intro to biology & chemistry for vet techs
1/116
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
117 Terms
systems biology
study of organisms and how they interact with their environment/other organisms
homeostasis
the stability of internal conditions
null hypothesis
initial hypothesis
element
a specific substance that cannot be broken down into simpler substances by chemical reactions
how many total elements are there?
118
how many elements are naturally occurring?
94
nucleus
central region of an atom
neutron
no charge particles within nucleus
proton
positively charged particles within nucleus
electrons
negatively charged particles scattered/orbiting around/outside the nucleus
atom
the smallest component of an element that retains all chemical properties of that element
the number of electrons is ____ to the number of protons
equal to
valence electrons
exist in the outer shell, important for chemical reactions
the number of protons is ____ to the atomic number
equal to
atomic mass
(number of protons) + (average number of neutrons)
isotopes
when atoms of the same element have the same number of protons but different numbers of neutrons
ionic bonds
involve 1 atom losing 1 or more electrons and another atom gaining the electrons that were lost by the first atom
ion
atom with a charge
anion
ion w/ negative charge
cation
ion w/ positive charge
covalent bond
when electrons are shared to achieve a full outer shell
molecules
the result of atoms being joined by covalent bonds (smallest unit of a compound)
compound
contain @ least two differeent elements
nonpolar covalent bonds
when two covalently bonded atoms share electrons nearly equally (ex: oxygen molecule O2)
polar covalent bonds
when electrons are shared unequally due to a difference in charge between the atoms, developing a slightly positive/negative charge at each pole of the bond (ex: water molecule H2O)
cohesion
water attracted to itself (ex: surface tension)
adhesion
water attracted to other substances (ex: towel soaking up water)
acid
substance that increases the concentration of hydrogen ions in a solution
base
provides hydroxide ions (or other negatively charged ions) that combine with hydrogen ions, therefore decreasing their concentration in a solution
pH
indicates acidity or alkalinity of a solution
weak bases or acids will only
partially dissociate
pH ranges from
0 to 14
on the pH scale of 0 to 14, 0 is ___ and 14 is ___
acidic, basic
7 on the pH scale is?
neutral
a pH change of one unit indicates a change of how much in hydrogen ion concentration?
tenfold
equation to solve for hydrogen ion concentration
[H+] = 10-pH
the 4 types of macromolecules
proteins, carbohydrates, lipids, nucleic acids
what atom forms the basis of life and is central to compounds and molecules that impact biological functions?
carbon
made of amino acids, these control the structure and function of cells
proteins
amino acids
molecules with a central carbon that binds to a hydrogen atom, two functional groups, and a variable side group (R group)
R group
unique chemicla properties that makes each amino acid its own
chains of many amino acids connected by peptide bonds becomes this:
a polypeptide chain
amino acids that can’t be synthesized by the body (and need to be obtained via diet) are considered
essential amino acids
peptides are made from
two or more amino acids
how are peptide bonds formed?
via a dehydration reaction, involving the loss of a water molecule
how are amino acids polymerized into peptides and proteins?
through the formation of a peptide bond
nonessential amino acids
made by the body
this class of proteins catalyzes reactions
enzymes
these proteins are essential for the immune system
antibodies
these proteins make up cells (ex: keratin forming hair and nails)
structural proteins
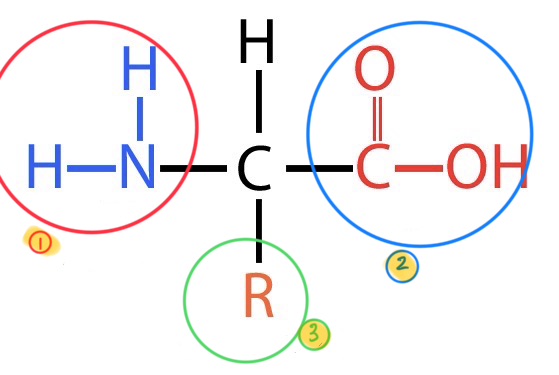
what does this image depict?
the basic breakdown of an amino acid
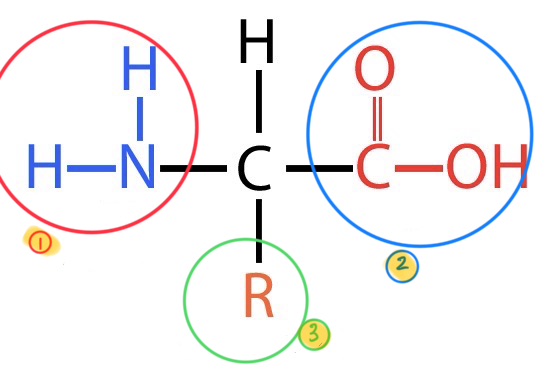
what is the #1 in this image?
amino group
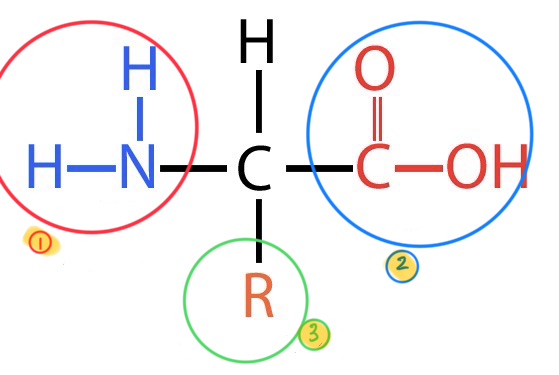
what is the #2 in this group?
carboxyl group
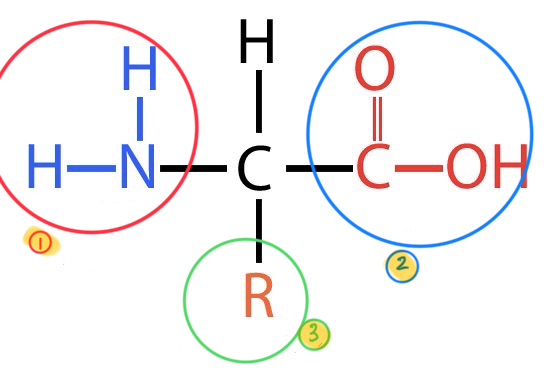
what is the #3 in this group?
the variable side group or R group
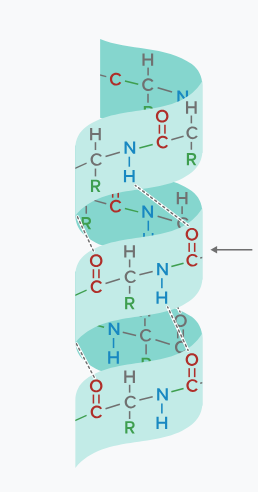
this image depicts what?
alpha-helix
alpha-helices occur when?
when hydrogen bonds between an amino hydrogen and a carbonyl oxygen causes the peptide to form a spiral or helical structure
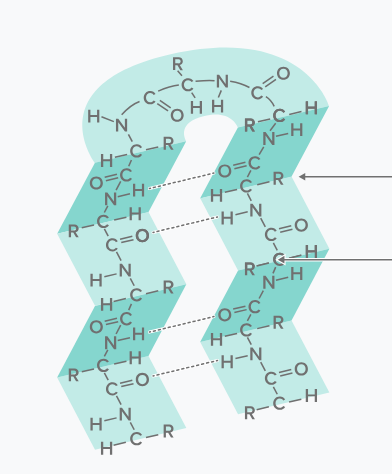
this image depicts what?
beta-pleated sheet
beta-pleated sheets occur when?
there is hydrogen bonding between amino acids and carbonyl oxygens, causing portions of the polypeptide chain to turn back on itself like an accordion
what determines the function of a protein?
its tertiary structure
the three-dimensional shape (or tertiary structure) of a protein is largely driven by what?
the hydrophobic areas of the protein that prefer to be buried inside of a cell structure, or due to interactions between different R groups associated with the amino acids
some proteins also have a quaternary structure which is?
the arrangement of multiple polypeptide chains to form a larger protein
true or false: quaternary structures can also affect function (ex: hemoglobin, insulin, etc.)
true
protein structures are sensitive to changes in pH, temperature, and salt concentration. changes in these can cause what?
protein denaturation
protein denaturation
the process of a protein unfolding from its natural structure
what is the primary energy source for plants and animals?
carbohydrates
monosaccharides, disaccharides, and polysaccharides are all examples of what?
carbohydrates
monosaccharides
single sugars, the simples form of carbohydrate that cannot be broken down
monosaccharides have _____ on their carbons, allowing them to form hydrogen bonds with water, making them _____
alcohol groups, water-soluble
biopolymers
polymers of biological molecules
polysaccharides are a type of what?
biopolymer
glycogen
stores energy in the muscles and livers of humans/animals
starches
water-soluble, helical structures with alcohol groups readily available for hydrogen bonding. used to store energy in plants
cellulose
forms hydrogen bonds between its strands to form sheets, making it insoluble and more rigid
cellulose is the technical term for what?
dietary fiber
nonpolar, water insoluble molecules found in cells/tissues of living organisms are known as?
lipids
simple lipids contain only:
carbon, hydrogen, and oxygen
fatty acids
simple lipids that have long chains of carbon atoms bonded only to hydrogen, with a carboxyl group (-COOH) at the end
four major groups of lipids
fatty acids, phospholipids, steroids, waxes
waxes
fatty acids with long chain alcohols, often coating leaves of plants or functioning as a water repellent
triglycerides
fatty acids with glycerol
triglycerides are considered ___ if they’re solid at room temperature, and ___ if they’re liquid at room temperature
fats, oils
unsaturated fats
contain at least one double bond, obtained from foods such as fish/avocados/tree nuts
saturated fats
fats without any carbon double chain bonds, therefore “saturated with” hydrogen atoms
phospholipids
important building blocks of the cell membrane. similar to fatty acids but have a phosphate group at the end
steroids
simple lipids that serve as important components of cell membranes for membrane fluidity and as signaling molecules. characterized by a base structure of four connected hydrocarbon rings
four important steroids
testosterone, estrogen, cholesterol, prednisone
nucleotides are made of what three components?
a 5-carbon sugar (ribose), a phosphate group, and a nitrogenous base
the phosphate group
is the backbone to which all sugars and bases are bonded
RNA
copies genetic information, often a single helix, and has oxygen on one of the ribose carbons in its polymer chain
what monomers/nitrogenous bases make up RNA and DNA?
adenine, guanine, cytosine, and uracil or thymine
what nucleotide base is RNA specific?
uracil
what nucleotide base is DNA specific?
thymine
cytosol
a jelly-like fluid in which intracellular organelles are suspended
organelles
specialized cellular strucutres that have specific functions within the cell
cytoplasm
space within the cell that contains the cytosol
prokaryotes are defined by what two primary characteristics?
unicellular, lack of nucleous
eukaryotes are defined by what three defining features?
membrane-bound nucleus, membrane-bound organelles, chromosomes
cells that serve to absorb nutrients from things like food and blood use what to do so?
microvilli
DNA is stored in the?
nucleus
nuclear envelope
double membrane that surrounds the nucleous offering an extra layer of protection against possibly harmful bacteria/enzymes/pathogens