Orgo Test 3 (past reactions)
1/67
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
68 Terms
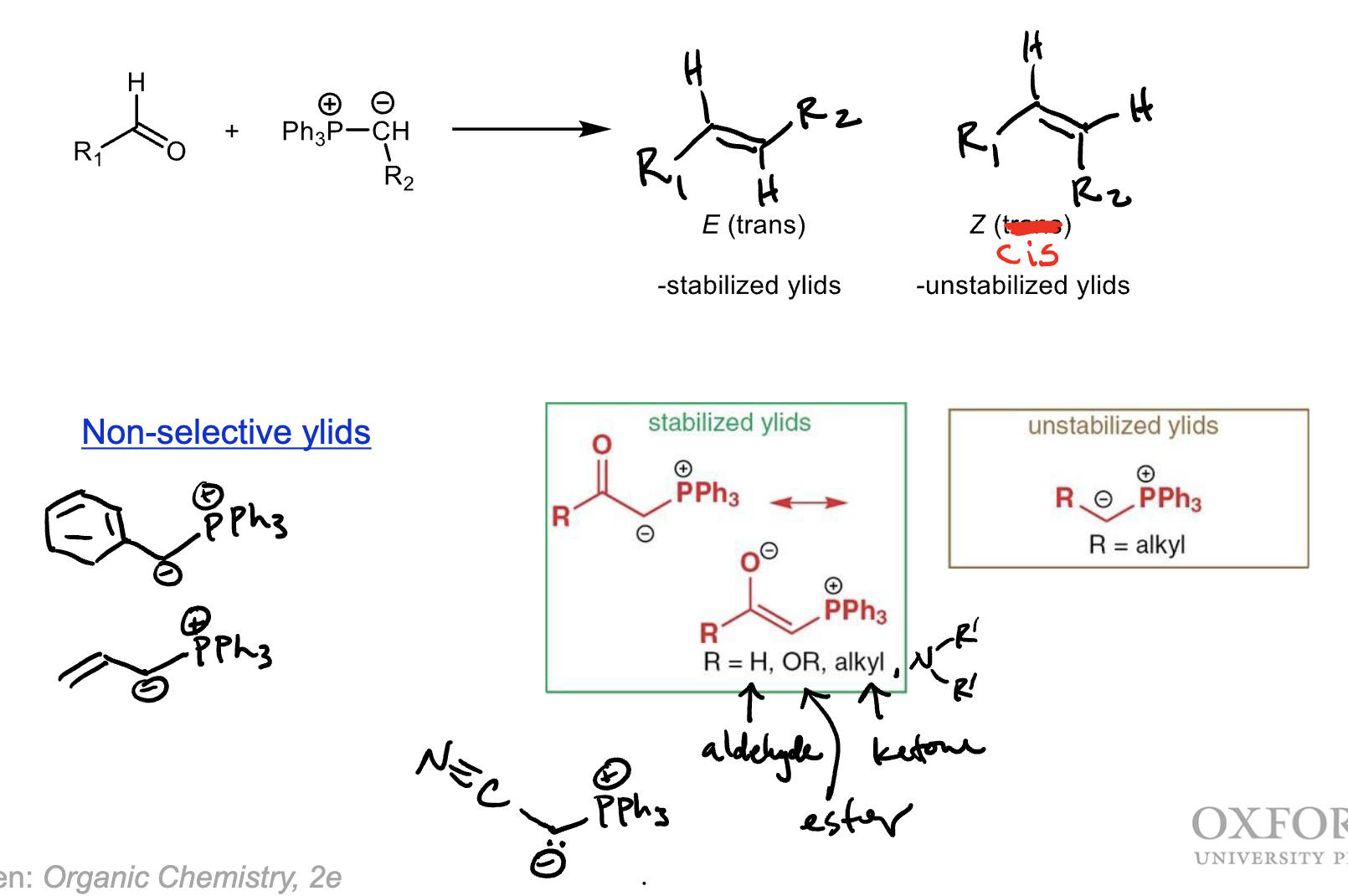
Name the stabilized, un-stabilized, and non-selective phosphorous ylids. What alkene stereoisomers do they produce?
Reagents and product for alkene reduction.
Reagents: H2, Pd
Product: alkane
Reagent’s for alkyne reduction to an alkane.
H2, Pd
Reagents for cis alkyne reduction.
Lindlar’s catalyst, H2
Reagents for trans alkyne reduction.
NH3, Li
Reagents and product for aldehyde reduction.
Reagents: NaBH4, LiAlH4, LiBH4
Product: primary alcohol
Reagents and product for ketone reduction.
Reagents: LiAlH4, LiBH4, and NaBH4
Product: secondary alcohol
Reagents and product for ester reduction.
Reagents: LiAlH4, LiBH4
Product: primary alcohol
Reagents and product for carb. acid reduction.
Reagents: LiAlH4, BH3
Product: primary alcohol
Reagents and product for amide reduction.
Reagents: LiAlH3, BH3
Product: amine
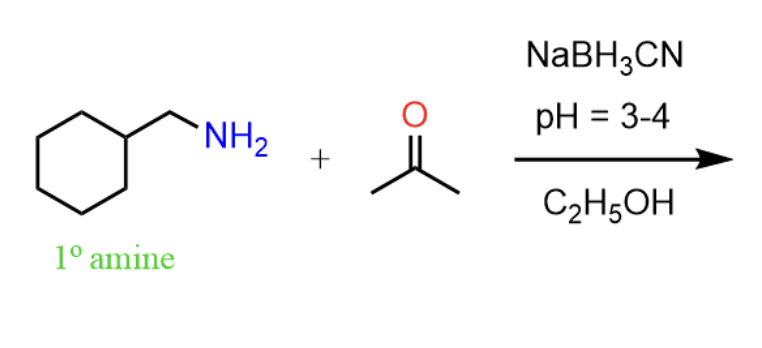
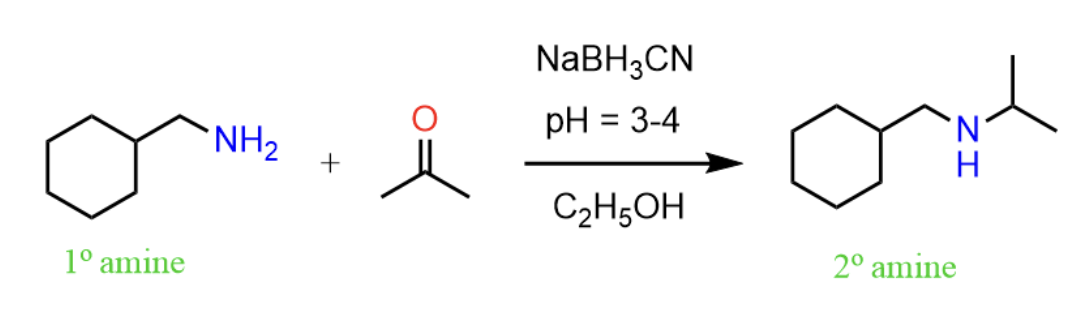
Reactants, reagents, and products of reductive animation.
Reactants: ketone/aldehyde, amine
Reagents: LiAlH4, LiBH4, BH3, NaBH4, or NaCNBH3
Product: amine
Primary alcohol to aldehyde reagents.
Weak oxidizing agent (PCC, DMP, PDC, TPAP, NMO) w/ CH2Cl2
Reagents for primary alcohol to carboxylic acid.
Strong oxidizing agent (H2CrO4, KMnO4)
Reagents and product of secondary alcohol oxidation
Reagents: oxidizing agent, typically H2CrO4, KMnO4 can also be DMSO (via Swern oxi.) or Dess-Martin
Product: ketone
Reagents for alkene oxidation to aldehyde/ketone
O3, Me2S
Reagents for alkene to carb. acid
O3, H2O2 or KMnO4
Reagents for alkene to diol
MnO4 or OsO4
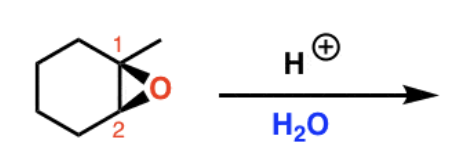
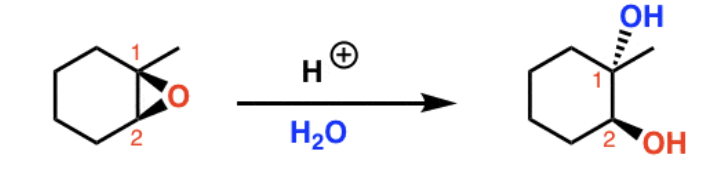
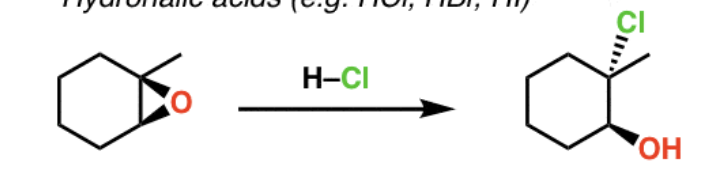
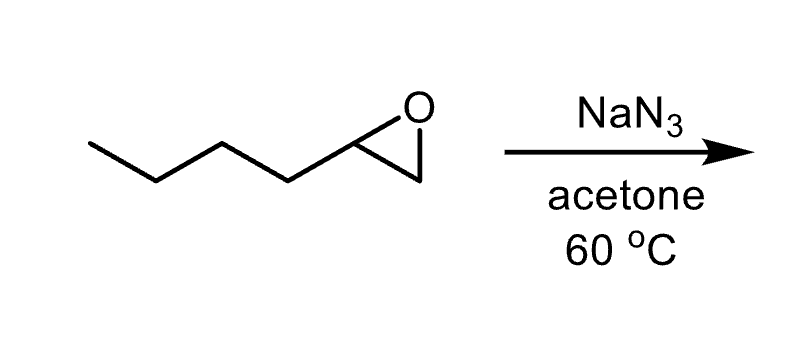
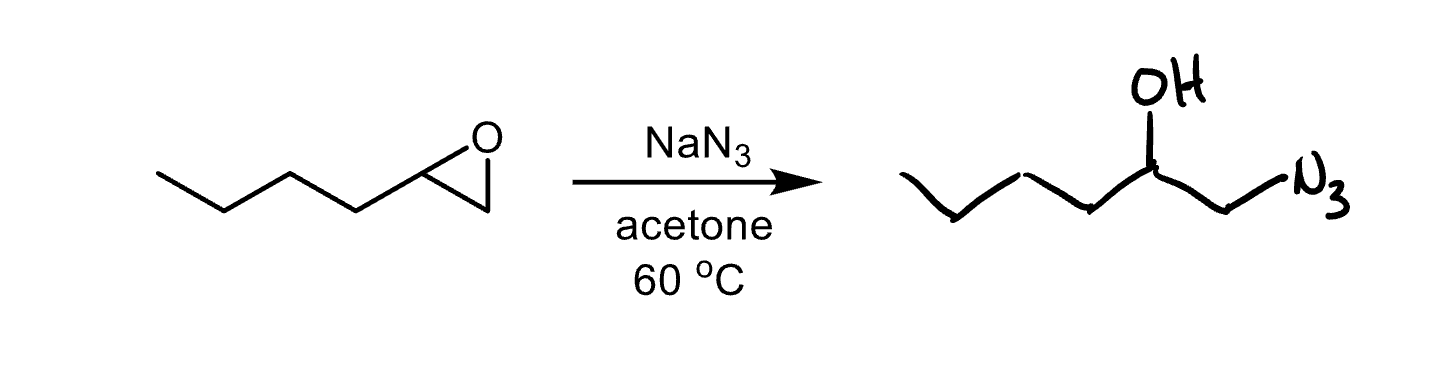
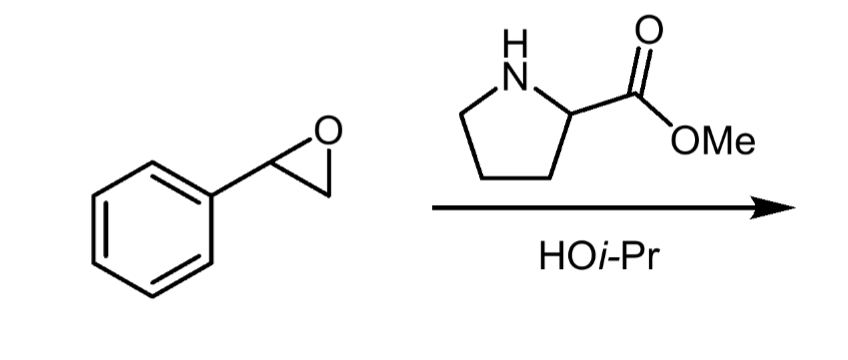
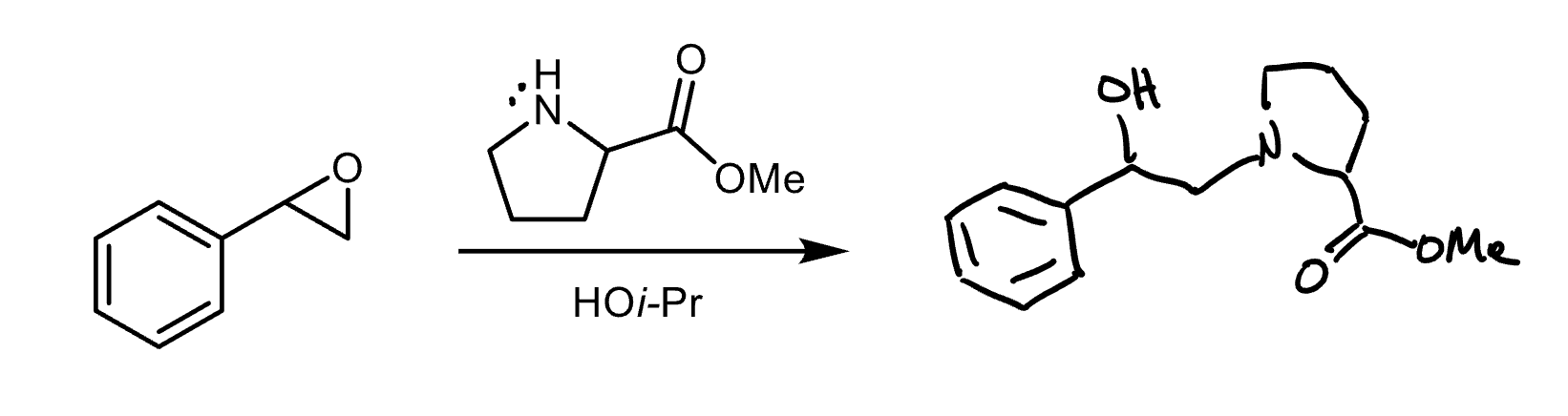
Reagents and product of epoxide opening
Reagent: nucleophile
Product: alcohol, diol if nucleophile is water
Reagent for synthesis of an aldehyde from an ester.
DIBAL
Reagent for epoxidation of alkene
m-CPBA
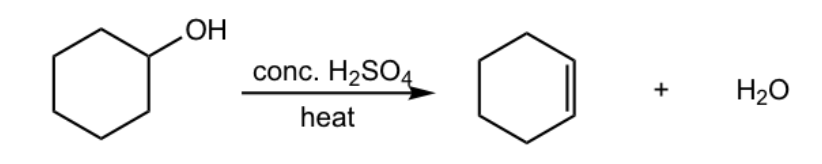
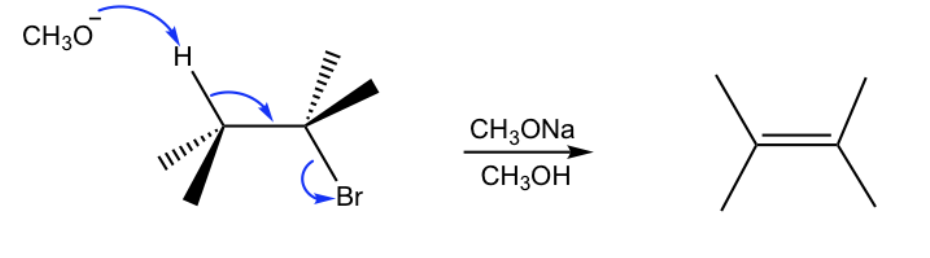
Reactants and reagents for E2 to form an alkene.
Reactants: alkyl halide or alcohol
Reagents: base (alkyl halide) or acid (alcohol)
What do benzyl ethers protect? How are they installed? How are they removed?
They protect alcohols. They are installed through NaH and BnBr. They are removed through H2, Pd/C, and HBr.
Reagents for carboxylic acid to acid chloride.
SOCl2 or PCl5
Reagent for acid chloride to acid anhydride.
carboxylic acid

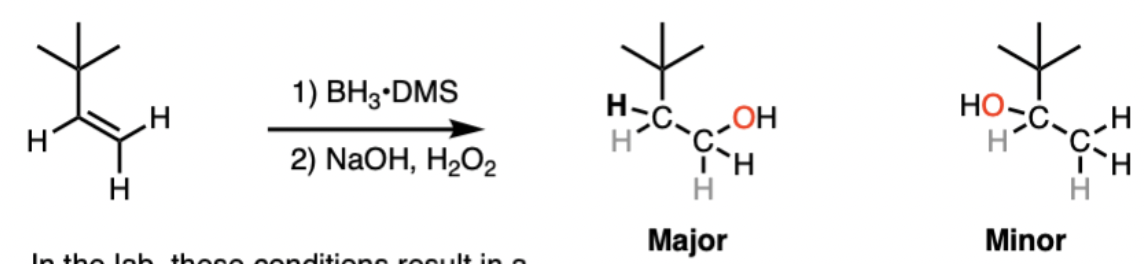
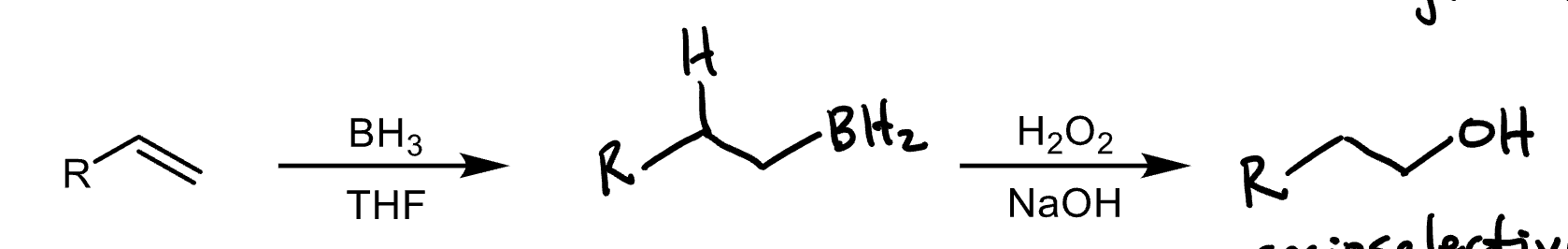
Alkene Hydroboration/Oxidation reagents and products
Reagents: 1) BH3, B2H6 2) oxidant (H2O2) in the presence of base (NaOH, KOH)
Product: alcohol
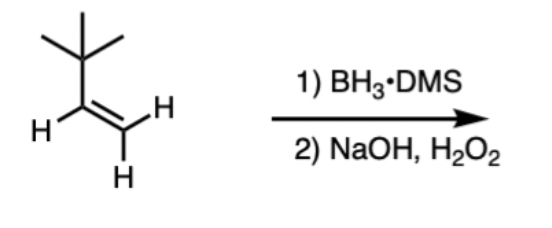
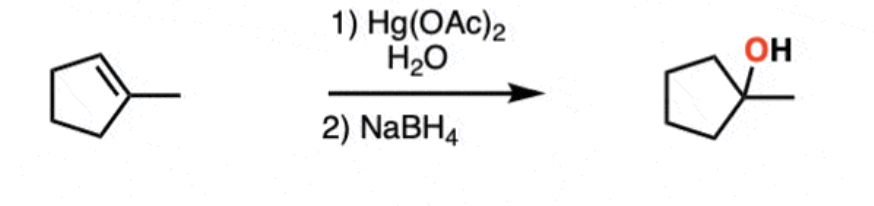
Reagents and products of alkene oxymercuration
Reagents: 1) mercury salt (Hg(OAc)2) and water) 2) NaBH4
Product: alcohol
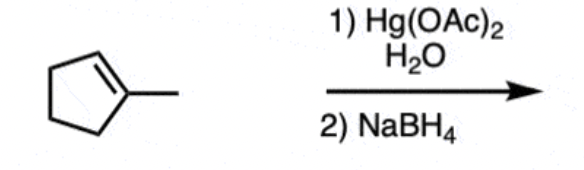
Reagents for alkylation of 1,3-dicrbonyl
1) LDA 2) alkyl halide
Reagents for alkyne alkylation
Base (NaNH2) & alkyl bromide OR BuLi & alkyl chloride

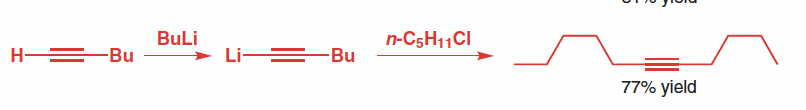
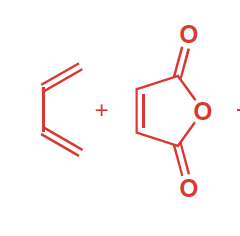
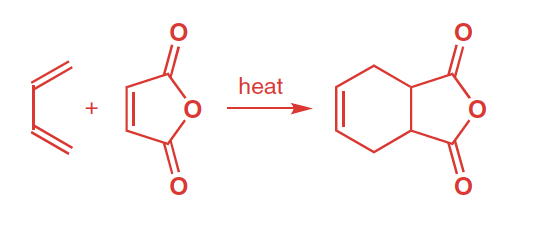
Reactants, reagents, and products of Diels–Alder Reaction
Reactants: diene (conjuagted, cis confirmation) , pi bond (dienophile)
Reagents: heat
Product: six-membered ring w/ one pi bond
Reactants and reagents for friedel-craft alkylation.
Reactants: benzene, alkyl halide
Reagents: Lewis acid (AlCl3)
Reactants and reagents for friedel-craft acylation.
Reactant: benzene, acyl group
Reagents: Lewis acid (AlCl3)
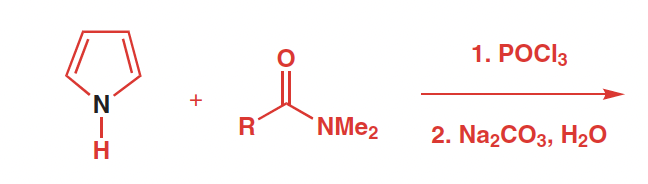
Ractants, reagents, and products of Vilsmeier–Haack Formylation
Raeactants: aromatic ring or pyrrole and an amide
Reagetns: POCl3, DMF
Products: aryl aldehyde
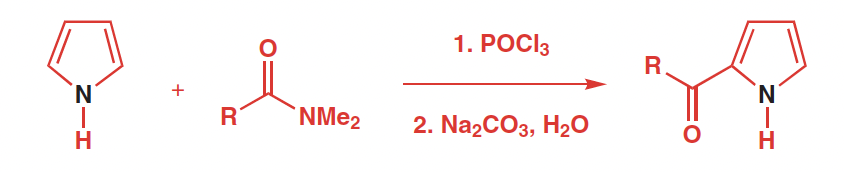
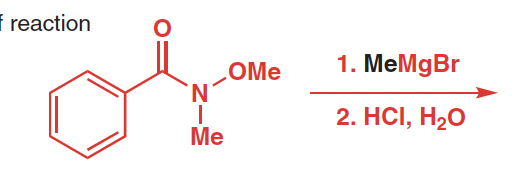
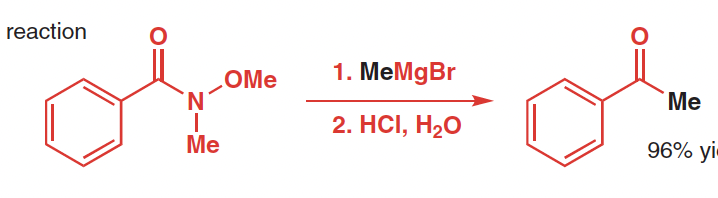
Reactants, reagents, and products of Weinreb Ketone Synthesis
Reactants: N-methoxy-N-methyl amide
Reagetns: organomagensium or organolithium reagents, acid
Product: ketone
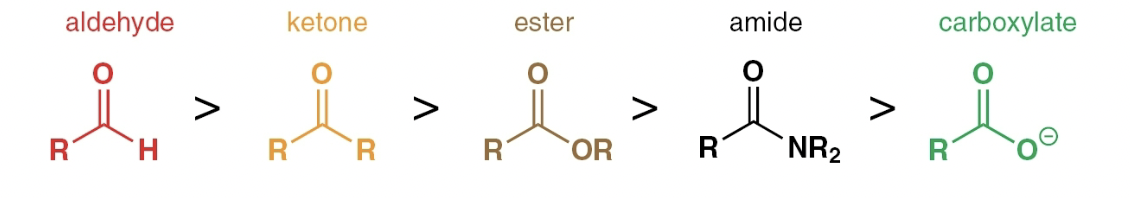
Order of carbonyl reactivity
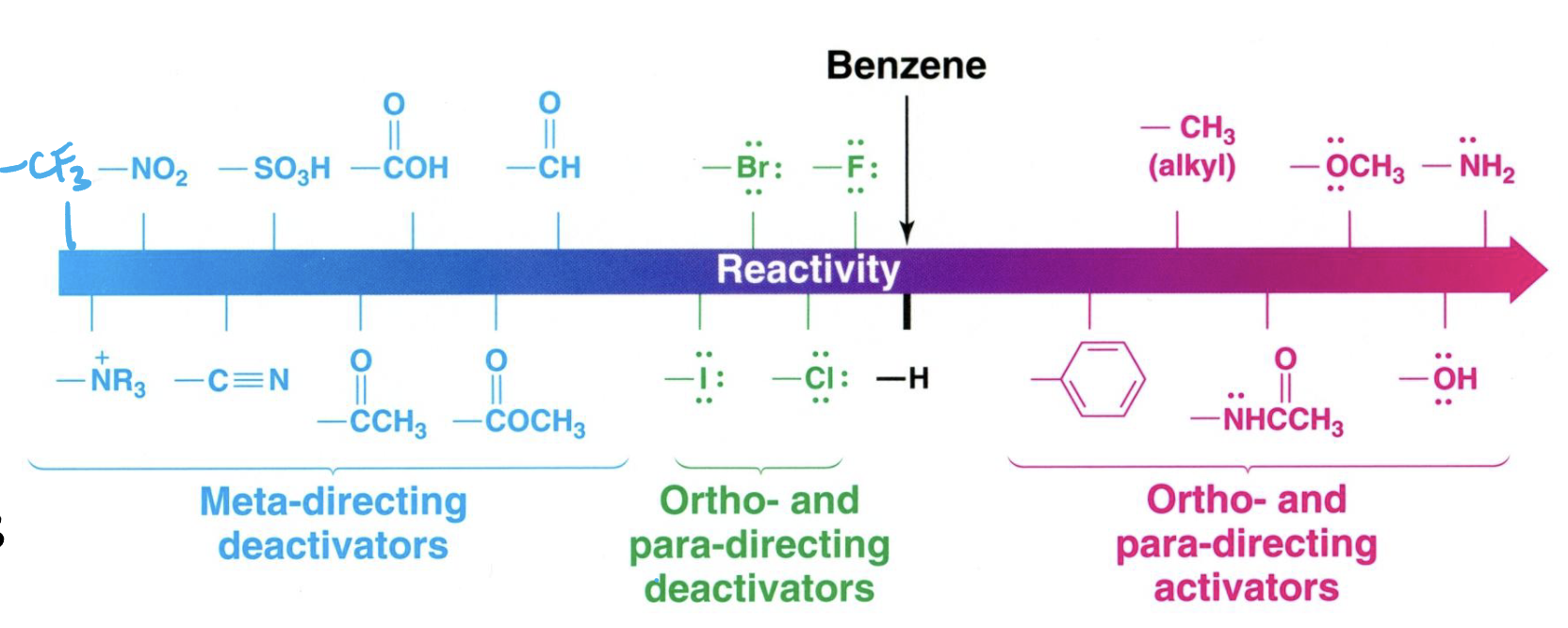
Give a list of benzene functional groups from least reactive to most reactive. State if they are meta-, ortho-, or para-directing activators/deactivators.
Reactants, reagents, and products for nitration reaction.
Reactants: benzene, nitric acid
Reagens: H2SO4
Products: nitrobenzene
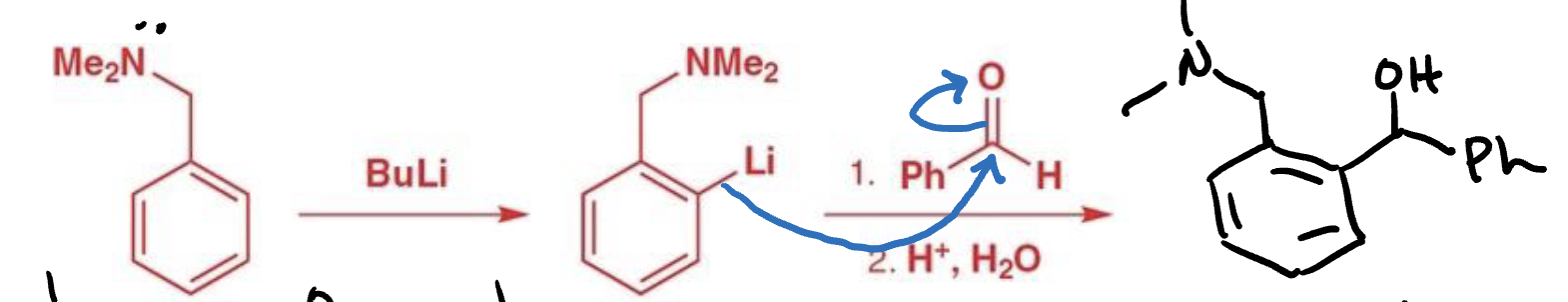
Reactants and reagent for ortholithiation
Reactant: mono-substituted benzene
Reagent: Bu-Li

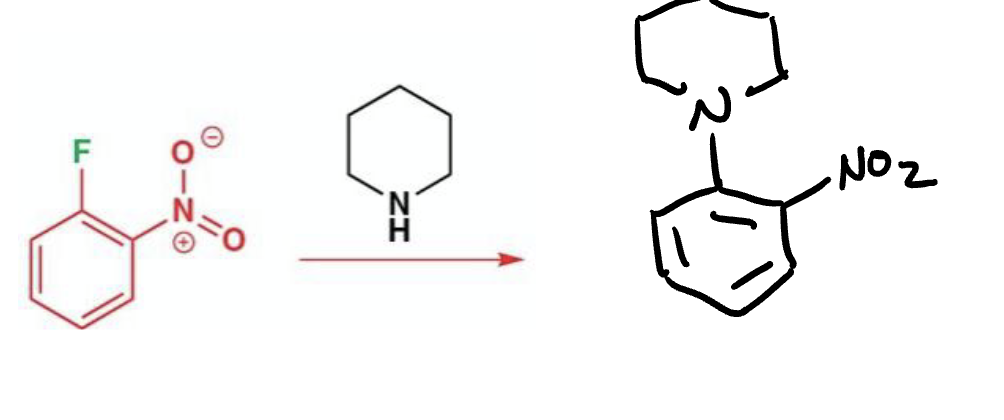
What are the typical components of a nucleophilic aromatic substitution?

What nucleophiles produce steric selectivity in epoxide opening?
Basic nucleophiles attack the less substituted carbon. Acidic nucleophiles attack the more substituted carbon.
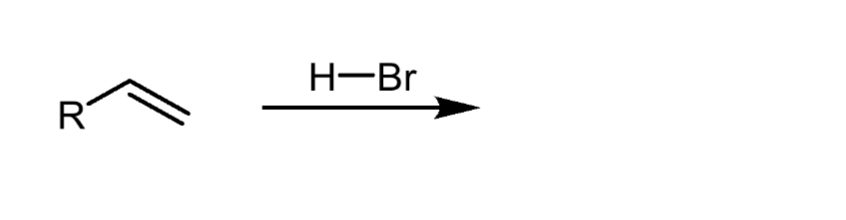
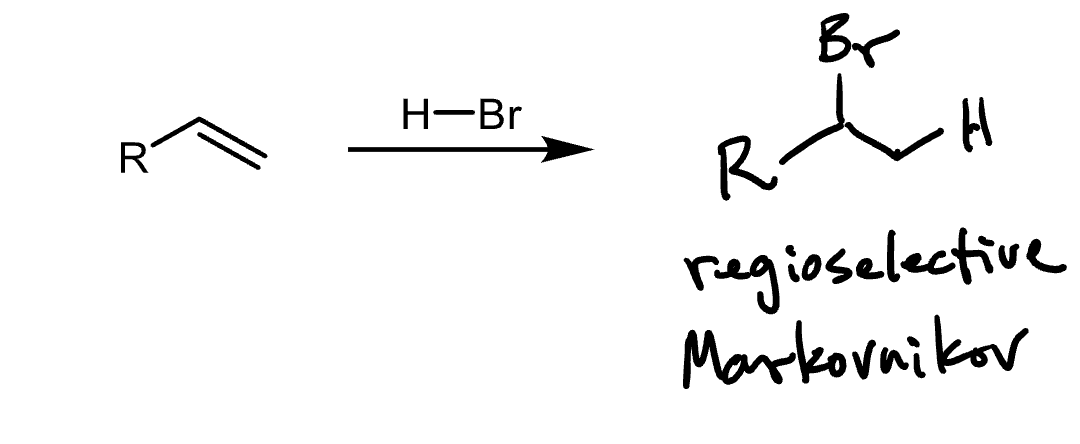
Reactants, reagents, and products of markinov’s reaction. Describe selectivity. What us an exception to rule?
Reactants: alkene
Reagents: HX
Product: alkyl halide
The halide will attach to the most substituted carbon, unless Borane is being added, in which case BH2 will be added to the least substituted carbon.

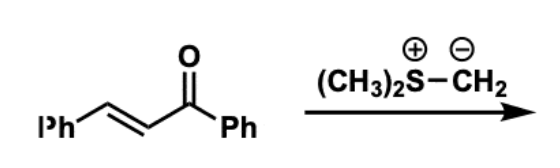
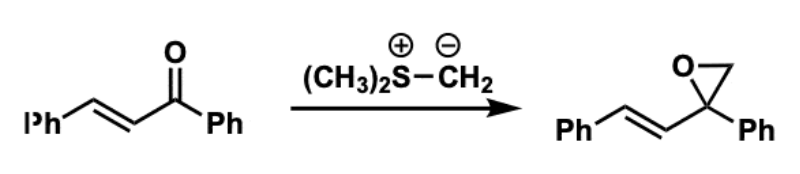
Corey−Chaykovsky Epoxidation reactants, reagents, and products.
Reactants: aldehydes/ketones
Reagent: sulfonium or sulfoxonium ylid
Product: epoxide
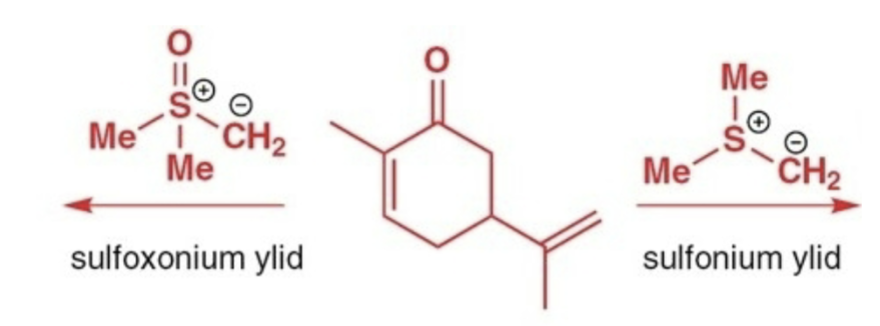
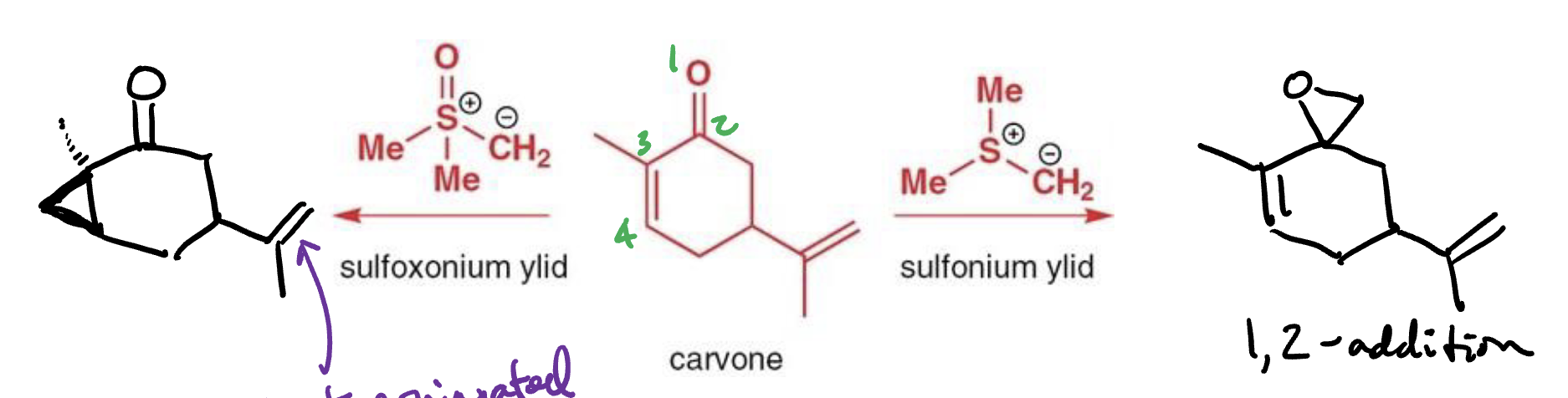
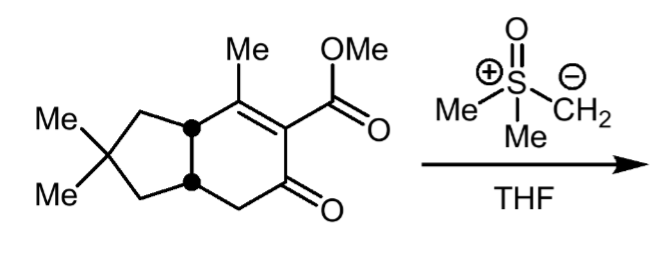
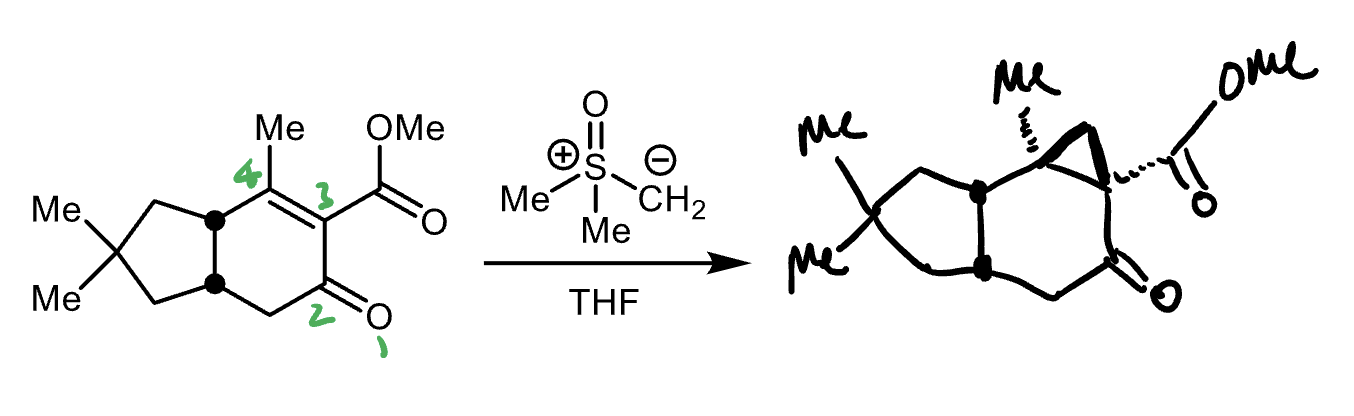
Corey−Chaykovsky cycloproponation reactants, reagents, and products.
Reactants: 1,3- conjuagted keton/aldehyde
Reagent: sulfoxonium ylid
Product: cyclopropane
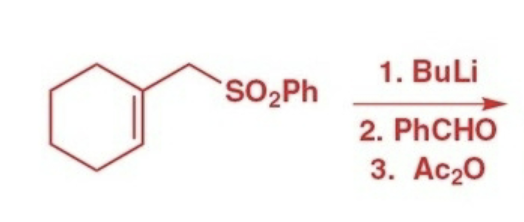
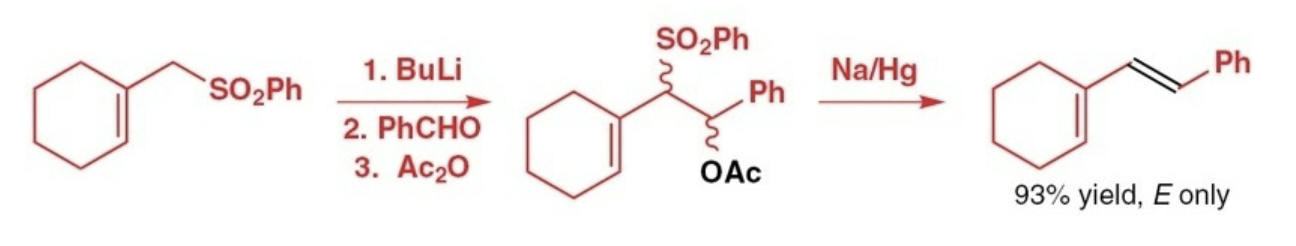
Julia olefination reactants, reagents, and products.
Reactants: phenyl sulfone
Reagents: 1) BuLi 2) aldehyde/ketone 3) acetic anhydride
Product: trans alkene
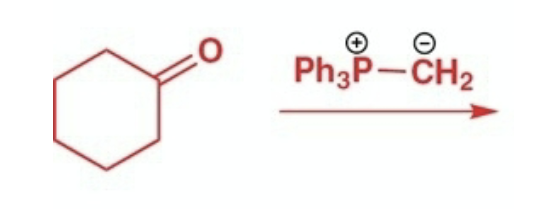
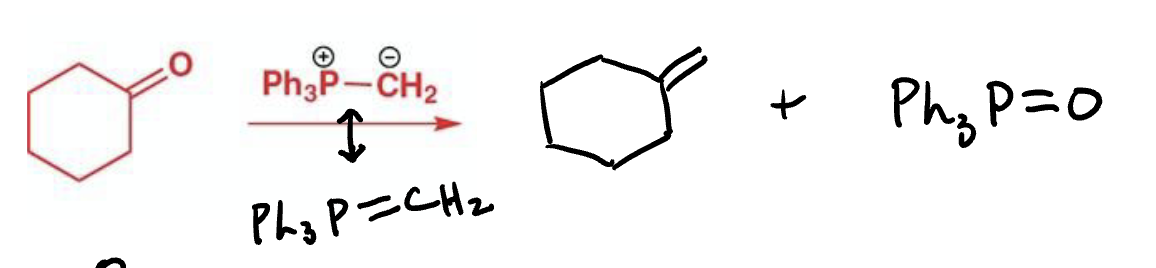
Wittig reactants, reagents, and products.
Reactants: ketone/aldehyde
Reagents: phosphonium ylid
Product: cis/trans alkene depending on ylid