Unit 3 Chem
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
the smallest representative particle of an element
atom
the three particles the atom is made of
protons, neutrons, and electrons
letter for atomic number
Z
letter for atomic mass
A
the weighted mass of all the isotopes of an element
average atomic mass
equation for average atomic mass
average atomic mass = %A/100 x (mass A) + %B/100 x (mass B) + …
positive ion
cation
negative ion
anion
which particle determines the radioactivity of the atom
electron
particles occupying the nucleus
nucleons
atoms of the same element with different masses
isotopes
forces of attraction between nucleons
strong forces
force of repulsion between protons
electrical force
spontaneously emits a particle and decays into another element
a radioactive isotope
the changing into another element through radioactive decay
transmutation

what symbol is this?
alpha

what symbol is this?
beta

what symbol is this?
gamma
what is the chemical symbol for alpha?
2/4 H or 2/4 alpha
what is the chemical symbol for beta?
0/-1 e or 0/-1 beta
what is the chemical symbol for gamma?
0/0 gamma or gamma
which type of radioactive emission is pure energy
gamma
which type of radioactive emission is the weakest
alpha
which type of radioactive emission is in the middle for strength
beta
which type of radioactive emission is the strongest
gamma
the time it takes for half the mass of a radioactive sample to decay
half-life
bombardment of a stable isotope to force it to decay
artificial transmutation
heavy nuclei are bombarded with neutrons and split
nuclear fission
combination of two nuclei into one with release of energy
nuclear fusion

which scientists model is this?
Dalton

which scientists model is this?
Thomson
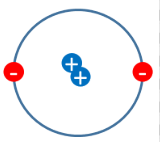
which scientists model is this?
Rutherford
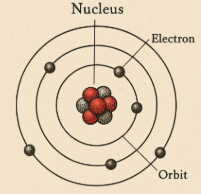
which scientists model is this?
Bohr