Chem 141 chapter 1
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
A law
tells you what
A theory
tells you why
Sizing from largest to smallest
Cells.Molecule.atom
Graphite AFM
A way to see atoms
Atoms originally were found by
The greeks.. It was based on philosophy
Brownian motion
Random motion of atoms and particles
What did the greeks believe that was incorrect
They believed that shape determines properties
Molecular level structure determines…
Properties
Greeks were right about
atoms in constant random motion
Atoms characteristics
Neutral. 2 protons 2 e-
compound
2 or more different atoms (EX: 2 He atoms and 1 H atom)
Daltons atomic theory
Elements are composed of small, indivisible particles called atoms. all atoms of an element are the same

whos model is this
daltons model
what are in the nucleus
Protons and neutrons
JJ thompson discovered
the e-
how did jj thompson discover the e-
By using the cathode ray tube, when he shot particles toward the + and - anodes, the particles bent towards the + anode, showing that there is a - particle in an atom

whos model is this
Thompsons model
Ernest rutherfords experiment
Gold foil experiment. evidence that the nucleus exists
alpha particle
2 protons, 2 neutrons, positively charged
rutherford experiment shows
atoms are mostly empty space and theres small, dense, nucleus in atoms center
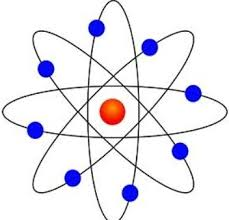
whos model is this
Rutherfords planetary model
Neutrons were the ____ atoms to be found
last
why were neutrons the last to be discovered?
because they have no charge and cant be manipulated with magnetic fields
coulombs law
opposite charges attract, like charges repel. F=(q1q2)/r2
Gravity
attraction between objects that have a mass
electromagnetic force
responsible for repulsion/attraction between charged particles
as Mass Increases, gravity..
increases
As distance increases, gravity…
decreases
electromagnetic force is ____ than Gravity
stronger
____ is too weak to be detected at atomic/molecular level
Gravity
Energy
change in energy caused by changes in forces. Energy is conserved
system
the part you’re looking at
surrounding
everything else
Kinetic energy
energy of motion. KE=1/2MV2
Increase velocity =_____
increase in kinetic energy
Potential energy
stored energy.
energy can be ….
transfered and transformed
noble gasses…
do not react with anything
breaking a bond=
energy absorbed
when distance decreases electrostatic _____
is stronger
deeper PE well =
stronger interaction
Larger atomic radii =
shift right on the PE