Chapter 7: Monosaccharides
1/42
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
Carbohydrates are some of the most __ molecules on earth
Abundant!
Why do bacteria need rigid cell walls?
If they didn’t, water would flood their cells and they would burst due to osmotic pressure
Name a few functions of carbohydrates
Fuel
Energy storage
Structure
Barrier/lubricant
Cell signaling
Cell adhesion
Cellular identity
What is sialic acid used for?
To determine the “age” of RBC in our system, to know it if should be turned over or not
What decorates the surface of the COVID spike protein?
Carbohydrates! They help the virus evade our immune system
Saccharides carbonyls come in what two forms?
Aldehydes or ketones (aldoses and ketoses)
The simplest monosaccharide contains how many C?
3!
Glucose is a __hexose. Fructose is a __hexose.
aldohexose, ketohexose
Draw D-glucose
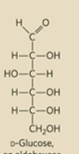
Draw D-fructose
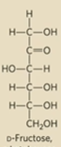
What are the L and D names based on?
The stereochemistry of the simplest saccharide, Glyceraldehyde
What does a horizontal line mean on a Fisher Projection? What about vertically?
Horizontal: towards us
Vertical: plane of paper
In a Fisher Projection, how can you tell L or D stereochemistry?
D: Carbonyl at top, most distant -OH towards right
L: Carbonyl at top, most distant -OH towards left
The most distant is NOT the CH2OH
How many isomers exist for a monosaccharide with n chiral centers?
2n
What configuration are most sugars in? What configuration are most amino acids in?
Sugars: D
Amino Acids: L
Epimer definition
A different configuration at only ONE carbon, and NOT the one that determines L or D.
How to tell the difference between an aldose and a ketose in the name of the sugar?
a -ul is inserted in the ketone name
Ex: D-Ribose vs D-Ribulose
Ribulose is a ketone, ribose is an aldehyde
Draw D-Ribose
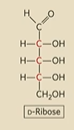
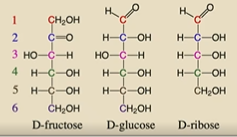
What 3 sugars do we need to remember?
D-fructose, D-glucose, D-ribose
Which form is dominant in aqueous solution?
Cyclic
The formation of sugar rings is an…
intramolecular cyclization
What do we call the two possible anomers of a cyclic form of a saccharide? Why can we get get these two forms?
Alpha and Beta. The -OH group can attack the carbonyl either from the front or back.
What is the most abundant form of glucose in solution?
B-D-glucopyranose
Pyranose=? Furanose=?
6 membered ring, 5 membered ring
Both with one O as part of the ring
How do we define alpha or beta? How do we determine L or D in cyclic form?
L: Terminal carbon (the CH2OH carbon) is pointing down
D: Terminal carbon is pointing up
Beta: -OH pointing same way
Alpha: -OH opposite same way (ANTIparallel)
Mutarotation definition
Interconversion of alpha and beta anomers (ring opens and closes all the time, alpha and beta forms in a certain equilibrium, often favoring one of them)
Draw a phosphate group
Draw alpha-D-deoxyribose
How are disaccharides formed?
The -OH of one monomer attacks one of the carbons of another, forming an O-glycosidic bond
Formation of a disaccharide is an example of what kind of reaction?
Condensation
What is the reducing end of a multiple saccharide chain?
the hemiacetal residue, the anomeric -OH is still available to attack another monomer
Name the abbreviations for fructose, glucose and ribose
Fru, Glc, Rib
What does GlcNAc mean
N-acetylglucosamine
What does GlcN mean
Glucosamine
What are the 3 rules for naming di saccharides?
Put the non-reducing residue at the left
Name the reducing sugar on the right
Describe the linkage of the glycosidic bond
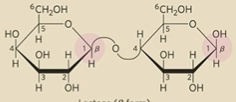
What type of glycosidic bond is lactose
Gal(B1 →4) Glc
What is interesting about sucrose?
It’s a disaccharide with no reducing end
How does Benedicts solution work?
If there is a reducing end present, the Cu+2 in solution will oxidize, and the solution will become red. Needs the linear form though but that’s fine because the ring is always opening and closing
What is a homopolysaccharide?
Polymer made of only one monosaccharide
Name the glycosidic linkages of glycogen, starch, cellulose and chitin
Glycogen: (alpha1→4)Glucose
Starch: (Alpha1→4)glucose
Cellulose: (B1→4)Glucose
Chitin:(B1→4)GlcNac
Sooo why can’t we eat our pants?
Don’t have a digestive enzyme for the B1→4 linkage
How can O-glycosidic bonds be broken?
Boiling with acid or heating up in general
How can you analyze the type of glycosidic bond present?
Exhaustive methylation of all the -OH groups, cleavage with acid, then see which carbons have an -OH still (those were the ones involved in the linkage)