Unit 2.2 Understanding Macromolecules
1/131
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
132 Terms
Define macromolecules
Large biological molecules
Define polymers
Molecules that are created by many molecules put together
What are polymers made up of?
Monomers
What are monomers?
Single units put together to form a polymer
What is a good analogy of monomers-polymers?
A beaded necklace! Each bead is a monomer while the entire necklace is a polymer
What type of macromolecules are we studying in this course? (3 things)
Carbon-based, biological molecules that contain functional groups
What are functional groups?
Functional groups have specific properties that they provide to the molecules they are in
What are 2 properties of the functional groups we are examining? Plus a bonus category
Polar and hydrophilic; some are acidic or basic
What is a mnemonic device to remember the functional groups and where are they called?
Harry Styles Can Colour All Pictures- hydroxyl, sulfhydryl, carbonyl, carboxyl, amino, and phosphate
What is the general feature of a Hydroxyl functional group and its properties?
H and O; polar; hydrophilic
What is the general feature of a Sulfhydryl functional group and its properties?
S; polar; hydrophilic
What are the general features of a Carbonyl functional group and its properties?
Central C and O; bound to 2 side groups, double bond to oxygen; polar; hydrophilic
What is the general feature of a Carboxyl functional group and its properties?
Central C bound to O and OH; acidic; polar; hydrophilic
What is the general feature of an Amino functional group and its properties?
N; polar; hydrophilic; basic
What is the general feature of a Phosphate functional group and its properties?
P; polar; hydrophilic; acidic
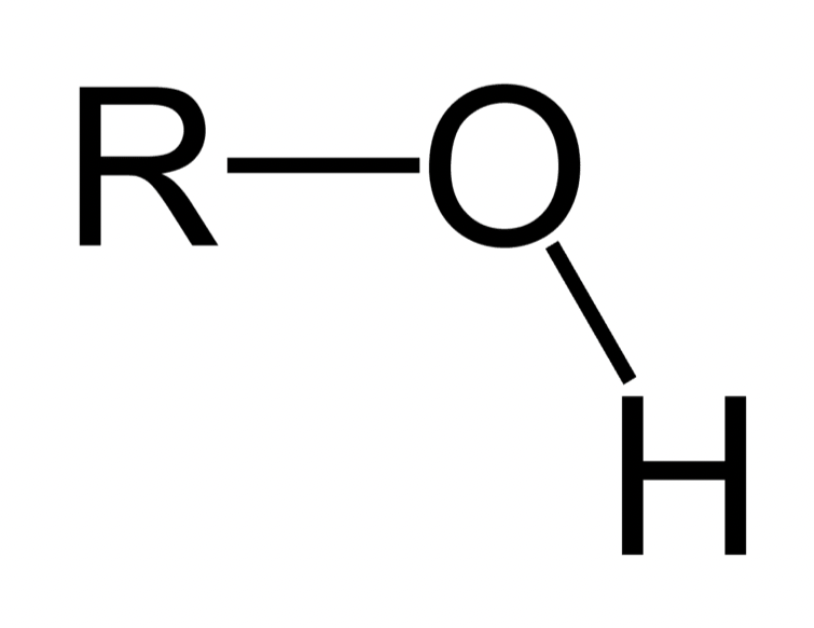
Which functional group is this?
Hydroxyl
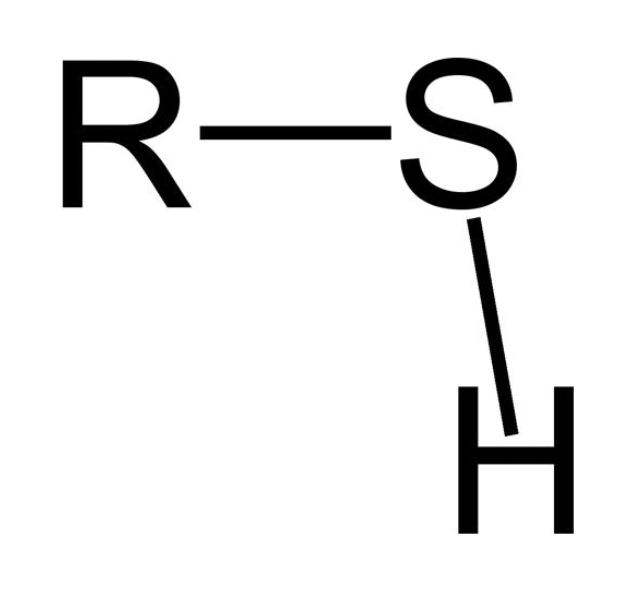
Which functional group is this?
Sulfhydryl
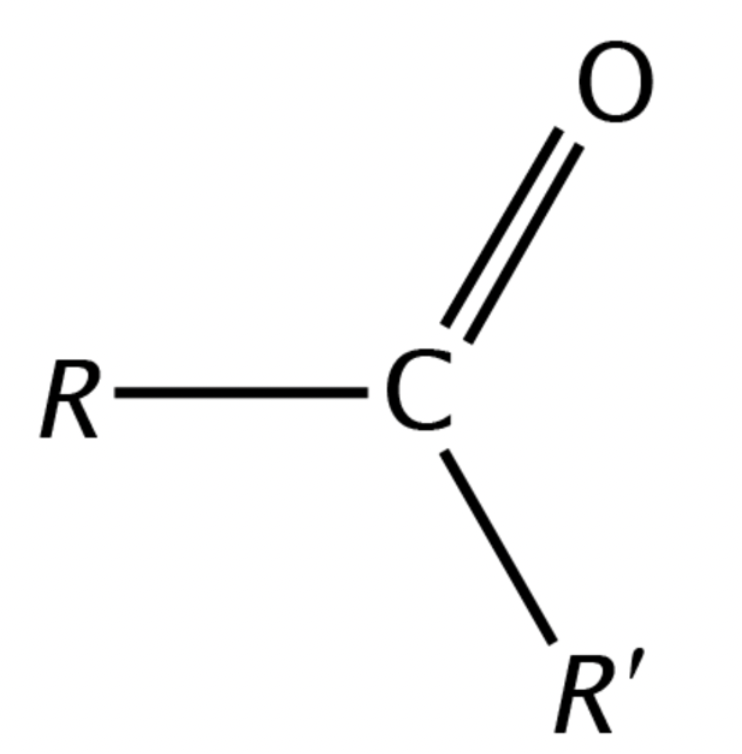
Which functional group is this?
Carbonyl
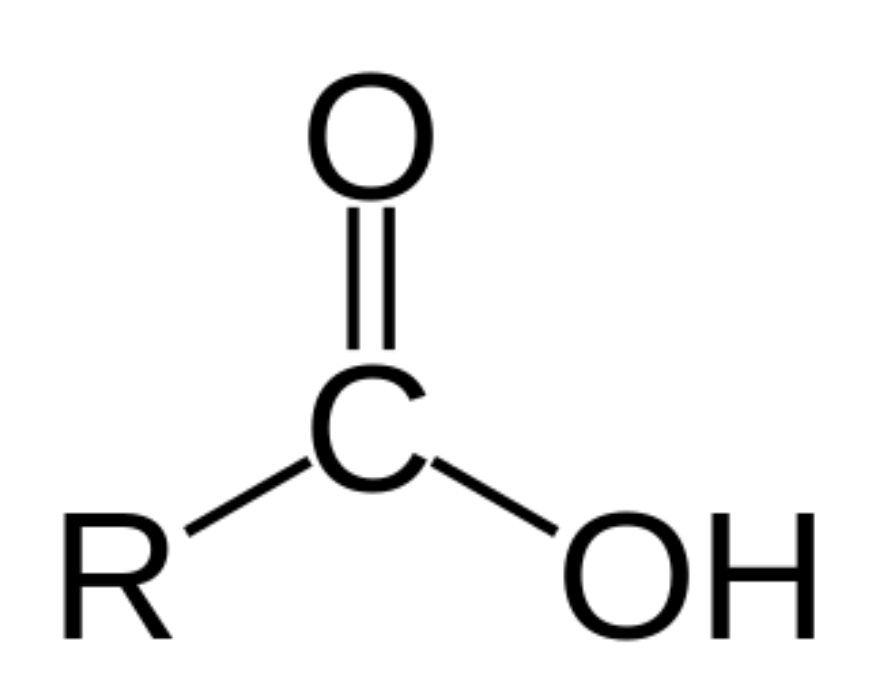
Which functional group is this?
Carboxyl
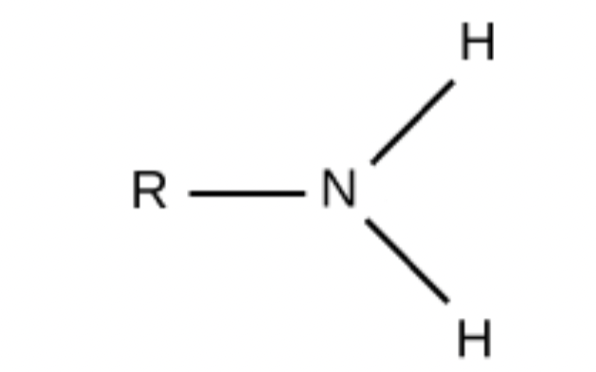
Which functional group is this?
Amino
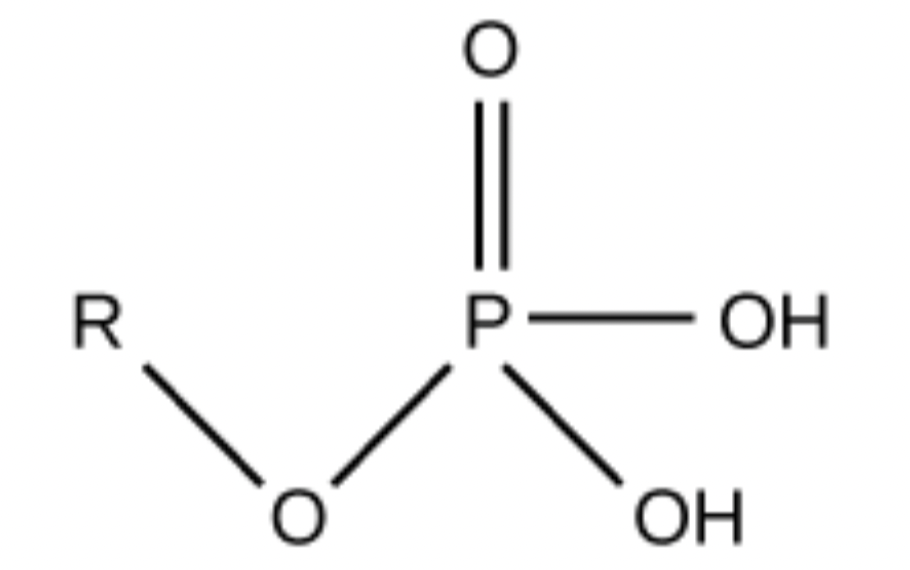
Which functional group is this?
Phosphate
What is the → and ← relationship between monomers and macromolecules?
Biochemical reactions use monomers (smaller repeating units) as building blocks to build macromolecules. Similarly macromolecules are broken down to form monomers
What must be removed to form macromolecules?
Water
How is water removed in the process of building macromolecules?
Through condensation/dehydration synthesis
What is the difference between anabolic and catabolic?
Anabolic builds up while catabolic breaks down
Is the creation of macromolecules from monomers an anabolic reaction or a catabolic reaction?
Anabolic- build up
What is a very basic equation of making a macromolecule/condensation (dehydration) reaction?
Monomer + Monomer - Water = Macromolecule
Are all macromolecules polymers?
No. All polymers are macromolecules but not all macromolecules are polymers. A lipid is a macromolecule but not a polymer because it doesn’t have repeating subunits
What is hydrolysis?
A catabolic (breaking down) reaction to break down macromolecules by adding water. Macromolecules are broken down into its smaller monomer units
What is a very basic equation of breaking down a macromolecule/hydrolysis reaction?
Macromolecule + Water = Monomer + Monomer
Besides water, what is required for a hydrolysis reaction?
Enzymes
What are enzymes?
Protein catalysts that speed up chemical reactions
Are condensation/dehydration and hydrolysis reactions important?
YES. Vital biological reactions necessary to make or break the macromolecules of life
What are the 4 macromolecules of life and a mnemonic device to remember them?
Can Louis Pick Niall- Carbohydrates, Lipids, Proteins, and Nucleic acids
What are the 3 main functions of carbohydrates?
Energy, cell identification, cell communication
What 3 elements do carbohydrates contain and in what ratio? Think alphabetical
Carbon, Hydrogen, and Oxygen; 1:2:1 ratio
What are some examples of carbohydrates and why do we need them?
Bread, cereal, pasta, and grains; vital to remain healthy and active
What 2 groups are carbohydrates classified into?
Simple carbohydrates and complex carbohydrates
What are the 3 types of simple carbohydrates?
Monosaccharides, disaccharides, and oligosaccharides
What is a “mono”saccharide?
“One sugar”. Simple chains that can form rings when they dissolve in water. Contain one subunit of carbohydrate
What are the 2 types of monosaccharides?
Aldoses and ketoses
How do the 2 monosaccharide types differ?
Differ by the location of the carbonyl functional group
What are 3 examples of aldose monosaccharides?
Ribose, glucose, and galactose
What are two types of ketose monosaccharides?
Ribulose and fructose
What is the most important monosaccharide and why?
Glucose. The body’s preferred source of energy
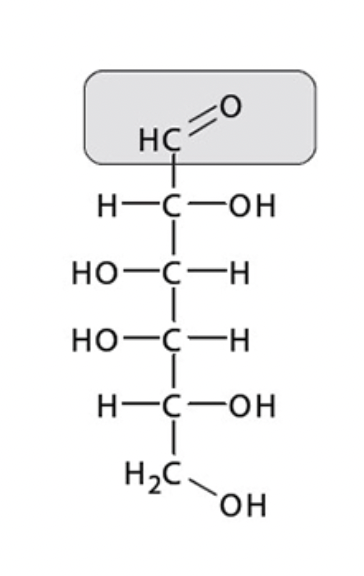
Is this an example of an aldose or a ketose?
Aldose
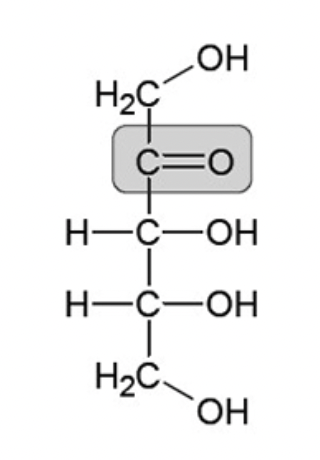
Is this an example of an aldose or a ketose?
Ketose
Glucose and galactose are aldose monosaccharides and fructose is a ketose monosaccharide but they are all isomers of each other. What does this mean?
Means they have the same chemical formula but a different arrangement of atoms
What is an isomer?
Molecules that have the same chemical formula but a different structural arrangement of atoms AND possessing different chemical properties
What is the difference between alpha and beta glucose?
Isomers; in alpha glucose the hydroxyl group points down; in beta glucose the hydroxyl group points up
What happens to a monosaccharide structure with 5 or more carbons?
Will form a ring structure when dissolved in water and a linear chain structure when dry
How are disaccharides formed?
Formed when 2 monosaccharides combine by a covalent bond
What is the linkage called when 2 monosaccharides combine to form a disaccharide? Which type of reaction is it?
A glycosidic linkage through a condensation/dehydration reaction
How many simple sugars are involved in a oligosaccharide? Their linkage? Reaction?
3-10 simple sugars; glycosidic linkage; condensation/dehydration reaction
What do we get when combining glucose + glucose?
Maltose + H2O
What do we get when combining glucose + fructose?
Sucrose + H2O
What do we get when combining glucose + galactose?
Lactose + H2O
What are polysaccharides and are they simple or complex?
Complex carbohydrate. They are polymers composed of several 100 to several 1000 monosaccharide subunits held together by glycosidic linkages
What are 2 reasons why complex carbohydrates are important in our diet?
Important for our daily energy requirement and to convert the excess into storage energy
Besides humans, who else are complex carbohydrates important for?
Plants, insects, and crustaceans
What are the 4 types of complex carbohydrates and a mnemonic device to remember them?
Songs Get Crazy Colourful- Starch, Glycogen, Cellulose, and Chitin
What is starch made of?
Amylose and amylopectin
Starch is the primary energy storage molecule for who?
For plants
What type of foods have a lot of starch and what do we use it for?
Root vegetables like carrots and potatoes have a lot of starch which we digest for energy
Glycogen is the main energy storage molecule for who?
Animals
What is the main function of glycogen?
Regulates blood glucose levels
How does glycogen regulate blood glucose levels?
When glucose increases, liver and muscles convert excess glucose to glycogen. When glucose is needed, enzymes break glycogen molecules apart for use
Why is cellulose useful in plants?
Cellulose forms long chains that are tough- ideal building material for the cell walls of plants
Can humans digest cellulose?
No. It is a form of fibre so good for egestion
What is chitin used for?
Used by insects and crustaceans to form their hard exoskeletons!
What are lipids?
Lipids are fats! Organic molecules that dissolve in non-polar substances and are insoluble in water
What are lipids good for?
Efficient energy storage molecules that yield twice the amount of chemical energy per gram compared to carbohydrates and proteins!
Besides energy, what are 3 uses of lipids?
Physical/thermal insulation; components in cell membranes; raw materials for the synthesis of hormones
What 3 elements make up lipids?
Hydrogen, Carbon, and Oxygen
What are the 4 families of lipids?
Fats, phospholipids, steroids, and waxes
What is a fat made of?
One glycerol molecule reacts 1-3 fatty-acid chains
Which reaction forms a fat molecule?
Condensation/dehydration synthesis
In the synthesis of a fat molecule, after water is removed, what is the linkage called when the fatty acid chains attach to the glycerol? What is the process called?
Ester linkage; esterification
What is a monoglyceride?
When a fat molecule is made up of only one fatty acid chain
What is a diglyceride?
When a fat molecule is made up of two fatty acid chains
What is a triglyceride?
When a fat molecule is made up of three fatty acid chains
Fatty acids can be classified as either-
Saturated or unsaturated
What are the 3 properties of Saturated fatty acids and give 2 examples
Saturated with Hydrogen atoms; no double bonds; solid at room temperature; lard and butter
What are 3 properties of Unsaturated fatty acids and give an example
Not saturated with Hydrogen; one or many double bonds; liquid at room temperature; olive oil
Where are phospholipids found and describe their permeability
Make up cell membranes of animal cells; phospholipid bilayer in cells is virtually impermeable to macromolecules, relatively impermeable to charged ions, and quite permeable to small lipid soluble molecules
Describe the physical properties of phospholipids
Hydrophilic head, hydrophobic two-stranded tail; made of one glycerol, two fatty acids, and a highly polar phosphate group
What are sterols?
A subgroup of steroids. They are compact hydrophobic molecules containing four fused hydrocarbon rings
Cholesterol is what type of lipid?
A steroid
For what 3 reasons is cholesterol important for cell membranes?
Affects fluidity; provides membrane support; messenger in cell communication during development
What can cholesterol be converted into?
Vitamin D
What happens if there is too much cholesterol in your diet?
Can lead to clogged arteries and cardiovascular disease
What are waxes?
Long chain fatty acids; hydrophobic; form waterproof coatings (paraffin, cuticle on plant leaves)
What are 5 examples of proteins?
Enzymes, immunoglobin, hemoglobin, keratin, and fibrin
What are proteins made of? Are they all the same?
Amino acid monomers; number, type, and arrangement of the amino acids will determine the type of protein
Amino acids are the building blocks of-
Proteins
How many amino acids make up proteins? What is their structure in a protein?
20 different amino acids; 3 distinct parts; attached to a central carbon
Every amino acid contains what 3 things?
Amino group, carboxylic acid group, radical group (also called a side chain)
Which part makes each amino acid unique and how?
The radical group. They are a group of atoms that make the amino acid polar, non-polar, or charged
Of the 20 different amino acids, where do they come from?
9 amino acids are essential and we get them from our diet. The others are manufactured in our cells!
What reactions do amino acids go through to form proteins? What are these bonds called?
Dehydration synthesis; peptide bonds.