Lecture 9 (Redox, Reduction Potentials & Electron Carriers)
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms

What is oxidation vs reduction?
Oxidation = electrons lost
Reduction = electrons gained

What are redox reactions?
Coupled oxidation and reduction reactions that occur simultaneously

What is an oxidant vs reductant?
Oxidant causes oxidation and is reduced
Reductant causes reduction and is oxidized
Why is electron movement central to biological energy?
Electron transfer between species with different electron affinities creates electromotive force (emf) that can store/release energy and perform work
Why is glucose oxidation exergonic?
Glucose is reduced
As it is oxidized, electrons flow to O2 (exergonic) because O2 has higher electron affinity than carrier intermediates
How does mitochondrial electron flow drive ATP synthesis?
Electron flow pumps H+ across inner mitochondrial membrane → proton-motive force
ATP synthase uses H+ flow to make ATP

How can an overall redox reaction be analyzed?
Split it into oxidation and reduction half-reactions

What is a conjugate redox pair?
Electron donor/acceptor pair in a half-reaction
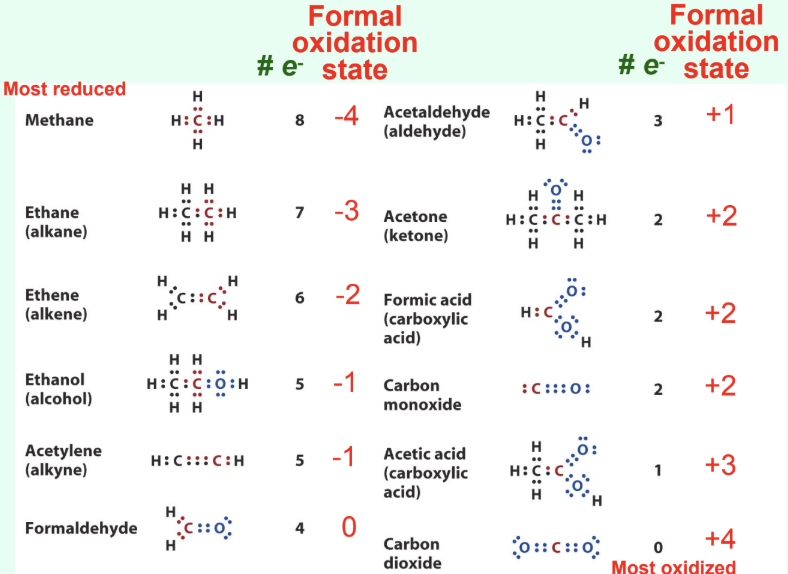
What is the electronegativity order?
H < C < S < N < O
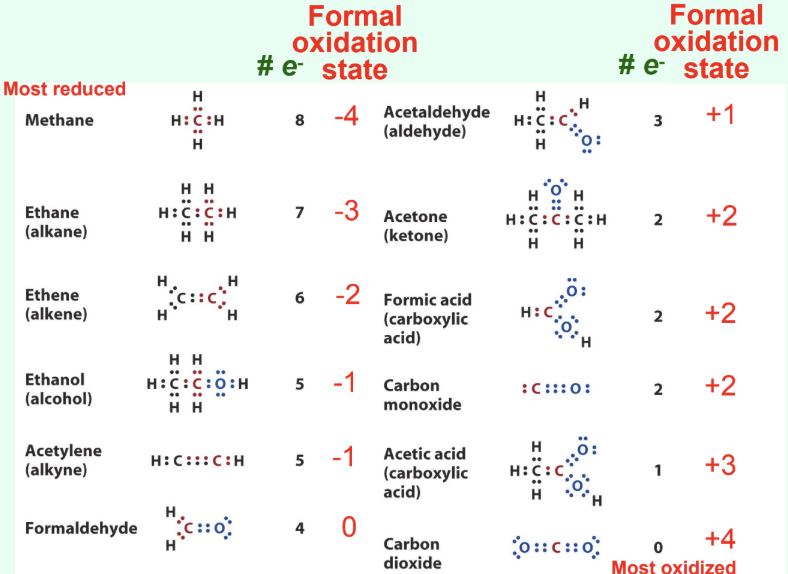
How does carbon become oxidized?
Formal loss of electrons
Often loss of H or gain of O
What key carbon facts should you know?
C has atomic number 6, is tetravalent (able to form up to 4 covalent bonds), and has 4 valence electrons
What are signs of biological oxidation?
Loss of electrons
Loss of hydrogen
Or addition of oxygen
What are the 4 biological electron-transfer modes?
Electrons
H atoms
Hydride ions (:H−)
Direct combination with O2
What is a reducing equivalent?
One electron equivalent transferred as e−, H atom, hydride (:H− = 2e−), or oxygen involvement

What is reduction potential, E?
Affinity of an electron acceptor for electrons in a redox pair (measured in volts)

What is biochemical standard reduction potential, E°′?
E measured under biochemical standard conditions:
55 M water
1 M solutes
101.3 kPa gases
pH 7
298 K

How do electrons flow by E°′?
From lower E°′ to higher E°′

What does a more positive E°′ mean?
Greater electron affinity
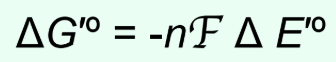
What is n in ΔG°′ = −nFΔE°′?
Number of electrons transferred
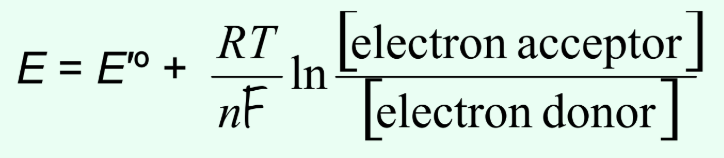
What does the Nernst equation describe?
Actual E depends on concentrations of electron acceptor and donor
Why is biological glucose oxidation controlled?
Energy is released in stages so it can be captured as ATP
What is a coenzyme?
Low-molecular-weight compound required for catalytic activity
What are the universal reversible redox coenzymes?
NAD+/NADH
NADP+/NADPH
FMN/FAD
What are other electron carriers?
Lipid-soluble quinones
Iron-sulfur proteins
Cytochromes (contain heme groups)
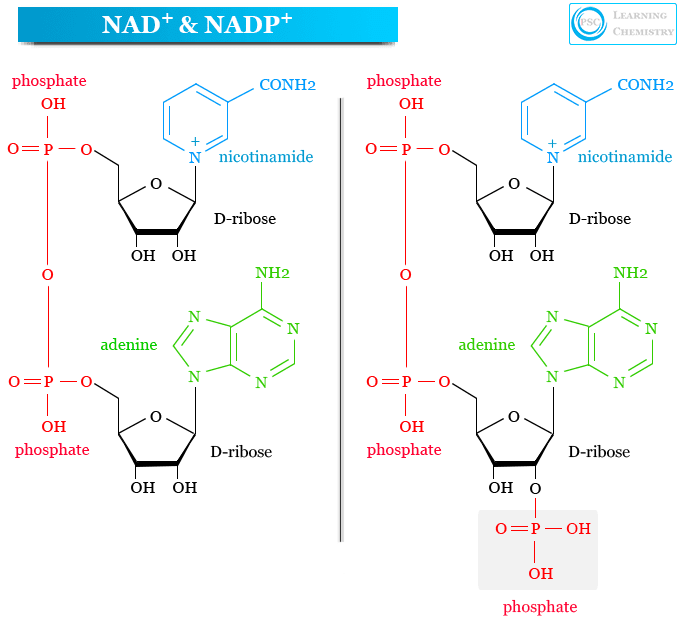
What are NAD+ and NADP+?
Soluble electron carriers
NAD+ = nicotinamide adenine dinucleotide
NADP+ = phosphorylated NAD+
What vitamin makes NAD(P)+?
Niacin, vitamin B3
How are NAD+ and NADP+ reduced?
By hydride transfer: NAD(P)+ + 2e− + H+ → NAD(P)H
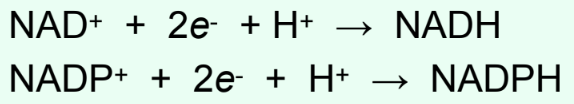
Do NAD(P)+ coenzymes stay bound?
No
They dissociate after each reaction cycle
What is NAD+ mainly used for?
Catabolism (oxidation breakdown of organic molecules)
NAD+/NADH mainly in mitochondria
What is NADPH mainly used for?
Anabolism/reduction (e.g. synthesis of complex molecules)
NADP+/NADPH mainly in cytosol
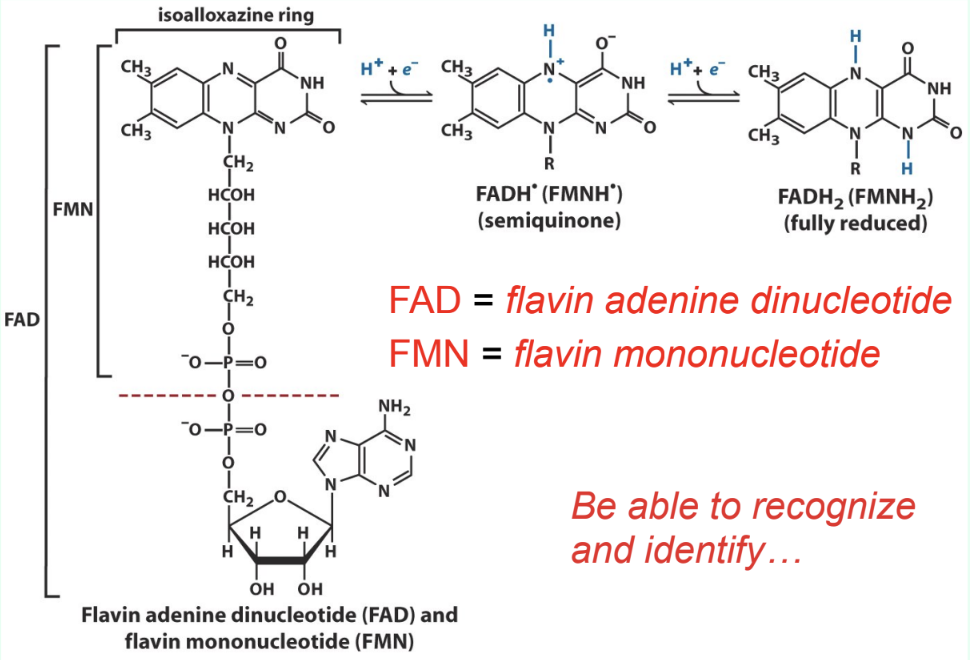
What are FAD and FMN?
Flavin redox coenzymes:
FAD = flavin adenine dinucleotide
FMN = flavin mononucleotide
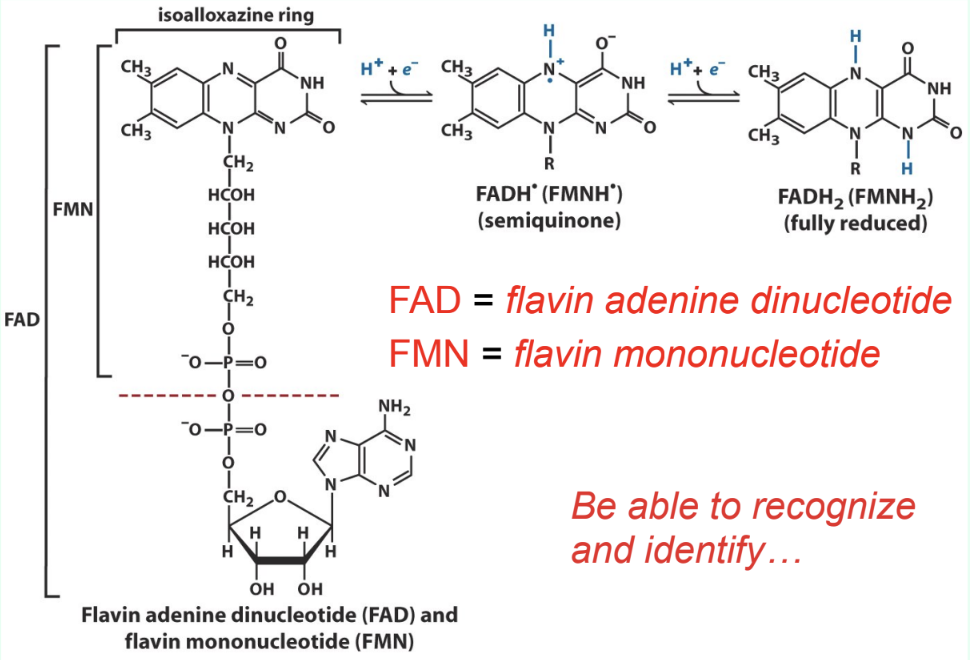
How are FAD/FMN bound in enzymes?
Tightly bound prosthetic groups of flavoproteins
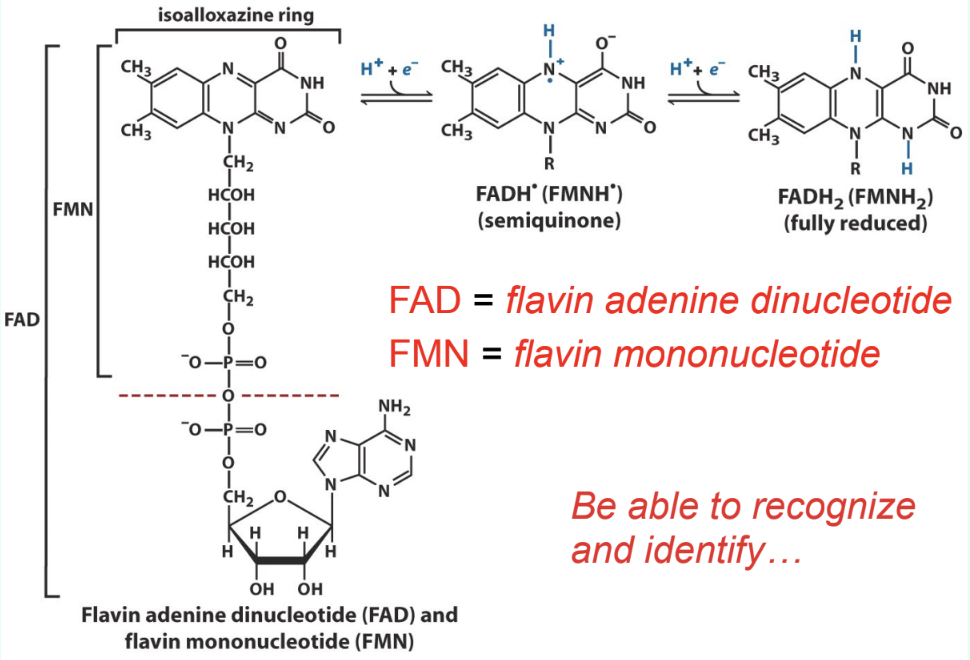
Do FAD/FMN dissociate each redox cycle?
No
They remain bound and do not dissociate