Collision theory
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
What is the rate of reaction?
How fast a reaction is occuring
What do you need for a fast reaction rate?
Plenty of rapidly moving particles in a small volume
What does the steric factor mean?
Means that the location of a collision matters.
For example, if you were trying to react the OH bond in ethanol (CH3CH2OH), a collision with the CH3 group wouldn't do anything.
What does collision theory state is needed for a chemical reaction to take place?
For particles to collide with each other in the right position and with enough energy
What is the collision frequency?
The number of collisions pe unit of time
What are the 5 factors that increase the rate of reaction?
Increasing temperature
Increasing concentration of a solution
Increasing the pressure of a gas reaction
Increasing the surface area of solid reactants
Using a catalyst
How does increasing temperature increase the rate of reaction?
Increases the speed of the molecules which increases both their energy and the number of collisions
How does increasing the concentration of a solution increase the rate of reaction?
More particles present in a given volume so more collisions occur.
Why does the rate of reaction drop as the reaction goes on?
The reactants are used up and so their concentration falls
How does increasing the pressure of a gas reaction increase the rate of reaction?
Considering the the ideal gas law: pV=nRT - increasing pressure reduces the volume of the gas.
This means the molecules are closer together and they will collide more often
There are more molecules or atoms in a given volume so collisions are more likely
How does increasing the surface area of solid reactants increase the rate of reaction?
The greater the total surface area of a solid, the more of its particles are available to collide with molecules in a gas or a liquid.
What is a catalyst?
A substance that can change the reaction rate without getting consumed in the process
How does using a catalyst increase the rate of reaction?
They provide an alternative pathway with a lower activation energy. If the activation energy is reduced, more reactants can cross the energy barrier easily
What is the minimum energy needed to start a reaction called?
Activation energy, Ea
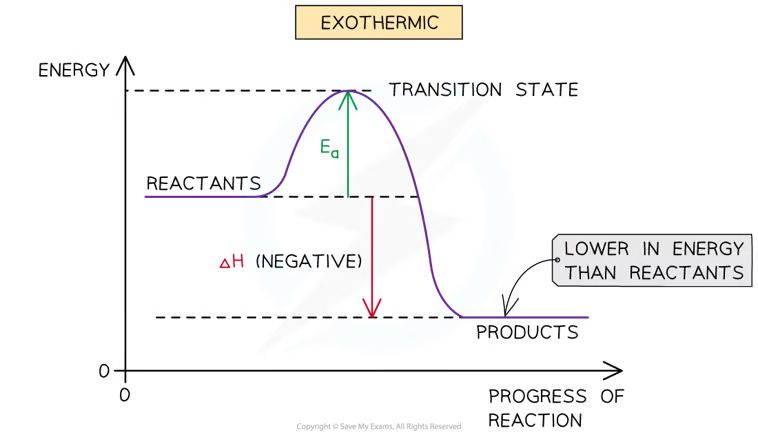
Exothermic reaction profile
The reactants are higher in energy than the products
Endothermic reaction profile
The reactants are lower than the products
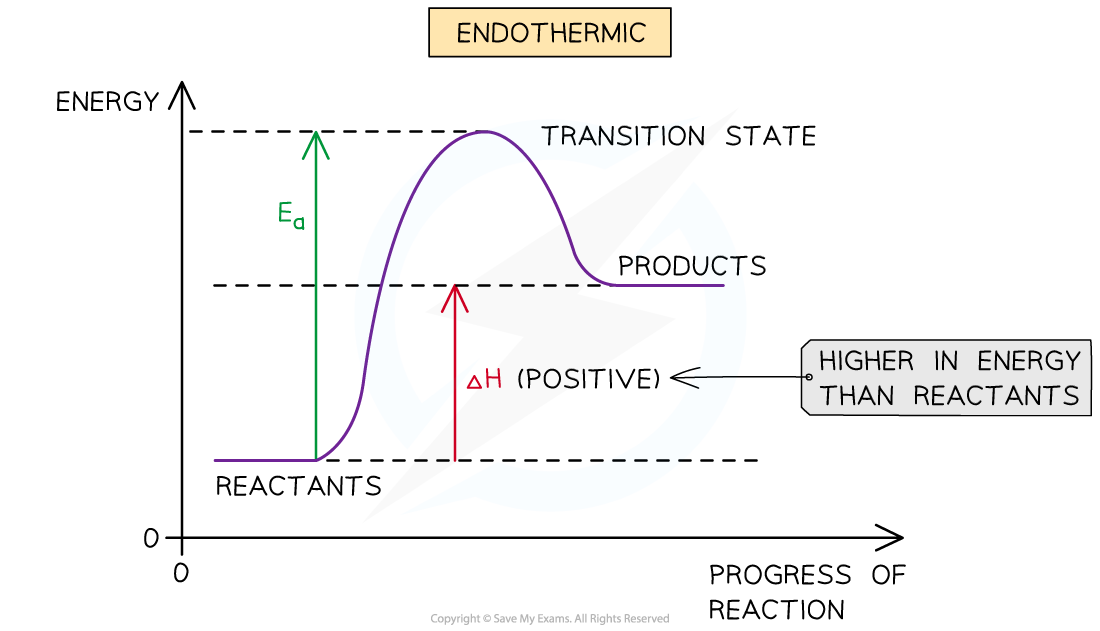
How is the Ea in endothermic reactions different to that of exothermic reactions?
The Ea in endothermic reactions is relatively larger than in exothermic reaction
What is the transition state?
When some bonds are in the process of being made and being broken