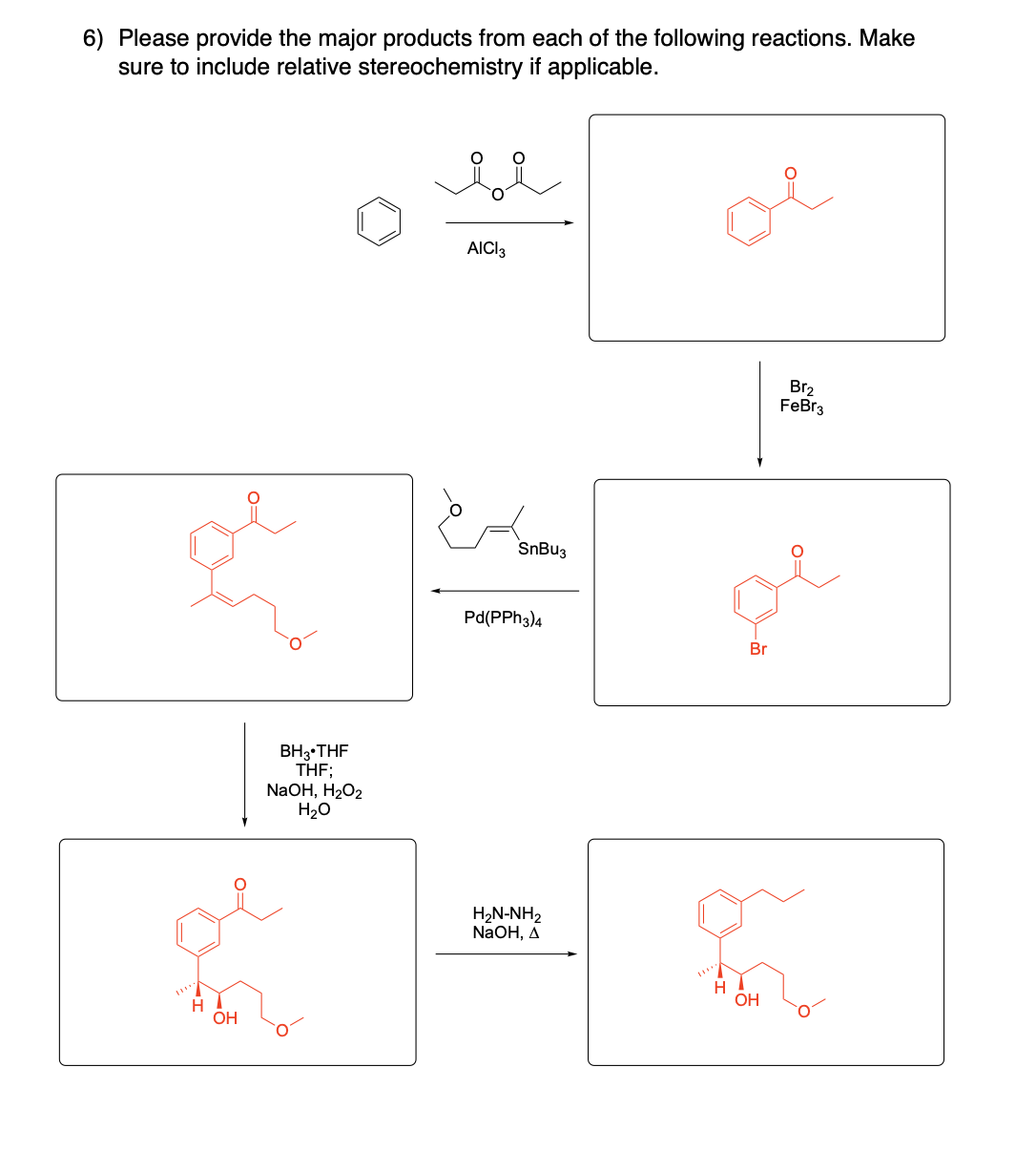
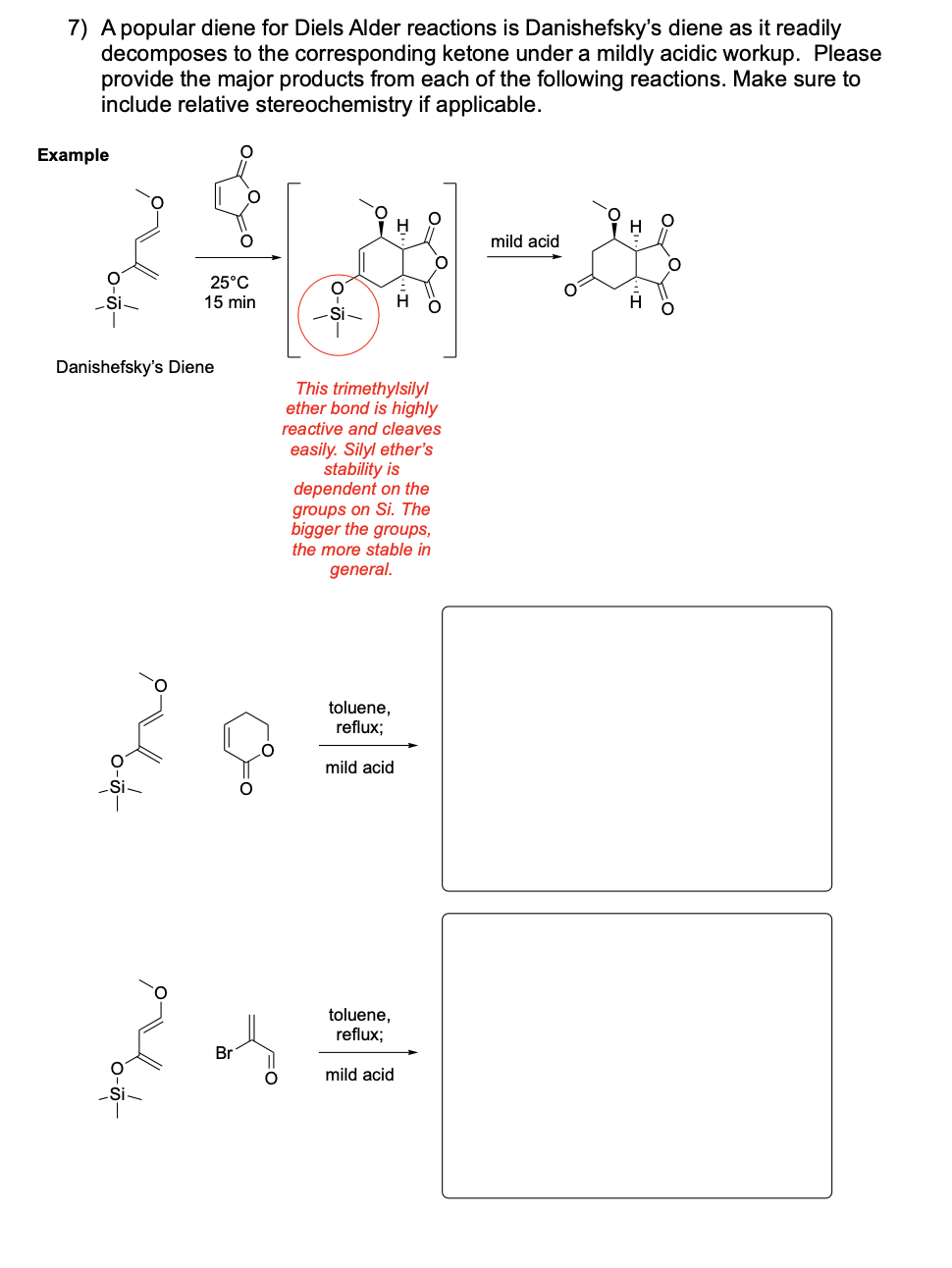
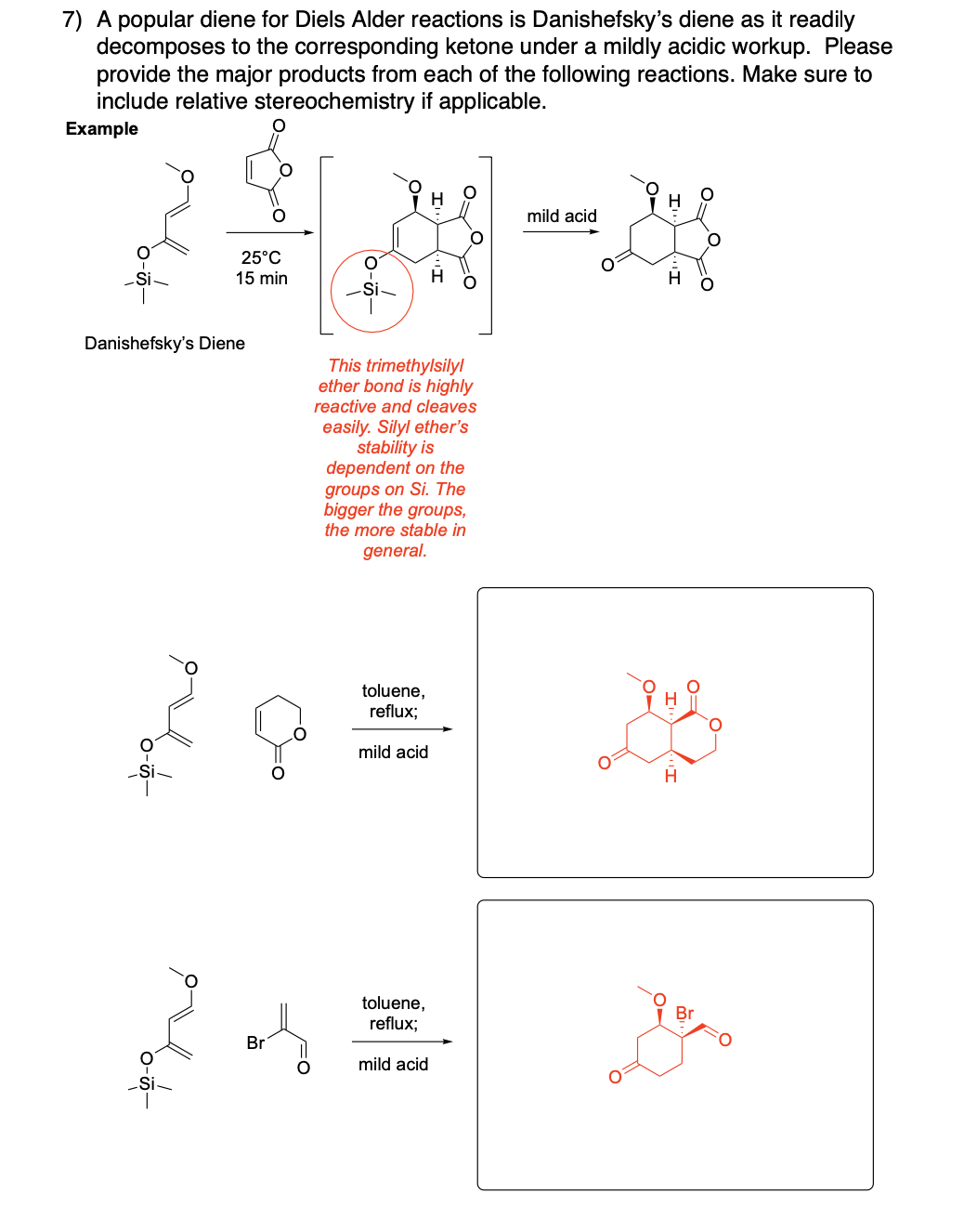
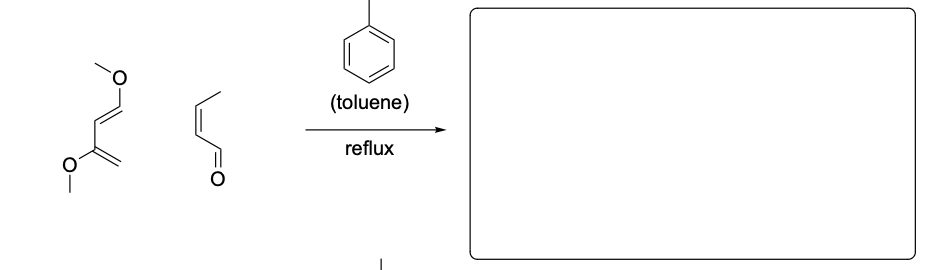
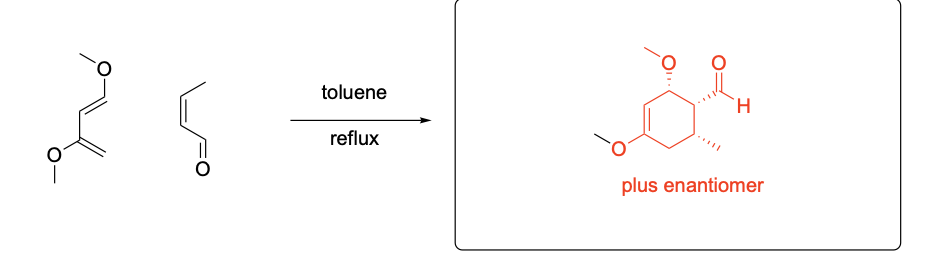
Please provide the major products from each of the following reactions. Make
sure to include relative stereochemistry if applicable.
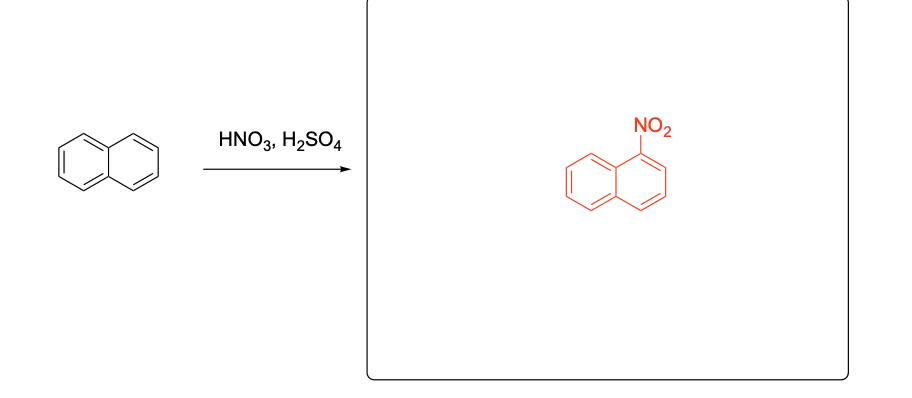
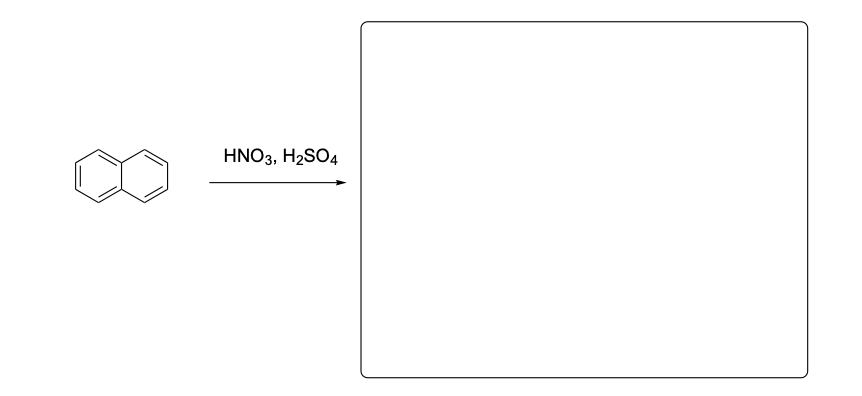
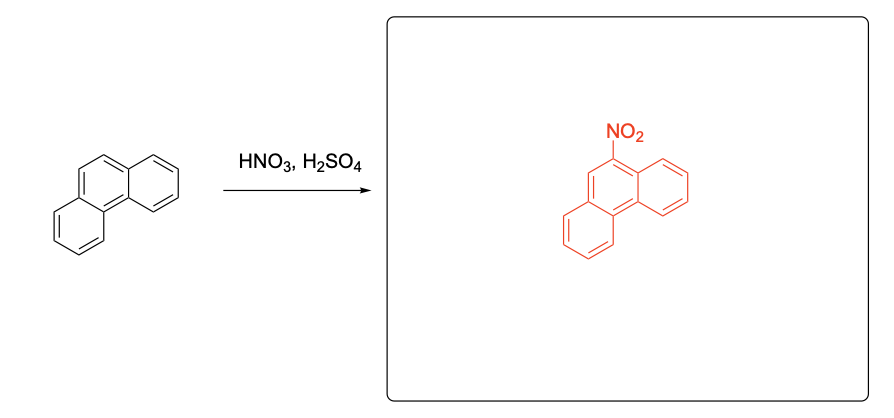
Please provide the major products from each of the following reactions. Make
sure to include relative stereochemistry if applicable.
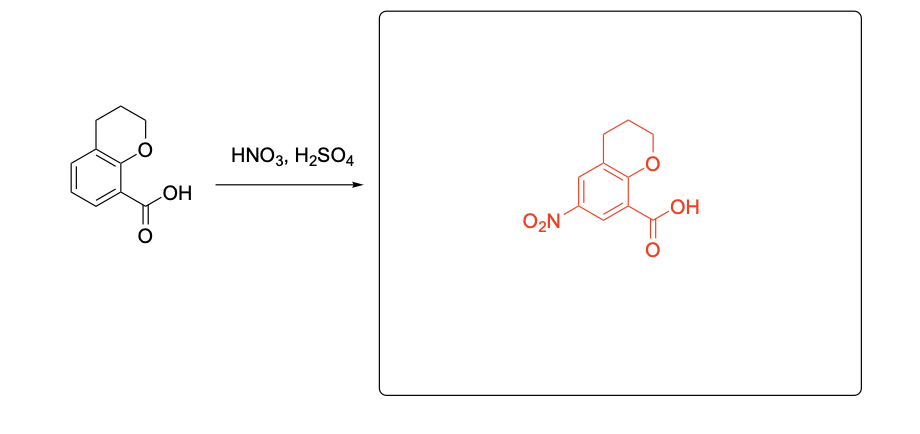
Please provide the major products from each of the following reactions. Make
sure to include relative stereochemistry if applicable.
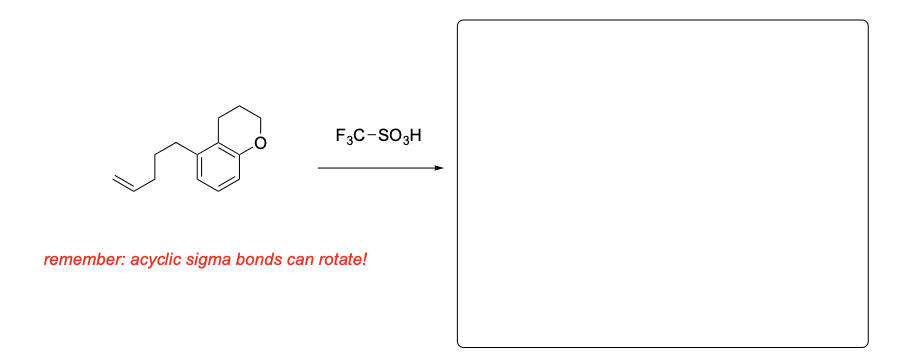
Please provide the major products from each of the following reactions. Make sure to include relative stereochemistry if applicable.
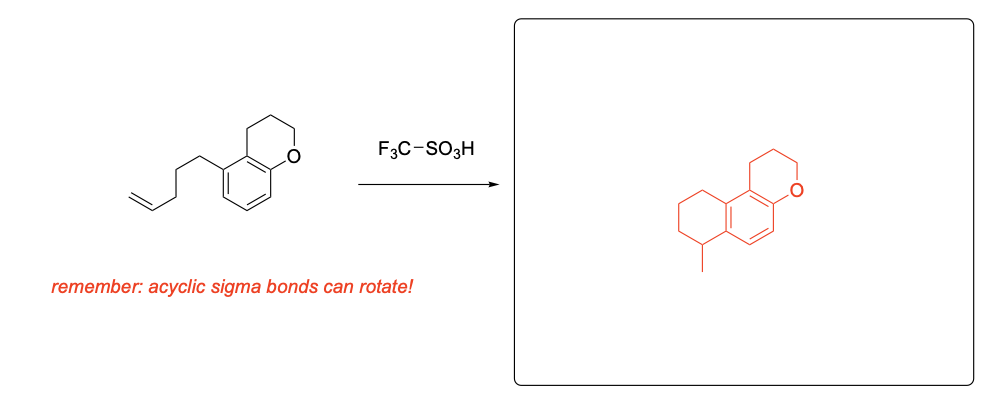
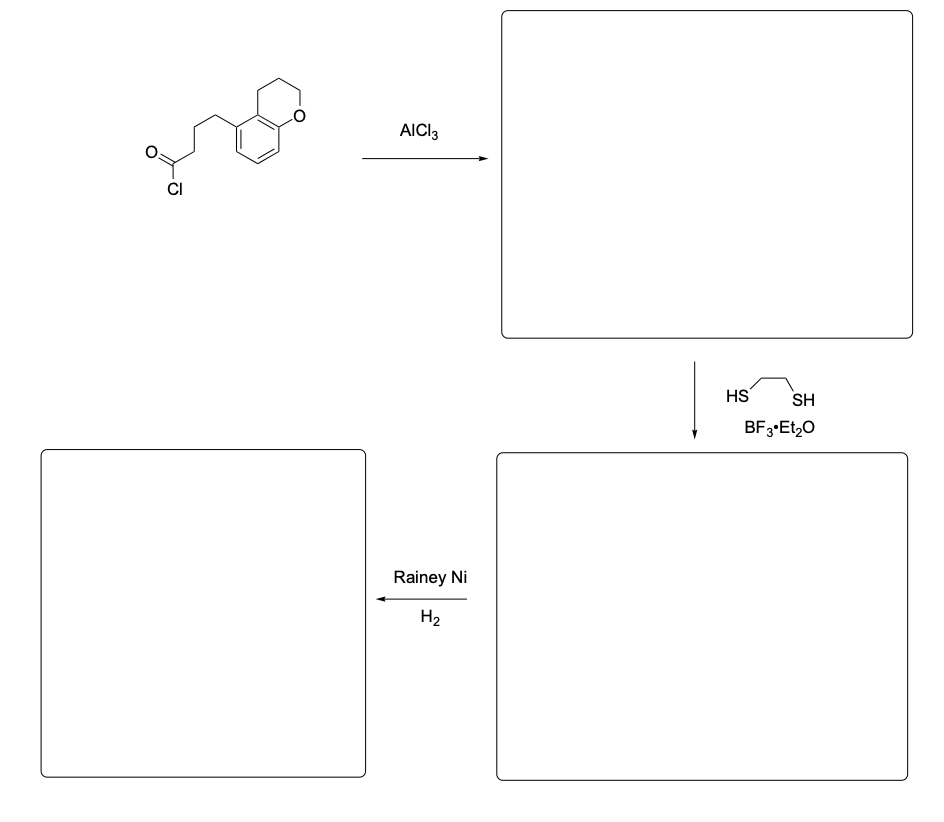
Please provide the major products from each of the following reactions. Make sure to include relative stereochemistry if applicable.
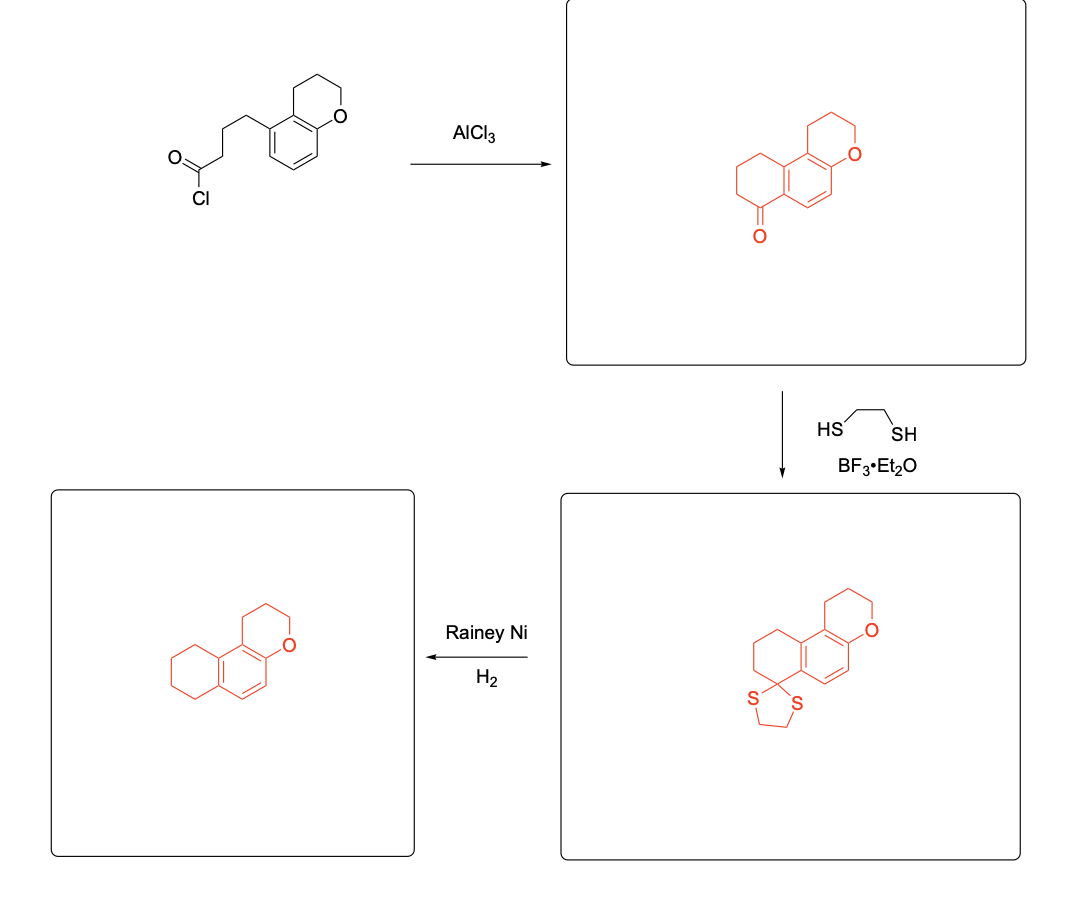
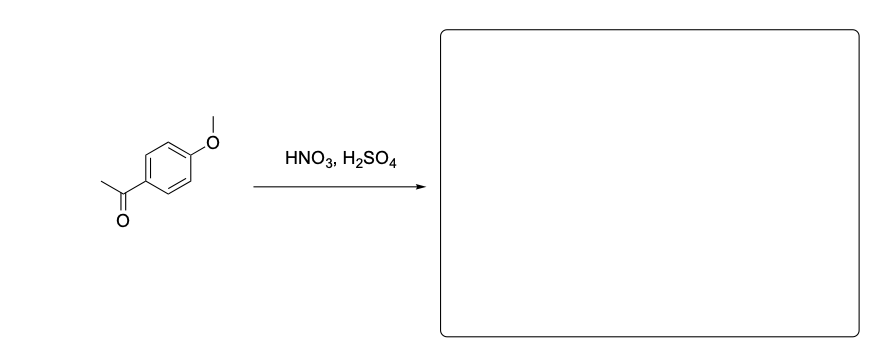
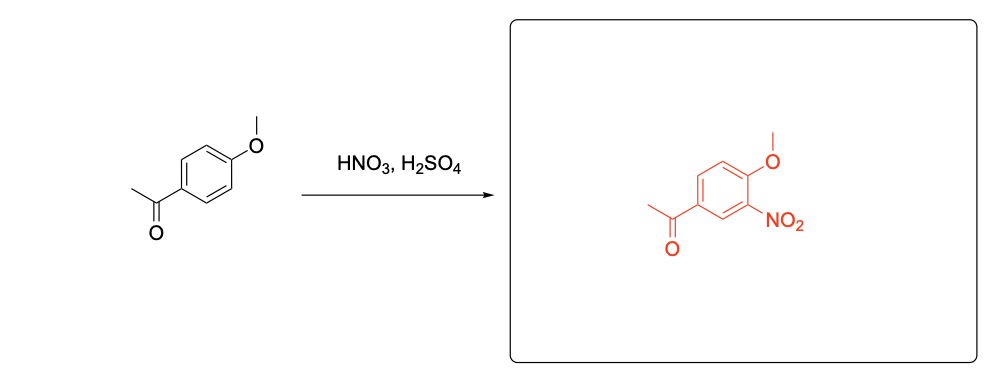
Please provide the major product from the following reaction. Make sure to include relative stereochemistry if applicable.
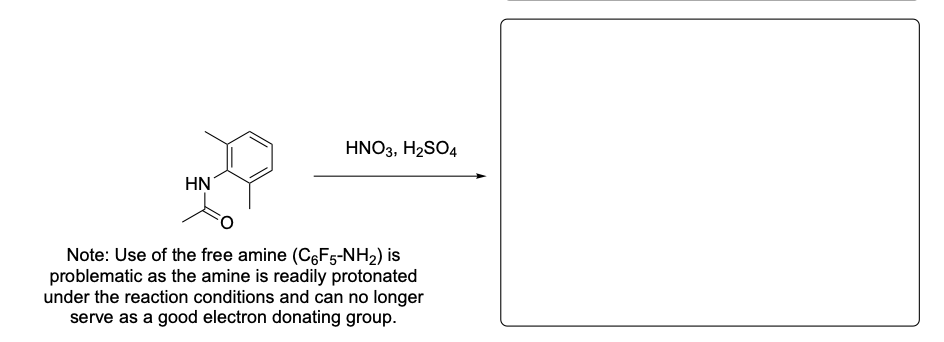
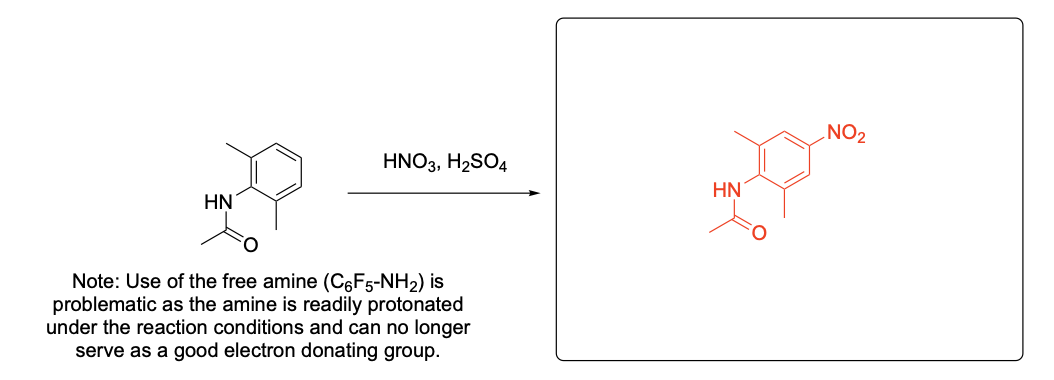
Please provide the major product from the following reaction. Make sure to include relative stereochemistry if applicable.
Please provide the major product from the following reaction. Make sure to include relative stereochemistry if applicable.