Chem exam 3 chapter 20
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
Radioactive decay types
If on Right and it’s -1 for Z (atomic charge) it’s….
If on left and it’s -1 for Z, it’s…
If it’s -4 and -2, the element identity on right is Helium, and it’s…
If it’s +1 to Z on right, it’s…
On right, -1 = positrion emission
On left, -1 = Electron capture
-4 and -2 (helium) = alpha particle decay
+1 on right to Z, is beta particle decay
Review 20.1 if you don’t get it but it’s pretty simple once you get hang of it.
Top number is molar mass
Bottom number if atomic number from periodic table
ID unstable Isotopes and predict their modes of decay for
37Cl, 222Rn, 26Mg, 66Co, 116I
ID # of protons and neutrons in each element
a. Proton = atomic #
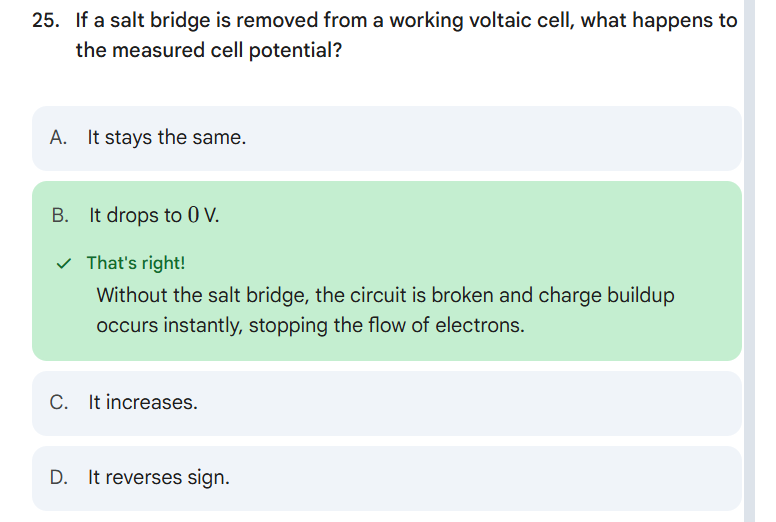
b. Neutron = The number above the element - protons
ID stability
More than 82 protons = non-stable
Odd # of Protons/Neutrons = non-stable
Even number of Protons/Neutrons = Stable
Magic # for either proton or neutron = Stable
Protons: 2, 8, 20, 28, 50, or 82
Neutrons: 2, 8, 20, 28, 50, 82, or 116
For those that are unstable, predict the mode of decay
If protons are greater than 82, it’s alpha decay.
Compare value above the unstable isotope to its atomic mass on periodic table. If the value above the unstable isotope is higher than the number for its atomic mass, it will be beta decay. (above belt)
Compare value above unstable isotope to its atomic mass, if the value above the unstable isotope is lower than its atomic mass in periodic table, you can predict it will be positron emission or electron capture (below belt)
Beta particles have a mass number of ___.
Beta particle formation is accompanied by the conversion of a _____ into a _____.
Beta particles have mass numbe of 0
Beta particle formation is accompanied by the conversion of a neutron into a proton.
List position and values for Alpha, Beta, Positrion Emission, and Electron capture
Alpha: Right, subtract 4 and 2 for He
Beta: Right, add 1 to Z
Positrion emission: Right, subtract 1 from Z
Electron capture: Left, subtract 1 from Z
The mass of a nucleus is always _____ the sum of the masses of its nucleons
mass of nucleus is always less than sum of its nucleons
What’s the equation for finding half lives
ln(initial/final) = (ln2/half life)t
If you gotta solve for half life, what do you gotta do
mutliply 2 by ln2, then solve for half life
Of elements that have non-radioactive isotopes and are stable, which has highest atomic #?
- lead
iron
- radium
uranium
Answer is lead
It’s basically asking, which is going to be stable.
Radium and uranium have more than 82 protons based on atomic number, therefore they’re not stable.
Lead has highest atomic number compared to iron, and is still stable, therefore its lead.
As soon as you see Voltaic cell, what does this tell you?
Spontaneous redox reaction
+E and negative G
Voltaic/Galvanic (same thing) cells must be ______.
The one that’s Oxidized will have more ____ E value
The one that’s Reduced (cathode) will have a more ____ E-cell
Voltaic/Galvanic cells MUST be Spontaneous! (+E, -G, K>1)
Oxidized will have more negative E value in Voltaic cells
Reduced (cathode) will have a more positive E value in voltaic cells
Write overall reaction between AgNO3 and Solid Zn in a Voltaic cell
Identify the Anode and Cathode
Anode = more negative E value in Voltaic/Galvanic cell
Cathode = more positive E value in Voltaic/Galvanic cell
Zinc —> Zinc(s)
AgNO3 —> Ag+ + NO3-
Write out Anode
Zn(s) —> Zn+2 + 2e-
Write out Cathode
Ag+ + e- —> Ag(s)
Balance out electrons
2Ag + 2e- —> 2Ag(s)
Assemble net ionic equation
Zn + 2Ag —> Zn+2 + 2Ag
Add back in the Spectator ions
Zn(s) + 2AgNO3 —> Zn(NO3)2 + 2Ag
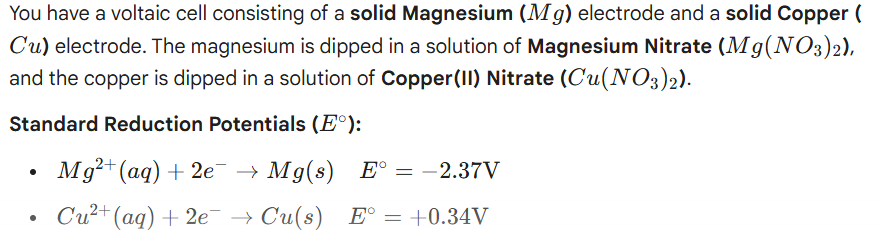
ID the Anode and Cathode for the voltaic cell
Balance out the electrons for both, multiply if necessary to get them to cancel out
Write out the net ionic equation where the charges are equal (i.e +2 = +2)
Write the overall equation where you plug back in the placeholders on the values that have charges to get them to cancel out
i.e
from the start
Mg —> Mg+2 + 2e-
Cu+2+ 2e- —> Cu(s)
Net ionic equation
Mg + Cu+2 —> Mg+2 + Cu(s)
we have a +2 on Cu, and a +2 on Mg
and we want overall charge on both sides to be 0
So we add back in the placeholder value of NO3- onto both, but NO3- is only -1, so we need 2. so it becomes
Mg(s) + Cu(NO3)2 —> Mg(NO3)2 + Cu(s)
s
How do you know which aqueous ion concentration to increase to drive the reaction forward?
Take the Magnesium/Cadmium reaction you just solved:
Mg(s)+Cd2+(aq)→Mg2+(aq)+Cd(s)
Driving the reactant ion conc forward will increase forward direction for more voltage, more E, more negative G, more K!
vice versa for product ion conc

For Electrolysis, you have 2 reactions fighting for cathode, and 2 fighting for anode
I.E here, it’s Water reaction vs Br reaction
and Water reaction vs K reaction
ID which is the Anode and Cathode by lookiong at the sign of the element
I.E KBr in H2O
K = +, therefore If we write it as K+ —> K(s) it would be reduced
Br = -. therefore if we write it as Br- —> Br, it would be oxidized
Look at the 2 reactions that are displaying Oxidation , choose the one that is more negative.
This is the anode!
Look at the 2 reactions that are displaying reduction, choose the one that is more positive
That will be your
Cathode!
So ID which are anode and cathode by looking at question and assigning signs to the molecular formula i.e KBr = K+ --> K(s) reduction (cathode) and Br- --> Br2 (anode) oxidation
2. For choosing oxidation vs whatevers in solution like water, choose the one that's more negative. I.E, if Br is -0.5, and H2O is -0.6, then H2O will be anode
3. For choosing reduction vs whatevers in solution like water, choose one thats more positive. If K is +47, and h2o is + 2, choose K as cathode.
Which pair of substances will produce the greatest EMF when used as electrodes in 1M solutions of their ions
Reactivity
Na > Zn > Fe > Pb > H > Cu > Hg> Ag
The most reactive will be the anode, and have the most negative reductipn potential
The least reactive will be further out to left and have most positive reduction potential
i.e Na + Ag would have greatest reactivity together
Most reactive = ______, and has most _____ reduction potential
Least reactive = _____, and has most _____ reduction potential.
Most reactive = Anode, and has most negative reduction potential
Least reactive = Cathode, and has most positive reduction potential
Strongest Oxidizng agent =
Strongest Reducing agent =
What about on Standard Oxidation potentials chart?
Strongest oxidizng agent = Most positve value
Strongest reducing agent = Most negative value
ON STANDARD REDUCTION POTENTIALS CHART
ON STANDARD OXIDATION POTENTIALS CHART
Strongest Oxidizing agent = negative
Strongest reducing agent = positive
cancel log
cancel ln
cancel log
10value
cancel ln
evalue
Equation for amps and mass and shit
m = I x t x MM / nF
m = mass
i = amps
t = time
MM = molar mass
n = electrons
F = 96485
For a Concentration cell, what is the value of Ecell always???
Why is this important for determining cell potential?
Ecell is 0.
Still need to use nernst equation to solve, but you can remove the Ecell from the equation.

Write out equation from anode/cathode
ID the reactant ion