33 - carboxylic acid and derivatives
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
production of benzoic acid
Benzoic acid is the simplest aromatic carboxylic acids with the molecular formula of C6H5COOH
Benzoic acid and its derivatives are often used as reagents in the synthesis of esters
The compounds can be produced from the oxidation of alkylbenzenes
Oxidation of alkylbenzenes
The alkyl side-chain in alkylbenzenes, such as methylbenzene, can be oxidised to a carboxylic acid
The alkylbenzene is heated under reflux with a solution of hot alkaline KMnO4 (this is the oxidising agent)
The purple colour of the Mn7+ ions disappears as they are reduced to Mn4+ions
A brown precipitate of MnO2 is formed
The mixture is then acidified with dilute acid (such as hydrochloric acid or sulfuric acid) to protonate the organic product form and produce a benzoic acid
Making benzoic acid from methylbenzene

Alkylbenzenes such as methylbenzene undergo oxidation to form benzoic acid
reactions of carboxylic acids to produce acyl chlorides
Acyl chlorides are compounds with the functional group -COCl
They look similar in structure to carboxylic acids but have a Cl atom instead of an -OH group attached to the carbonyl (C=O)
Acyl chlorides are more reactive than their corresponding carboxylic acids and are therefore often used as starting materials in the production of organic compounds such as esters
They can be prepared from the reaction of carboxylic acids with:
Solid phosphorus(V) chloride (PCl5)
Liquid phosphorus(III) chloride (PCl3) and heat
Liquid sulfur dichloride oxide (SOCl2)
For example, the acyl chloride ethanoyl chloride can be formed from ethanoic acid in the above reactions
Using ethanoic acid to form ethanoyl chloride
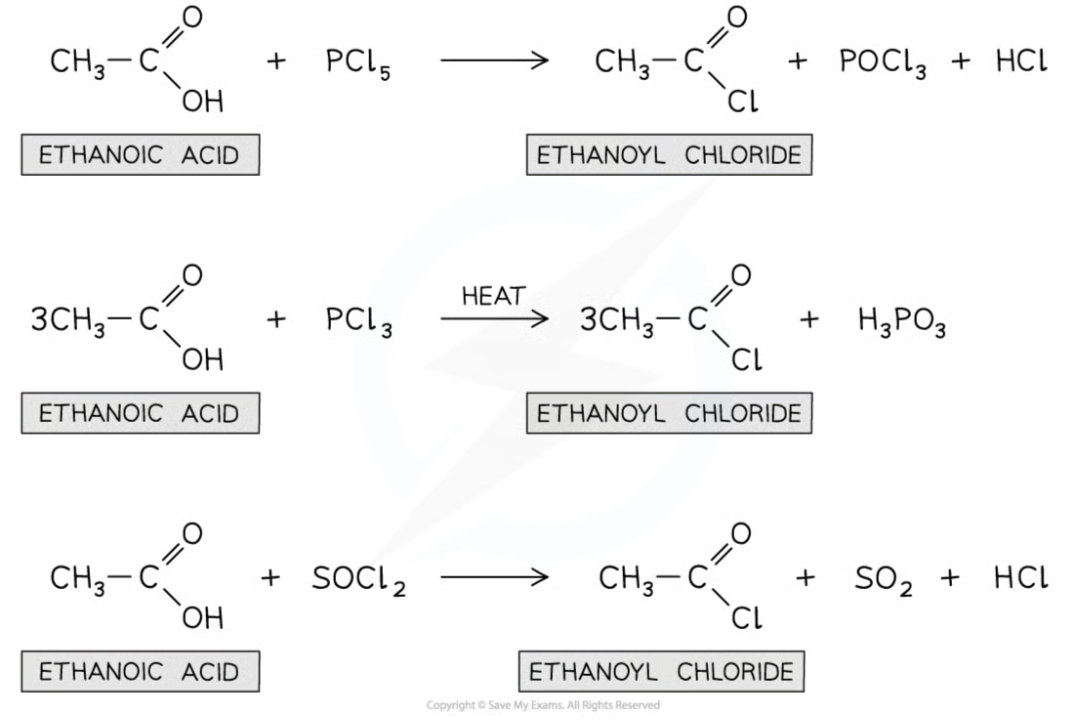
Ethanoic acid can be used to produce ethanoyl chloride with different by-products depending on the reagent used
further oxidation of carboxylic acids
Carboxylic acids can be formed from the oxidation of primary alcohols
The primary alcohols are firstly oxidised to aldehydes and then further oxidised to carboxylic acids
Some carboxylic acids can get even further oxidised
Methanoic acid
Methanoic acid is a strong reducing agent and gets further oxidised to carbon dioxide (CO2)
The oxidation of methanoic acid can occur by:
Warming methanoic acid with mild oxidising agents such as Fehling’s or Tollens’ reagent
In a Fehling’s solution, the Cu2+ ion is reduced to Cu+ ion which precipitates as red Cu2O
With Tollens’ reagent, the Ag+ is reduced to Ag
Using stronger oxidising agents such as acidified KMnO4 or acidified K2Cr2O7
The purple KMnO4 solution turns colourless as Mn7+ ions are reduced to Mn2+ ions
The orange K2Cr2O7 solution turns green as the Cr6+ ions are reduced to Cr3+ ions
Ethanedioic acid
Another carboxylic acid that can get further oxidised is ethanedioic acid
A strong oxidising agent such as warm acidified KMnO4 is required for the oxidation of ethanedioic acid to carbon dioxide
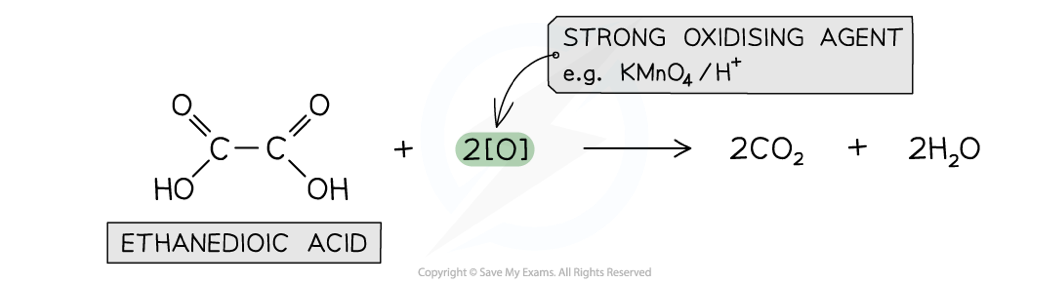
relative acidities of carboxylic acids, phenols and alcohols
Carboxylic acids are compounds with a -COOH functional group
They can act as acids and lose a proton (H+ ion) in an aqueous solution to form carboxylate salts and water
Carboxylic acids forming carboxylate salts

Carboxylic acids dissociate in aqueous solutions to form carboxylate salts and water
However, carboxylic acids are only weak acids as the position of equilibrium lies well over to the left-hand side
The pKa values of carboxylic acids, phenols, and alcohols suggest that carboxylic acids are stronger acids than alcohols and phenols
The pKa is a measure of the relative strength of a species as an acid
The smaller the pKa value, the stronger the acid
Relative acidity of ethanol, phenol & carboxylic acids table
Acid | Dissociation | pKa at 25 oC |
|---|---|---|
Ethanol | C2H5OH (aq) C2H5O– (aq) + H+ (aq) | 16 |
Phenol | C6H5OH (aq) C6H5O– (aq) + H+ (aq) | 10 |
Ethanoic acid | CH3COOH (aq) CH3COO– (aq) + H+ (aq) | 4.8 |
Benzoic acid | C6H5COOH (aq) C6H5COO– (aq) + H+ (aq) | 4.2 |
This order of relative acidities can be explained by looking at the strength of the O-H bond and the stability of the conjugate bases of the acids
explaining the relative acidities of carboxylic acids, phenols and alcohols by looking at the strength of the O-H bond
In carboxylic acids, the electrons in the O-H bond are drawn towards the C-O bond
The electrons in the C-O bond are drawn towards the C=O bond
Overall, the O-H bond is weakened due to the carbonyl (C=O) group removing electron density from it and drawing it towards itself
Carboxylic acids can therefore more easily lose a proton compared to phenols and alcohols which lack this electron-withdrawing carbonyl group
Comparing OH bond strength of carboxylic acids, ethanol and phenol

The carbonyl group in carboxylic acids draws the electrons away from the O-H bond causing it to become weaker compared to the O-H bond in phenols and alcohols
explaining the relative acidities of carboxylic acids, phenols and alcohols by looking at the stability of the conjugate bases of the acids : carboxylate ions
The conjugate base of carboxylic acids is the carboxylate ion
The charge density on the oxygen atom is spread out over the carboxylate ion
This is because the charge is delocalised on an electronegative carbonyl oxygen atom
As a result, the electrons on the oxygen atom are less available for bond formation with an H+ ion to reform the undissociated acid molecule with -COOH group
The position of the dissociation equilibrium lies more to the right compared to alcohols and phenols
The equilibrium position of a carboxylic acid and its carboxylate ion

The carboxylate ion is stable due to the delocalisation of the charge density on the electronegative oxygen
explaining the relative acidities of carboxylic acids, phenols and alcohols by looking at the stability of the conjugate bases of the acids : alkoxide ions
The conjugate base of alcohols is the alkoxide ion
The alkyl group in the ion is an electron-donating group that donates electron density to the oxygen atom
As a result, the electron density on the oxygen atom is more readily available for bond formation with an H+ ion
Alkoxide ions also lack the ability to delocalise the charge density on the entire ion
The conjugate bases of alcohols are therefore less stable than the alcohols themselves and are more likely to reform the alcohol
This means that alcohols are weaker acids compared to carboxylic acids and phenols
The position of the dissociation equilibrium lies more to the left
The equilibrium position of an alcohol and its alkoxide ion

The electron-donating alkyl groups in alkoxide ions increase the electron density on the oxygen atom which is, therefore, more likely to bond with a H+ ion and reform the alcohol
explaining the relative acidities of carboxylic acids, phenols and alcohols by looking at the stability of the conjugate bases of the acids : phenoxide ions
In the phenoxide ion (which is the conjugate base of phenol) the charge density on the oxygen atom is spread out over the entire ion
This delocalisation of electrons stabilises the phenoxide ion
As a result, the electrons on the oxygen atom are less available for bond formation with a proton (H+ ion)
The conjugate base of phenols is therefore more stable than phenol
However, since the delocalisation of charge density is on carbon atoms and not on electronegative oxygen atoms like in the carboxylate ion, phenoxide ions are less stable than carboxylate ions
Therefore, phenols are weaker acids relative to carboxylic acids
The position of the dissociation equilibrium lies more to the right compared to alcohols and more to the left compared to carboxylic acids
The equilibrium position of phenol and the phenoxide ion

The charge density is delocalised on the entire benzene ring in the phenoxide ions
relative acidities of chlorine - substituted carboxylic acids overview
Electron-withdrawing groups bonded to the carbon attached to the -COOH group make the carboxylic acids stronger acids
This is because the O-H bond in the undissociated acid molecule is even further weakened as the electron-withdrawing group draws even more electron density away from this bond
Furthermore, the electron-withdrawing groups extend the delocalisation of the negative charge on the -COO- group of the carboxylate ion
The -COO- group is now even more stabilised and is less likely to bond with an H+ion
Chlorine-substituted carboxylic acids are examples of carboxylic acids with electron-withdrawing groups
pKavalues of ethanoic acid and chlorine-substituted derivatives table
Acid | pKa at 25 oC |
|---|---|
Ethanoic acid, CH3COOH | 4.8 |
Chloroethanoic acid, CH2ClCOOH | 2.9 |
Dichloroethanoic acid, CHCl2COOH | 1.3 |
Trichloroethanoic acid, CCl3COOH | 0.6 |
The pKa values of ethanoic acid and chloro-substituted derivatives show that the more electron-withdrawing groups there are on the carbon attached to the -COOH group, the stronger the acid
Comparing the relative acidities of chlorine substituted derivatives of ethanoic acid
Trichloroethanoic acid is the strongest acid as:
The O-H bond in CCl3COOH is the weakest since there are three very strong electronegative Cl atoms withdrawing electron density from the -COOH group
When the O-H is broken to form the carboxylate (-COO-) ion, the charge density is further spread out by the three electron-withdrawing Cl atoms
The carboxylate ion is so stabilised that it is less attracted to H+ ions
The equilibrium of trichloroethanoic acid and the trichloroethanoate ion

Relative acidity of trichloroethanoic acid
Ethanoic acid is the weakest acid as:
It contains an electron-donating methyl group which strengthens the O-H bond
The methyl group donates negative charge towards the -COO- group which becomes more likely to accept an H+ ion
The equilibrium of ethanoic acid and the ethanoate ion

Relative acidity of ethanoic acid
Production of Esters by Reacting Alcohols With Acyl Chlorides
Esters are organic compounds with an -COOR functional group
They have characteristic smells and are used in perfumes, cosmetics and as solvents
Esters can be prepared from the condensation reaction between alcohols and carboxylic acids
This is also called an esterification reaction
A more effective way of preparing esters is from the condensation reaction between alcohols and acyl chlorides instead
Unlike the reactions with carboxylic acids, acyl chlorides are more reactive (so the reactions happen faster) and their reactions go to completion (so no equilibrium mixture is formed and the yield of the ester is maximum)
Examples of esterification reactions include:
Formation of ethyl ethanoate from ethanol and ethanoyl chloride
Formation of phenyl benzoate from phenol and benzoyl chloride
Formation of esters from the reaction of alcohols with acyl chlorides

The first part of the ester name comes from the alcohol and the second part of the ester name comes from the acyl chloride
production of acyl chlorides
Due to the increased reactivity of acyl chlorides compared to carboxylic acids, they are often used as starting compounds in organic reactions
Acyl chlorides are compounds that contain an -COCl functional group and can be prepared from the reaction of carboxylic acids with:
Solid phosphorus(V) chloride (PCl5)
Liquid phosphorus(III) chloride (PCl3) and heat
Liquid sulfur dichloride oxide (SOCl2)
Propanoyl chloride can this way be prepared from propanoic acid using the reactions above
Using propanoic acid to form propanoyl chloride

Propanoic acid can be used to produce propanoyl chloride with different by-products depending on the reagent used
overview of the reactions of acyl chlorides
Acyl chlorides are reactive organic compounds that undergo many reactions such as addition-elimination reactions
In addition-elimination reactions, the addition of a small molecule across the C=O bond takes place followed by elimination of a small molecule
Examples of these addition-elimination reactions include:
Hydrolysis
Reaction with alcohols and phenols to form esters
Reaction with ammonia and amines to form amides
hydrolysis of acyl chlorides
The hydrolysis of acyl chlorides results in the formation of a carboxylic acid and HCl molecule
This is an addition-elimination reaction
A water molecule adds across the C=O bond
A hydrochloric acid (HCl) molecule is eliminated (hydrogen chloride gas but it is dissolved in solution)
An example is the hydrolysis of propanoyl chloride to form propanoic acid and HCl
Hydrolysis of acyl chlorides

Acyl chlorides are hydrolysed to carboxylic acids
formation of esters with acyl chlorides
Acyl chlorides can react with alcohols and phenols to form esters
The reaction with phenols requires heat and a base
Esters can also be formed from the reaction of carboxylic acids with phenol and alcohols however, this is a slower reaction as carboxylic acids are less reactive and the reaction does not go to completion (so less product is formed)
Acyl chlorides are therefore more useful in the synthesis of esters
The esterification of acyl chlorides is also an addition-elimination reaction
The alcohol or phenol adds across the C=O bond
A HCl molecule is eliminated
Esterification reactions using acyl chlorides

Acyl chlorides undergo esterification with alcohols and phenols to form esters
formation of amides
Acyl chlorides react with ammonia or primary amines to form amides in a condensation reaction.
A lone pair on the nitrogen atom attacks the carbonyl carbon in the acyl chloride.
The reaction proceeds via a nucleophilic addition–elimination mechanism:
The nucleophile adds to the C=O bond
A chloride ion (Cl⁻) is eliminated
Hydrogen chloride (HCl) is formed
What happens to the HCl?
The HCl formed does not remain unreacted.
It is immediately neutralised by a second molecule of ammonia or amine present in excess.
This forms an ammonium salt (e.g. NH₄Cl, CH₃NH₃Cl).
Why 2 molecules are needed
The 1st molecule of ammonia/amine forms the amide
The 2nd molecule of ammonia/amine neutralises the HCl and forms ammonium salt
Examples
Reaction with ammonia
Product: Primary amide (propanamide) and ammonium chloride
CH3CH2COCl + 2NH3 → CH3CH2CONH2 + NH4Cl
Reaction with methylamine
Product: Secondary (substituted) amide and methylammonium chloride
CH3COCl + 2CH3NH2 → CH3CONHCH3 + CH3NH3Cl
Reaction with ethylamine
Product: Secondary (substituted) amide and ethylammonium chloride
CH3COCl + 2CH3CH2NH2 → CH3CONHCH2CH3 + CH3CH2NH3Cl
Summary for formation of amides
All reactions form HCl, which is not observed as a separate product
The HCl is neutralised by excess ammonia or amine
The final products are an amide and an ammonium salt
addition elimination in acyl chloride reactions mechanism
Acyl chlorides undergo addition-elimination reactions such as hydrolysis, esterification reactions to form esters, and condensation reactions to form amides
The general mechanism of these addition-elimination reactions involves two steps:
Step 1 - Addition of a nucleophile across the C=O bond
Step 2 - Elimination of a small molecule such as HCl or H2O
mechanism of hydrolysis of acyl chlorides
In the hydrolysis of acyl chlorides, the water molecule acts as a nucleophile
The lone pair of the oxygen atom from water carries out an initial attack on the carbonyl carbon
This is followed by the elimination of a hydrochloric acid (HCl) molecule
Reaction mechanism of the hydrolysis of acyl chlorides

The two-step addition-elimination reaction mechanism of propanoyl chloride to form propanoic acid
mechanism for formation of esters with acyl chlorides
In the esterification reaction of acyl chlorides, the alcohols or phenols act as a nucleophile
The lone pair of the alcohol / phenol oxygen atom carries out an initial attack on the carbonyl carbon
This is again followed by the elimination of an HCl molecule
With phenols, the reaction requires heat to proceed and needs to be carried out in the presence of a base
The base deprotonates the phenol to form a phenoxide ion which is a better nucleophile than the phenol molecule
The phenoxide ion carries out an initial attack on the carbonyl carbon
A small molecule of NaCl is eliminated
Reaction mechanism of the esterification of acyl chlorides with alcohols

The two-step addition-elimination reaction mechanism of propanoyl chloride and ethanol to form ethyl propanoate and water
Reaction mechanism of the esterification of acyl chlorides with phenols


The three-step addition-elimination reaction mechanism of propanoyl chloride with phenol to form phenyl propanoate
mechanism for the formation of amides
The nitrogen atom in ammonia and primary/secondary amines act as a nucleophile
The lone pair of the nitrogen atom carries out an initial attack on the carbonyl carbon
This is followed by the elimination of an HCl molecule
Both reactions of acyl chlorides with ammonia and amines are vigorous however there are also differences
With ammonia - The product is a non-substituted amide and white fumes of HCl are formed
With amines - The product is a substituted amide and the HCl formed reacts with the unreacted amine to form a white organic ammonium salt
Reaction mechanism of the formation of amides from acyl chlorides with ammonia

The two-step addition-elimination reaction mechanism of propanoyl chloride and ammonia to form propanamide
Reaction mechanism of the formation of amides from acyl chlorides with primary amines


The addition-elimination reaction mechanism of propanoyl chloride and methylamine to form methylpropanamide
Reaction mechanism of the formation of amides from acyl chlorides with secondary amines


The addition-elimination reaction mechanism of propanoyl chloride and dimethylamine to form dimethylpropanamide
overview of the ease of hydrolysis for different organic compounds and how it can be explained
Hydrolysis is the breakdown of a compound using water
The ease of hydrolysis for different organic compounds may differ
For example, the ease of hydrolysis, starting with the compounds most readily broken down, is: acyl chloride > alkyl chloride > aryl chloride
This trend can be explained by looking at the strength of the C-Cl
strength of c-cl bond in acyl chlorides
Acyl chlorides are hydrolysed most readily at room temperature
This is because the carbon bonded to the chlorine atom is also attached to an oxygen atom
There are two strong electronegative atoms pulling electrons away from the carbonyl carbon, leaving it very δ+
The C-Cl bond is therefore weakened and nucleophilic attack of the carbonyl carbon is much more rapid
Hydrolysis of acyl chlorides

The hydrolysis of acyl chlorides occurs most readily
strength of the c-cl bond in alkyl chlorides
The carbonyl carbon in alkyl chlorides is only attached to one electronegative atom which pulls electrons away from it
This carbon atom is therefore not very δ+ and the C-Cl bond is stronger than the C-Cl bond in aryl chlorides
The hydrolysis of alkyl chlorides, therefore, requires a strong alkali (such as OH-) to be refluxed with it
An OH- ion will hydrolyse the alkyl chloride as it is a stronger nucleophile than H2O
Hydrolysis of alkyl chlorides

The hydrolysis of alkyl chlorides requires a strong nucleophile
strength of the c-cl bond in aryl chlorides
In aryl chlorides, the carbon atom bonded to the chlorine atom is part of the delocalised π bonding system of the benzene ring
One of the lone pairs of electrons of the Cl atom overlaps with this delocalised system
The C-Cl bond, therefore, has some double-bond character causing it to become stronger
As a result, the C-Cl bond is difficult to break and hydrolysis will not occur
Hydrolysis of aryl chlorides

Due to the strong C-Cl bond in aryl chlorides, these compounds will not undergo hydrolysis