3.1.6.2 equilibrium constant Kc for homogenous systems
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
for a homogeneous equilibrium system, what can we write an expression for? + how?
for an equilibrium constant Kc in terms of the concentration of the reactants + products involved
what is Kc?
a ratio of the concentration of the products to the reactants
what is a homogeneous equilibrium?
a system in which the reactants + products are in the same phase
what is Kc deduced from?
the equation for a reversible reaction
give + explain the general expression for an equilibrium constant, Kc, for any reaction

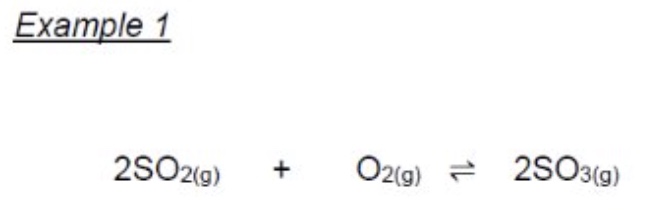
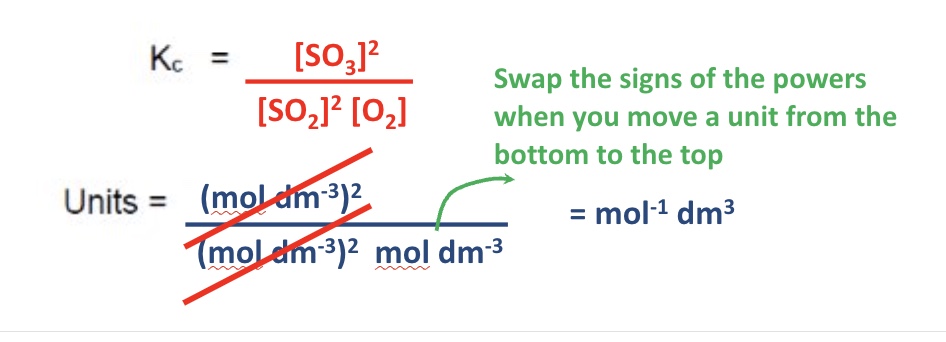
give the Kc expression + units for this equation
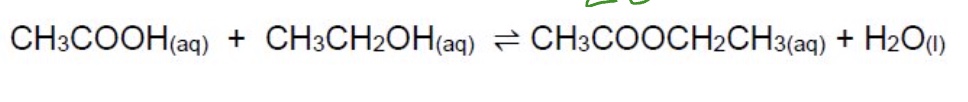
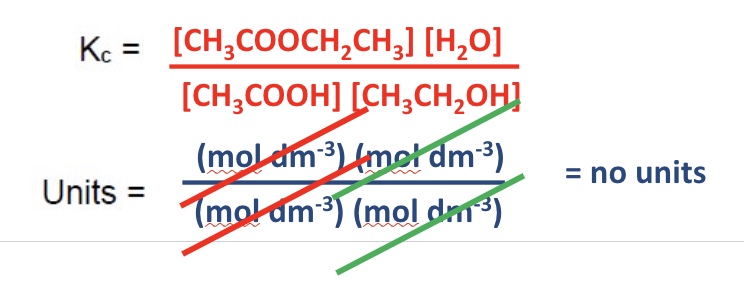
give the Kc expression + units for this equation

calculate a value for Kc + give the units


In this example, why can moles be used to find the value of Kc rather than concentration?
because there are equal numbers of moles on both sides of the equation + so the volume terms cancel out in the Kc expression
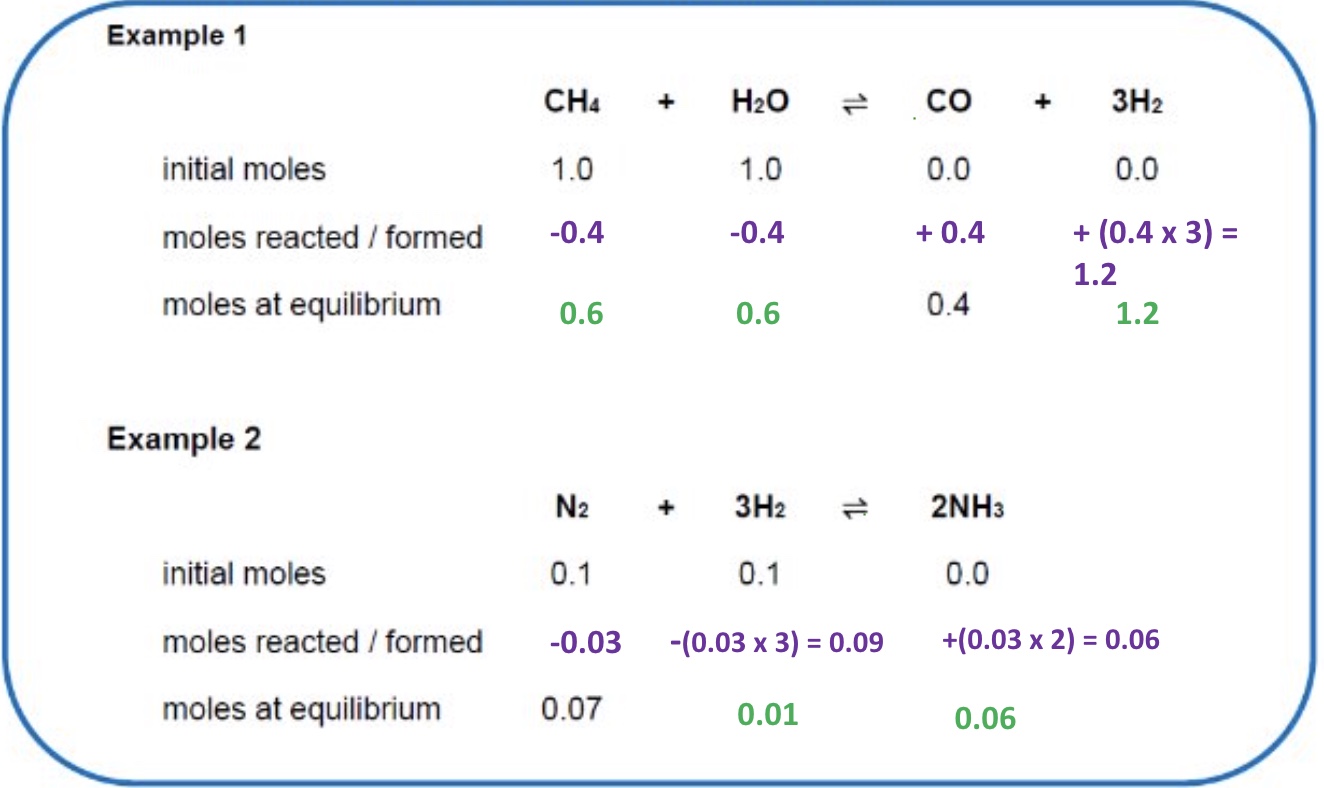
the moles at equilibrium are usually not given to you, how can you calculate them?
using initial moles + the moles of one the species at equilibrium (using ICE)

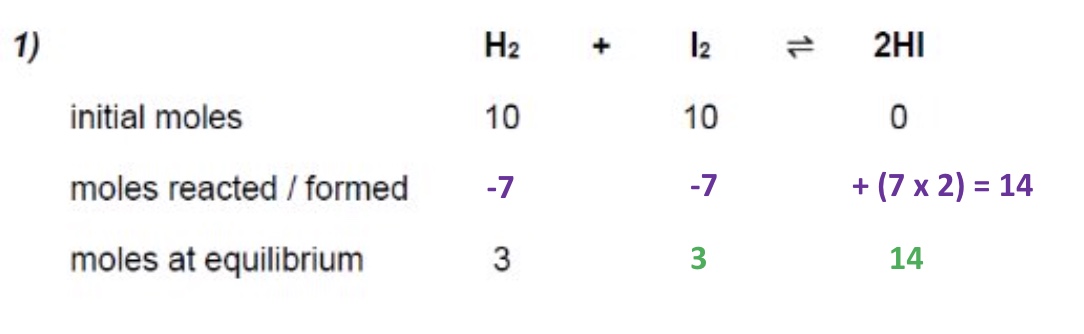
calculate the moles at equilibrium of iodine + hydrogen iodide:

in terms of x, calculate the moles at equilibrium of nitrogen + hydrogen

what are the two steps to calculate Kc using initial moles?
use ICE to find moles
use Kc expression to find Kc

using ICE + Kc expression, calculate a value for Kc with units
as number of moles is equal on both sides, we can just use moles to calculate Kc instead of using the to calculate concentration first


using ICE + Kc expression, calculate a value for Kc with units


using ICE + Kc expression, calculate a value for Kc with units
number of moles on both sides is not equal, so moles at equilibrium will be used to calculate concentrations

along with calculating Kc, what else do you need to be able to do with Kc calculations?
rearrange Kc calculations to find the concentration of a specific substance

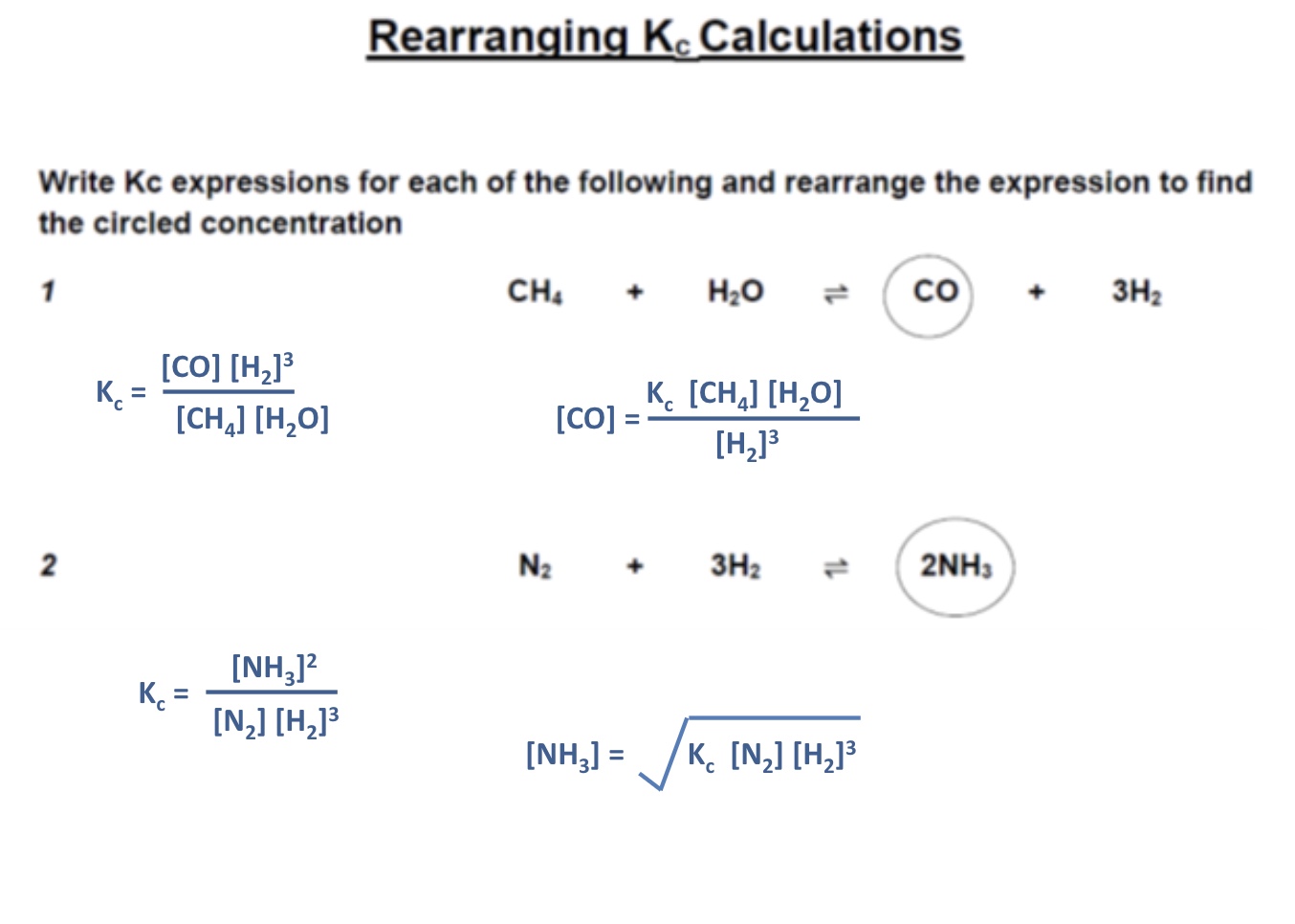

example 1 of rearranging Kc
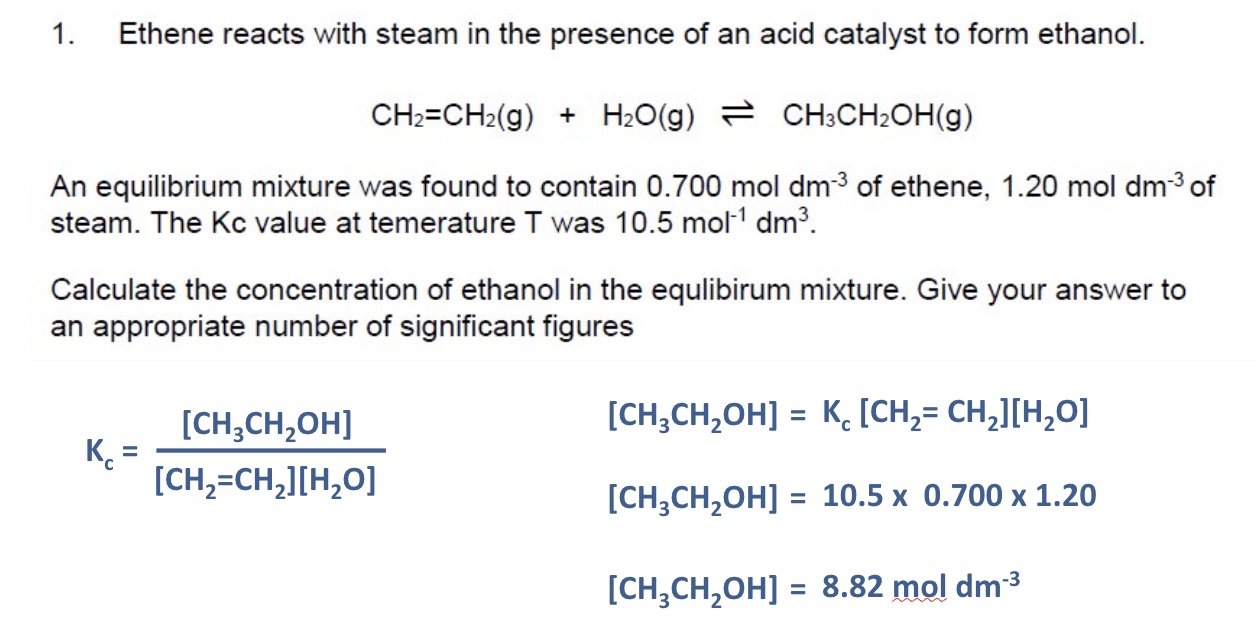
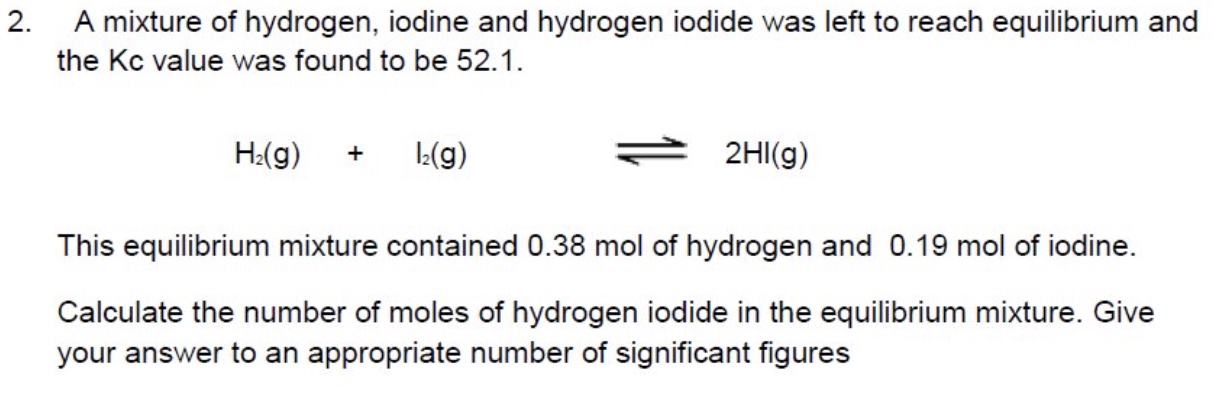
example 2 of rearranging Kc

the value of the equilibrium constant, Kc is what?
temperature-dependent
what does temperature-dependent mean?
its only affected by temperature
how does a change in temperature affect Kc?
a change in temp changes the values of Kc
an increase in temperature always shifts the equilibrium in….
the endothermic direction
what shifts the equilibrium in the exothermic direction?
a decrease in temperature
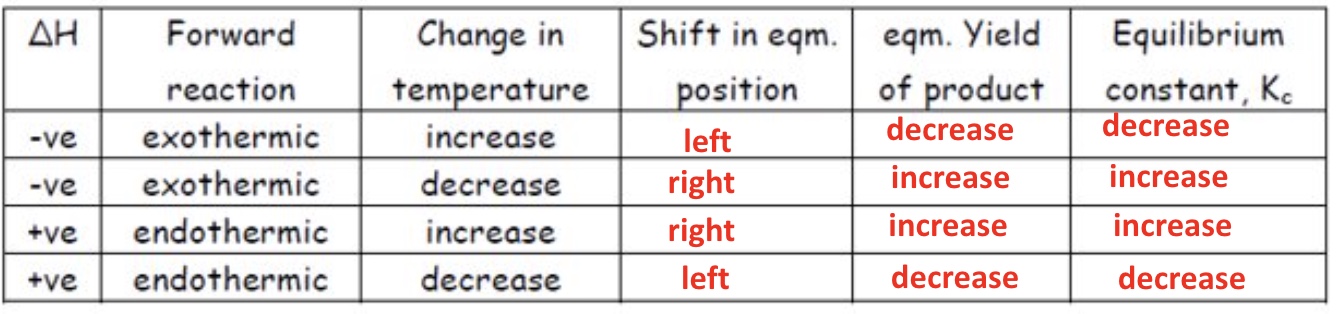
describe the effect of increasing the temperature on Kc if the forward reaction was exothermic
if the temperature is increased, the equilibrium shifts to the left → therefore the yield of products would decrease
the top part of the expression becomes smaller + the bottom part of the expression becomes larger, therefore the value of Kc would decrease
describe the effect of increasing the temperature on Kc if the forward reaction is endothermic
if temperature is increased, the equilibrium shifts to the right + therefore the yield of products would increase
the top part of the expression becomes larger + the bottom part becomes lower → therefore the value of Kc would increase
summary of changes in temperature
*value of Kc is only affected by temperature
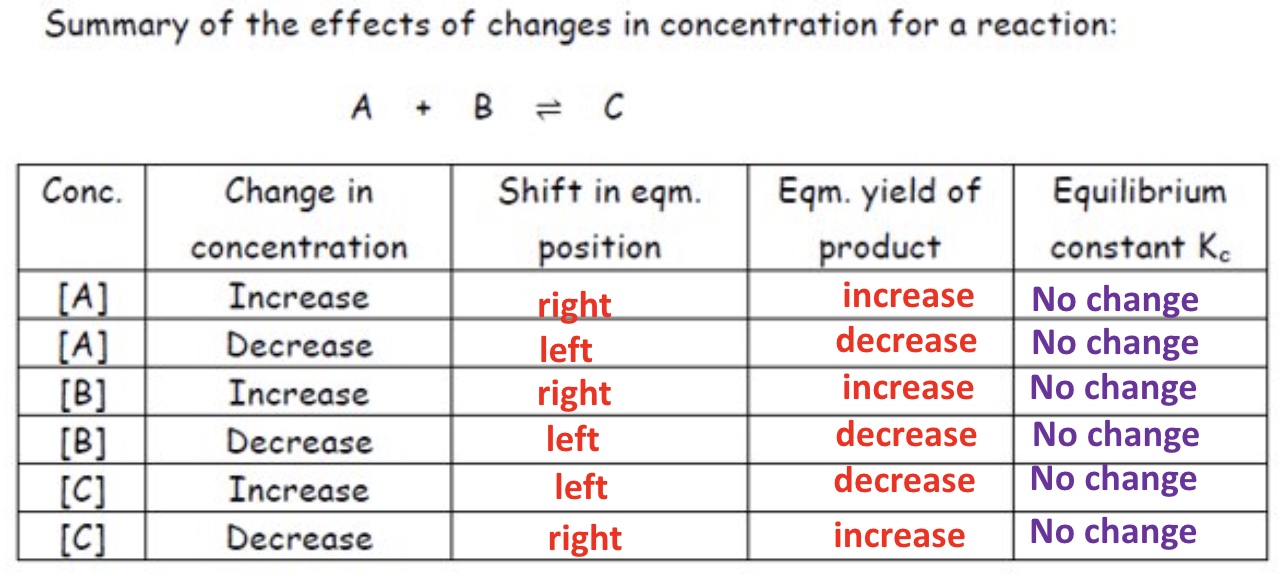
explain the effect changes in concentration as on Kc
Kc is unchanged by changes in the concentration
because if the concentration of any species involves in equilibrium is changed then the concentrations of other species will change so that the value of Kc remains constant
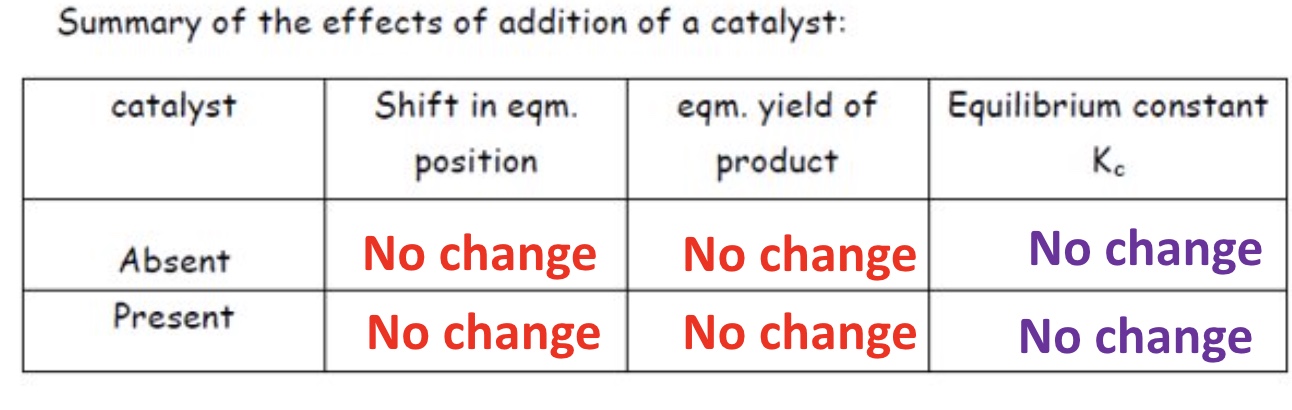
outline the effect a catalyst has on:
Kc
position of equilibrium
yield
it has not effect on the Kc
neither does it have any effect on the position of equilibrium in a chemical reaction
hence, catalysts do not affect the yield of a chemical processes
explain the effect changes in pressure has on Kc
Kc is unchanged by changes in pressure as the only factor that affects it is temperature
changing the pressure will cause equilibrium to shift if there are different numbers of moles on either side of the balanced equation