Reaction + Reagents/Product [Alcohol, Oxidation, Reductions]
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
Alcohol Chemisitry (MAIN GOAL)
Turn OH into Good LG
Trap Oxygen lone pair in Resonance {aka Pseudohalides}
Protonate -OH for positive charge
Use enthalpy/entropy to make it so that reaction can’t resist breaking the C-O bond
Pseudohalide Mechanism
Reagents: 1. Ms/Ts-X, Pyridine 2. (Nucleophile)- +(spec ion)
Product: OH replaced with Nucleophile
Dehydration
Reagents: H2SO4 (Strong Acid), heat
Product: OH removed and creates alkene (double bond)
Enthalpy (Sn2)
Reagents: PCl3 or PBr3
Product: Alcohol removed and replaced with Cl or Br in flipped orientation (ex: OH was dashed, Cl is Wedged)
Enthalpy (E2)
Reagents: POCl3 or POBr3, Pyridine (2 equivalent)
Product: OH and beta H removed to create double bond/alkene between beta carbon and alpha carbon
Entropy (Sn2)
Reagents: SOCl2, Pyridine
Product: OH replaced by Cl in flipped orientation + gaseous SO2Cl
Oxidation Synthesis
Carbon losing electrons/decreasing in oxidation number
Primary OH —> Aldehyde
Secondary OH —> Ketone
Hydrate —> Carboxylic Acid
PCC
OXIDATION:
Reagent: PCC
Product: Ketone or Aldehyde (depending on if reactant was primary or secondary)
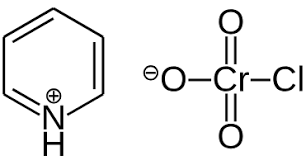
Dess Martin Synthesis
Reagent: DMP, CH2Cl2 (Aprotic Solvent)
Products: Ketone or Aldehyde (depending on if reactant was primary or secondary)
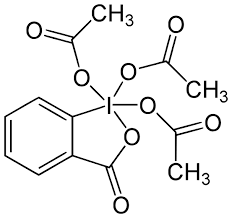
Jones Oxidation
Reagents: H2SO4, H2O2, H2O, Me2O (Acetone), CrO3
Product: Carboxylic Acid (from a PRIMARY ALCOHOL)
Reduction Synthesis
Cabron gaining electrons/Increasing Oxidation Number
“Reverse Oxidation” (Ketone —> Secondary OH; Aldehyde —> Primary OH)
Hydride
REDUCTION
Reagents: 1. NaBH4 or LiAlH4, MeOH (solv) 2. H3O+
Products: Ketone / Aldehyde become primary or secondary OH
Organometallic Reduction
Reagents: 1. Alkyne - + Na, gringard reagent (R-MgX) or Organolithium (R-Li; more reactive), THF (polar, aprotic solvent) 2. H3O+
Product: Tertiary or Secondary OH