Alkyne Reaction Mechanisms
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
Alkylation
1. NaNH2
2. R'-X
X= Cl, Br, I
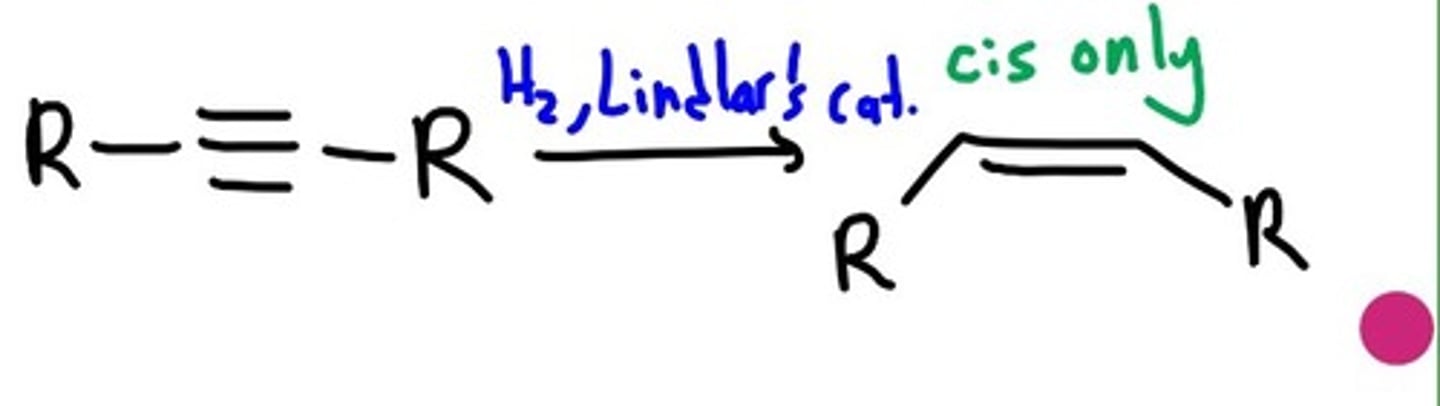
Poisoned Hydrogenation
CIS
1. H2
2. Lindlar's Cat
ends at alkene product
sun addition= cis alkene
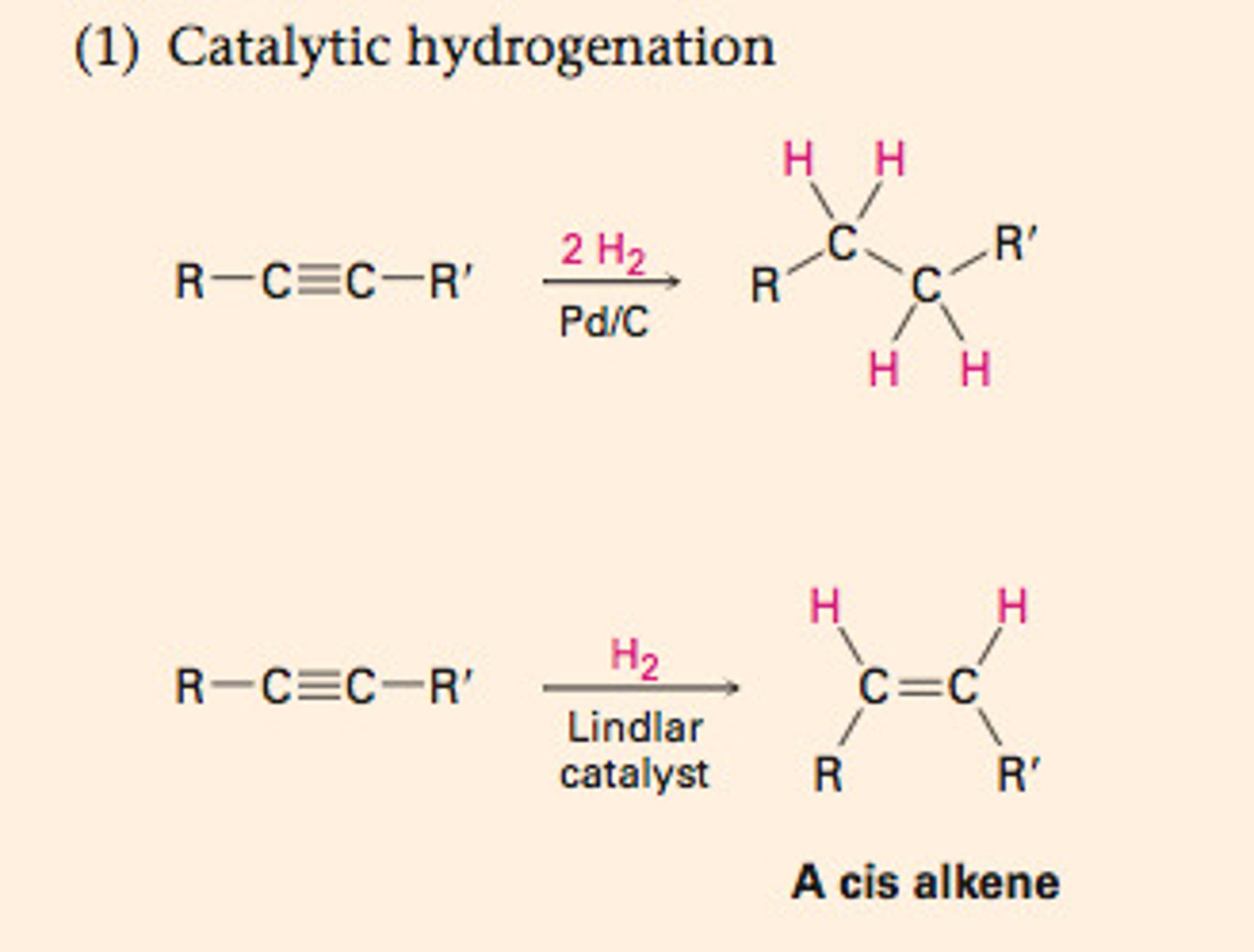
Catalytic Hydrogenation
1. H2
2. Pt
forms alkane
syn addition
not selective
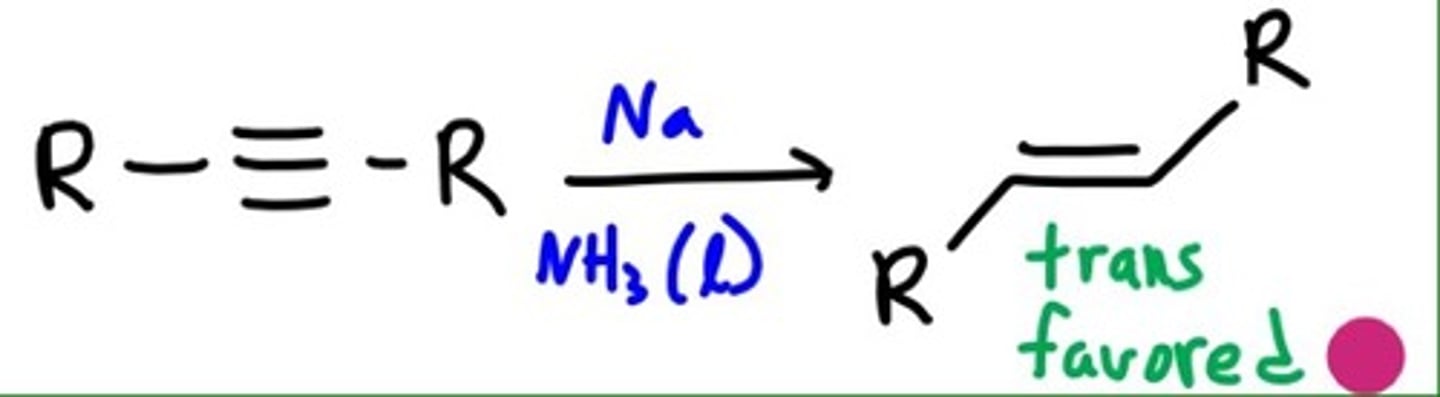
Dissolving Metal Reduction
TRANS
Synthesis:
1) Na,
2) NH3 [liquid]
anti addition
trans alkene only
part polar part rad
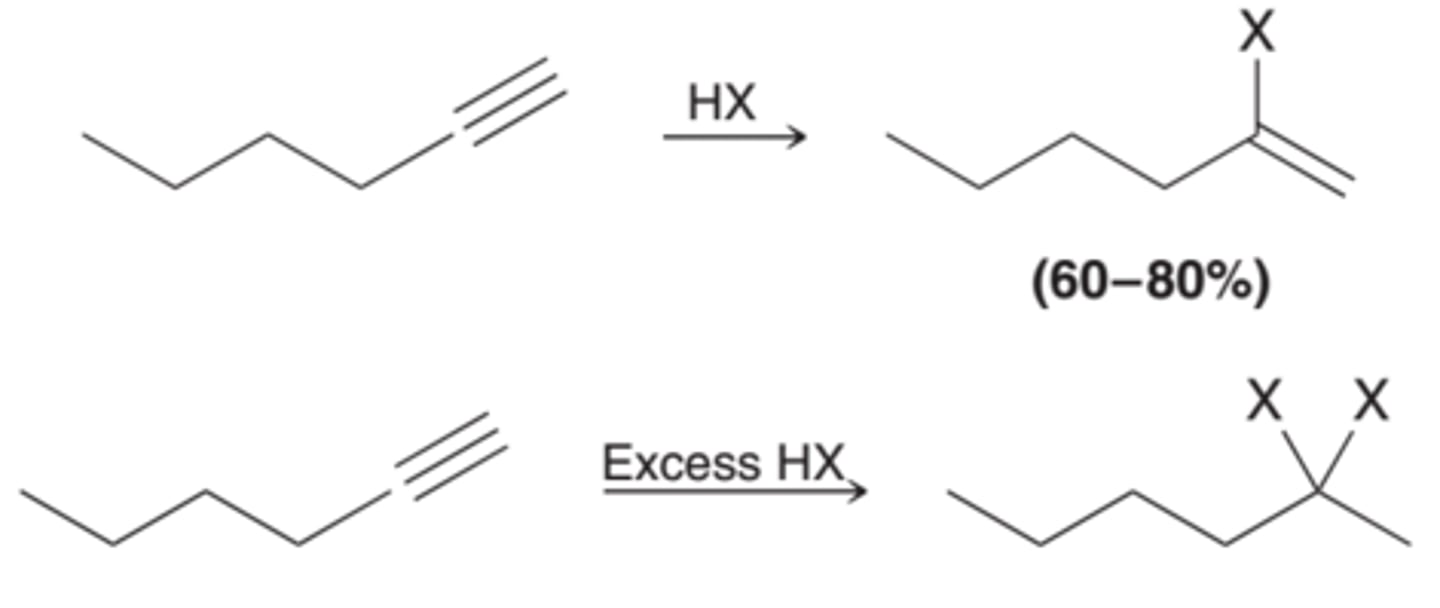
Hydrohalogenation POLAR
1. HBr but also HCl in xs
MARK
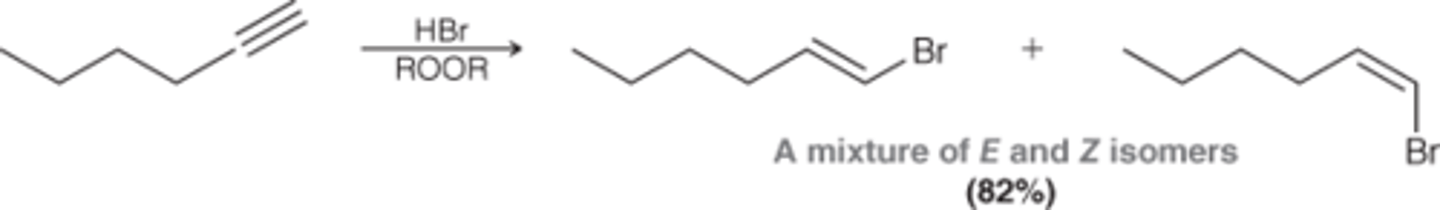
Hydrohalogenation RADICAL
1. HBr
2. ROOR
xsHBr
ROOR
anti mark
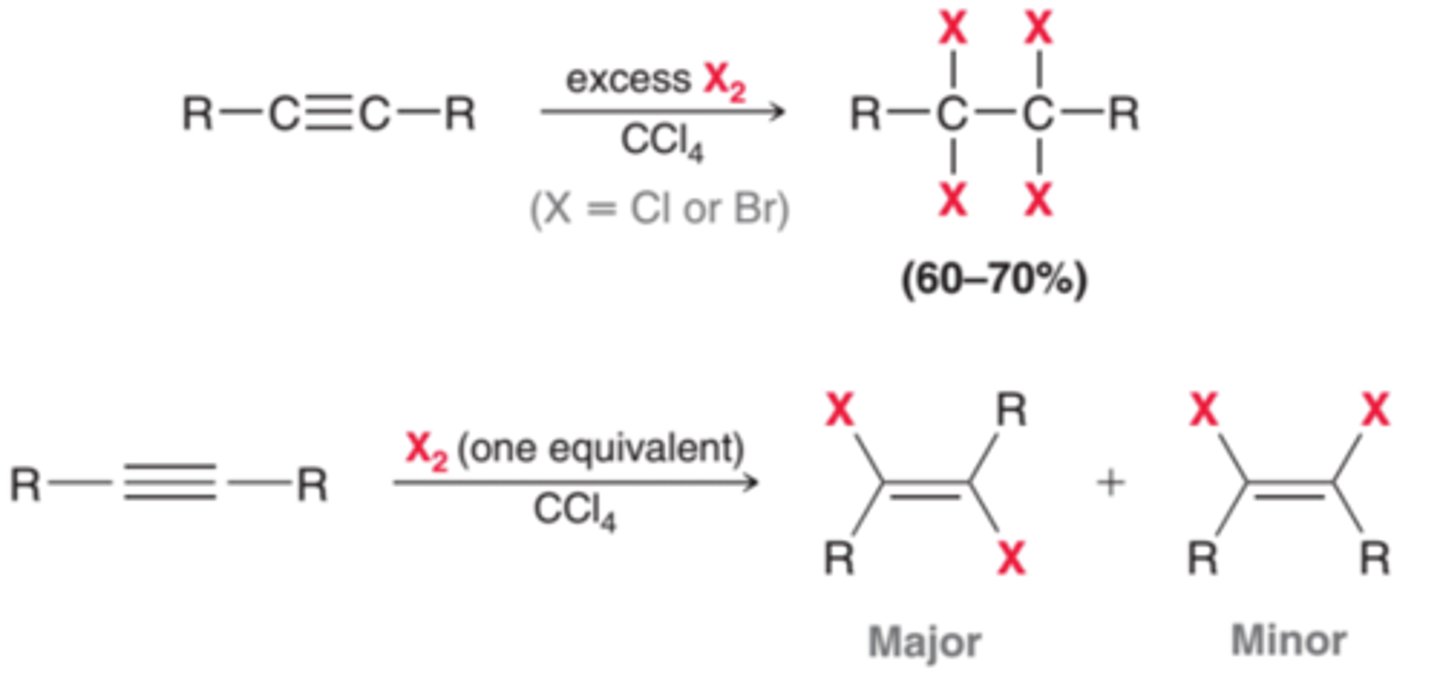
Halogenation
1. X2 (br or cl)
2. CCl4
xsX2
CCl4
-Reagents: Br2 or Cl2 in CCl4
-Addition
-First addition is usually trans
-Markovnikov addition
-Excess X2 ---> tetrahalides
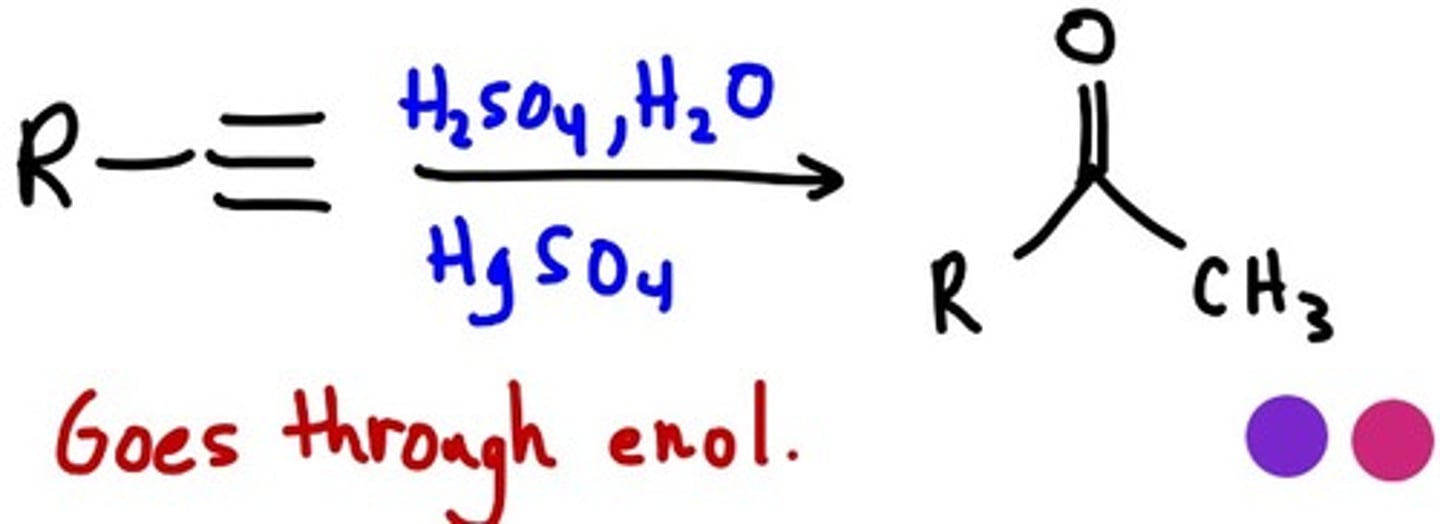
Acid Catalyzed Hydration
1.H2SO4, H2O
2. HgSO4
goes through enroll which is where OH single bond becomes O double bond
MARK= H least OH most
enol: C=C becomes C=O (Me Ketone)
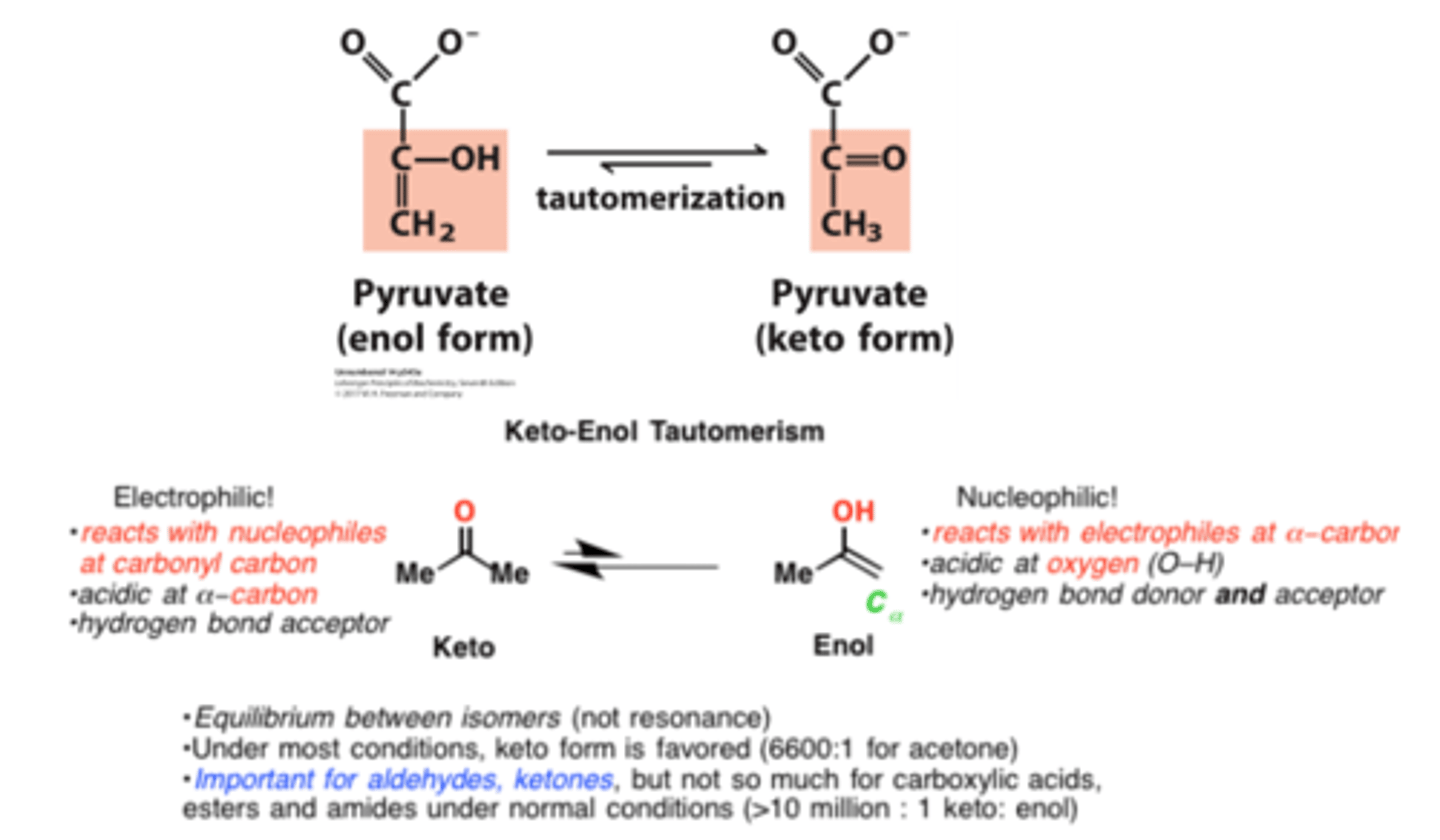
tautamerization
constitutional isomers
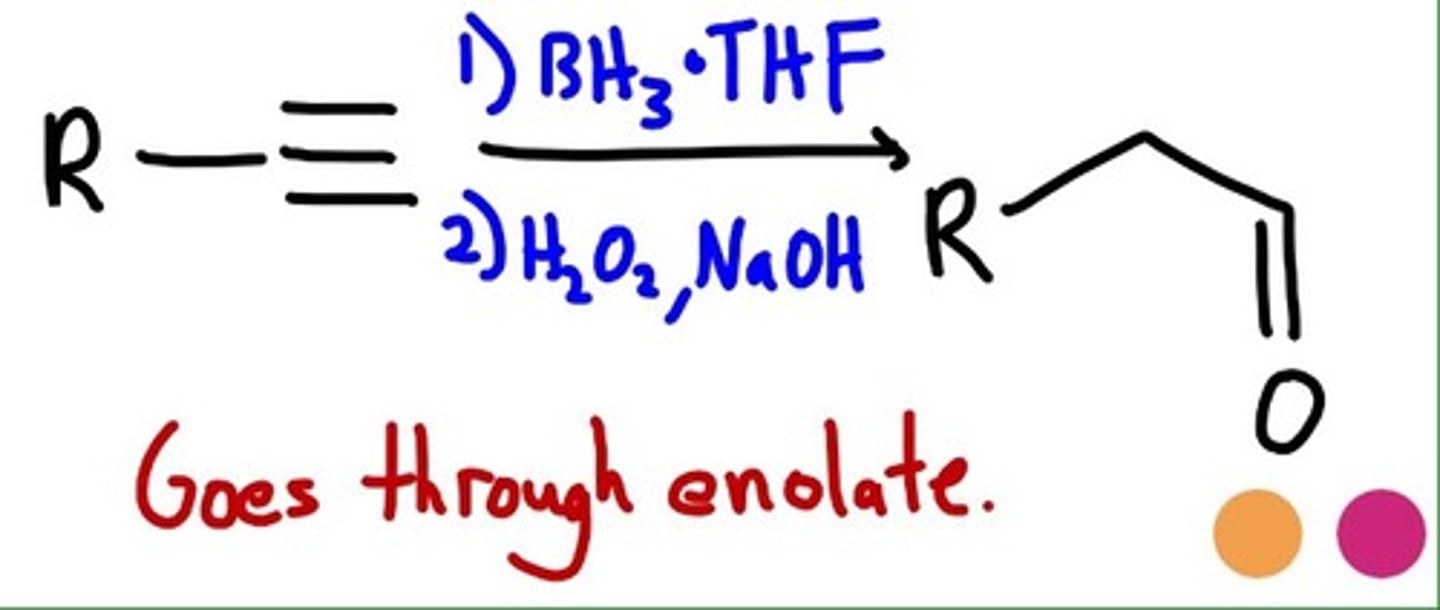
Hydroboration-Oxidation
1. BH3, THF
2. H2O2, NaOH
1. BH3, THF
2. H2O2, NaOH
forms aldehyde (vs AC hydration which forms ketone)
ANTIMARK= H most and OH least
enol is formed goes through ketone/enol tautamerisation
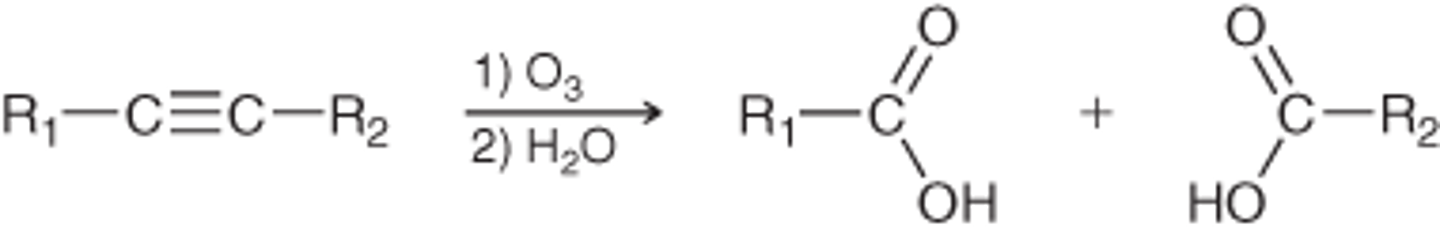
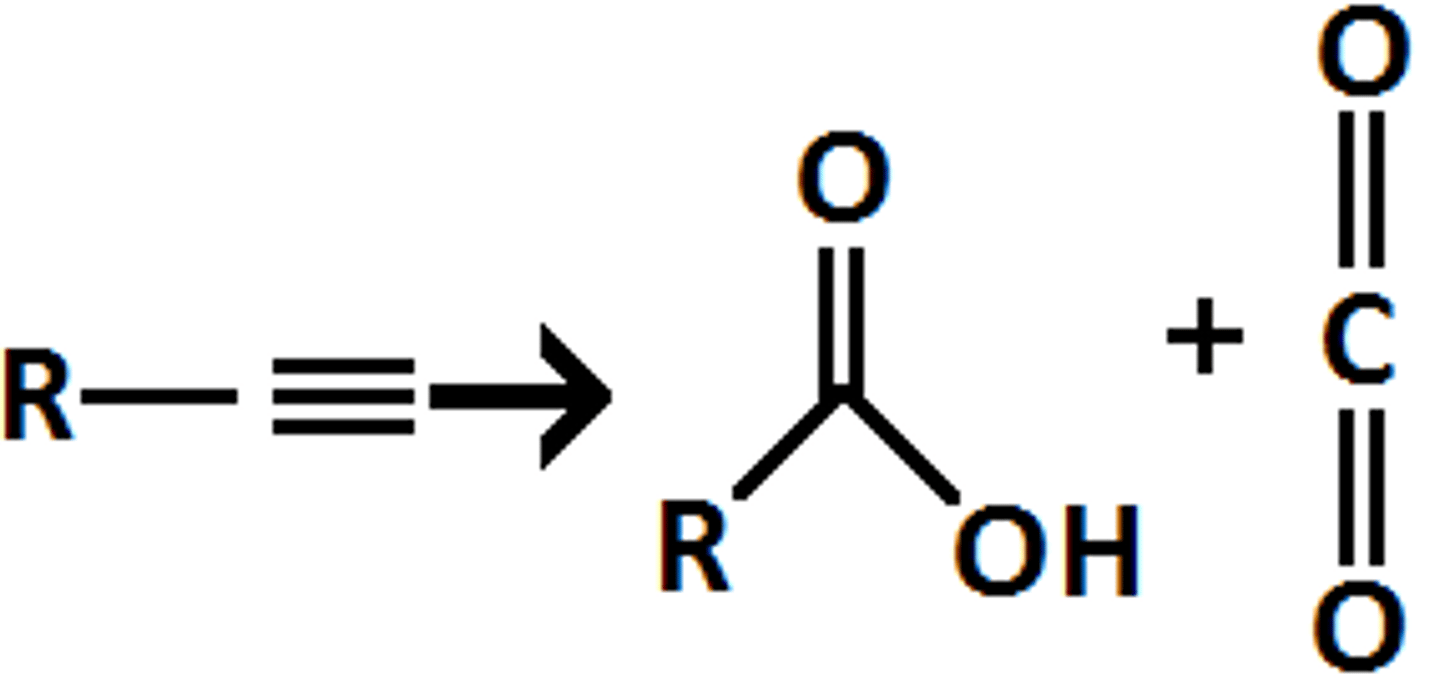
ozonolysis (terminal alkyne)
1. O3, DMS
2. H2O
1CO2 forms with 1carbox acid
R3H
ozonolysis (internal alkyne)
1. O3, DMS
2. H2O
2 carboxylic acids form
R3R'
2 carboxylic acids form