Chapter 10: Oxidation & Reduction
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
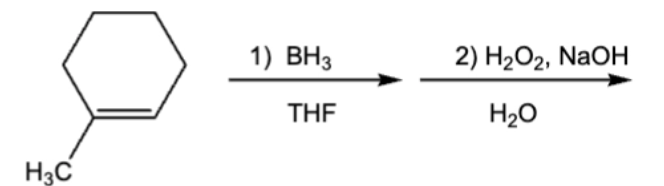
10.1 Anti-Markov,cis/syn, Hydroboration-oxidation (addition of H and OH)
1st step is Markov, but Bh2 is replaced by OH which makes it anti-mark
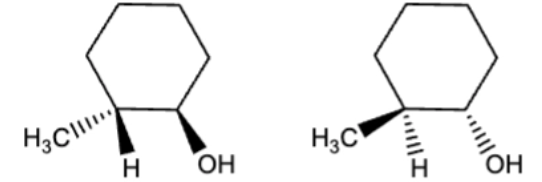
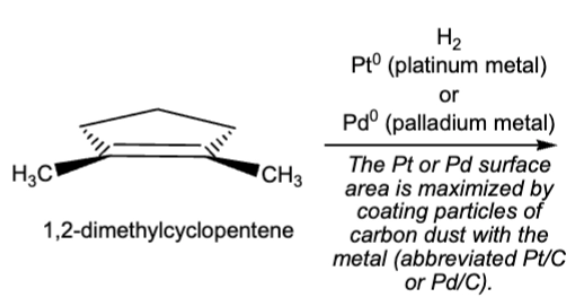
10.2 Catalytic hydrogenation, syn/cis addition of H onto the original double bond carbons
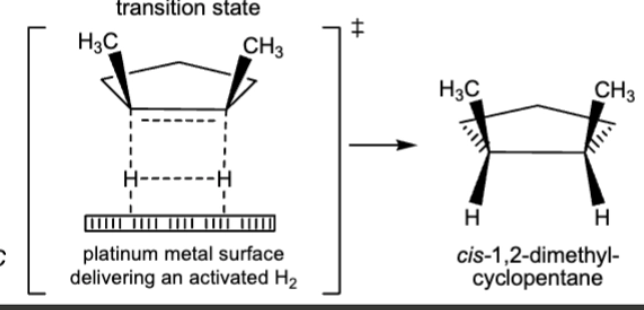
H2 can be D2 (deuterium)
double and or triple bonds become single
always assume excess
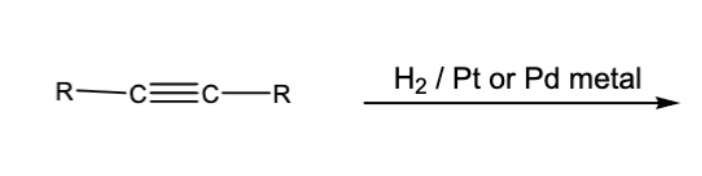
10.3 steroselective alkyne reductions. Triple or double bonds become single
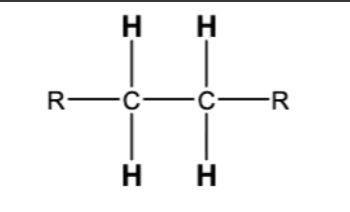
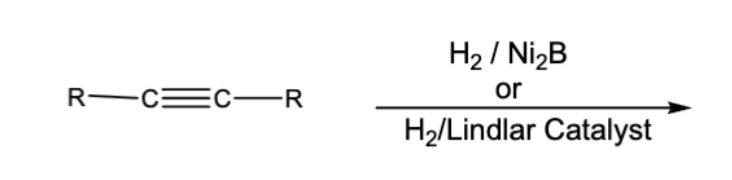
10.4 Steroselective Alkyne Reductions. Triple bond reduces to double, cis/syn addition of H
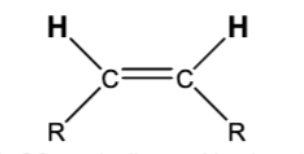
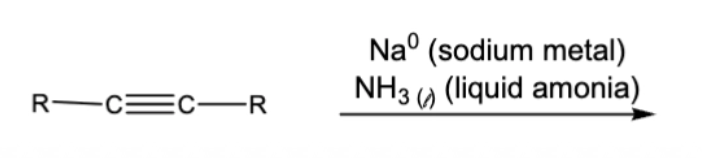
10.5 Steroselective Alkyne Reductions
triple bond becomes double
trans/anti addition of H
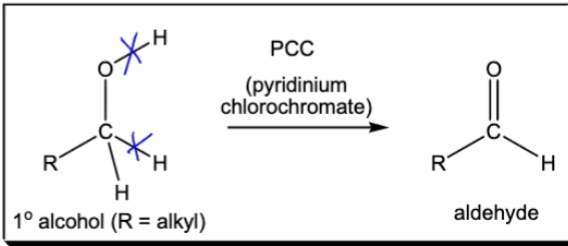
Oxidation of 1º Alcohol into aldehyde. PCC breaks O-H and C-H bonds
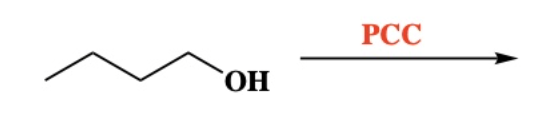
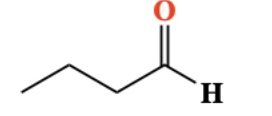
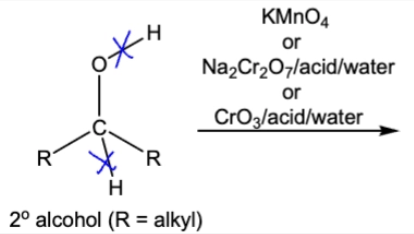
10.7 Oxidation of 2º Alcohol into ketone. breaks O-H and C-H bonds
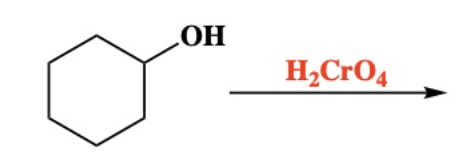
10.7 Oxidation of 2º Alcohol into ketone. breaks O-H and C-H bonds
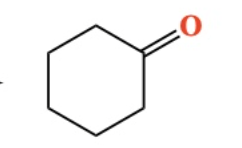
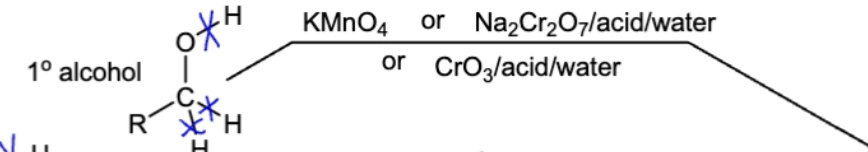
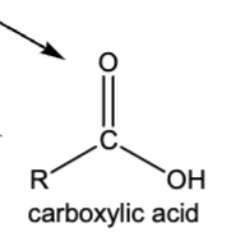
10.8 Oxidation of 1º Alcohol into a carboxylic acid
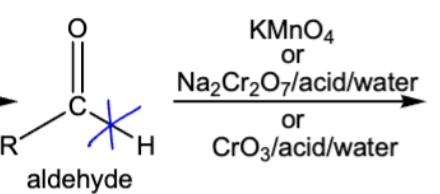
10.8 Oxidation of aldehyde into carboxylic acid

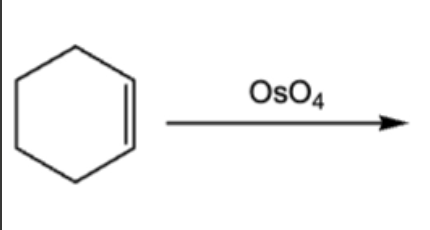
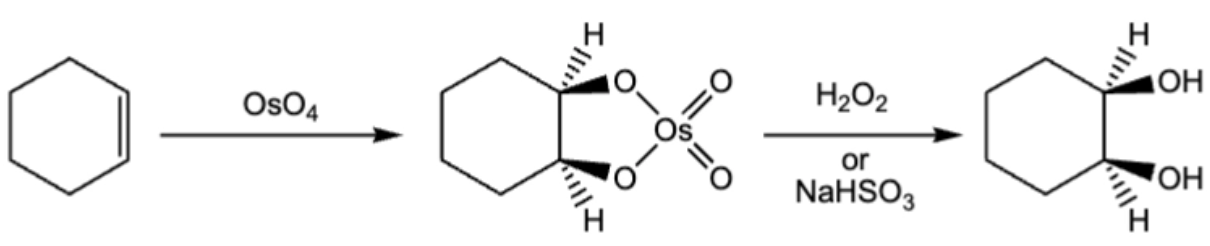
10.9. Syn/cis addition of 2 OH group to a pi bond
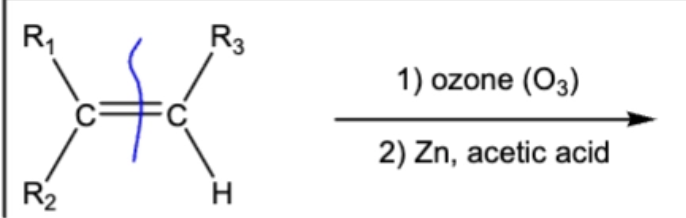
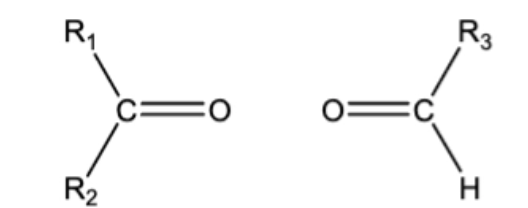
10.10 Ozonolysis. C=C bond breaks, ends have O attached
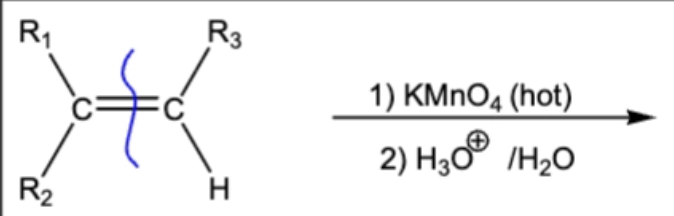
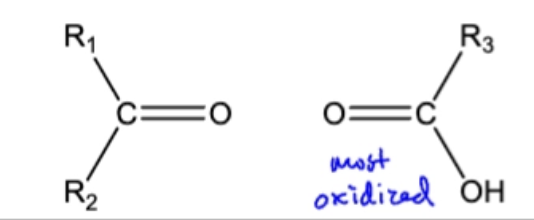
10.11 Permangante Oxidation. C=C bonds broken, O attached, Oxidation of H (H → OH)
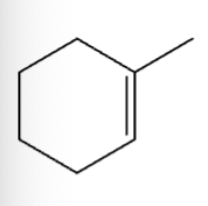
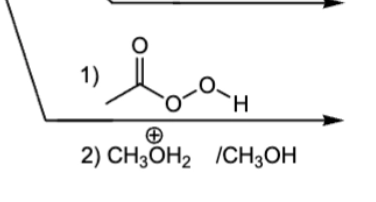
1) MBCPA epoxide ring formation
2) acidic conditions, Markovnikov addition of CH3O and OH


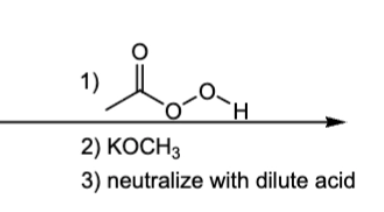
1) MBCPA
2) basic conditions, Anti-Mark addition of OCH3 and H (from dilute acid)
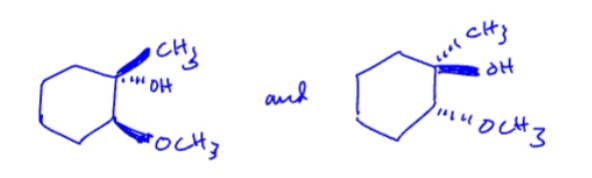
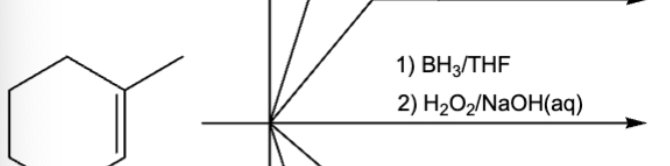
10.1 Anti-Markov,cis/syn, Hydroboration-oxidation (addition of H and OH)
1) addition of BH2 and H
2) BH2 replaced by OH. OH ends up on less substituted C
