3.1.1 - atomic structure
1/66
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
67 Terms
relative charges of protons, neutrons and electrons
proton = +1
neutron = 0 (neutral)
electron = -1
relative masses of protons, neutrons and electrons
proton = 1
neutron = 1
electron = 1/1840 or effectively 0
what are isotopes
atoms with the same number of protons, but different numbers of neutrons
why do isotopes have similar chemical properties
why do they have slightly varying physical properties
they have the same electron configuration
they have different masses
can an ion be an isotope
Yes, an atom can be an isotope and an ion simultaneously. For instance, a specific isotope of chlorine, like Chlorine-37, can lose or gain electrons to form an ion, such as the chloride ion (Cl⁻).
what does a time of flight (TOF) mass spectrometer measure (2)
how heavy the different ions formed by a sample are (this is called m/z, where m = Ar of the ion and z = charge on the ion)
how much of a particular ion there is (i.e. relative abundance)
because it measures mass of isotopes and abundance of isotopes, this can be used to find the r________ a_______ m____ and therefore identify the e________.
because it measures mass of isotopes and abundance of isotopes, this can be used to find the relative atomic mass (Ar) and therefore identify the element.
in substances made of molecules, a mass spectrometer can help us find the r_________ m____________ m____.
relative molecular mass
what happens in TOFMS (3)
particles of the substance are ionised to form 1+ ions
these = accelerated so they all have the same Ek
time taken to travel a fixed distance = used to find mass of each ion in the sample
name the 5 steps in a TOFMS
ionisation
acceleration
ion drift
ion detection
data analysis
diagram of the TOF mass spectrometer
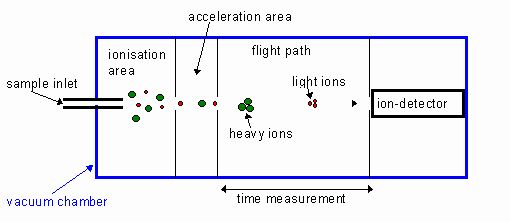
STAGE 1: IONISATION
what are the 2 ways the sample can be ionised
electron impact
electrospray ionisation
STAGE 1: IONISATION
in electron impact, sample being ionised is v__________. then, high energy e-s are f______ at it. this usually knocks off o___ e________ from each particle, forming a ___ ion.
in electron impact, sample being ionised is vaporised. then, high energy e-s are fired at it. this usually knocks off 1 electron from each particle, forming a 1+ ion.
STAGE 1: IONISATION
general eq. w/ state symbols to show ionisation of an atom
X (g) —→ X+(g) + e-
STAGE 1: IONISATION
what fires the high energy electrons
an electron gun (a hot wire filament w/ a current running through it that emits electrons)
STAGE 1: IONISATION
where are the 1+ ions then attracted towards
a negative electric plate
where they are accelerated
STAGE 1: IONISATION
what elements and substances is electron impact used for
ones with a low Mr
electron impact is also used when the substance is an ELEMENT, not a molecule
STAGE 1: IONISATION
when molecules are ionised with electron impact, what is the 1+ ion formed known as
give an example equation for this, w/ state symbols, for methane
a molecular ion
CH₄(g) → CH₄⁺(g) + e⁻
STAGE 1: IONISATION
what happens in electronspray ionisation
(this is a perf mark scheme answer)
sample dissolved into a volatile solvent
then injected through a fine hypodermic needle at high voltage
(this gives a fine mist (aerosol))
sample gains a proton / H+
(next few flashcards are adding info to this, but this is pretty much all you need)
STAGE 1: IONISATION
what is the tip of the needle attached to
the + terminal of a high-voltage power supply
STAGE 1: IONISATION
how are the particles ionised
by gaining a proton (e.g. a H+ ion which is just 1 proton) from the solvent as they leave the needle
this produces XH+ ions (ions w/ a single positive charge and a mass of Mr + 1)
STAGE 1: IONISATION
general equation for electronspray ionisation w/ state symbols
X(g) + H+ —→ XH+ (g)
STAGE 1: IONISATION
what happens to the solvent as the XH+ ions are attracted towards a negative plate (where they are accelerated)
it evaporates away
STAGE 1: IONISATION
what substances is electrospray ionisation used for
why is this
ones with a high relative formula mass (including many biological molecules, e.g. proteins)
so the molecules do not fragment
STAGE 2: ACCELERATION
the positive ions are accelerated using a…
why?
electric field (there is an attraction between the + charged ions and the - charged plates)
so that they all have the same Ek
STAGE 2: ACCELERATION
you’ll need to use the kinetic energy equation. give this and the units used.
(you need to know how to rearrange this too!)
Ek = 1/2mv²
Ek in J, mass in kg, velocity in m s-1
STAGE 2: ACCELERATION
given that all the particles have the same Ek, the velocity of each particle depends on its…
mass
STAGE 2: ACCELERATION
l________ particles have a f________ v_________, and h_________ p___________ have a s________ v__________.
lighter particles have a faster velocity, and heavier particles have a slower velocity. (think about the equation)
STAGE 3: FLIGHT TUBE
the positive ions travel through a h____ in the n__________ c________ p______ into a t_____.
the positive ions travel through a hole in the negatively charged plate into a tube.
STAGE 3: FLIGHT TUBE
what does the TOF of each particle through this flight tube depend on
velocity of particle
(which in turn depends on its mass)
STAGE 3: FLIGHT TUBE
to find time of flight, we need what 2 equations?
kinetic energy eq (Ek = 1/2mv2) and distance speed time eq (d = v x t, where d = metres, v = m s-1, and time = seconds)
STAGE 3: FLIGHT TUBE
lighter ions t______ f________ and reach detector in l____ t_____ than the h_________ p__________ that m_____ s_________ and t____ l__________ to reach detector.
lighter ions travel faster and reach detector in less time than the heavier particles that move slower and take longer to reach detector.
STAGE 4: DETECTION
what happens when the positive ions hit the detector plate
what does this generate
they are discharged (by gaining electrons from plate)
a movement of electrons, and hence an electric current that is measured
STAGE 4: DETECTION
what does the size of the current give a measure of
no. of ions hitting the plate (and therefore abundance)
STAGE 5: DATA ANALYSIS
a computer uses the data to produce a…
mass spectrum
what 2 things does this show
mass to charge (m/z) ratio
abundance of each ion that reaches the detector
since all ions produced by electrospray ionisation and most ions produced by electron impact = have a 1+ charge, what does this mean the m/z effectively is?
the mass of each ion (y / 1 = y)
formula for relative atomic mass (Ar)
Ar = (mass no. of iso. 1 x relative abundance of iso. 1) + (mass no. of iso. 2 x relative abundance of iso. 2) / combined abundance of all isotopes (might be 100, might not be!)
(the values in the Ar formula can be found on the mass spectrum. remember, don’t divide the percentages by 100!)
how to do a TOF calculation for 2 isotope ions traveling down the same flight tube (look in notes for examples)
the ions will be accelerated to the same kinetic energy. so Ek for one ion = Ek for another ion. then set up up eqs. with the substituted values to be equal to each other (1/2 mv2 = ½ mv2). cancel out common terms on both sides (you don’t need to find masses on both sides separately). then rearrange and solve
energy levels are divided into…
sub-levels consist of a set number of…
orbitals are a region of space that can hold up to…
you can’t say w____ an e- is in an orbital and where it’s going n___
sub-levels
orbitals
2 e-
you can’t say where an e- is in an orbital and where it’s going next
how many sub-levels are there?
give the names of these
3
s, p and d
how many orbitals does each sub-level hold
s = 1
p = 3
d = 5
therefore, how many e- are in each sub-level
s = 2
p = 6
d = 10
give the order of sub-levels for e- configs.
1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s…
here is a diagram of e- configs. in boxes.
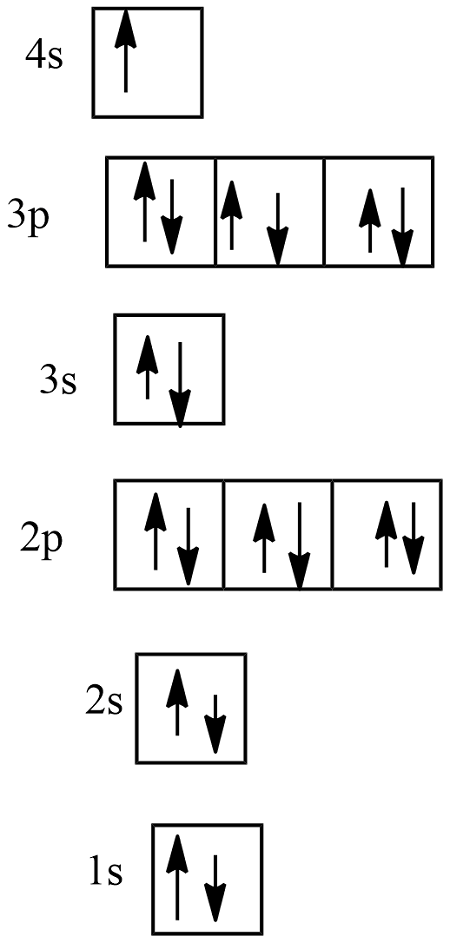
in this, what is an e-?
what is an orbital?
what is a sub-level?
what is an energy-level?
one of the arrows in a box (called a single-headed arrow, they look more like the data sign than in this diagram)
a box
a row
a group of sub-levels (e.g. the rows 2s and 2p make up the 2nd energy level)
in the e- config., what do you write next to each of the sub-levels?
how many e- are in that sub-level (amnt. of orbitals x2)
REMEMBER: when filling up the boxes, put one arrow in each box FIRST and then go back and fill them so they each have 2.
how do you write simplified versions of e- configs.?
find the total number of e- in your config., then find a noble gas with the highest possible atomic number that could fit into that. then write the noble gas symbol in square brackets, and if there are any sub-levels left, write those afterwards too.
for this diagram, give the e- config
then give the simplified e- config
then name what element this is
1s2 2s2 2p6 3s2 3p6 4s1
[Ar] 4s1
potassium (K) because atomic number (Z) = 19
elements in the periodic table are in different…
what are these blocks called?
where is each block on the periodic table?
blocks
s, p, d or f block
(see photo)
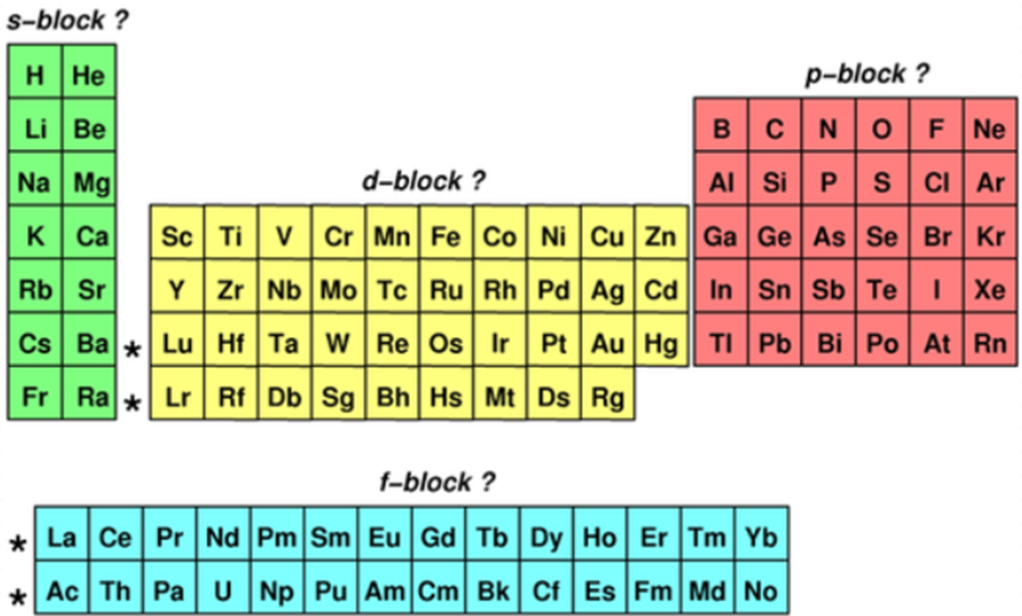
in the s-block (according to their e- configs.), what do atoms have in common in terms of their outermost e-?
the outermost e- are in an s sub-level
in the p-block (according to their e- configs.), what do atoms have in common in terms of their outermost e-?
the outermost e- are in a p sub-level
in the d-block (according to their e- configs.), what do atoms have in common in terms of their outermost e-?
the final e- enters a d-orbital (research this)
how would you change the e- config. of an atom to be the e- config. of an ion?
add or subtract e- (as an ion is an atom that has lost or gained e-)
there’s a catch for d-block elements, though. with d-block elements, it’s the ___ electrons which are lost f____.
with d-block elements, it’s the 4s electrons which are lost first (even though the 4s orbital is filled before the 3d orbital during electron configuration).
e.g. e- config. for Fe atom (Z = 26) → [Ar] 4s² 3d⁶
then e- config. for Fe²⁺ ion = [Ar] 3d⁶
COVER IONISATION ENERGIES
what 3 factors affect IEs
distance between the nucleus and the e- being removed
shielding (repulsion) by e-s in inner shells between nucleus and outer e-s
nuclear charge
1st ionisation energy def
energy required to remove one mole of e-s from one mole of gaseous atoms
if first IE is low
electron is lost more easily
1st, 2nd, 3rd IE
Na (g) —→ Na+ (g) + e- (this ion is formed after the 1st IE has been measured)
Na+ (g) —→ Na2+ (g) + e-
Na2+ (g) —→ Na3+ (g) + e-
explaining significant jumps in ionisation energies (ENERGY LEVELS AND THEIR DISTANCE FROM THE NUCLEUS)
(number of, e.g. 1st) e- is removed from the (number of, e.g. 3rd) energy level / (name of, e.g. 3s) sub-level
which is further / closer to the nucleus
more / less shielded by inner shells of e-s
and therefore more / less tightly held
than the (number of, e.g. 2nd) e-
which is removed from the (number of, e.g. 2nd) energy level / (name of, e.g. 2p) sub-level
general trend in 1st IEs going across period 3 (INCREASING NUCLEAR CHARGE, BUT SAME SHIELDING)
as atomic no. increases —→ first IE of period 3 elements also increases
bc nuclear charge increases
outer e- has a stronger force of attraction to the nucleus
more and more energy is required to remove it
even though there is little change between distance between outer e- and nucleus and amount of shielding between them
what are always (?) the anomalies in this trend
group 2-3 and group 5-6 (3 and 6 are lower)
why is this (SUB LEVELS AND DISTANCE)
outer e- in Al is lost from the 3p sub level
this is further away from the nucleus
less shielded by inner shells of e-s
so the outer e- is less attracted to the nucleus than the outer e- of Mg
which is removed from the 3s sub-level
why is the 1st IE of S (grp 6) lower than P (grp 5) (ORBITALS)
in S, 2e-s are in 3p orbital
these repel each other
takes less energy to remove one of the electrons from this pair than it does to remove the 1st e- of P
where all the 3p orbitals are singly occupied