MedChem2 Exam 1: Fletcher Content
1/175
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
176 Terms
What is an acid?
Proton Donor
What is a base?
Proton Acceptor
The pKa of an acid is the pH where is is exactly ______ dissociated
half
Acidic functional groups
Imide, phenol, sulfonamide
Carboxylic acid, acylfulfonamide
Sulfonic acid, HCl
Acid strength and pKa
Acid strength comes from the stability of the conjugate base
The more the negative charge can be delocalized and over more electronegative atoms, the more stable is the conjugate base and hence the stronger is the parent acid (the power it's pKa)
Which is more acidic? Alcohol or Phenol? Why?
Phenol (pKa 10) is more acidic than alcohol (pKa 15), this is because phenol has 4 resonance structures, whereas alcohol only has 1 anion structure
Which is more acidic: Imide or Amide? Why
An Imide is more acidic than an amide. Amides have 2 resonance structures with a pKa of 15; whereas Imides have 3 resonance structures and a pKa of 10
Why are carboxylic acids so acidic if they only have 2 resonance structures?
They are more acidic due to the charge being delocalized onto 2 electronegative atoms (oxygen), which helps stabilize the conjugate base
Which is more acidic: sulfonamides or acylsulfonamides? Why?
An acylsulfonamide is more acidic than a sulfonamide, due to the conjugate base being more stable with 4 resonance structures, sulfonamides only have 3 resonance structures
Basic functional groups
Aniline, pyridine, imidazole
primary, secondary, and tertiary amines
Guanidine
Why are amides not basic?
Amides are neutral due to the long pair of electrons being delocalized into the adjacent carbonyl (not available for acid/base reactions)
Why are pyrroles not basic?
The lone pair of electrons is pulled into (delocalized) the conjugated ring to contribute to the aromaticity
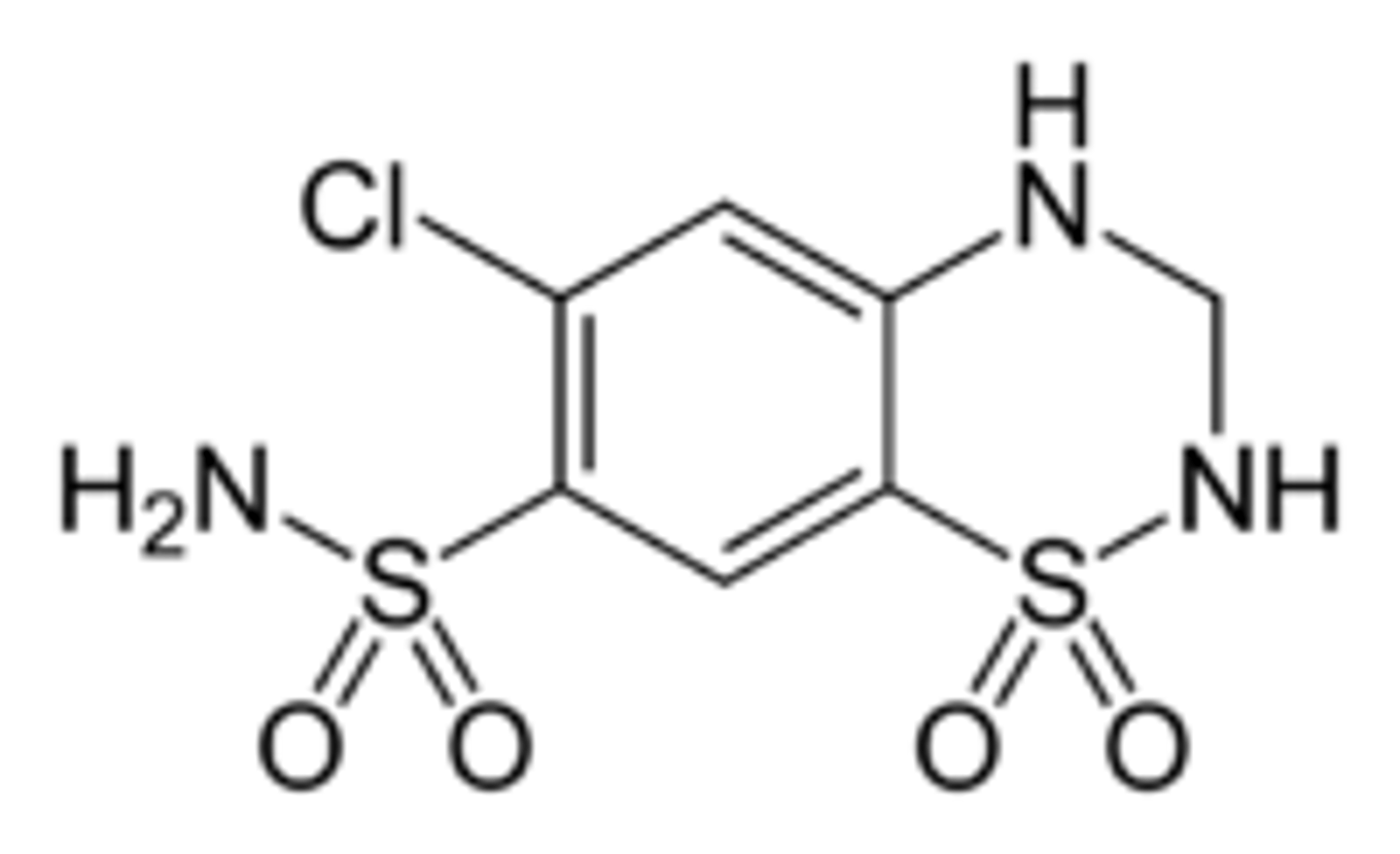
Hydrochlorothiazide "N" group
HCTZ contains a secondary amine group with electrons being delocalized into an aromatic ring
This is an aniline derivative with a pKa of 5
Soluble at pH 3 and below
Do benzoic acid and sodium benzoate make good injectables? (in blood pH buffer system)
Yes, both species are ionized/soluble at pH 7+
This is due to both are soluble at the same buffered pH because the same equilibria are established in each case
Do tertiary amines and HCl salts make good injectables? (in buffered solution)
Yes, both are soluble at pH 6 and below and the same equilibria are established allowing for complete dissolution
T/F: water acts as a buffer and a species dissolved in water will not affect pH
False!! Water is NOT a buffer, the species dissolved in water will affect pH
How do you determine the pH of the resulting solution upon dissolution of a salt in water?
Add pKa values of the parent and base of the salt and then divide by 2
What is the pH of a solution of NaCl in water?
NaCl (salt) = HCl + NaOH
HCl pKa = 0, NaOH pKa = 14
(0+14)/2 = 7
How do you know if two salts in an admixture dissolve completely (i.e. compatible) in BUFFER?
If both salts (look at the parent compound) are independently soluble in the buffer, then they will also be soluble as an admixture
Look at parent structure of each salt, determine what pH it will be soluble at, then compare to given buffer
How do you know if two salts in an admixture dissolve completely (i.e. compatible) in WATER?
Add the pKa values of the parent acid and base of the salt and divide by 2 (do for each salt)
Then add these 2 salt pH's together and divide by 2 to determine the resulting pH of the solution
Last, determine if the parent compound of each salt is soluble in the resulting pH
No -> incompatible; Yes -> compatible
Bioisosteres for Carboxylic Acids
Chemical substituents or groups with similar physical and/or chemical properties that produce broadly similar biological properties
Reasons to use bioisosteres
Reduce toxicity
Change bioavailability
Modify activity
Alter metabolism
Why might someone opt to use tetrazole as a bioisostere for a carboxylic acid?
Carboxylic acid has a negative charge making it poorly cell permeable and is prone to metabolism
Tetrazole is more cell permeable as the charge can be delocalized, additionally, it is more resistant to metabolism and has the same pKa as a carboxylic acid
Bioisosteres for carboxylic acids:
tetrazole
acyl sulfonamide
3-hydroxyisoxazole
Drug Incompatibility
Interactions between two or more substances, which leads to changes in therapeutic, physical, and chemical properties of the pharmaceutical dosage form
Therapeutic Incompatibility
Modification of the therapeutic effect of one drug by another drug (drug-drug interactions) i.e. synergism/antagonism
Physical Incompatibility
Interaction between two or more substances, which leads to a change in color, odor, taste, viscosity and/or morphology (often called pharmaceutical incompatibilities)
Often occurs as a result in pH
Chemical Incompatibility
A chemical reaction between two or more substances, which leads to a change in chemical properties of the drug
Chemical Incompatibilities: Types
Oxidation
Hydrolysis
Factors leading to oxidation:
oxygen
light
temperature (higher)
pH (basic)
dosage form
unsaturated bonds
How do we protect drugs from oxidation?
Addition of anti-oxidants
Addition of chemicals that form complexes with metals
Protection from light
Pharmaceutical dosage forms (solid vs solution)
Storing at low temperature
Storage under inert (nitrogen) atmosphere
What are some indications that a drug has undergone oxidation?
Change in color, odor, viscosity of dosage form
Pink and stinks!
What chemical groups are prone to oxidation?
phenols
catechols
pyridines
thiols
sulfides
thiophenes
Factors leading to hydrolysis
water
faster at pH extremes
accelerated by high temperatures
How do we protect drugs from hydrolysis?
storage in a dry place
use buffered solution ( close to 7) if must be aqueous solution
package with materials that absorb water
What chemical groups are prone to hydrolysis?
Esters
Amides
Factors leading to drug-drug reactions
Combination of electrophilic and nucleophilic drugs
What is drug metabolism?
When a drug enters the body and is altered by metabolic enzymes to be more easily excreted
May be metabolized into an active, inactive, or toxic metabolites
Phase I Metabolism
Drug metabolism where the drug is converted into more polar (water-soluble) molecules that can be excreted
Often done by CYP450 enzymes
Generally involve oxidation, reduction and hydrolysis
Mostly occurs in the liver
Chemical groups prone to oxidation in phase I metabolism
N-methyl groups
Aromatic rings (para position is preferred)
Alkyl groups (terminal positions)
Alicylic rings (most exposed position)
Chemical groups prone to reduction in phase I metabolism
Nitro groups
Azo groups
Aldehyes
Ketones
Chemical groups prone to hydrolysis in phase I metabolism
Esters
Amides
How can you block N-oxidation (i.e. of a pyridine/amine)
Lower the pH and block the N lone pair (proton will now be bonded to lone pair so oxidation is no longer possible, or at least now much slower
Heteroatom Dealkylation
Requires abstraction of an alpha H radical
Then a-hydroxylation can occur
N groups become semi-aminals, O groups become hemi-acetals (both are unstable groups)
These further break down into formaldehyde and their respective O/N groups
Phase II Metabolism: Conjugation Reactions
A polar molecule is attached to a suitable polar "handle" that is already on the drug or was attached during phase I
Generally occurs in the liver
Resulting conjugate has greatly increased polarity
Phenols, alcohols, and carboxylic acids undergo glucuronidaiton reactions
Sulfation is another type of Phase II reactions
Types of conjugates added during phase II reactions
Glucuronyltransferases for phenols, alcohols, and carboxylic acid gorups
Sulfotransferases for phenols, alcohols, and amines
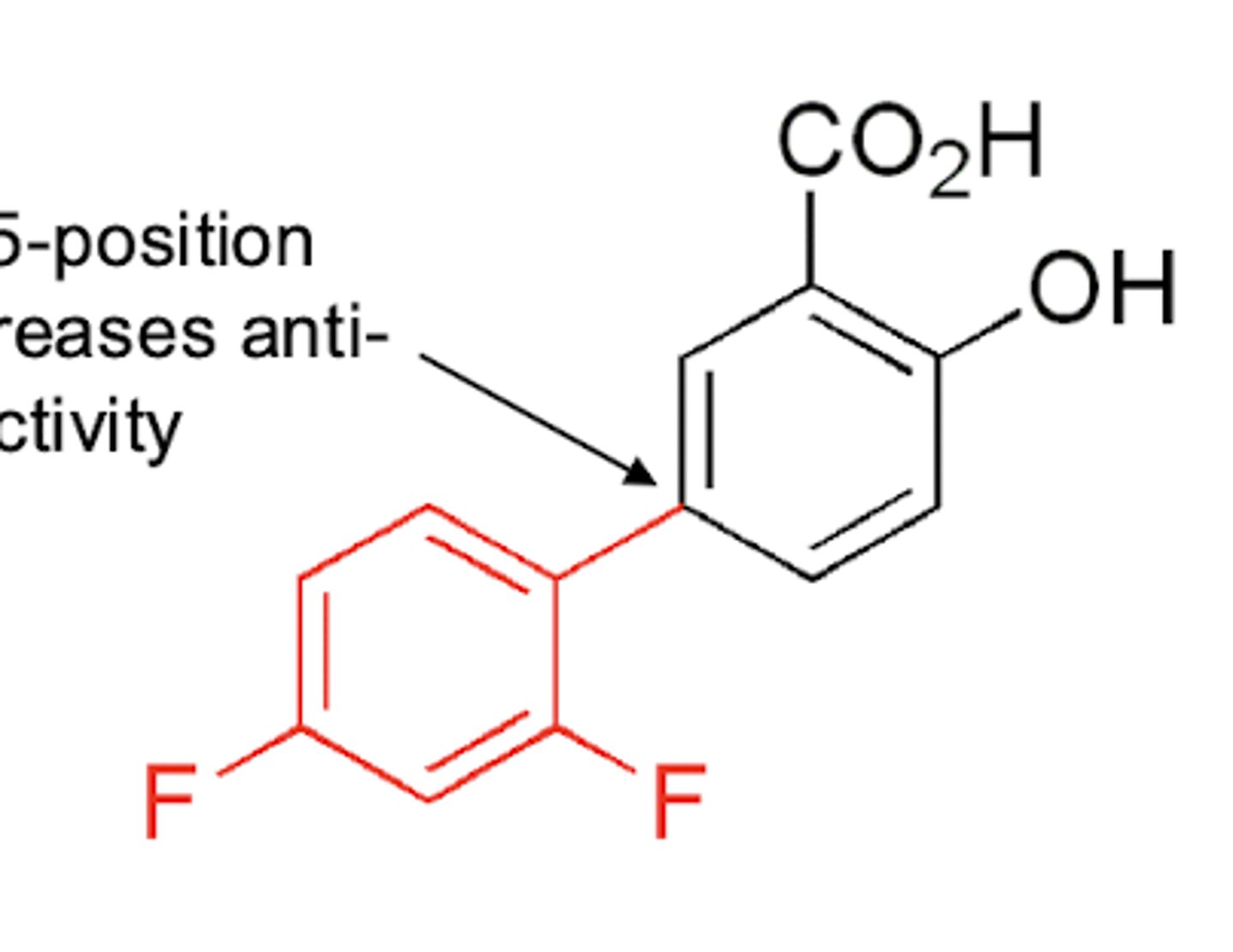
How do we block Para-Hydroxylation?
By using fluorine (versus H) to inhibit hydroxylation from occuring
This works due to H and F being roughly the same size
How do we block metabolism of an aromatic alkyl group?
By replacing CH3 with CF3 or Cl, these are isometric in size with CH3 and reduce metabolism
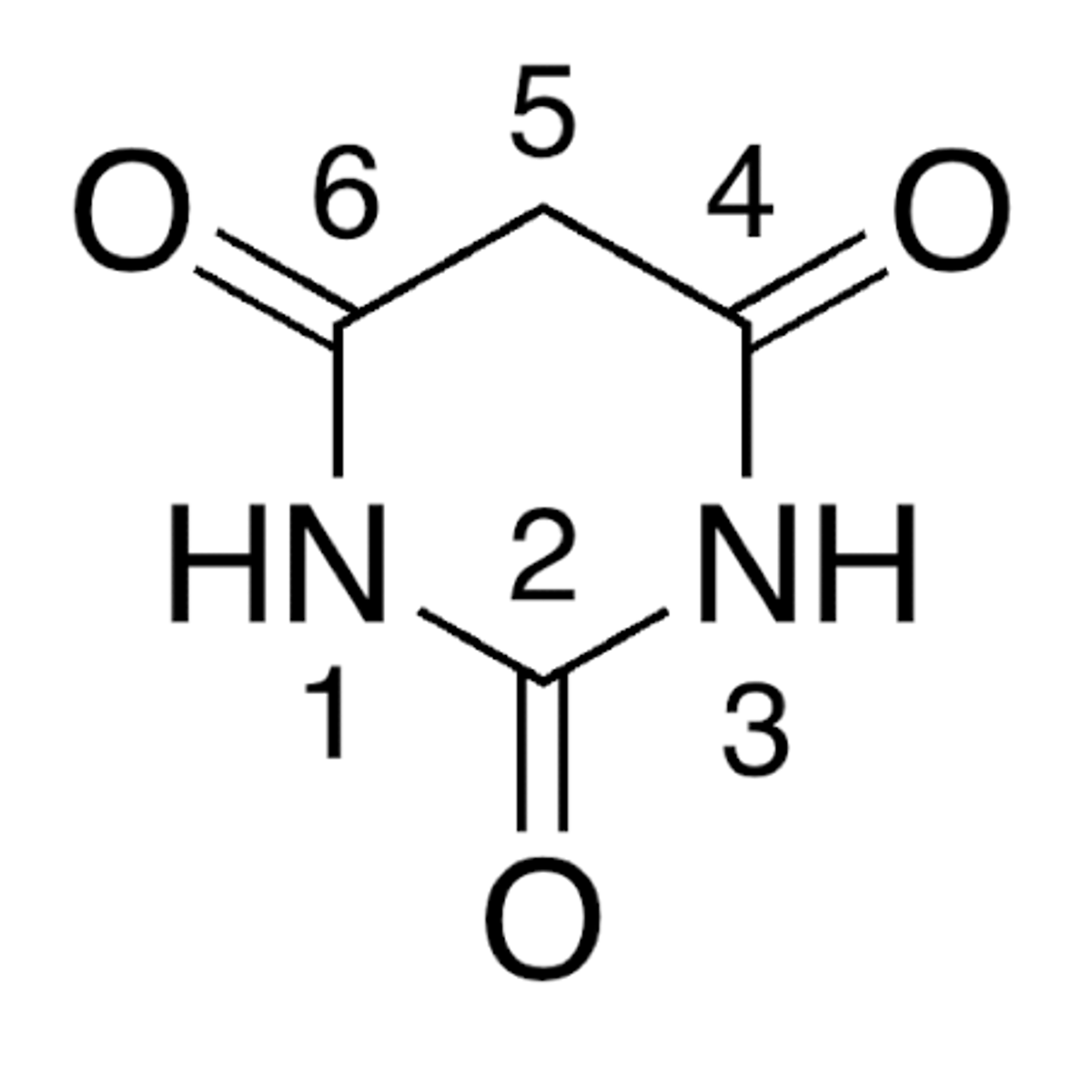
Why does barbituric acid have a low pKa of 4?
Tautomerism
When deprotonated the aromatic anion is stabilized
Barbituric Acid Pharmacophore
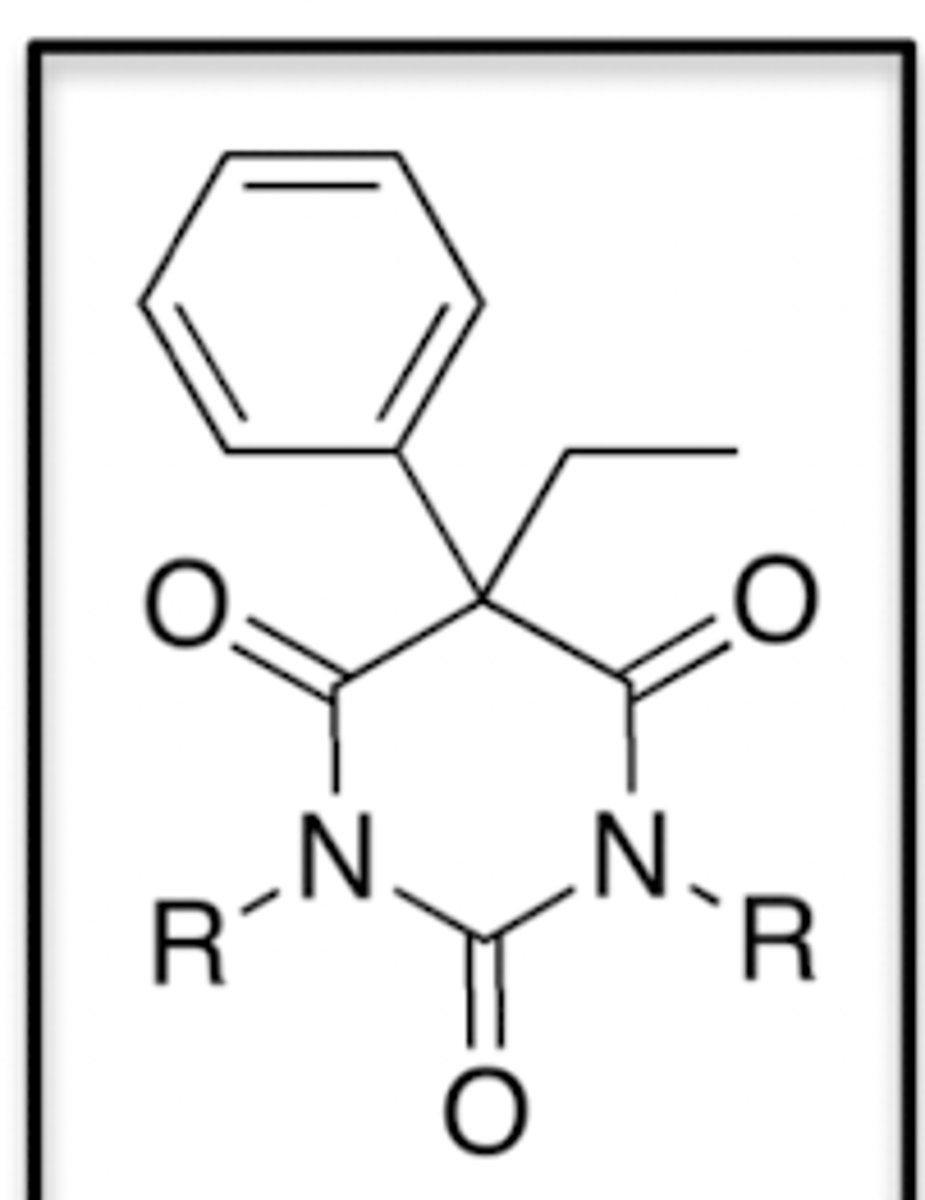
Important SAR pieces for Barbiturates: 5' substituents
The combined number of carbon atoms of the 5' substituents should be between 6-10
Unsaturation (double bonds) is better than saturated
Branched is better than unbranched
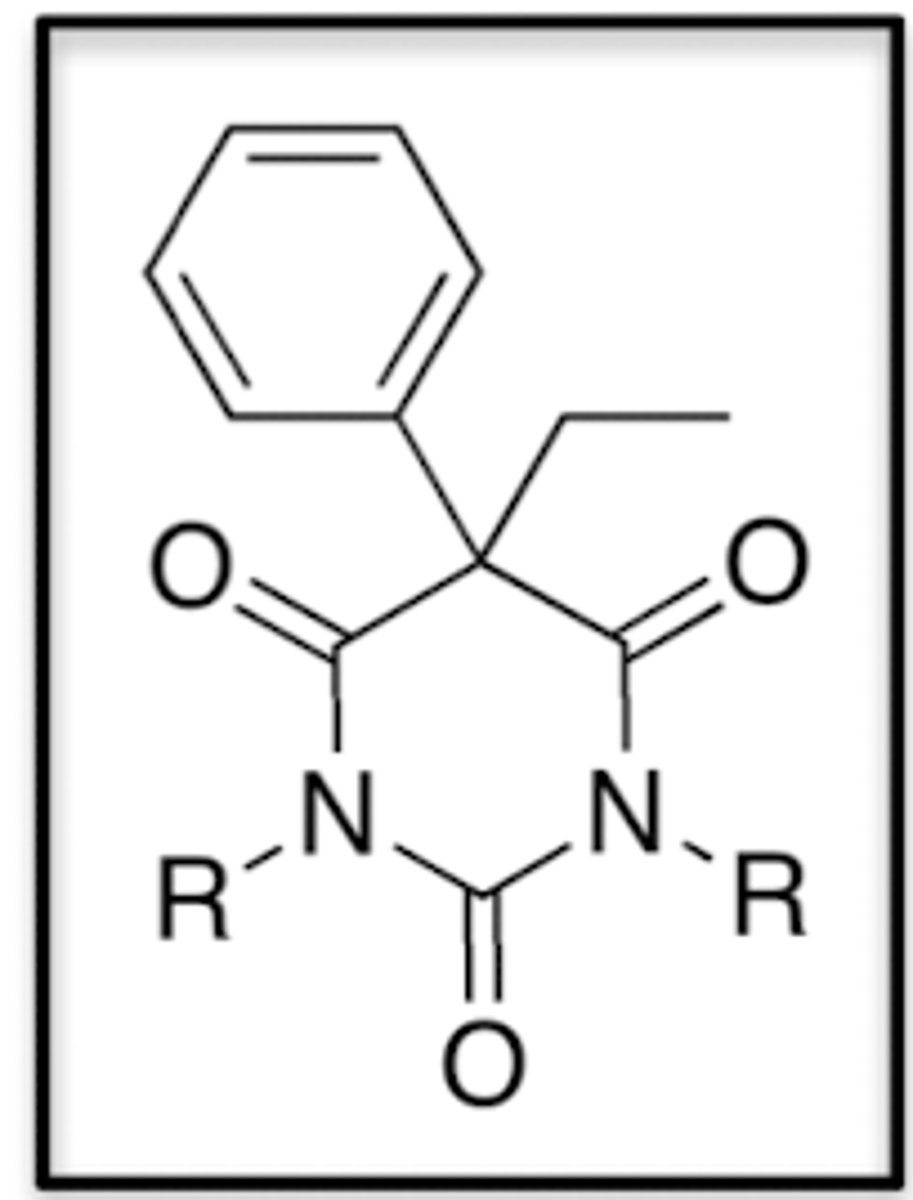
Important SAR pieces for Barbiturates: Acidic NH
At least one acidic NH is required for activity at t=0
If no acidic NH, then the molecule is not active at t=0
If an acidic H can be formed via time-delay metabolism then the molecule can be active (via N-alkylation)
Important SAR pieces for Barbiturates: 2' substituents
Replacement of the 2-substituted oxygen with sulfur increases lipid solubility
Since maximal thiobarbiturate levels are quickly reached, onset of activity is rapid
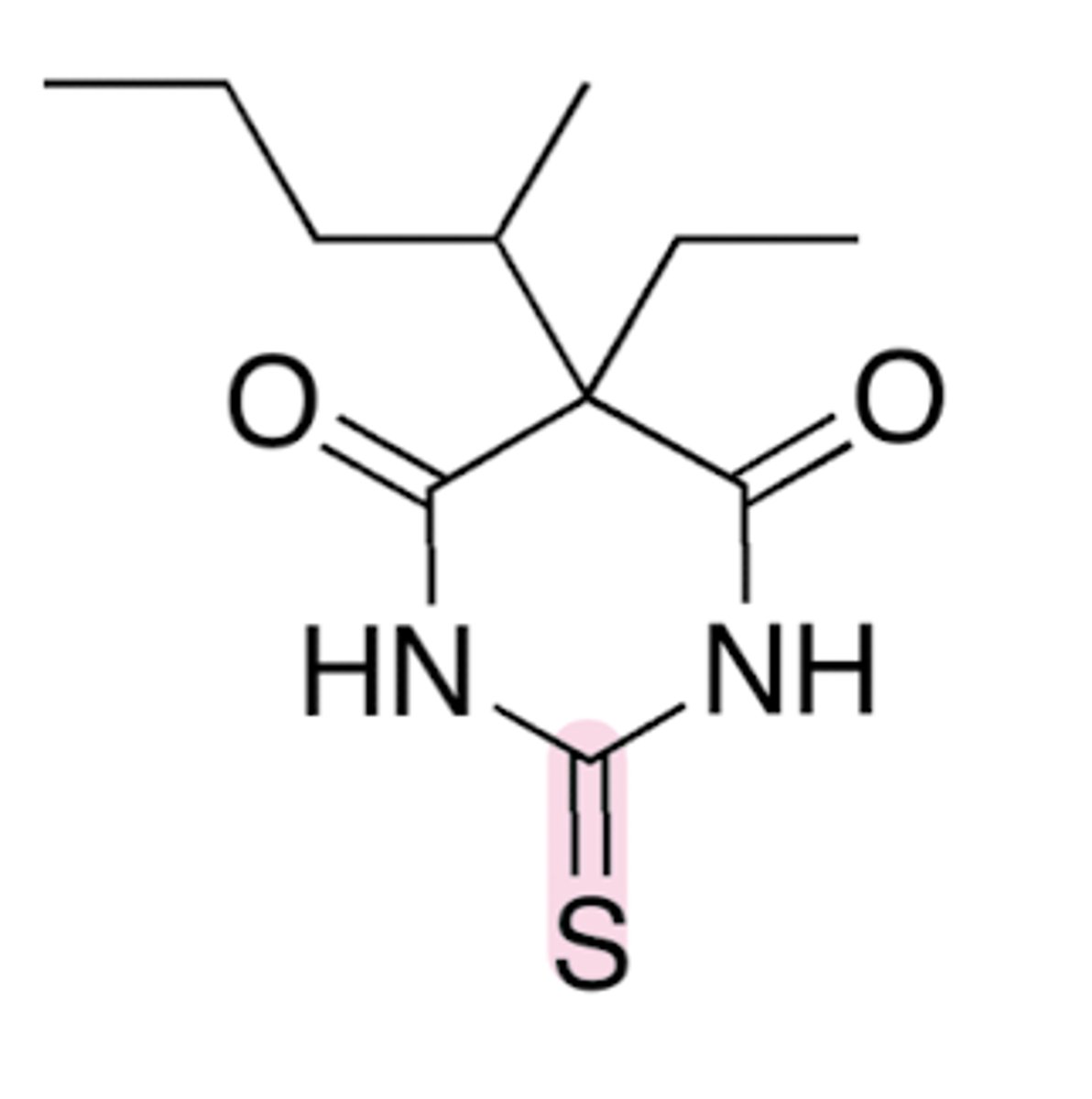
Why do we need prodrugs of Barbiturates?
They are poorly water-soluble
We can utilize the N-dealkylation mechanism of N-methylated barbiturates to make them water-soluble prodrugs (inactive)
What are the two ways to activate an Succinate Ester Barbiturate Prodrug?
Ester hydrolysis
Intramolecular decomposition
How are Phosphate Ester Barbiturate Prodrugs activated?
Ester hydrolysis
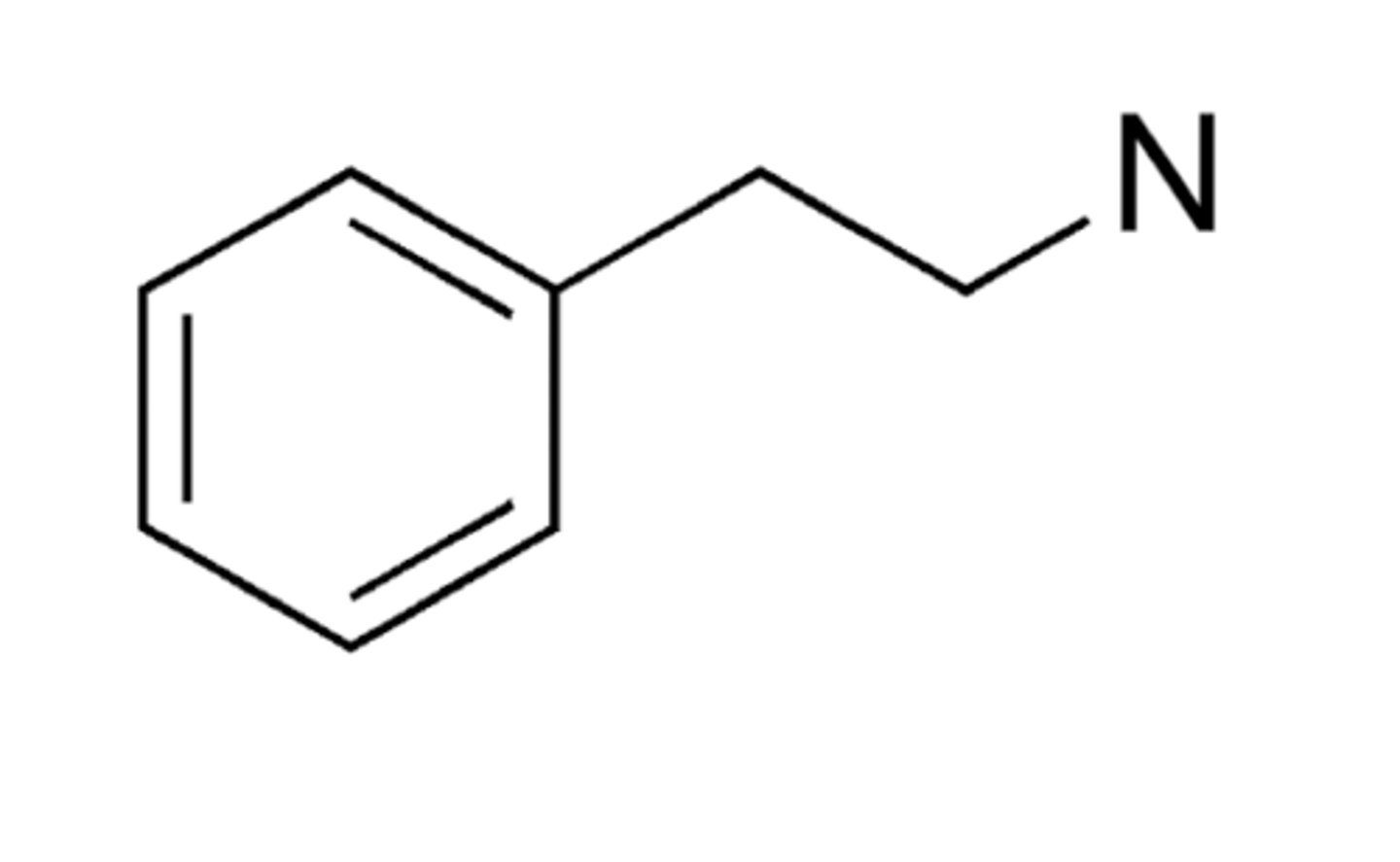
Pharmacophore of an Amphetamine
Aromatic ring
2-C linker
Basic amine
Amphetamine Pharmacophore: Beta Position
Beta -OH: reduces potency
Beta =O: retains potency
Amphetamine Pharmacophore: Aromatic Substitution
Unsubstitution of the aromatic ring is preferred (can be H/F, these are isosteric)
Amphetamine Pharmacophore: Chiral Center
S is preferred over R
Amphetamine Pharmacophore: Terminal Amine
NHMe > NH2 > NHR > NR2
Amphetamine Pharmacophore: alpha-methyl group
Homologation decreases potency, replacement by H decreases potency
Requirements for N-dealkylation
Nitrogen in the alpha position
Abstractable hydrogen radical
Internal Salt
A molecule with both an acidic and basic group that allows the molecule to be soluble in water, but is insufficient having one of those groups alone for water-solubility
How do we determine if an internal salt is soluble in water?
Calculate molecules resulting pH by adding the pKa's of the acid and basic group and divide by 2.
Then see if one of these groups is completely ionized at this pH
If one is soluble, then it is soluble in water
T/F: an HCl salt of an amino acid-based compound is more resistant to oxidation than is a sodium salt
True
Why are HCl salts more resistant to oxidation compared to sodium salts?
HCl salts can protonate amino groups and make the electrons less available to oxidation processes
Anticonvulsant General Pharmacophore
Barbiturates: X= CONH
Hydantoins: X= NH
Oxazolidinediones: X = O
Succinimides: X= CH2
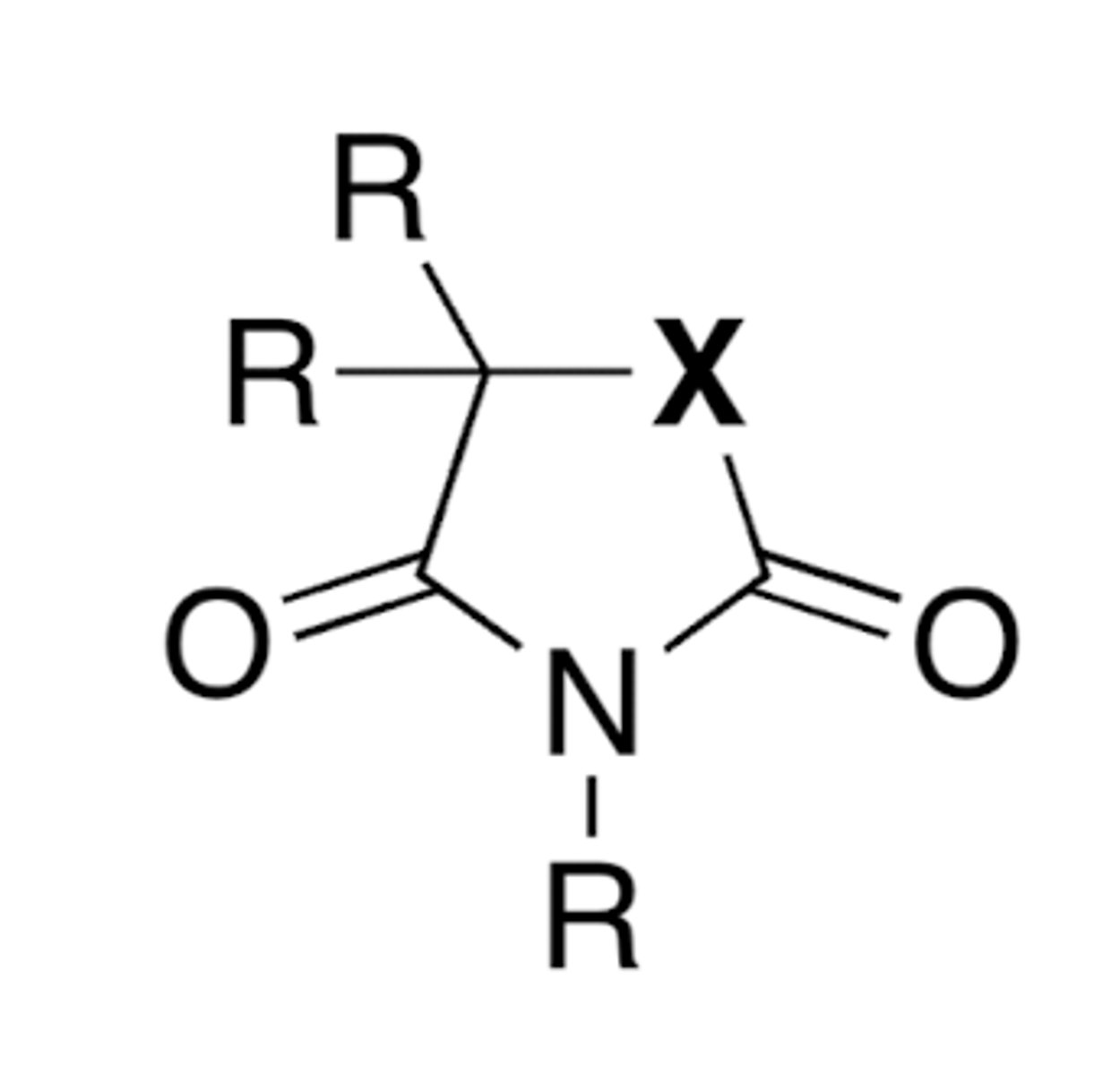
Barbiturates: X= CONH: Receptors + Drugs
Work on GABA receptors and Na+ channels
Drugs: phenobarbital and mephobarbital
Hydantoins: X= NH: Receptors + Drugs
Works on Na+ channels
Drugs: phenytoin, mephenytoin, and ethotoin
Oxazolidinediones: X= O: Receptors + Drugs
Works on Ca2+ channels
Drugs: trimethadione and paramethadione
Succinimides: X= CH2: Receptors + Drugs
Works on Ca2+ channels
Drugs: ethosuximide, phensuximide, and methsuximide
Is Vibagatrin water soluble? (internal salt)
NO!
Resulting pH: 6.5
Neither a carboxylic acid nor primary amine is ionized at this pH
Is Tiagabine water soluble (internal salt)
NO!
Resulting pH: 6.5
Neither a carboxylic acid nor primary amine is ionized at this pH
Is a HCl Salt soluble in water? (tertiary amine + HCl)
Yes!
Resulting pH: 4
Both groups will be ionized
NSAID Definition
A drug that exerts analgesic, antipyretic, anti-inflammatory, (and anti-platelet) properties
Example: Aspirin
Is acetaminophen an NSAID?
No, it doesn't have anti-inflammatory effects
It does have analgesic and antipyretic affects
Aspirin Structure
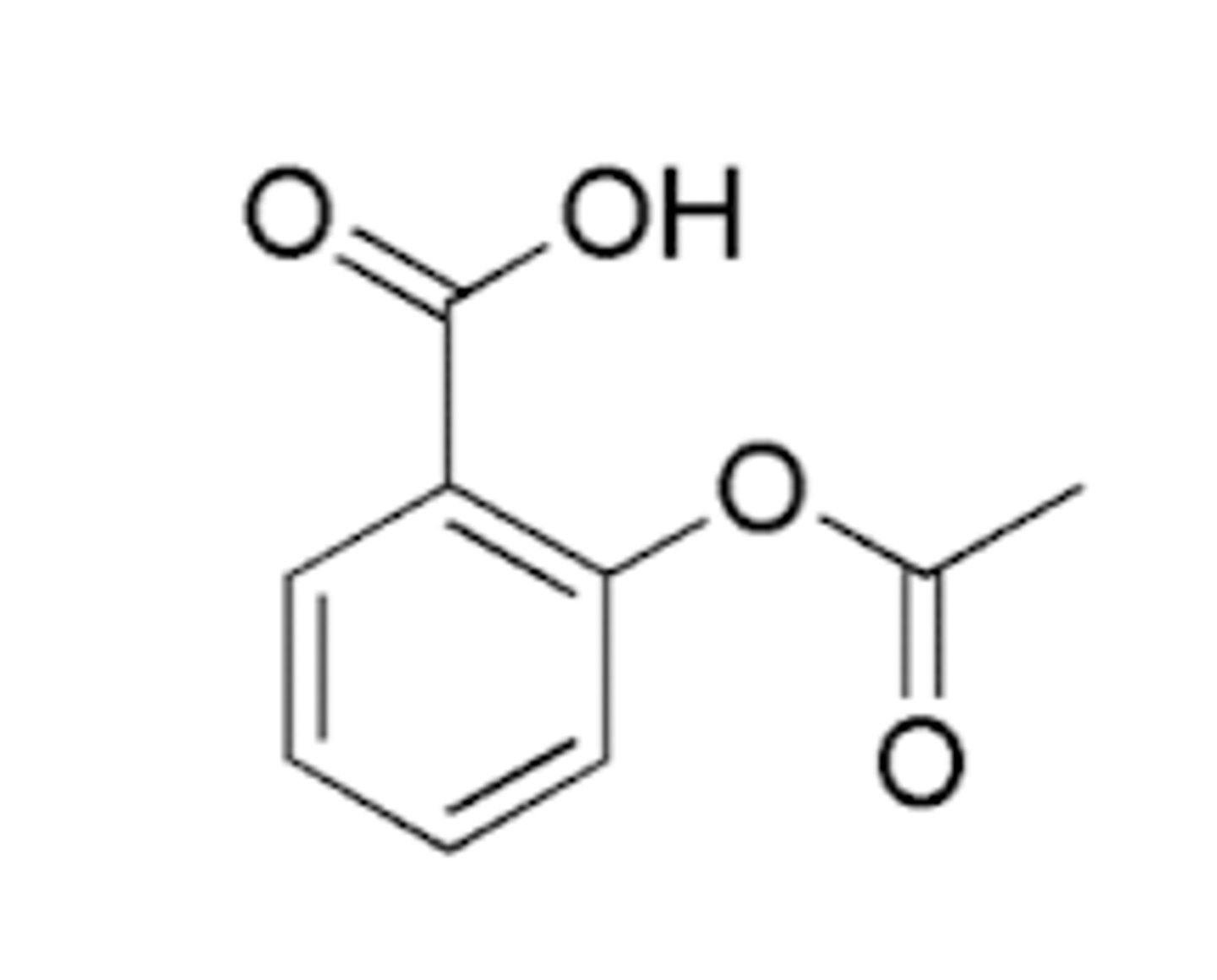
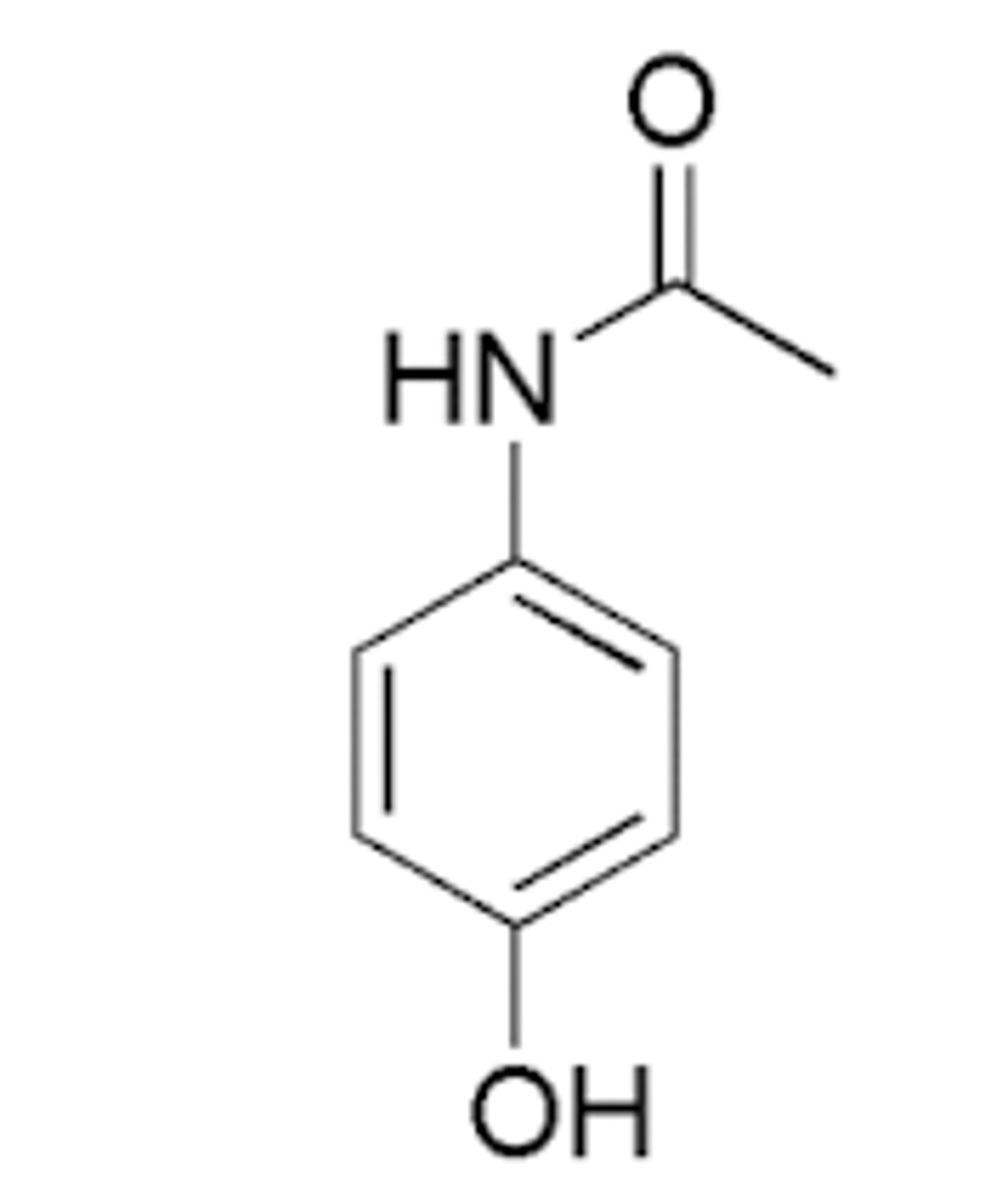
Acetaminophen Structure
Ibuprofen Structure
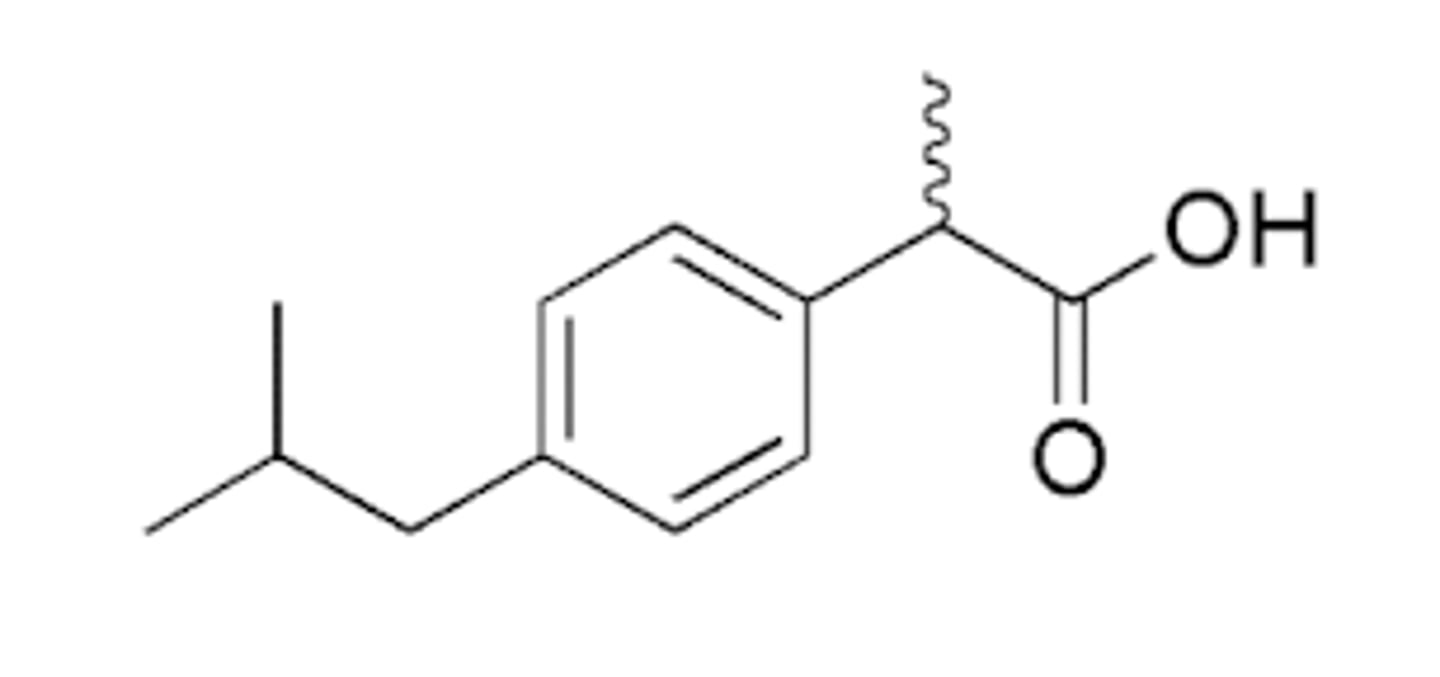
How do NSAIDs work?
NSAIDs inhibit PG synthesis through binding the active site of COX-1 and COX-2
T/F: Aspirin is a reversible COX(1/2) inhibitor
False, it is irreversible (but is more potent against COX-1 than COX-2)
COX-1
Constitutive enzyme
Responsible for maintenance and protection of the GI tract
COX-2
Inducible enzyme
Synthesized for inflammation and pain
Active sites of COX enzymes
Oxidation of arachidonic acid (AA) to PGG2
Reduction of PGG2 to PGH2
Pharmacophore of Salicyclates
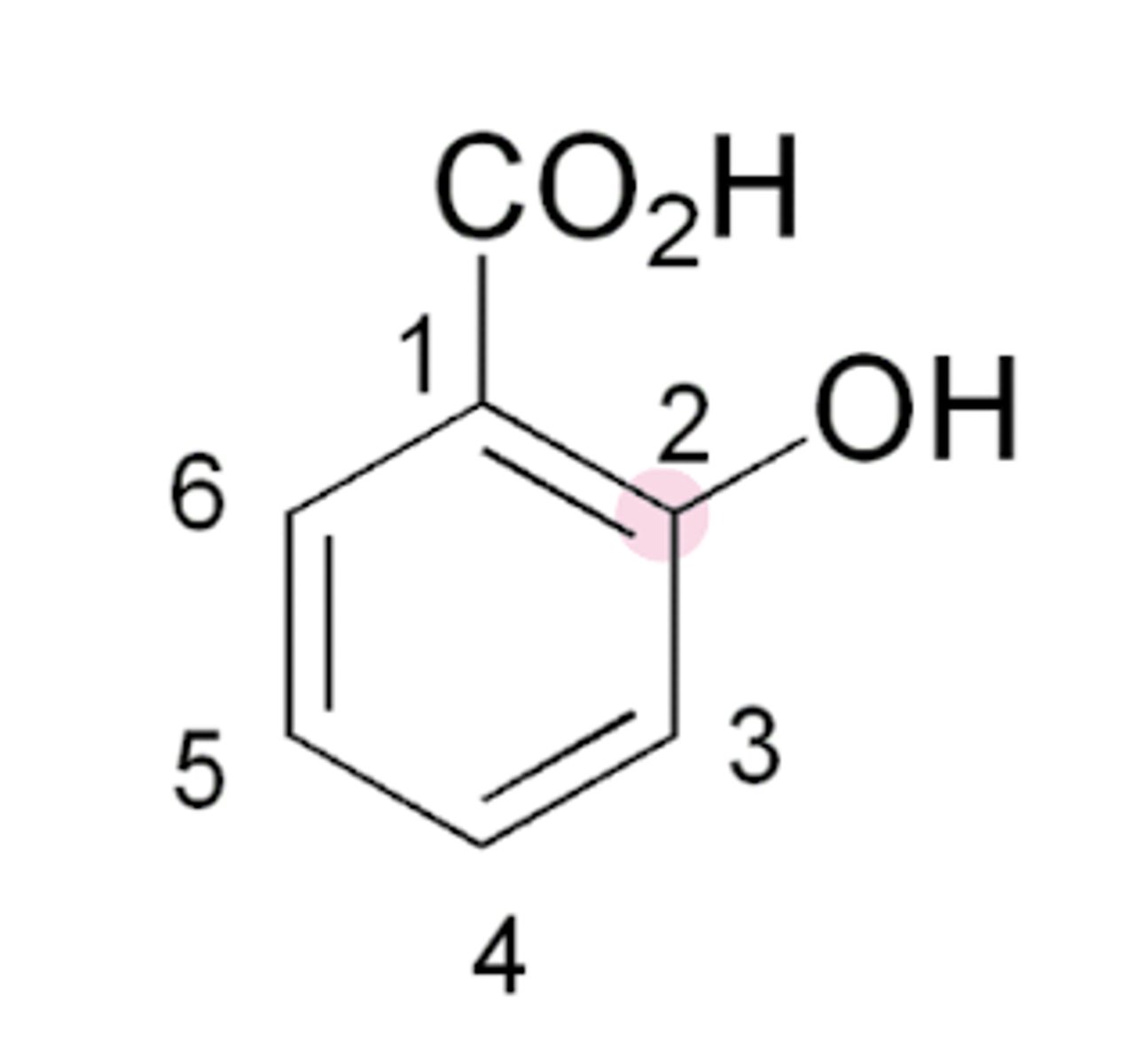
Groups needed for salicylate anti-inflammatory activity
CO2H and OH groups ortho/next to one another
What diminishes/abolishes salicylate activity
No OH group ortho to COOH
Strongest-Weakest: Order of salicylate strength
Aspirin > Salicylic acid > Benzoic acid
Aspirin: why is it so potent?
Aspirin is potent due to the acetyl group which allows it to irreversibly inhibit COX1/2
Why is Salicylamide as effective as aspirin, but exhibits no anti-inflammatory properties?
Has a CONH2 group instead of a CO2H group
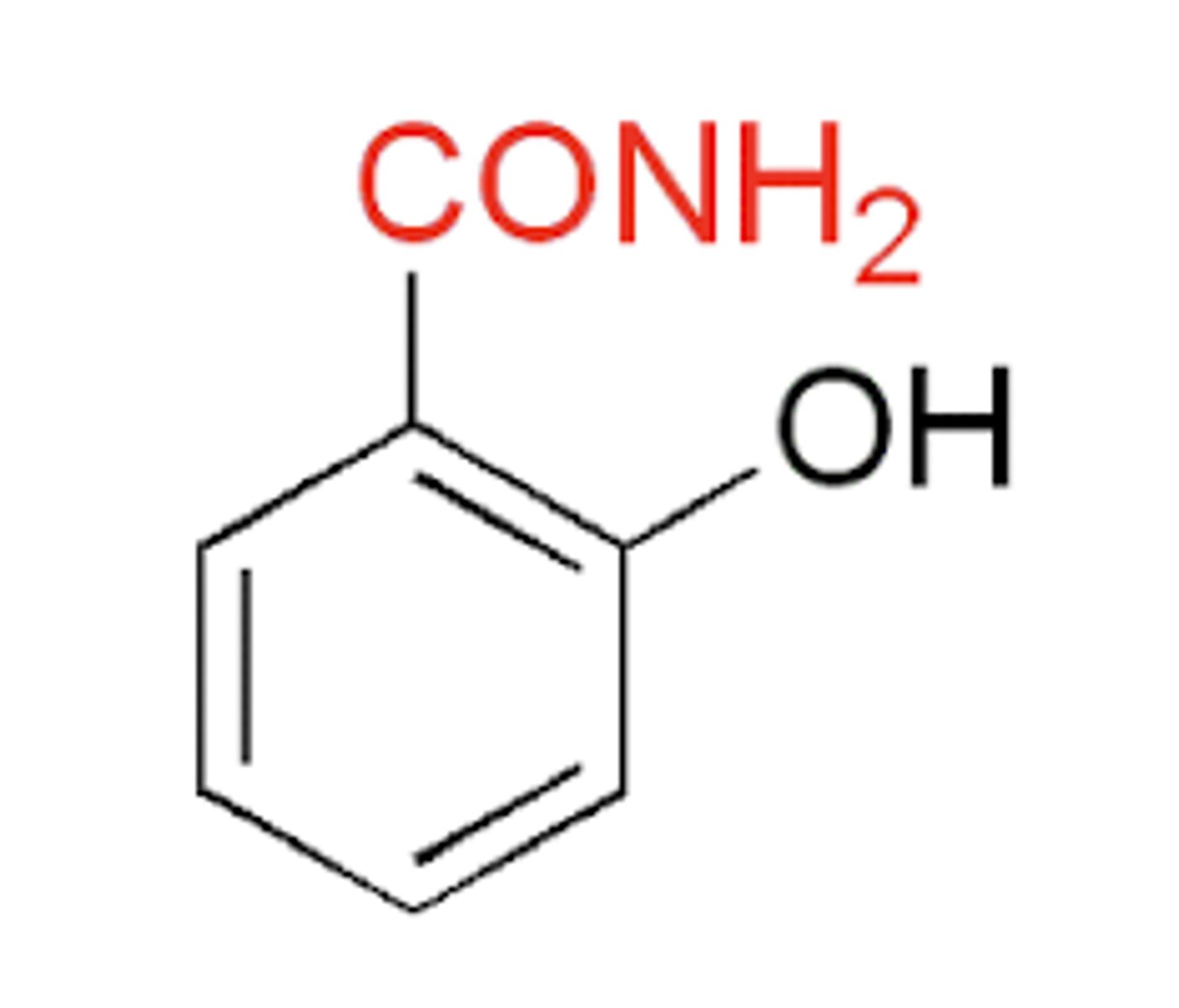
How can you increase anti-inflammatory properties of a salicylate?
First it requires the CO2H and OH groups ortho
Then substitution at the 5-position increase anti-inflammatory properties
Addition of the OH meta to the COOH also increases potency
How do NSAIDs inhibit COX?
They reversibly inhibit COX via a salt bridge (electrostatic interaction) using the Arg120 residue
Blocks the channel so AA cannot access, which temporarily inhibits PG synthesis
How does Aspirin irreversibly inhibit COX?
Aspirin irreversibly inactive the enzyme via covalent modification of Ser-530 (COX-1) and Ser-516 (COX-2)
Cannot react with AA to make PGs after this occurs
Also binds reversibly as salicylate (Arg120 salt bridge)
NSAIDs cause gastric damage by Dual-Insult mechanisms, what are they?
1: ion trapping/direct acid damage
2: inhibition of PG synthesis especially PGE2
Primary NSAID Insult: Ion Trapping
When a neutral molecule is permeable and crosses into a different pH causing it to become ionized, resulting in it being trapped, allowing for accumulation and toxicity
Secondary NSAID Insult: COX1/2 Inhibition
NSAIDs generally inhibit PG formation, this includes cytoprotective PGE2 and PGI2 which inhibit gastric secretion (both) and secretion of alkaline mucus (PGE2)
Causes vomiting, heartburn, GERD, and PUD
Which drug is the most toxic: acetanilide, phenacetin, or acetaminophen?
Acetaminophen due to the formation of a Michael acceptor -> NAPQI
How is APAP metabolized?
Major: sulfation
Minor: glucuronidation
<5% CYP450 -> NAPQI
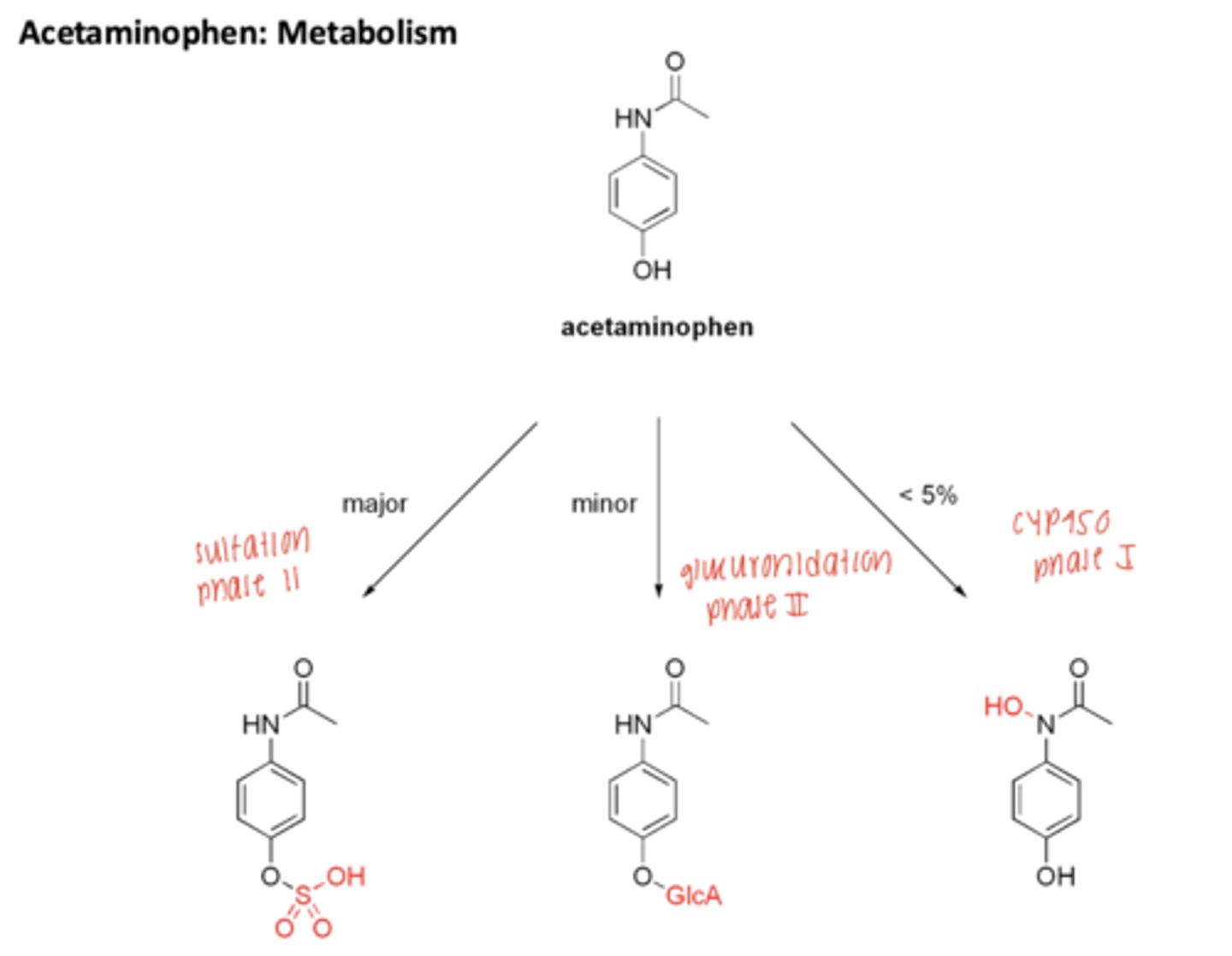
Metabolism of APAP: 3 Routes
(normal dosage- small amounts of NAPQI made; high doses- larger amounts of NAPQI
Route 1: toxic metabolite is excreted after conjugation with glutathione
Route 2: if followed with antidote NAC, is metabolized and excreted renally
Route 3: No NAC, hepatic necrosis, renal failure, and death