Electrode Potentials Theory
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
if you place two metals in a salt solution and connect them together, what happens?
electric current flows so that electrons pass from the more reactive metal to the less reactive.
why is a salt bridge used instead of a piece of wire in an electrical circuit
a wire only transfers electrons wheres a salt bridge transfers ions
what 2 metals form a Daniell cell?
zinc and copper
a stronger reducing agent will - electrons
lose electrons
porous pot can act as what
a salt bridge
what is used to compare the tendency of different metals to release electrons
the Standard Hydrogen Electrode
list all the components of the standard hydrogen electrode
platinum wire, hydrogen gas bubbled in (100kPa), finely divided platinum black, 1moldm^-3 HCl, salt bridge
explain the function of the salt bridge (2)
the ions in the ionic substance in the salt bridge move through the salt bridge to maintain charge balance
equation for hydrogen-oxygen fuel cell operating under alkaline conditions
O2 + 4H+ + 4e- → 2H2O
suggest the major advantage of using hydrogen as a fuel for cars via fuel cells instead of an internal combustion engine
internal combustion engine wastes a greater proportion of the available thermal energy
give the standard hydrogen electrode cell representation (as if it were in a full cell representation, use / as lines)
//H+(aq)/H2(g)/Pt(s)
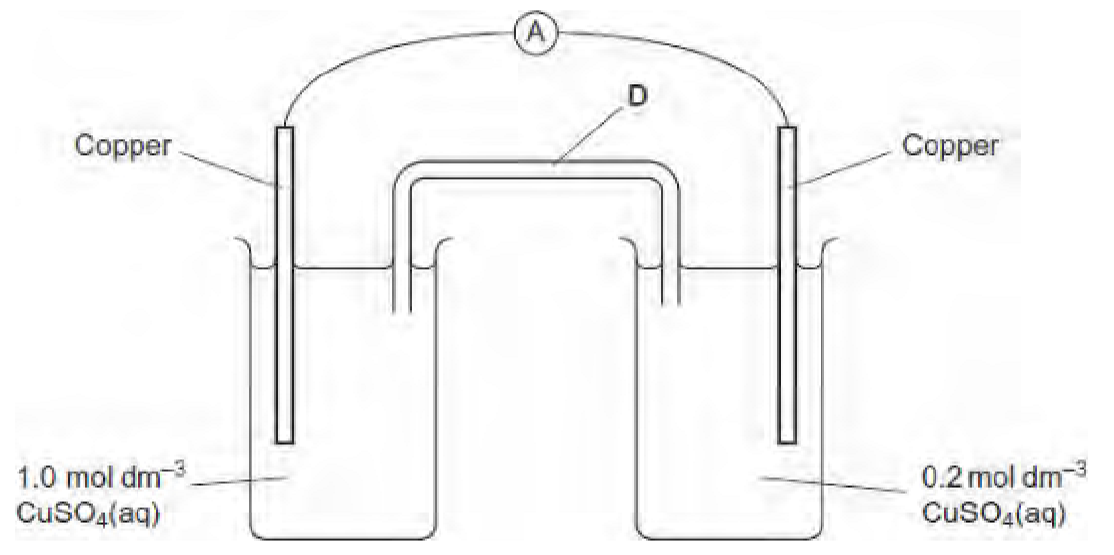
why would the current fall to zero after the cell has operated for some time?
eventually the concentration of CuSO4 in each electrode will be the same
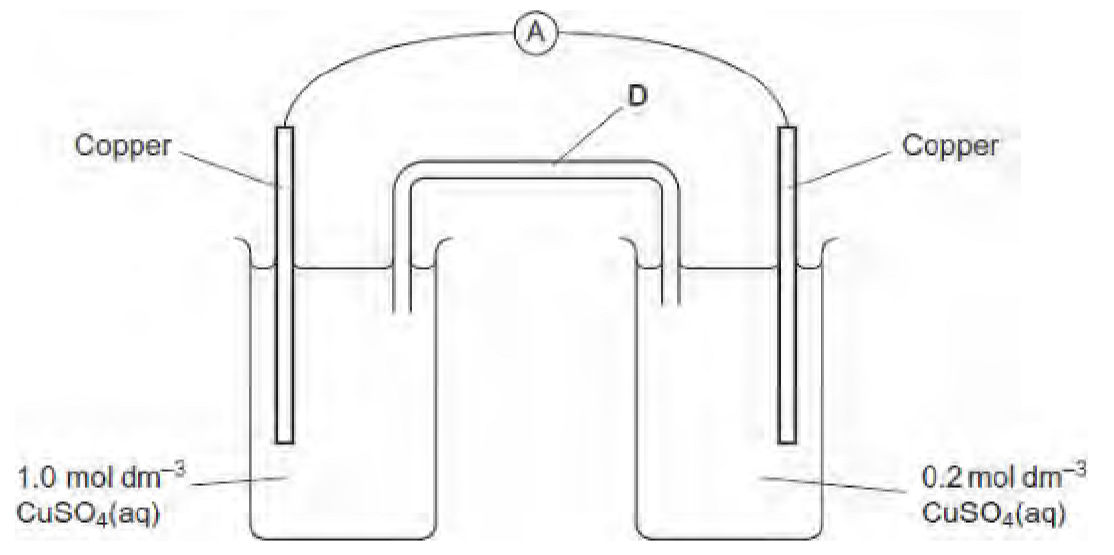
why would the electrons move from right to left?
the oncentration of Cu2+ ions is higher in the left half-cell. reaction of Cu2+ with 2e- will occur in preference at the left hand electrode.
give the half equations for the electrode reactions in an alkaline hydrogen-oxygen fuel cell:
H2 + 2OH- → 2H2O + 2e-. O2 + 4e- + 2H2O → 4OH-
how is an electric current generated in a hydrogen oxygen fuel cell
hydrogen electrode produces electrons. oxygen electrode accepts electrons.
why does a fuel cell not need to be recharged
there is a continuous supply of fuel
two major hazards associated with the use of a hydrogen-oxygen fuel cell in a vehicle
hydrogen is flammable, H+ is corrosive
why are rechargeable cells connected to solar cells which provide electrical energy for illuminated road signs?
solar cells do not supply energy all the time. rechargeable cells can store electrical energy for when the cells are not working.
give one reason why many waste disposal centres contain a separate section for cells and batteries
pollution of the environment by toxic substances
what electrode is used for the Fe3+ Fe2+ half cell
platinum
conditions for measuring emf
1kPa, 298K
deduce one essential property of the non-reactive porous separator labelled in the diagram
allows ions to pass through it
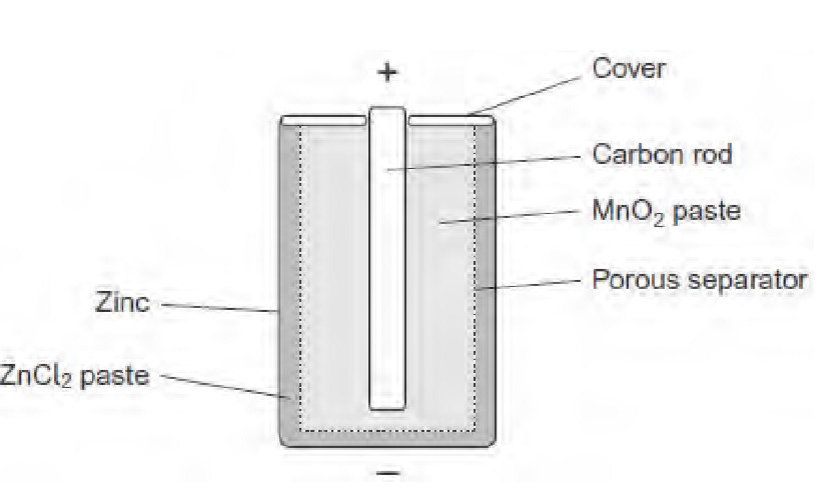
function of the carbon rod in this cell
inert electrode
why would a cell leak after being used for a long time
the metal container for the cell has reacted
why would the emf of a lead acid cell change after several hours
reagents are used up
why does the voltage remain constant in a fuel cell
the concentration of reagents remains constant
what flows in and out of a hydrogen oxygen fuel cell?
hydrogen and oxygen in, water out
what connects the two half cells in a hydrogen-oxygen fuel cell?
an electrolyte which carries charge
how does an electrolyte connect the half cells in an alkaline hydrogen oxygen fuel cell
facilitating the flow of OH- ions from the oxygen half cell to the hydrogen half cell
what is the only part of the fuel cell which remains in the fuel cell?
the electrolyte
what is the only part of the fuel cell which may need to be occassionally replaced?
the electrolyte
which is the negative terminal of the hydrogen oxygen fuel cell?
the hydrogen electrode
what reaction occurs in an alkaline hydrogen oxygen fuel cell at the negative electrode
2OH-(aq) + H2(g) → 2H2O(l) + 2e-
what is the positive terminal of an alkaline hydrogen-oxygen fuel cell?
the oxygen electrode
what reaction occurs in an alkaline hydrogen-oxygen fuel cell at the positive terminal?
1/2O2(g) + H2O(l) +2e- → 2OH-(aq)
what is the full equation for the hydrogen oxygen fuel cell?
1/2O2(g) + H2(g) → H2O(l)
how does an electrolyte connect the half cells in an alkaline hydrogen oxygen fuel cell
facilitates the flow of H+ ion from the hydrogen half cell to the oxygen half cell
what reaction occurs in the hydrogen half cell of an acidic hydrogen-oxygen fuel cell
H2(g) → 2H+(aq) + 2e-
what reaction occurs in the oxygen half cell of an acidic hydrogen-oxygen fuel cell
1/2O2(g) + 2H+(aq) + 2e- → H2O(l)
what is the overall reation for the acidic hydrogen oxygen fuel cell
1/2O2(g) +H2(g) → H2O(l)
why might hydrogen-oxygen fuel cells NOT be regarded as a carbon neutral source of energy
H2 may be made using an energy source that is not carbon neutral
when the standard electrode potential for Fe3+/Fe2+ is measured, a platinum electrode is requried. what is the function of the platinum electrode.
provide a reaction surface
SHE always goes on the
left
the electrode with the more negative E standard value always goes on
the left
reduction occurs at which electrode?
positive
oxidation occurs at which electrode
negative
for the Fe2+/Fe3+ electrode, what 2 key elements are needed
platinum electrode, mixture of soluble Fe2+ and Fe3+ compounds both at 1 moldm^-3
3 marks for measuring change in mass of an electrode
allow cell to discharge until [reagent] has halved. use an experiment to determine [reagent], i.e. colorimetry. measure electrode mass before and after.
why may the recharging of the lithium cell produce CO2
energy needed to recharge may come from the combustion of carbon containing compound like fossil fuels
why would K2CO3 not be used for a salt bridge when the SHE is used
the carbonate ions react with the acid present
the reaction which occurs at the positive electrode of the lithium cell
Li^+ + CoO2 + e^- → Li^+[CoO2]^-
the reaction which occurs at the negative electrode of the lithium cell
Li → Li^+ + e^-