Full Chemistry Paper 1
1/523
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
524 Terms
Charge of a proton
+1
Where is a proton found in an atom?
In the nucleus of an atom
What is the relative mass of a proton?
1
What is a neutron?
A subatomic particle with no charge
Relative charge of a neutron
0 (neutral)
Where is a neutron found?
In the nucleus of an atom
What is the relative mass of a neutron?
1
What is an electron?
A subatomic particle that has a negative charge
What is the relative mass of an electron?
1/1836
Whole atomic structure
a positively charged nucleus, consisting of protons and neutrons, surrounded by negatively charged electrons in shells, with the nuclear radius much smaller than that of the atom and with almost all of the mass in the nucleus
why are atoms electrically neutral
as they have the same number of electrons as protons
The negative charge of an electron exactly cancels out the positive charge of a proton
describe the dalton model of the atom
dalton thought atoms were solid spheres that made up all substances, with no internal structure - this model came before the discovery of electrons, protons, or neutrons
J.J. Thompson's model of the atom
the model made by JJ. Thompson, the atom was thought to consist of negatively charged electrons (the ‘plums’) in a positively charged ‘dough’ or ‘pudding’ - the atom was thought to be overall neutral as the negative electrons balanced out the positive ‘dough’
who discovered the electron and how
J.J Thompson discovered the electron by using a cathode-ray tube - he conducted an experiment which identified the electron as a negatively charged subatomic particle, hence proving that atoms are divisible
What was Ernest Rutherford's Gold Foil Experiment and what did it lead hum to discover
rutherford directed a beam of positively charged alpha particles at a thin gold foil and around the foil, he placed a fluorescent screen to detect where the alpha particles went after hitting the foil. most particles went straight through, some deflected and few reflected which led him to hypothesise that most of an atom’s mass is concentrated in a region of space at the centre of the atom called the nucleus
What are alpha particles?
2 protons and 2 neutrons; a helium nucleus with a 2+ charge
James Chadwick's atomic discovery
Discovered the neutron in 1932
Bohr Model
electrons orbit the nucleus in fixed shells or orbitals located at set distances from the nucleus
Each orbital has a different energy associated with it, with the higher energy orbitals being located further away from the nucleus
timeline of changing models of the atom
dalton model
plum pudding model
nuclear model
bohr model
Radius of atom
10⁻¹⁰m
Radius of atom's nucleus
10-¹⁴ m
Isotopes
Atoms of the same element that have the same number of protons but a different numbers of neutrons
mass number
the total number/relative mass of protons and neutrons in a nucleus.
atomic number
the number of protons in the nucleus of an atom, unique to each element
Why might the number of electrons be misleading to the atomic number?
Electrons can be gained/lost via ionic bonding, but protons stay the same.
Why might the relative atomic mass of elements in the periodic table be displayed as a decimal?
it depends on the mass number and abundance of isotopes
how to calculate number of neutrons in an atom
mass number - atomic number
How to calculate relative atomic mass
(isotopic mass 1 x % of 1) + (isotopic mass 2 x % of 2) / 100
similarities of isotopes
- same number of protons
- same number of electrons
- same chemical properties
Differences in Isotopes
- Different masses
- Different number of neutrons
- Nuclear stability may vary
- physical properties
electronic configuration
the arrangement of electrons in an atom
Why can't chemical reactions be used to create different elements?
chemical reactions don't affect atom nuclei.
Which subatomic particles take part in chemical reactions?
electrons
mass number vs relative atomic mass
mass number is a whole number as there is a whole number amount of protons and neutrons, however RAM can be a decimal as it is an average of the relative mass and abundance of isotopes
The vertical columns on the periodic table are called
groups
The horizontal rows on the periodic table are called
periods
Electronic configuration of first 3 shells
1st shell - 2
2nd shell - 8
3rd shell - 8
What do elements in the same group have in common?
same number of outer shell electrons and similar chemical properties
What do elements in the same period have in common?
same number of energy shells
How did Mendeleev organize his periodic table?
he organised the elements into vertical columns based on their properties and the properties of their compounds. he then started to arrange them horizontally in order of increasing mass number and as he worked, he found that a pattern began to appear in which chemically similar elements fell naturally into the same columns
how did mendeleev predict the existence of undiscovered elements
He used the properties and trends of other elements in the group with the gap to predict the properties of these undiscovered elements
differences in modern periodic table and mendeleevs table
mendeleev organised elements by increasing mass number, modern is by increasing atomic number
mendeleev left gaps for undiscovered elements
Ionic bonding occurs between...
metals and non-metals
metals tend to form ------- when ionically bonding
cations (positive ions)
this means they have lost electrons
non-metals tend to form ------- when ionically bonding
anions (negative ions)
this means they have gained electrons
Why do electrons transfer in ionic bonding?
To create a full outer shell of both elements
dot and cross diagrams
Used to model the electrons in chemical bonding.
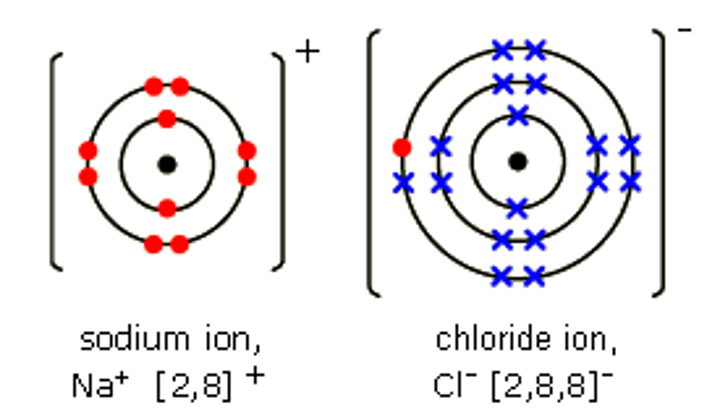
How is the transfer of electrons shown in dot and cross diagrams?
Arrows are used to show the transfer of electrons from metal to non-metal.
What notation is used to indicate the charge of atoms in dot and cross diagrams after electron transfer?
Enclosing the atom diagrams in square brackets with the charge at the top.
Ion
electrically charged atom or group of atoms formed by the loss or gain of electrons
If the electron number of a 2+ ion is 68, what is the atomic number?
70
Ionic structure
Giant ionic lattice, consisting of a regular arrangement of ions, held together by the strong electrostatic forces of attraction between oppositely charged ions
How do non metals in an ionic compound differ in their naming
they have an -ide ending, for example, silicide, fluoride, chloride
Compound ions with oxygen end in..
-ate
Sulfate charge
SO₄²⁻
Carbonate charge
CO₃²⁻
Nitrate charge
NO₃⁻
Ammonium charge
NH₄⁺
hydroxide charge
OH⁻
Silver charge
Ag⁺
Zinc charge
Zn²⁺
lead charge
Pb²⁺
Copper charge
Cu²⁺(usually)
Group 1 charge on ion
1⁺
Group 2 charge on ion
2⁺
Group 3 charge on ion
3⁺
Group 5 charge on ion
3⁻
Group 6 charge on ion
2⁻
Group 4 charge on ion
4⁺/4⁻
Group 7 charge on ion
1⁻
Where does covalent bonding occur?
between 2 non-metals
Molecule
two or more atoms held together by covalent bonds
how does a covalent bond form
Non-metal atoms can share electrons with other non-metal atoms to obtain a full outer shell of electrons. When atoms share pairs of electrons, they form covalent bonds
diatomic molecule
A molecule consisting of two atoms
how to remember diatomic molecules
BrINClHOF
bromine, iodine, nitrogen, chlorine, hydrogen, oxygen, flourine
What holds covalent bonds together?
electrostatic attraction between the positively charged nuclei of two atoms and the negatively charged shared pair(s) of electrons located between them
What is a correlation between the number of shared pairs of electrons and the ionic charge of the element?
they are equal
For example nitrogen has a 3- charge, and shares 3 pairs of electrons
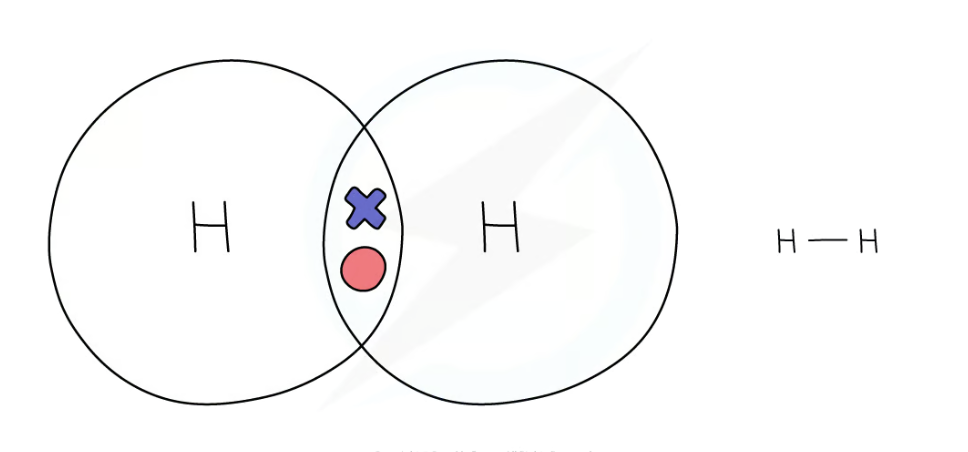
dot and cross diagram for hydrogen
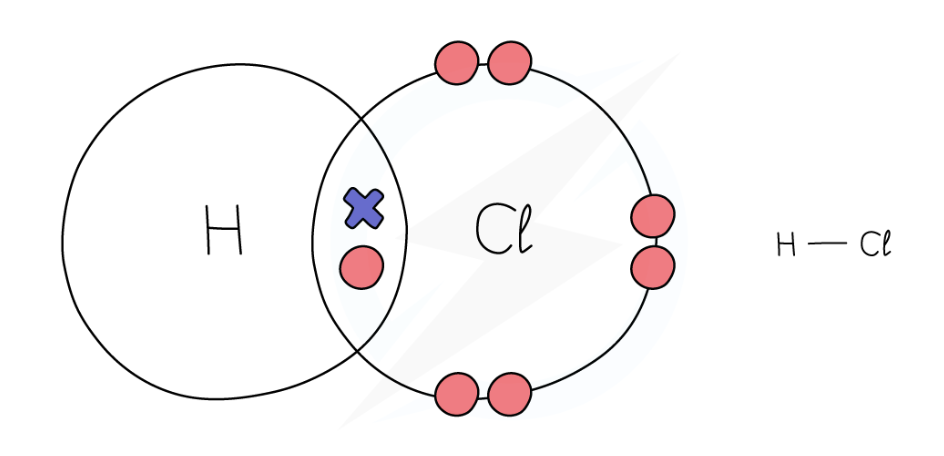
dot and cross diagram for hydrogen chloride
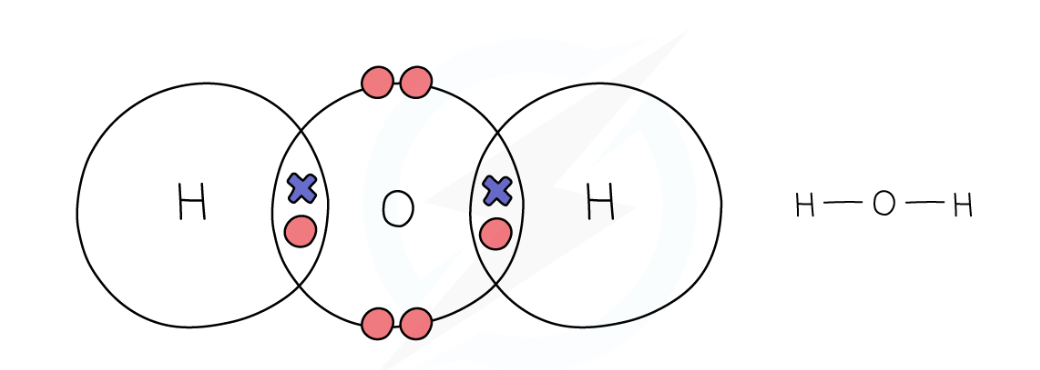
dot and cross diagram for water
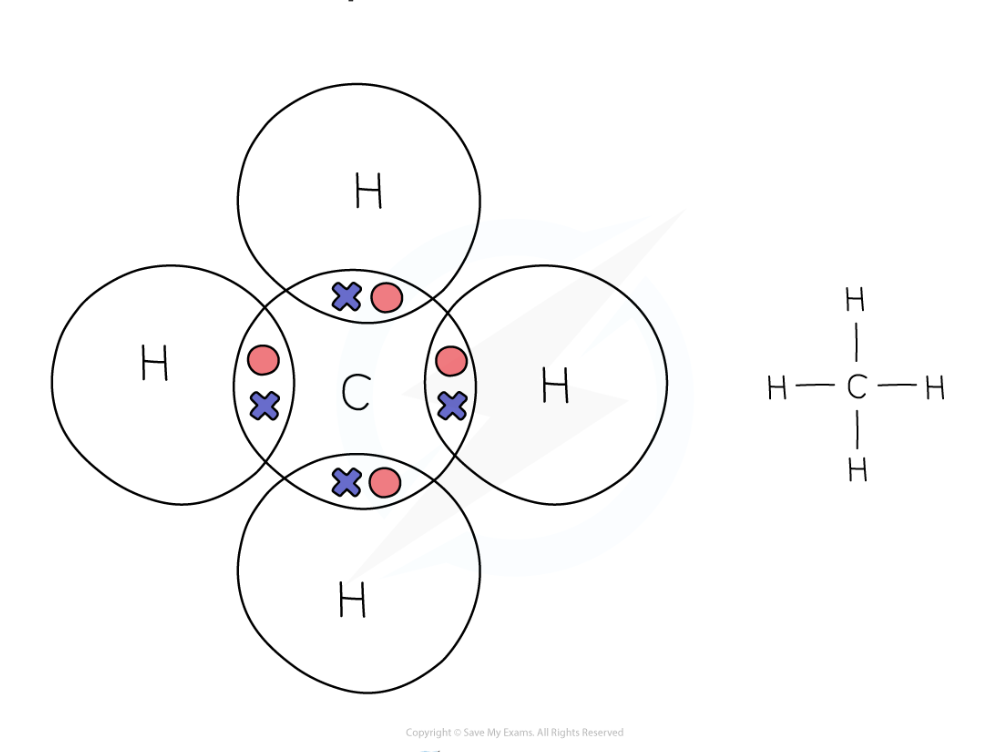
dot and cross diagram for methane
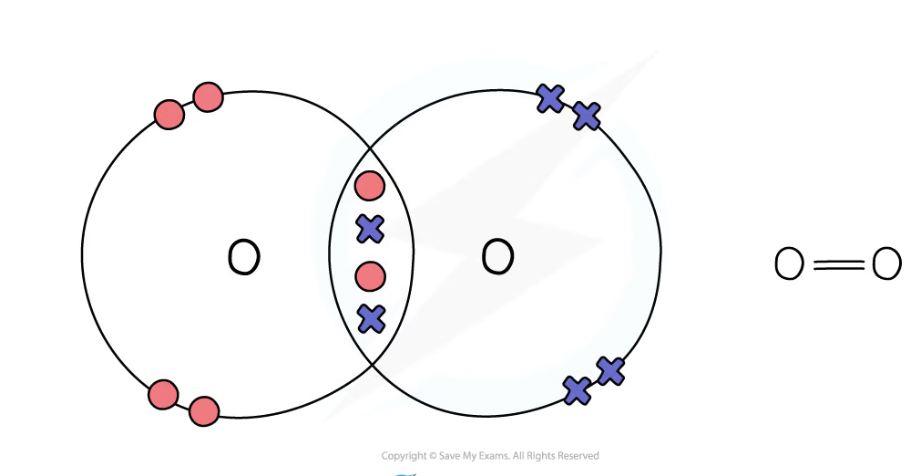
dot and cross diagram for oxygen
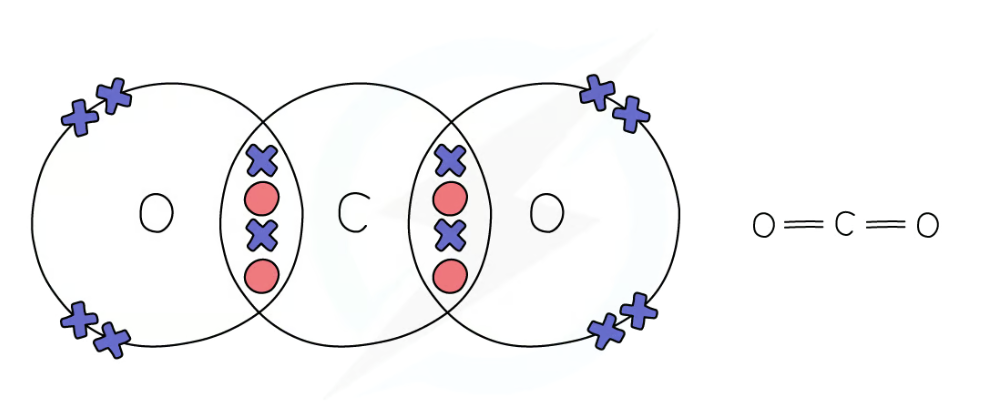
dot and cross diagram for carbon dioxide
properties of ionic compounds
high mps and bps
water soluble
can conduct electricity
why do ionic compounds have high mps and bps
A lot of energy is required to break the strong electrostatic forces of attraction between oppositely charged ions
why are ionic compounds water soluble
due to the attractions between polar water molecules and the ions, which allows the water molecules to pull the ionic lattice apart
why do ionic compounds conduct electricity (when molten)
Ionic compounds can conduct electricity in the molten state or in solution as they have ions that can move and carry charge
properties of covalent substances
low mps and bps so covalent compounds are usually liquids or gases at room temperature
insoluble
cannot conduct electricity
why do covalent substances have low mps and bps
the weak intermolecular forces are easily overcome, meaning that these compounds are usually volatile
why are covalent substances insoluble
they tend to be non-polar but can dissolve in organic solvents
why do covalent substances not conduct electricity
all electrons are involved in bonding so there are no free electrons or ions to carry the charge
Allotropes
different molecular structures of the same element in the same state
allotropes of carbon
diamond, graphite, graphene, buckminsterfullerene
Giant covalent structure
many non-metals atoms bonded to other non-metal atoms via strong covalent bonds
properties of giant covalent substances
high mps and bps
most cannot conduct electricity
why do giant covalent substances have high mps and bps
as they have many strong covalent bonds and large amounts of heat energy are needed to overcome these forces and break down bonds
why do most giant covalent substances not conduct electricity
Most cannot conduct electricity as they do not have free electrons but there are some exceptions such as graphite
structure of metals
Metals consist of metal atoms held together strongly by metallic bonding
Within the metal lattice, the atoms lose their valence electrons and become positively charged
The valence electrons no longer belong to any metal atom and are said to be delocalised, creating what is known as a sea of free electrons
The free electrons move freely in between the positive metal atoms
properties of metals
very strong - high mps and bps
insoluble in water
can conduct
malleable and ductile
why are metals strong and have high mps and bps
as a result of the attraction between the positive metal ions and the negatively charged delocalised electrons. A lot of heat energy is needed to overcome forces and break these bonds