Peter Slater y2
1/21
Earn XP
Description and Tags
d-block chemistry
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
oxidation states of transition metals
early/late transition metals have few oxidation states, those in the center have a wide range
chem of 1st series TMs dominated by M2+ and M3+ ions
2nd and 3rd series TMs
lower oxidation states are dominated by M-M interactions
high oxidation states form molecular species and covalent bonds - high oxidation states are most stable for 2nd and 3rd row
CFSE
tetrahedral - e (2 orbitals) lower, T2 (three orbitals) higher energy - oct opposite but t2h and eg
only get high spin for 1st series TMs, 2nd and 3rd always low spin
jahn teller distortion - occurs to remove degeneracy of electronic states eg. Cu2+ d9 and d4 hs eg. Mn4+ (exaggerated version for d8 square planar complexes)
explains lattice enthalpies for MCl2 and hydration energies of divalent metal ions
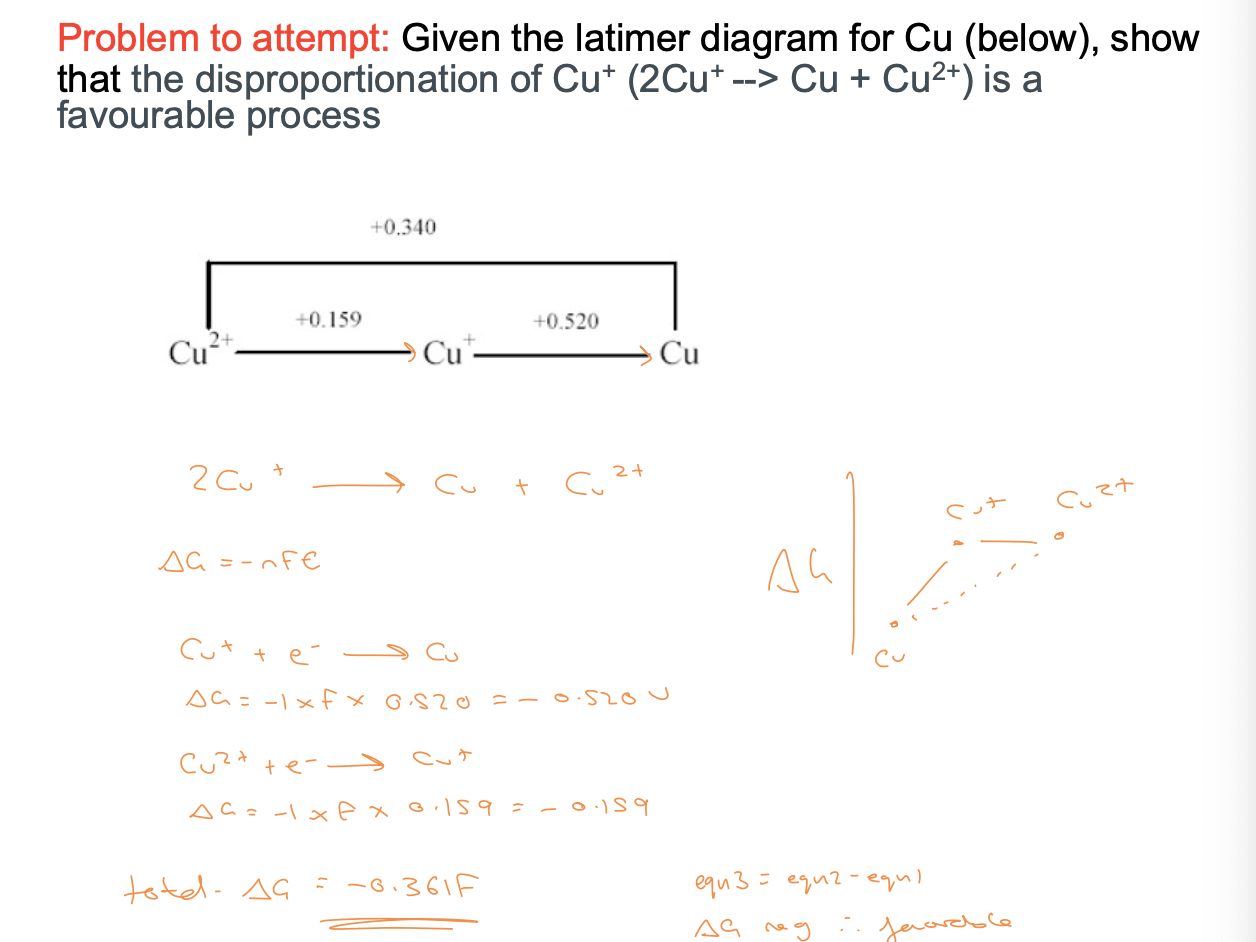
determining the spontaneity of a reaction
∆G = -nFE
combine any two standard reduction reactions by reversing one of the couples and adding the potentials together
if E is =ve the reaction as written is spontaneous and ∆G = -ve
if E is negative then the reverse reaction is spontaneous
E0 : most positive = most oxidising
latimer diagrams
used to monitor relative (thermodynamic) stabilities of all oxidations states of a transition metal
potentials are given as reduction potentials
can add branches on to describe a metal in the presence of other species
what factors change the redox potentials?
generally anything that alters ∆G0
concentration
temperature
other reagents that arent inert
pH
in aqueous chemistry the most important is pH change hence it’s conventional to construct latimer diagrams for the two extremes of pH=0 and pH=14 (1 M acid and 1 M base)
frost diagrams
plot nE (V) against oxidation state
determine oxidation states and plot nE using the equation nE = -∆G/F
interpreting frost diagrams
if the line connecting higher and lower lying species has a +ve slope then the higher lying species is an oxidising agent - if -ve the higher lying species is a reducing agent
steeper slope = stronger oxidising/reducing agent
species at the bottom of the graph have low ∆G0 and have little tendency to react
the lowest species on the graph are the thermodynamic final product
determining disproportionation and comproportionation from frost diagrams
a species lying above the line connecting its neighbours is thermodynamically unstable to disproportionation and will disproportionate to the more stable species either side
species likely to undergo comproportionation to a third species are located to the left and right of a point which lies below the line connecting two species
dis - a single species is unstable and an autoredox reaction can occur at any time
com - required both reactants to be present - if one is missing the other species remains stable
frost diagrams of the 1st row
all metals are potent reducing agents except Cu
reducing strength of M0 decreases from Ca to NI
early TMs favour the +3 oxidation state
for late TMs +2 is more stable
elements in the middle (Mn + Fe) have the largest range of accessible oxidation states
only Ti in its highest state (+4) has virtually no oxidising power
trends in behaviour down group 6
highest oxidation state for Cr (+6) is strongly oxidising, Mo (+6) mildly oxidising but completely non-oxidising for W
most stable oxidation states are +3 for Cr, +4 for Mo and +6 for W
higher oxidation states become more favourable in 2nd and especially 3rd transition series
in the +5 state the energy of Mo and W species are the same which leads to very similar behaviour
behaviour is fairly representative of trends down any d-block group
effects of ligands
eg. Fe2+ much more stable in presence of CN- - weka field ligand (H2O) subbed for strong field ligand so change in CFSE
stabilisation of high oxidation states
high oxidation states are very strong oxidising agents and so are highly susceptible to reduction
in order to stabilise TMs in high oxidation states they must be complexed by other species which are even stronger oxidising agents
O2- and F- are the most commonly found coordinating species for high oxidaiton state TMs
most hihg ox states are only accessible as fluorides
bonding in high ox state halides is prodominantly covalent
group 8: can only get Fe6+ but can get Ru 8+ and Os 8+
high oxidation states in solution
mononuclear oxo complexes
eg. CrO42- (CrVI) - high oxidation states are stabilised by the O2- ligand
strong polarisation of e- density on water of hydroxyl O by high oxidation state cations - increases bronsted acidity of H2O and OH-
oxo complex formation also preferred by high pH
what are the key methods of synthesising mixed metal oxides containing transition metals?
standard solid state reaction
coprecipitation
sol-gel synthesis
hydrothermal synthesis
mechanochemical synthesis
standard solid state reaction
direct reaction of a mixture of solid starting materials (use carbonates for group 1/2 and oxides for other groups)
most widely used synthesis method
needs high temperatures, typically >900ºC and long reaction times - several hours/days with intermediate grinding
need to have materials in correct ratios otherwise cannot remove any impurities
why are high temperatures and long synthesis times needed for standard solid state reactions?
formation of product nuclei is difficult
usually large distances between reactants and products
large amount of structural reorganisation (bond breaking and reforming) involved in forming the product
growth of product layer may be even more difficult
need counter-diffusion of cations through existing product layer to the new reaction interfaces (basically if a lump of product of formed in the middle, the cations left to react are now even further away)
kinetic and thermodynamic considerations of standard solid state reactions
what is the rate determining step?
transport of ions to the reaction interface?
reaction at the surface/interface?
transfer of matter away from reaction interface (couples to 1)?
slow kinetics can lead to metastable products
not fast enough to form product
phase of interest may not be thermodynamically stable (relative to other possible products) at the temperature required for reaction
even slight differences can alter stability
disadvantages of standard solid state reaction method
high temp and long reaction times required - hence lots of energy and so not ideal for industrial synthesis
inhomogeneity of mixture: can lead to impurities or variations in composition (local excess of reagents even it total ratios are right) - regrind and reheat to help rectify
volatile products (eg. alkali metal oxides) means the high temperatures lead to a loss of reactant - add excess reactant and/or use seal tube to rectify
some phases only stable at low temperatures - use low temp synthesis route
coprecipitation
for a lower reaction temperature we need a high degree of homogenisation and a small particle size - this can be achieved using coprecipitation
eg. mix the correct molar ratios of aqueous strontium nitrate and iron nitrate. adding NaOH will then cause it to precipitate- precipitate can then be ground with Li2CO3 or LiOH and heated to 700-800ºC for 12 hours in air to give final product
problems arise if the reactants have different solubilities or do not precipitate at the same rate - means that you don’t get a homogenous mix and can end up with the incorrect reactant ratio
sol-gel synthesis
sol-gel method provides homogenous, small particle size mixtures and so lower reaction temperatures can be employed
eg.
dissolve metal salts eg nitrates in water
add a dicarboxylic acid and a dialcohol
heat → gel formation (metal ions dispersed in a poly-ester matrix)
further heat treatment (typically 200-300ºC lower than standard solid state synthesis temperatures) → product (ester network burns off)
alternative routes use alkoxides
evaluation of sol-gel synthesis
advantages
lower temperature
synthesis on new phases possible (including metastable phases)
capacity to form films or fibres and to control particle sizes and shapes
disadvantages
can be high cost - especially if using alkoxides
long processing times sometimes required
longer than standard solid state synthesis - can sometimes take weeks to form a gel
can be problems with use of alkoxides if different hydrolysis rates - issues with achieving a homogenous gel
hydrothermal synthesis
utilises water under pressure and at temperatures above its boiling point to speed up solid state reactions (usually 100-200ºC)
roles of water:
serves as a pressure transmitting medium
some or all of the reactants are partially soluble in H2O under pressure, and this enables reactions to take place in, or with the aid or liquid and/or vapour phases
reactions are normally carried out in a sealed teflon lined stainless steel pressure vessel connected to an external pressure control often known as an autoclave
allows reactions to occur that would normally required very high temperatures
particularly suited method to phases that are unstable at high temperatures eg. zeolites and MOFs
sample must be stable in water so cant use to prepare YBa2Cu3O7-x
mechanochemical synthesis
some compounds can be prepared by simply ball-milling the precursor powders (ball milling causes local heating of the powders
powders are milled in a ball mill for several hours- not all compounds can be prepared by this routec