ALDEHYDES, KETONES
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
simplest aldehyde
methanal (formaldehyde)
how to name aldehyde
no need to use number to locate aldehyde group
how to name unsaturated aldehyde
becomes enal
where is co (carbonyl) in aldehyde
end
where is co (carbonyl) in ketone
middle
polarity of ketone and aldehyde
polar (c=o is polar, O partially negative)
strength of intermolecular bond aldehyde and ketone
weak intermolecular attraction between partial positive charge of c=o and weak negative of another c=o
is hydrogen bonding possible between aldehyde and ketones and why
no.
no polar CO or NH bonds
boiling point ketone, aldehyde
lower than alcohol and carboxylic acid
how does aldehyde become carboxylic acid
oxidizing agents like potassium dichromate
what are liquid aldehydes sensitive to
oxidation via o2 air
how do ketones resist oxidation
oxidizing agents like potassium dichromate and molecular oxygen
reagent example for oxidation of aldehydes
tollens. silver dposits on walls on container as silver mirror
reduction (loss of oxygen gain of hydrogen) of aldehyde gives
primary alcohol. becomes C(H)OH group
reduction (loss of oxygen gain of hydrogen) of ketone gives
secondary alcohol. becomes C(H)OH group
reagent example for reduction of aldehyde/ketone
sodium borohydride (NaBH4)
hydrogen (hydride) adds to partially positive carbon in c=o
leaves negative charge on oxygen in c=o
what gives the primary/secondary alcohol with NaBH4
reaction with aqueous acid
what does NaBH4 not affect structurally
carbon carbon double bond, or aromatic ring
biological systems: agent for reduction of aldehydes and ketones
nadh (BIO HL TRAUMA.). delivers hydride to carbon in c=o of aldehyde/ketone
carbon bc: think of bio hl
describe reduction of pyruvate
done w nadh, gives lactate. end product of glycolysis (oxidation, lol!)
hemiacetal
addition of alcohol to carbon group of aldehyde/ketone (half acetal)
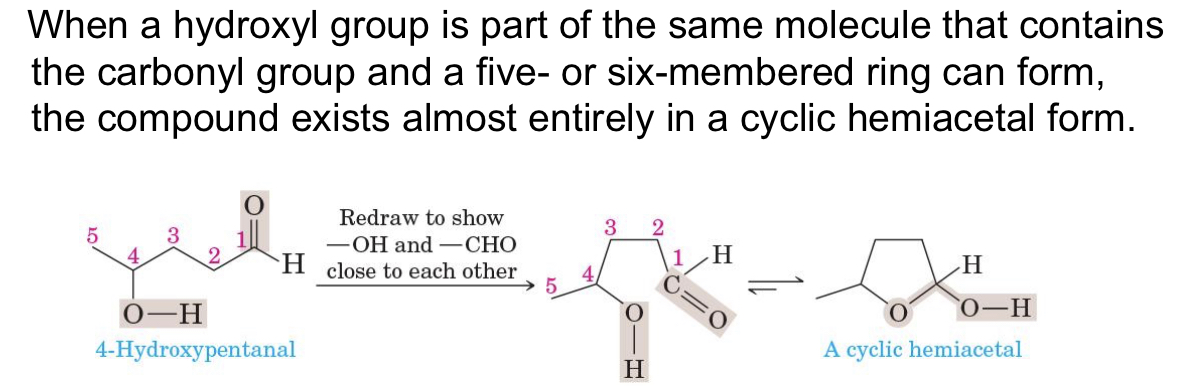
describe hemiacetals
unstable, minor component of equilibrium mixture
how does cyclic hemiacetal exist (3 things)
OH, C=O, 5 to 6 carbon ring
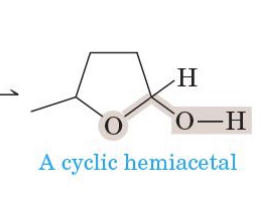
how are acetals formed
hemiacetal + alcohol. acetal formed along w water
all steps in hemiacetal and acetal formation are
reversible
enol
aldehyde/ketone with hdyrogen on alpha carbon, in equilibrium with constitutional isomer (different bonding)
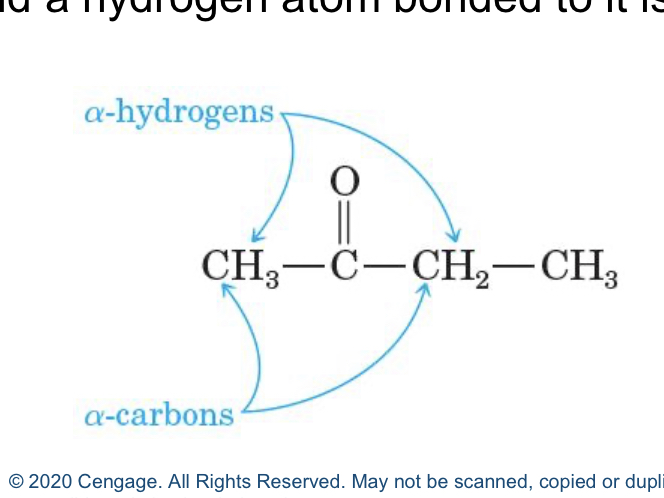
alpha carbon
carbon near carbonyl group
alpha hydeogen
hydrogen near the carbon that's near the carbonyl group
what form dominates in keto enol equilibrium
keto form
acetal formation (6)
hydrogen proton added to oxygen in c=o -> carbon in c=o becomes stronger electrophile but more susceptible to nucleophile attack
electrophile + nucleophile = covalent bond. adds first OR group required for acetal formation. intermediate = oxonium.
hydrogen proton transferred to another oxygen
bonds break to form stable molecules/ions
electrophile + nucleophile = covalent bond. OR group added to carbonyl carbon
proton talen away. regenerates H+ = acetal
general formula of aldehyde:
R-CHO (H attached near C=O) * is this true
oxidation: alcohol vs. aldehyde
primary alcohol oxidates -> aldehyde/ketone
aldehyde oxidates -> carboxylic acid
primary alcohol -> aldehyde -> carboxylic acid
reduction: alcohol vs. aldehyde
alcohol reduces -> alkane
aldehyde reduces -> primary alcohol
important reactions for structures: NAME THE PROCESSES (4)
oxidation
2. reduction
3. acetal formation
4. hemiacetal formation
how to understand structural reaction for aldehyde/ketone stuff
track carbonyl CARBON
⁃ oxidation adds oxygen or removes hydrogen