Lipids and Carbs
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
The fat that serves as the main lipid storage molecule is a:
Triacylglycerol
Also called triglycerides
What is triglycerides composed of
3 Fatty acids attached to a glycerol backbone
What are predominately used to form the Plasma membrane of cells
Phospholipids
What molecules are primarily stored in adipose tissue as stored energy
triglycerides
What does Saponification of lipids with fatty acids connected via an ester linkage include
Triacylglycerols
Sphingolipids
Phospholipids
What is Saponification
Esters are turned into soaps
Converts a larger lipid molecule into the salts of its component fatty acids
Sphingolipids participate in:
Cell signaling and Apoptosis
A type of lipid where fatty acids are attached to a sphingosine backbone
Act as signal-transducing molecules
What is the role of Cholesterol in the plasma membrane
Prevents lower temperatures from inhibiting fluidity
At high temp —> prevents increase in fluidity
At low temp —> Maintains membrane fluidity
What is the role of Cholesterol
Production of steroid hormones
Maintenance of cell membrane integrity
What are the properties of Unsaturated Fatty acids
They have Kinks due to the presence of double bonds
Increase membrane fluidity
What are the properties of Saturated Fatty acids
No double bond
Decrease membrane fluidity
What is the difference between long-chain fatty acids vs Short-chain fatty acids
LC Fatty Acids: more rigid which makes the membrane less fluid
SC Fatty Acids: Limited impact on membrane fluidity
What is the base molecule for steroid hormones
Cholesterol
For hormones such as estrogen and testosterone.
Are steroid hormones lipid soluble or insoluble
Lipid Soluble
What about steroid hormones
They are peptide hormones, derived from Amino acids
What are Terpenes
They are a class of unsaturated lipids
Commonly found in plants
Molecular formula (C5H8)n for n>1
What is the molecular formula for Isoprene
C5H8
What is the three precursor for prostaglandins?
Arachidonic acids (AA)
dihomo-y-linolenic acids(DGLA)
eicosapentaenoic acids(EPA)
Describe prostaglandins?
Group of lipids with hormone-like action that is produced in response to infection or tissue damage
What is a waxy substance often found in animals and vegetable fats
Stearic acid
Prostaglandins can be found as regulators of cell signaling in the:
Cardiovascular + Respiratory + Urinary system
What dies Nucleotides contain
5 carbon sugar
A phosphate group
Nitrogenous base
What are the purines
Adenine and Guanine
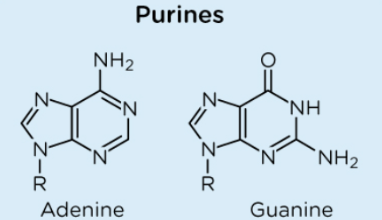
What are the pyrimidine
Cytosine
Uracil
Thymine
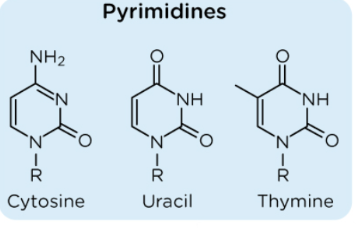
What nucleotides exist as Keto-enol tautomers?
Guanine, Uracil, Thymine
What nucleotides shift between amino form and imino form?
Adenine and cytosine
If adenine undergoes a tautomeric shift, it will bind to:
Cytosine
Adenine would be unable to hydrogen bond with thymine
What is a tautomeric shift?
The spontaneous isomerization of a nitrogen base to an alternative hydrogen-bond form, possibly resulting in a mutation
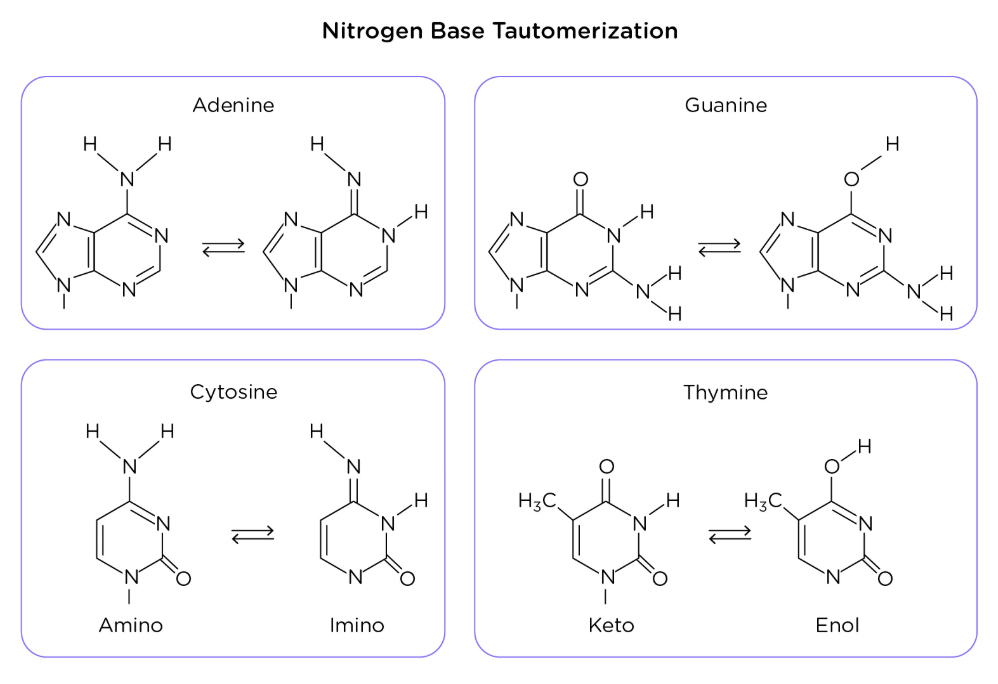
The Chargraff’s rules state that:
DNA should have 1:1 ratio of purines to pyrimidines
What vitamin would result in precipitate formation when the same amount of Vitamin powder is added to carbon tetrachloride in a beaker
Vitamin B
What are the fat-soluble vitamins
A,D,E,K
Have the capacity to be stored in the body for prolonged period
Susceptible to toxicity risks
What are the water-soluble vitamins
B-complex and Vitamin C
required for daily consumption as they are not stored in the body
What to know about Carbon tetrachloride (CCl4)
Has a tetrahedral structure around a carbon atom
Net-zero dipole moment
Non-polar solcent
Fat-soluble vitamins would dissolve in non-polar solvents and water would precipitate
Role of Sphingolipids
Major structural component of the nervous system
Involved in signal transmission and cell recognition
Can be found as ceramide, ganglioside and cerebroside.
Sphingolipids can be found as
ceramide
ganglioside
cerebroside.
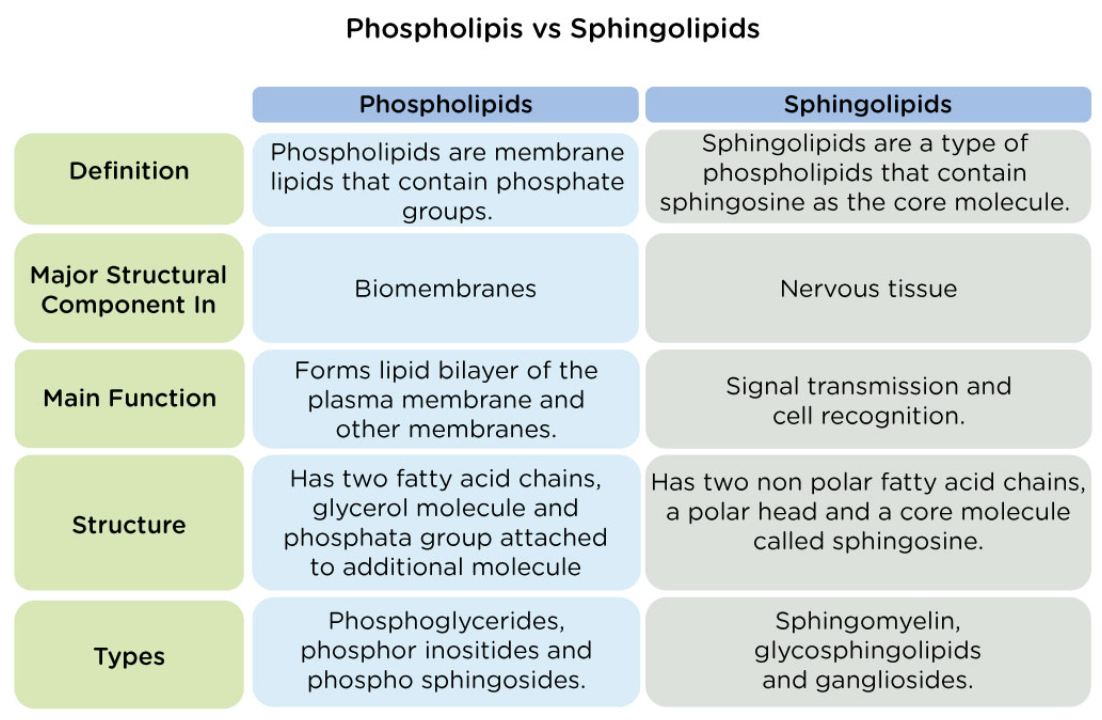
Phospholipids vs Sphingolipids
What are the four interactions that help stabilize teh tertiary structure of a protein
Ionic bonding
Hydrogen bonding
Disulfide linkages
Dispersion forces
Describe hydrophobic interactions in the case of tertiary structure interaction
This is the non-polar amino acid side chain cluster together in the protein’s interior to avoid water, aiding folding and stability
Describe hydrogen bonds in the case of tertiary structure interaction
The peptide backbone and polar amino acid side chains form hydrogen bonds stabilizing secondary structures and folding patterns
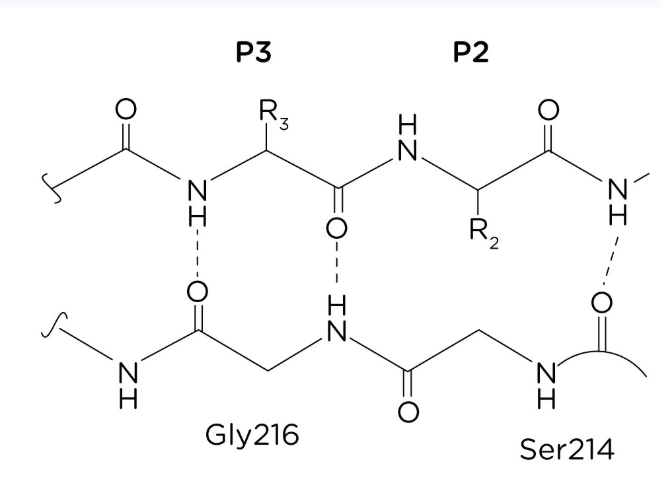
Describe Ionic interactions(salt bridges) in the case of tertiary structure interaction
Attraction between oppositely charged amino acids chains leads to salt bridge formation, stabilizing the structure
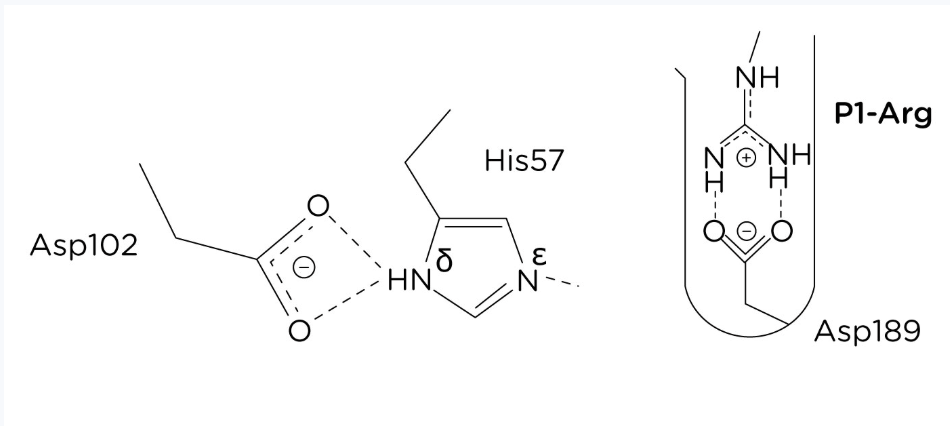
Describe Disulfide bonds in the case of tertiary structure
Covalent bonds between sulfur atoms of cysteine residue form disulfide bridges, adding stability
Describe Van der waals interaction in the case of tertiary structure
Weak, attractibe forces between nonpolar molecules contribute to compact folding
What binds to the allosteric site independently from substrate attachment
Noncompetitive substrate