BIO 245 Exam 3
1/207
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
208 Terms
what is drug resistance
microbes tolerate exposure to antimicrobial drugs which they were previously susceptible too
what is natural drug resistance
The ability of certain microorganisms to inherently resist the effects of specific antimicrobial drugs without prior exposure or genetic mutation.
antibiotic procedures
natural barriers (like the G- cell membrane)
This type of drug resistance is not a problem
how does drug resistance happen
1.) spontaneous mutations in chromosomal genes
2.) acquisition of new genes from other species
resistance factors
plasmids
what are the mechanisms of drug resistance
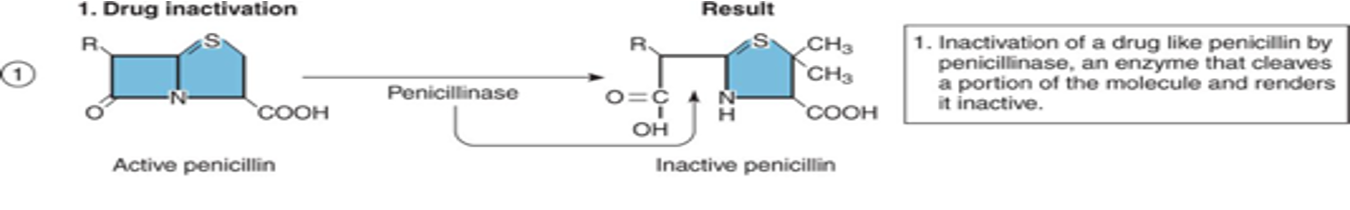
1.) Drug inactivation
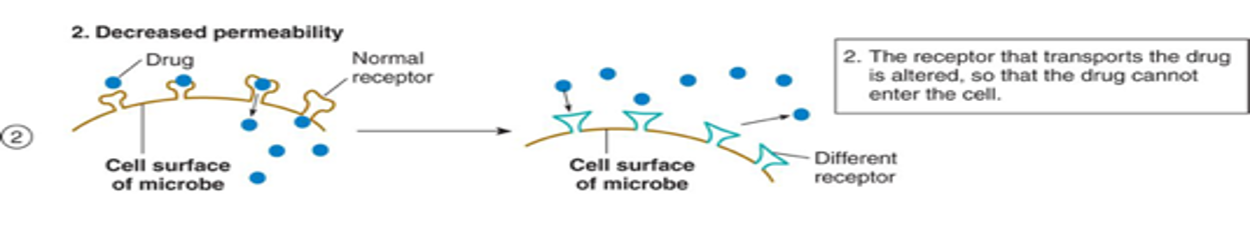
2.) Permeability
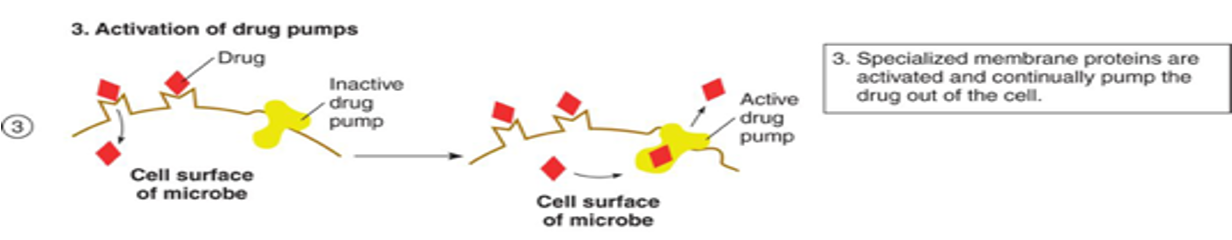
3.) Uptake
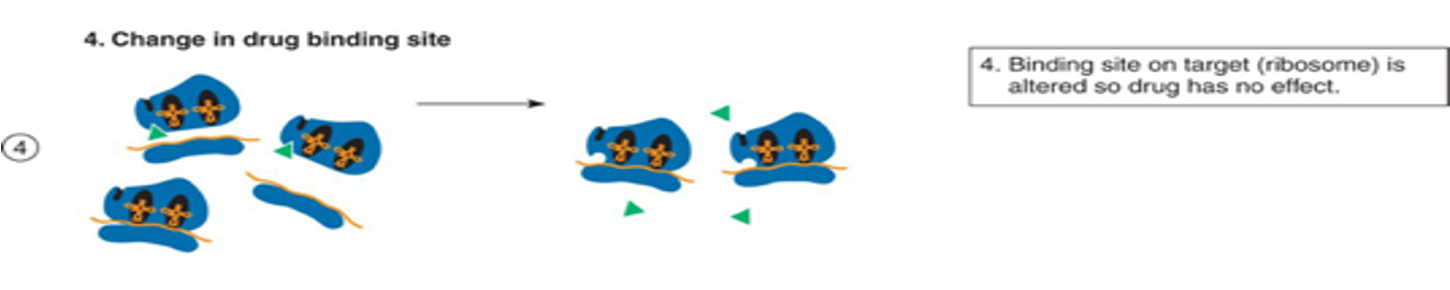
3.) Change in binding site
5.) Metabolism
Drug inactivation mechanisms
development of alternative enzymes that inactive a drug
requires new genes
decreased Permeability mechanisms
uptake of the drug is decreased or stopped
uptake (activation of drug pumps) mechanisms
microbe activates transport pumps to move the drug out of the cell
Change in drug binding site mechanisms
binding sites for the drug are reduced
can be due to mutation or acquisition of new genes
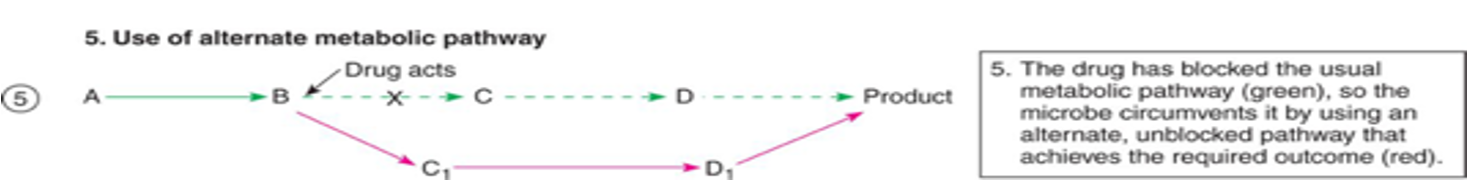
Use of alternate metabolic pathway mechanisms
the target metabolic pathway is shut down or an alternative is used
due to a mutation
drug example of drug inactivation mechanisms
different types of penicillin
drug example of permeability/uptake mechanisms
tetracycline and aminoglycosides ??
drug example of change in binding site mechanism
erythromycin
drug example of metabolism
sulfonamine and trimethroprim
what disease are considered the most urgent threats in terms of drug resistance by the CDC
Clostridium difficile
carbapenmen-resistant enterobacteriaceae
drug resistant neisseria gonorrhoeae
clostridium difficile
causes diarrhea and colon inflammation
life-threatening
largerly problematic for hospitalized patients recently treated with antibiotics
500,000 infections per year
15,000 deaths per year
Carbapenem-resistant Enterobacteriaceae
nightmare bacteria
members of the family enterobacteriaceae are resistant to almost all antibiotics
Klebsiella and E. coli
generally not a risk for healthy people
long-term facilities, etc.
9,000 infections per year
600 deaths per year
What are the major factors that contribute to the problem of drug resistance
greater use of antibiotics exerts selective pressure on susceptible bacteria and can favor survival of resistant strands
reduction in use could result in resistant bacteria being replaced with susceptible bacteria because resistant bacteria may be less fit than susceptible bacteria
inappropriate prescribing
patient non-compliance
use of antibiotics in agriculture
what are the 3 main host/drug interaction side effects
1.) damage to tissue through toxicity
2.) allergic reactions
3.) disruption of normal microbial flora
how do we remove microbes from a given environment
physical methods
heat
radiation
chemical methods
antiseptics
disinfectants
what are the two main ways we control microbial growth
physical controls
heat
radiation
filtration
chemical controls
Most work by: Disrupting cell membranes OR Altering protein and/or nucleic acid structure
Prion
infectious protein molecule
not considered alive
abnormal version of a normal protein
whats the easiest to decontaminate
bacterial vegetative cells
enveloped viruses
yeast
fungal spores and hyphae
protozoan trophozoites
whats moderately difficult to decontaminate
protozoan cysts
fungal sexual spores
naked viruses
unique vegetative bacteria
mycobacterium
gram negative pathogens
staphylococcus spp.
whats the most difficult to decontaminate
prions
bacterial endospores
sterilization
process that destroys or removes all viable microbes
generally only non-living
usually performed with heat
“-cide”
to kill
bactericide
a chemical agent that destroys bacteria
excludes endospores
fungicide
a chemical agent that destroys fungi
virucide
a chemical agent that destroys viruses
sporicide
a chemical agent that destroys bacterial endospores
bacteriostatic
prevent the growth of bacteria on surfaces
microbiostasis
the inhibition of microbial growth
stasis
to stand still
Germicide
any chemical agent that kills pathogenic microbes
can be used on living or non-living tissue
not effective against resistant organisms
disinfection
physical process or chemical agent that destroys vegetative pathogens
not effective against spores
not used on living tissue
also gets rid of toxins produced by pathogens
antiseptic chemical agents
agents that can be applied directly to living tissues to destroy vegetative pathogens
sepsis
infection of the blood
microbial load
microbial population size
sanitization
cleansing technique that removes debris, microbes, toxins, etc.
reduces possible spoiling or infection
soaps and detergents
degermation
reducing the load on living tissues
alcohol swabs
the death curve
a logarithmic curve: each increment is a 10x reduction in population size
not every cell dies right away
younger, more metabolically active cells die first
sterilization is the point at which survival is unlikely
factors that effects the slope of the death curve
the size of microbial load
mode of action of the agent
spores vs. vegetative cells
other possible effects
mixed vs. homogenous population
temperature and pH of the environment
concentration of the agent
presences of inhibitors, solvents, etc.
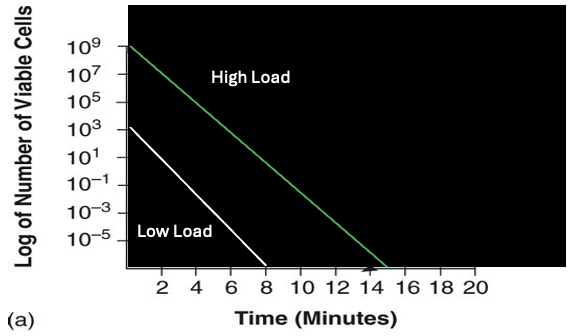
the microbial load size effect on the death curve
the more microbes initially, the longer it takes to kill all of the microbes
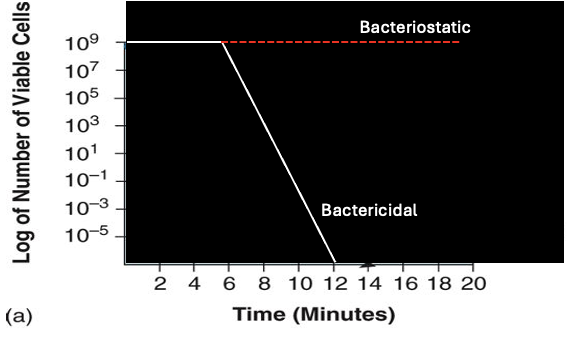
mode of action of the agent effect on the death curve
if an agent is bacteriostatic it inhibits growth without killing; if bactericidal, it kills bacteria, affecting the rate at which the death curve declines.
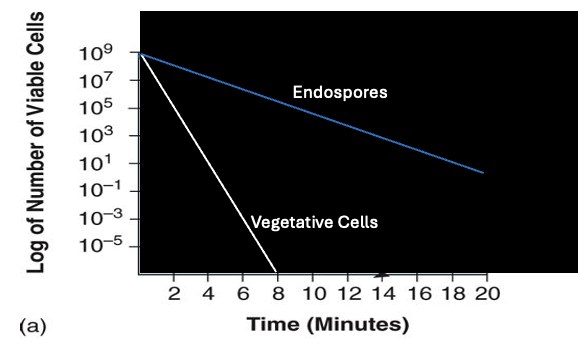
spore vs. vegetative cells effects on the death curve
Spores are more resistant to agents than vegetative cells, leading to a slower decline in the death curve.
Modes of Action
refers to the mechanisms through which antimicrobial agents inhibit growth or kill microbes, significantly influencing their effectiveness against different microbial populations.
how does targeting the cell wall mode of action work
prevents cell wall synthesis
digests the cell wall
breaks down the cell wall surface
how does targeting the cell membrane mode of action work
lowers surface tension of the membranes
how does targeting protein and nucleic acid synthesis mode of action work
bind to ribosomes and block translation
bind to DNA and block transcription
how does targeting protein function mode of action work
denatures proteins
breaks bonds in secondary and tertiary protein structure
what happens to the cell during the cell wall mode of action
cells become fragile and lyse
what happens to the cell during the cell membrane mode of action
cells loose ability to stop harmful molecules from moving in/out
cells lose ability to bring essential molecules in
what happens to the cell during protein and nucleic acid synthesis mode of action
cells can’t make proteins necessary for metabolism'
DNA can’t be replicated
genes can’t be expressed
what happens to the cell during the protein function mode of action
stops metabolism
examples of the cell wall mode of action
penicillin
detergents
alcohol
examples of the cell membrane mode of action
surfactants
alcohols
examples of the protein and nucleic acid synthesis mode of action
some antibiotics
radiation
examples of the protein function mode of action
heat
organic solvents
metals
different types of heat control
moist heat
steam under pressure
non pressurized steam
boiling water
Pasteurization
dry heat
incineration
dry oven
moist heat physical control
hot water, boiling water, or steam
60-135 degrees Celsius
denatures proteins and nucleic acis
dry heat physical control
ovens
160-1000 degrees celsius
denatures, oxidizes
steam under pressure
at sea level (15 psi), water boils at 100 degrees celsius
autoclave
nonpressurized steam
tyndallization
items are placed in chamber with steam from boiling water for 24 hours
repeated 3x
does not destroy endospores
we use this technique because come items are too sensitive to withstand heat from autoclaving
boiling water
boil at 100 degrees celsius
will not destroy endospores
easily recontaminated when removed from the water
pasteurization
application of heat to consumable liquids to kill infectious microbes
maintain integrity of beverage
Flash method
ultrahigh temperature pasteurization
flash method of pasteurization
exposed beverages to 71.5 degrees celsius for 15 seconds
doesn’t kill everything
20,000 cells/mL in milk is allowed
effective against some pathogens
Ultrahigh Temperature (UHT) pasteurization method
exposes milk to 134 degrees celsius for 2-5 seconds
completely sterilizes milk
incineration
bunsen burner - 1,870 degrees celsius
incinerator - 6,500 degrees celsius
dry over
150-180 degrees celsius
how do most chemical controls work
disrupting cell membrane or
altering protein and/or nucleic acid structure
what is the effectiveness of chemical control determined by
concentration
contact time
major groups of chemical controls
halogens
phenol and its derivatives
alcohols
hydrogen peroxide
aldehydes
gasses
detergents
heavy metals
halogens
microcidal
sporocidal with extend exposure time
examples:
flourine bromine, chlorine, iodine
limitations of halogens as a chemical control
exposure to light, alkaline pH, or organic matter can render halogens less effective
phenols
affect protein function and/or disrupt membranes
NOT sporidical
Alcohols
colorless hydrocarbons with -OH groups
ethyl and isopropyl alcohol are the only two used for microbial control
concentration >50% destroys cell membranes
concentration 50% - 95% denature proteins through coagulation
must be diluted at least 5% with water to denature proteins
NOT sporicidal
not very effective against viruses without an envelope
Hydrogen Peroxide
H2O2
forms free radicals (superoxides, hydroxyls), which are all toxic to cells
catalase breaks down H2O2 → H2O +O2
won't work against elevated concentrations used during disinfection
sporicidal at high concentrations
Aldehydes
organic substances with a -CHO on the terminal carbon
examples: gluaraldehyde and ortho-phthaldehyde (more potent)
causes cross linking of proteins on the cell surface to disrupt protein activity
sporicidal after 3 hours of exposure
becomes unstable in increased pH and temperature
Gases
Chlorine dioxide: disrupts proteins
used to treat drinking water, wastewater, medical waste, and buildings (large scale)
Ethylene oxide gas
reacts with DNA and proteins
very effective
explosive
carcinogen
detergents
solubilize membranes and disrupt proteins
Cationic detergents
positively charged
more effective
against G+, viruses, fungi, and algae
Nonionic detergents
soaps
not very microbicidal
Pseudomonas grows IN soap dishes
cleansing agents
Heavy Metals
mercury, silver, gold, copper, arsenic, and zinc have all been used for centuries
only mercury and silver now used
Oligodynamic
extremly toxic, easy to develop resistance, cause allergic reactions
bind to and inactivate proteins
oligodynamic
antimicrobial effects in very small amounts
thermal death time
the shortest exposure time necessary to kill all test microbes at a given temperature
thermal death point
the lowest temperature that can be used to kill all microbes in a sample in 10 minutes
radiation
energy emitted from atomic activity and dispersed at high velocity through matter or space
irradiation
bombardment with radiation
ionizing radiation
radiation that ejects orbital electrons from an atom
causes ions to form
harmful to DNA
gamma rays and X- rays
non-ionizing radiation
radiation that excites atoms, but does not ionize them
UV
mutates DNA
how does ionizing radiation work
cold sterilization: performed at low temperatures
penetrates solids and liquids
damages DNA and proteins by breaking bonds
exposure is harmful to humans
to the radiation
irradiated foods are perfectly safe for consumption
used to sterilize fruits, vegetables, meats
how does non-ionizing radiation work
germicidal lamp at 254 nm
doesnt penetrate solids or liquids very well
damages DNA through the formation of pyrimidine dimers
burns human skin
generally used to sterilize air, water
examples of control that are effective against endospores
Physical and chemical methods such as autoclaving, dry heat, and certain disinfectants like hydrogen peroxide and bleach.
primary pathogens
can cause infection and disease in a healthy hot
sometimes called a true pathogen
opportunistic
only causes infection and disease in non-healthy individuals or when introduces in a normally sterile party of the body
virulence
degree of pathogenicity of a parasitic microbe
virulence factors
properties of a pathogen that allow it to successfully invade and cause disease in a host
primary habitant of a disease-causing organisms
phases of an infection
1.) entering the host
2.) attaching to the host
3.) invading and becoming established
1st phase of infection
entering the host
common portals of entry
skin and mucous membranes
GI tract
respiratory system
urogenital tract
pregnancy and birth
skin and mucous membranes 1st phase of infection
usual entry points
damaged skin
mucosal membranes
alternatives
enzymes that break skin barrier
bites
artificial damage
conjunctiva
GI tract 1st phase of infection
must be ingested
pathogens must have a mechanism to survive digestive enzymes and acidic pH of the stomach
usually colonizes small or large intestines