8. B1.1 Carbohydrates and lipids
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
Main groups of biomolecules
Carbohydrates
Monosaccharides
Glucose
Galactose
Fructose
Ribose
Disaccharides
Maltose (glu + glu)
Lactose (galact. + galact.)
Sucrose (glu +fruct.)
Polysaccharides
Starch
Glycogen
Cellulose
Chitin
Proteins
E.g molecules
Enzymes
Antibodies
Peptide hormones
Lipids
Triglycerides
Fat stored in adipose cells
Phospholipids
Lipids forming a bilayer in cell membranes
Nucleic acids (DNA and RNA)
Steroids
Nucleotides
DNA
RNA
ATP
Carbon
Carbon compounds referred to as the building blocks of life
Carbon atoms has 4 electrons in its 2nd electron shell and can form 4 covalent bonds (borrows), allowing for a variety of stable compounds to exist
\
Covalent bonds
A type of bond in which a pair of electrons are shared between 2 neighbouring atoms.
Strongest bonds found in biomolecules
Provide stability
Only broken during specific chemical reactions with other molecules
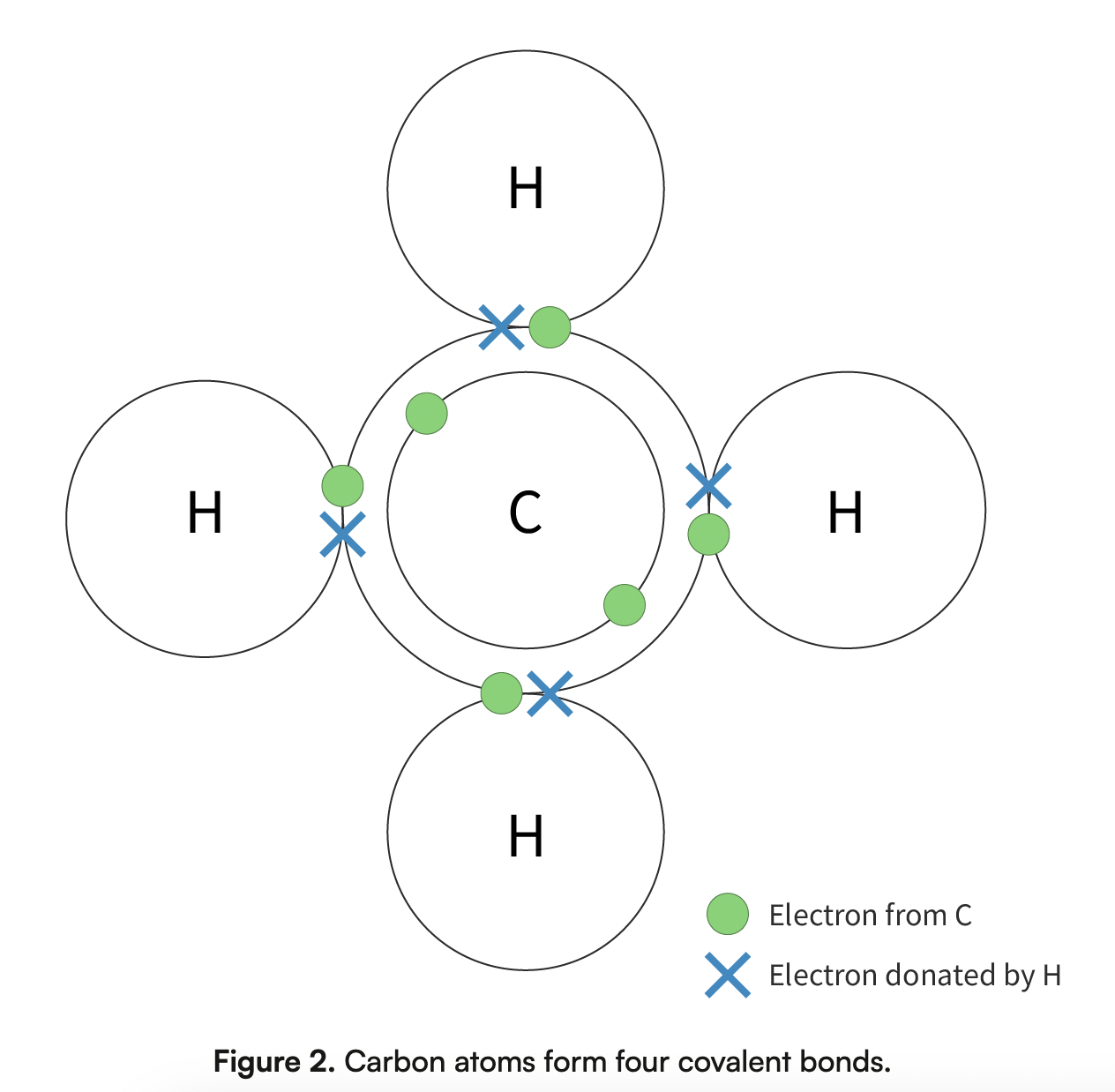
Covalent bonds formed by carbon
Carbon can form different types of covalent bonds with other carbon atoms or other types of atoms.
Giving rise to different forms
linear chain
branched chain
rings (cyclic) structures
Macromolecules
large molecules made up of smaller molecules called monomers
relative atomic mass of above 10,000 atomic units
Condensation / polymerisation reaction
chemical reaction that links one monomer to another
by-product of the reaction is water (removal)
catalysed by enzymes
E.g triglyceride formation
1 glycerol, 3 fatty acids
3 ester bonds formed
3 water molecules released
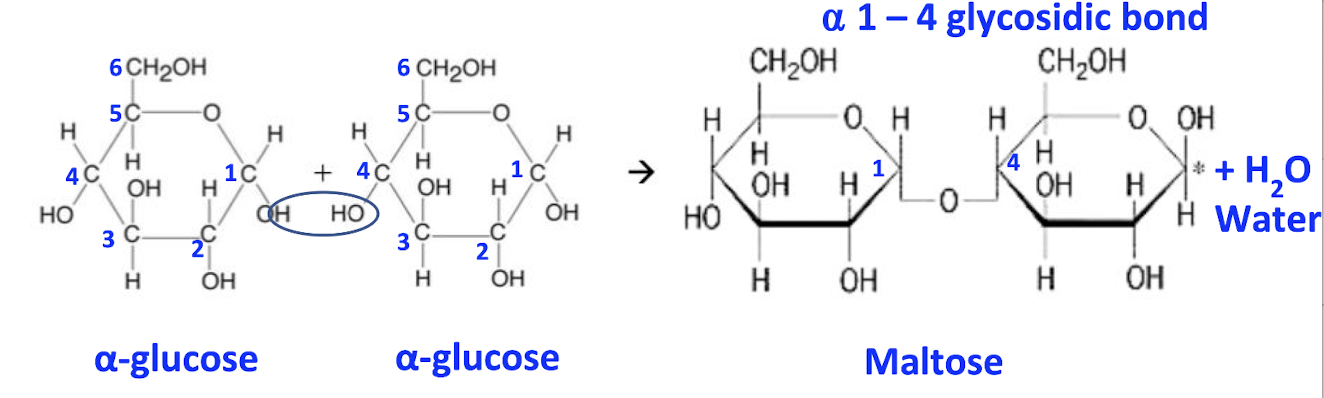
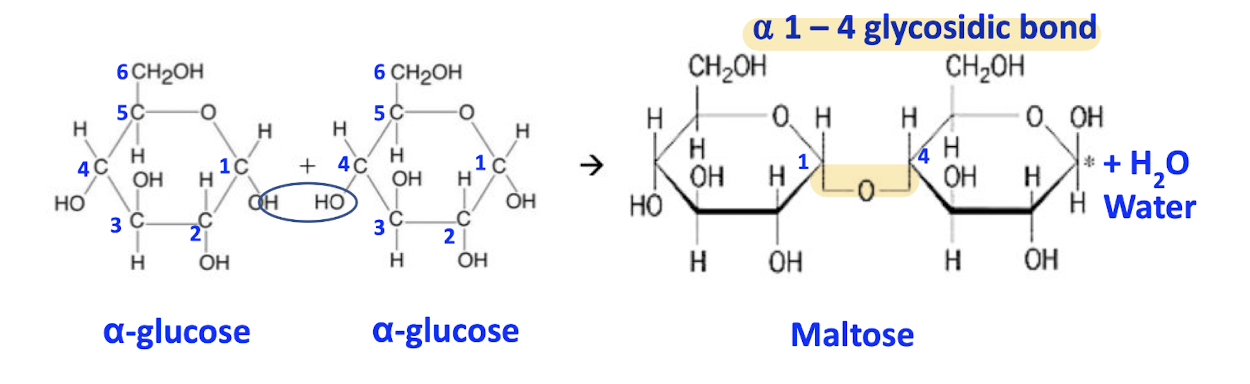
E.g maltose formation
1 glucose, 1 glucose
OH group on carbon-1 of one glucose attacks the -OH group on carbon-4 of the other glucose molecule
carbon-x, x is the position of the carbon in the compound in a clockwise direction!
1,4-glycosidic bond is formed
1 water molecule released
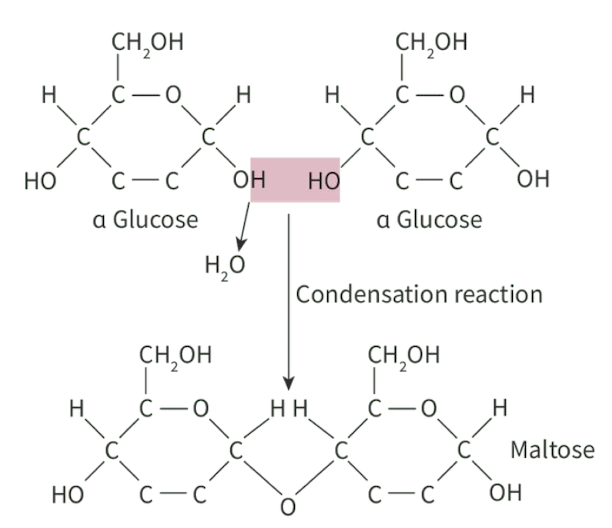
Label α-glucose on left side of eqn
Number the carbon atoms (6th is projected out)
Box up the hydroxyl group of the carbon-1 and carbon-4 atoms
Circle 2 H atoms and 1 O atom on the left that will form water molecule
Label maltose molecule
Add 1 water molecule on the product / right side of eqn
Name the bond formed
Hydrolysis (digestion)
Digestion of polymers can occur in
all cells as they can produce enzymes
in the gut of animals as enzymes are secreted
decomposers release enzymes into their environment to hydrolyse polymers around them so that they can absorb monomers
Hydrolysis requires water (add) and enzymes
Monosaccharides
Single unit of carbohydrate
Usually have 3 to 7 carbon atoms
Classified by the no. of carbon atoms they contain
Pentoses (5 carbon atoms) e.g ribose
Hexoses (6 carbon atoms) e.g glucose, galactose, fructose
Glucose
Hexose sugar
Formula: C6H12O6
Can exist in ring form or linear
2 isomers
α-glucose (alpha-glucose)
β-glucose (beta-glucose)
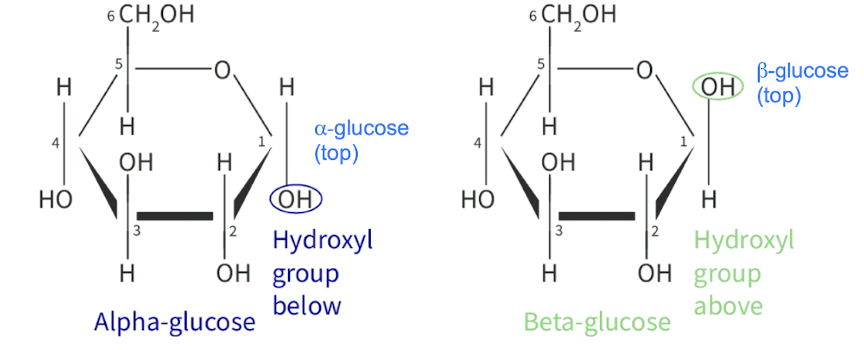
D-glucose and L-glucose
Difference is positioning of the 3 hydroxyl groups & 1 hydrogen group
D-glucose
RIGHT
L-glucose
LEFT
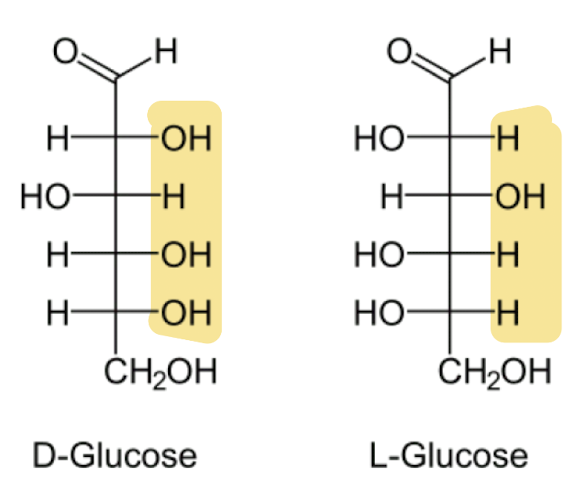
D-glucose and L-glucose
•α-L-glucose is the mirror image of α-D-glucose.
•β-L-glucose is the mirror image of β-D-glucose.
•*the “mirror” would be the plane of the ring, hence the oxygen and the carbon atoms do not change positions
Form and function of glucose
solubility in water
function an energy storage molecule
monomer / building block for polymers / polysaccharides
Solubility of glucose
Glucose is a polar molecule (i.e. having partial positive and negative charges)
Present of – OH groups enables formation of hydrogen bonds with water molecules
Hydrophilic
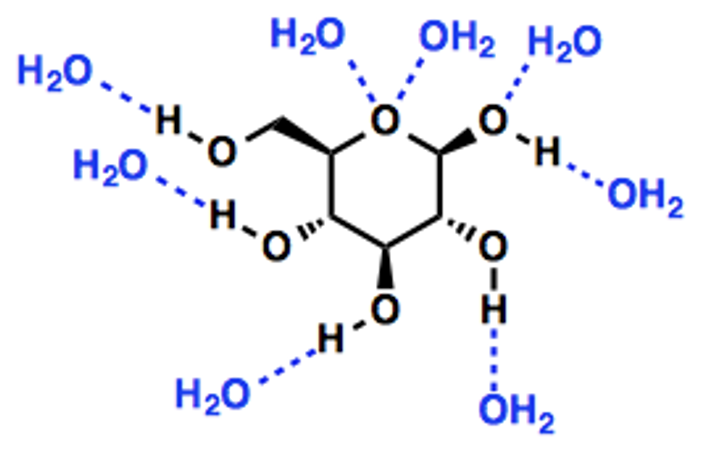
Energy storage
Glucose is a respiratory substrate used by most cells for cellular respiration to produce ATP for various functions
Energy stored in the C-C (carbon-carbon) and C-H (carbon-hydrogen) bonds of glucose
One molecule of glucose can produce a large amt. of ATP energy
water and carbon dioxide are the by-products
What are the components of starch?
2 polysaccharides
amylose
amylopectin (major component)
they are both composed of alpha-glucose monomers
Amylose and amylopectin allow more glucose molecules to be stored in a fixed volume
What are the characteristics of amylopectin?
branched due to the presence of alpha 1-6 glycosidic bonds
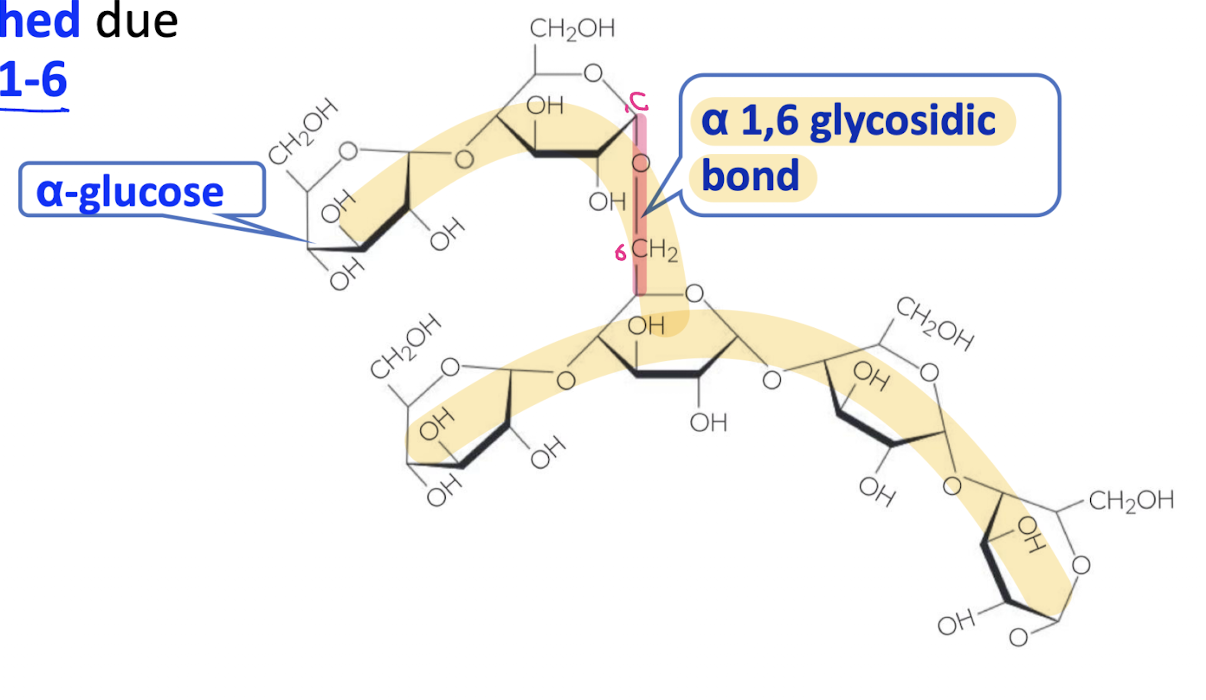
Storage of starch
in specialised plant structures (seeds, roots etc.)
starch is compact in structure due to its coiling and branching during polymerisation
allows for efficient storage in a small space
Amylose and amylopectin are relatively insoluble
large molecular size
maintain osmotic balance within organism
How is starch utilised?
plant in need of glucose
starch is broken down through hydrolysis
releases glucose molecules
glucose used as a source of energy
to carry out various cellular processes such as growth and photosynthesis
What is glycogen?
primary storage form of glucose and animals and yeast
relatively insoluble
large molecular size
helps maintain osmotic balance within an organism
found in the liver and muscles of animals
What is the components of glycogen?
monomer: alpha-glucose
joined by alpha 1-4 glycosidic bonds and alpha 1-6 glycosidic bonds
branched
more so than amylopectin despite being structurally similar
compact, enabling efficiency
Where is glycogen stored and how is it utilised?
liver
when blood glucose levels dropped
break down glycogen by hydrolysis
release glucose molecules into the bloodstream
muscle cells
store glycogen primarily to provide energy for muscle contraction during exercise
when energy is needed
glycogen broken down into glucose molecules which can be used for cellular respiration (produces energy by breaking down glucose, to generate ATP)
Draw the condensation reaction to form maltose from glucose
label the type of glucose molecules on the left side of the equation
number the carbon atoms of the 2 glucose atoms on the left
the condensation reaction involves the hydroxyl group of carbon -4 and the hydroxyl group of carbon 1 that are closest to each other — highlight these hydroxyl groups (-OH)
Circle 2Hs and 1O on the left side of the equation that will form the water molecule
Complete the equation by adding water on the product side
Label the disaccharide
Name the bond formed
Draw the condensation reaction to form the branch point in glycogen or amylopectin
Label the type of glucose molecules on the left side of the equation
Number the carbon atoms of glucose
The condensation reaction involves the hydroxyl group of carbon -1 and the hydroxyl group of carbon 6 that are closest to each other — highlight these hydroxyl groups (-OH) on the left side of the equation
Circle 2Hs and 1O on the left side of the equation that will form the water molecule
Add water on the product side
Label the bonds (1-4, 1-6)
What is cellulose?
complex polysaccharide that serves a structural function
monomer: Beta-glucose
an essential component of the plant’s cell wall
the molecule of glucose have to be vertically flipped in an alternating pattern
allows the OH (hydroxyl group) on carbon01 and carbon-4 of 2 B-glucose molecules to be closer to each other