Coulomb's law
1/39
Earn XP
Description and Tags
coulombs law, electrons, atomic radius, ionization, electronegativity, ions,
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
Columb’s law
The potential energy of two charged particles depends on their charges ( q1 & q2 ) and on the distance between them
Coulmbs law formula
is given by F = k(q1 * q2) / r²
F stands for
force
Ke stands for
coulombs constant 9×10 to the power of 9 NM squared/ C squared
q1 and q2 stands for
formula charges
r squared stands for
distance between the two charges
1) Alike charges
repel ( + and +, -and - )
2) Opposite charges
attract ( + and - )
3) forces of attraction and/ repulsion
-directly proportional to charge difference
-inversely proptinal to distance between
There are two types of electrons
valence electons and core electrons
valence electrons
e-s occupying the outer energy level
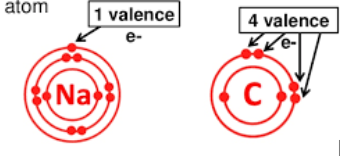
core electrons
all e-s not occupying the outer energy level
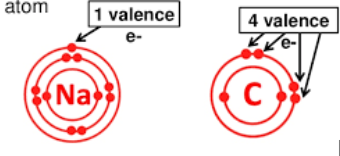
core electrons repel
valence electrons ( outer electrons)
more core electrons cause
-increase replusive forces on valence e-s
-increase shielding of + charge nucleus
The electron on the outer shell ( valence electrons) wont be as strong because
the first orbital of the electrons would repel the one above it
Atomic Raduis
half the distance between two chemically bonded homonucler atoms ( atoms of the same element)
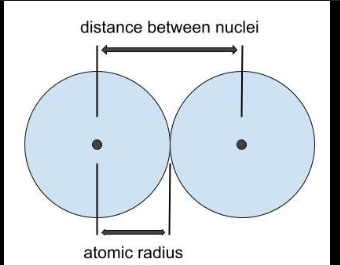
On the PE the atomic radius
-decrease from left to right across a period
-same number of core electrons ( repulsive force stays constant on valence electrons )
If # of protons increases
atrractive forces increase
in the PE increases from top to bottom down a group
-inc. number of electrons ( repulsive force inc. on valence e-s)
-more energy levels
Ionization Energy
-energy needed to remove e-s
-increas fro left to right across a period
-decreases from top to bottom down a group
Ion
atom ( group of atoms that have gained/ lost e-s
Increas from left to right across a period
same number of core e-s (repulsive force stays constant on valence e-)
number of protons increase ( attractive forces increases)
Decreases from top to bottom down a groups
inc. remember of core e-s ( repulsive force inc. )
more energy levels
Electron negativity
atrractiveness to an e- in a bond
increases from left to right across a period
decrease from top to bottom down a group
Every single element in row
18 has 0 electronegativity
Ions
an atom that has an electric charge
ions can not lose of
gain protons
ions valence e-s can be
lost or gained Ca
Cations ( Cat-ion)
lost e-
metals from cations
postively charged ion
metals tend to lose
e- ( oxidize)
non metals tend
to gain e- ( reduce))
Anions ( an-ion )
negativly charged ion
gained electron
non-metals from an-ions
metals gain/lose electrons to become a noble gases
in order to gain a valence electron
deterine charge
charge = # protons- # electrons
Atoms want a full valence shell
in other words a noble gas configuration
neon gases do not form ions
because already have a full valence shell
electron configuration can b determined
by the location of an element on the PT
period #
represents highest occupied EL ( for representive element )
block
reprsents last occupied sublevels ( S, p, d, f)
group
reresent number of e in the valence shell