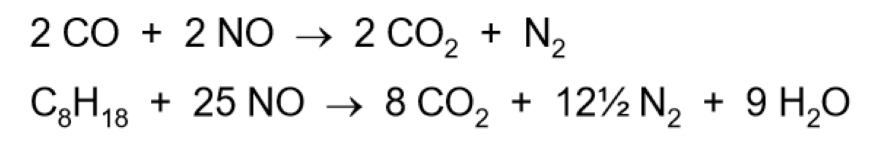Chemistry- alkanes learn eqs
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
Why are alkanes among the least reactive organic compounds
Their lack of a functional group
No significant diffference in electronegativity between the C and H atoms (2.5 and 2.1 respectively) making them un susceptible to attack by nucleophiles and electrophiles
2 uses of alkanes in industry
Fuels- short chain alkanes are very flammable and burn with a clean flame
They are feedstock (staring materials) for a range of other compounds eg solvents, lubricants, plastics etc
Fully describe fractional distillation process
Crude oil heated in furnace
A mixture of liquid and vapours pass into the bottom of the fractionating column where there is a temperature gradient
Shorter chain alkanes condense nearer the top of the column where it is cooler bc they have lower boiling points
Longer chain alkanes condense nearer the bottom of the column where it is hotter bc they have higher boiling points
What does the smaller the molecule mean in fractional distillation
low boiling point
Very volatile
Flows easily
Ignites easily
What does the larger the molecule mean in fractional distillation
high boiling point
Not very volatile
Does not flow easily
Does not ignite easily
What is the solubility of alkanes in water and in most organic solvents
Alkanes are non polar due to the similarities in electronegativity between carbon and hydrogen
and so are insoluble in water
But soluble in most organic (non polar) solvents
Trend in bp of alkanes as chain length increases
As chain length increases the boiling point of the alkanes increases as they get more carbons in their formula, the more points of contact between molecules creating more Van der Waals forces
This makes the Van der Waals forces stronger meaning more heat energy is required to separate the molecules when boiling
Trend in bp as branching increases
bp decreases
Less points of contact between molecules
Less van der waals forces induced
Weaker vdw forces
Less energy required to separate the molecules when boiling
What is cracking
The conversion of large hydrocarbons to smaller hydrocarbon molecules by breakage of C-C bonds
General equation for cracking
High Mr alkanes → smaller Mr alkanes + alkenes + hydrogen
2 types of cracking
Thermal cracking
Catalytic cracking
Conditions for thermal cracking and how does it occur
High temperature 700K-1200K / 400-900ºC
High pressure 7000 kPa
Occurs by homolytic fission creating free radical intermediates
Products of thermal cracking and uses
Produces mostly alkenes eg ethene used for making polymers and ethanol
Sometimes produces hydrogen used in the Haber Process and in margarine manufacture
Conditions for catalytic cracking
Low pressure- slightly below atmospheric
High temperature 720K/450ºC
Zeolite catalyst
Why are the temperatures much lower for catalytic cracking
The catalyst lowers the Ea of the reaction
Products of catalytic cracking, uses, and why
Produces branched and cyclic alkanes and aromatic hydrocarbons
Used for making motor fuels
branched and cyclic hydrocarbons burn more cleanly and are used to give fuels a higher octane number
Which is cheaper thermal or catalytic cracking?
Catalytic cracking as it saves energy as lower temperatures and pressures are used
What other reactions may take place during thermal cracking
Dehydrogenation- the removal or loss of hydrogen from alkanes to produce alkenes
Isomerisation- occurs when un branched alkanes are converted into branched isomers or when cyclic alkanes undergo rearrangement
Cyclisation- alkanes are converted to cyclic alkanes and aromatic hydrocarbons, often with the loss of hydrogen
Structure of zeolite catalyst
Honeycomb shape for a large surface area
Heterogenous catalyst
Adsorption desorption theory??
Why do alkanes make good fuels
Readily burn in the presence of oxygen
So highly exothermic combustion
Fuels release heat energy when burnt
When does complete combustion take place & products
When alkanes are burned in excess oxygen
Products are carbon dioxide and water
When does incomplete combustion take place & products
in limited amount of oxygen
Produces carbon monoxide (toxic) and or carbon (producing sooty flame)
Which releases less energy per mole complete or incomplete combustion
Incomplete
Environmental consequence of carbon
Carbon (soot) can cause global dimming- reflection of the sun’s light
How is sulfur dioxide gas formed from combustion
Sulfur containing impurities are found in petroleum fractions which produce SO2 when they are burned
Coal is high in sulfur content and large amounts of sulfur oxides are emitted from power stations
Environmental consequence of sulfur dioxide
Dissolves in atmospheric water and produces acid rain
How can SO2 be removed from waste gases from furnaces
Flue gas desulfurisation
The gases pas through a scrubber containing basic calcium oxide which reacts with the acidic sulfur dioxide in a neutralisation reaction
With CaO:
SO2(g) + CaO(g) → CaSO3(s)
With CaCO3:
CaCO3(s) + SO2(g) → CaSO3(s) + CO2(g)
What is the calcium sulphite formed from flue gas desulfurisation used for
Used to make calcium sulphate for plasterboard
Environmental consequence of release of unburnt hydrocarbons
Causes photochemical smog (caused by the reaction of unburnt hydrocarbons and nitrous oxide using sunlight)
Unburnt hydrocarbon + NOx → (UV light) photochemical smog
How are nitrogen oxides formed and equations and environmental consequences of products
From the reaction of N2 and O2 inside car engines
The high temp and spark in the engine provides sufficient energy to break the strong N2 bond
N2(g) + O2(g) → 2NO(g)
2NO(g) + O2(g) → 2NO2(g)
NO is toxic and can form acidic gas NO2
NO2 is toxic and acidic and forms acid rain
NO2 + H2O + 0.5O2 → 2HNO3(aq)
Consequence of carbon particles (particulates)
Exacerbate asthma and cause cancer
Respiratory problems
What do catalytic converters do and equations
Remove CO, NOx and unturned hydrocarbons (eg octane, C8H18) from the exhaust gases, turning them into ‘harmless’ CO2, N2 and H2O