ap chem unit 9
1/42
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
when does entropy (∆S) increase?
when matter becomes more dispersed (e.g. phase change from solid to liquid or liquid to gas)
when energy is dispersed (distribution of kinetic energy among gas particles as temp increases)
for a gas specifically, when does entropy increase?
when there is an increase in volume (at constant temp) and the gas molecules can move within a larger space
relationship between entropy and temperature
directly proportional
for what reactions does entropy increase for?
gases formed from liquids or solids
liquids or solutions (aq) formed from solids
reactions where there are more product gas molecules than reactant molecules

using particle-level reasoning, why does entropy decrease?
gas particles are more dispersed (have more microstates) than solids. the entropy decreses because the reactants, which include a gas, are converted into a solid product which is less dispersed than a gas.
the absolute molar entropy, S°, of Si(s) is 18 J/(K ∙ mol). Do you predict that the value of S° for H₂ (g) should be less than or greater than 18 J/(K ∙ mol)?
greater; H₂ (g) molecules have more microstates than solids which are in a fixed position
how do you calculate the entropy change for a process
∆S = ∑S°(products) - ∑S°(reactants)
standard gibbs free energy change
a measure of thermodynamic favourability
what temperatures will make ∆G° < 0 (favourable) if ∆H and ∆S are negative?
low temperatures
what temperatures will make ∆G° < 0 (favourable) if ∆H and ∆S are positive?
high temperatures
if ∆H is positive and ∆S is negative, what temperatures will make ∆G° < 0 (favourable)?
no temperatures
if ∆H is negative and ∆S is positive, what temperatures will make ∆G° < 0 (favourable)?
all temperatures
what are the units for ∆G?
KJ/mol
what should you do if you need to explain why entropy decreases or increases?
use particle-level reasoning
what is a common calculation mistake with the equation ΔG° = ΔH° – TΔS°?
not paying attention to the units and changing ΔS° from J/(K ∙ mol) to kJ/mol
the units of ΔH° and ΔG° are usually listed as kJ/mol, while the units of ΔS° are usually listed as J/(K ∙ molrxn)
under kinetic control
processes that are thermodynamically favoured but do not proceed at a measurable rate (it proceeds at a very slow rate)
what is a common reason for a process to be under kinetic control?
a high activation energy
when must you calculate ∆G to find if it is favourable (∆G < 0) or not?
if ∆H and ∆S are both negative or both positive
what is the value of R in the equation ∆G° = -RT ln K?
8.314 J mol K
what do processes with ∆G° < 0 favour in relation to K
processes with ∆G° < 0 favour products (K > 1)
what do processes with ∆G° > 0 favour in relation to K
processes with ∆G° > 0 favour reactants (K < 1)
how to solve for K with the equation ∆G° = -RT ln K?
K=e^-(∆G°/RT)
how can connections between K and ∆G° be made?
through estimation
when ∆G° is near zero, what will the equilibrium constant K be close to?
K will be close to 1
when ∆G° is much smaller or larger than RT, what will the value of K do?
deviate strongly from 1
what are three factors that ∆G° for the dissolution of a substance reflect?
the breaking of interparticle attractions that hold the solid together
the reorganization of the solvent around the dissolved species
the formation of interparticle attractions between solute and solvent
how can an external source of energy be used to make a thermodynamically unfavorable process occur.
electrical energy to drive an electrolytic cell or charge a battery
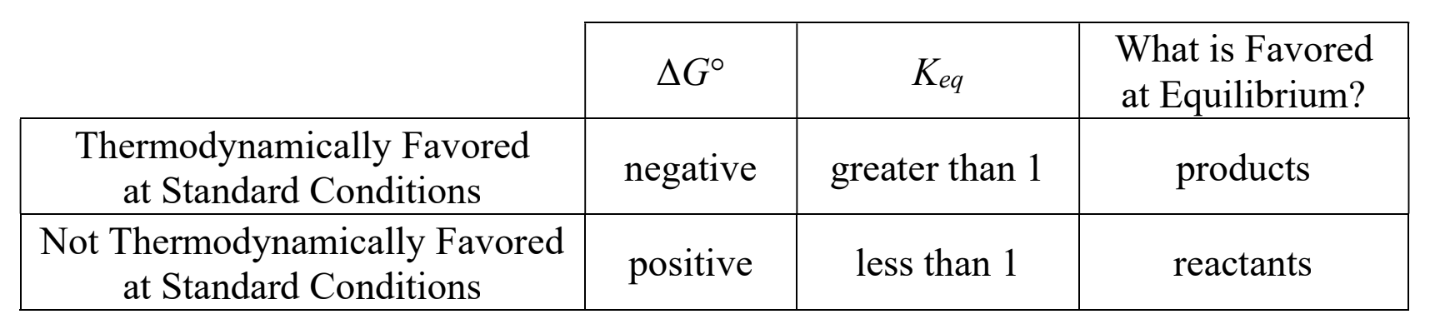
thermodynamically favoured at standard/non-standard conditions
remember
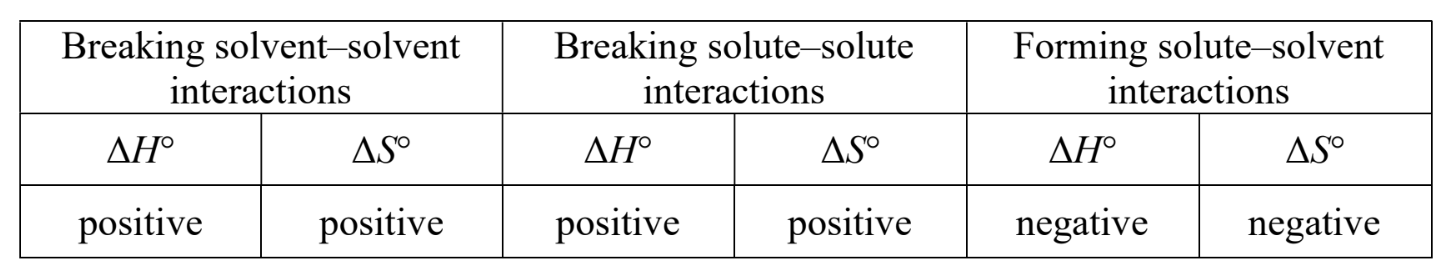
∆H and ∆S
remember
number of arrangements for molecules in pure water
large number of arrangements