BIOCH 310 Set 1 (First Midterm Content)
1/55
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
Thermodynamics drvies
Reaction direction/favourability. Only tells us the probability of a rxn happening, does not actually tell us anything about speed.
Kinetics (Enzymes) drives
The reaction rate (SPEED).
Flux (J)
J = Vforward - Vreverse. Tells us the direction which a reaction will proceed in, based on the difference between the speed the forward reaction is occuring at compared with the reverse reaction
Flux at equilibrium
0 (Vf = Vr)
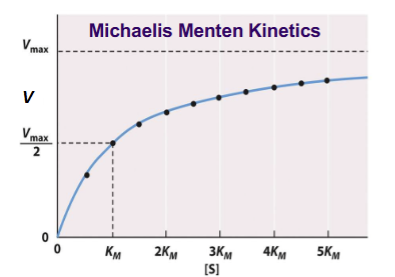
Decreasing Km
Increases affinity (shifts curve to the left); causes the substrate to be better at binding to its enzyme. Often close to the physiological concentration a particular substrate. Activator impact
Increasing Km
Decreases affinity (shifts curve to the right); causes the substrate to be worse at binding to the enzyme. Inhibitor impact.
Km
Can be found on graphs at Vmax/2.
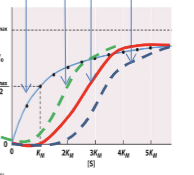
Allosteric enzyme binding curves
Sigmoidal in shape (takes a bit to form the complex, before exponential growth). Able to be saturated.
First law of thermodynamics
Energy cannot be created or destroyed, only change forms (relates to needing energy in order to generate more energy)
Second law of thermodynamics
Entropy must be maximized in all circumstances (chaos is favoured). To have “order”, you need to put energy in to the system, and it must then also generate energy.
Gibbs free energy
ΔG = Gproducts - Greactants. When it is negative, this is a exergonic (favourable reaction) = products have less energy than the reactants = energy being released = catabolic. When it is positive, this is a endergonic (unfavourable reaction) = products have more energy than reactants = energy is needed = anabolic.
Actual free energy
Depends on the standard free energy and the concentrations of the reactants/products.
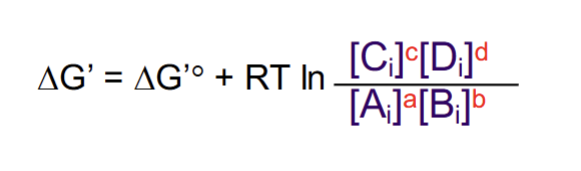
How to generate a more negative free energy (exogeronic rxn)
Increasing reactants or decreasing products - causes the fraction to grow and become smaller in value, which when taken to the natural log becomes negative.
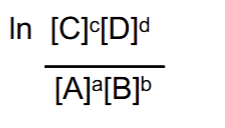
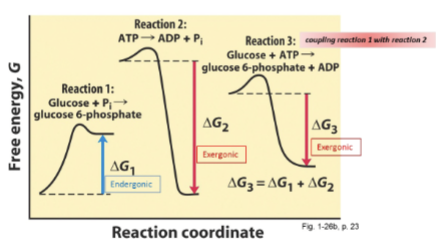
Direct coupling
Using the same enzyme and enzyme active site to perform two consecutive rxns, with one driving the next. Pi is the common intermediate.
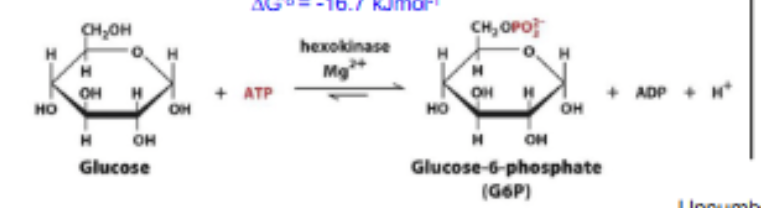
Hexokinase
An example of direct coupling. Using the common Pi intermediate (and water). Hydrolyzes ATP in the first step. Phosphorylates Glucose in the second. Overall rxn still negative G due to coupling.
H2O + ATP → Pi + ADP (exergonic: -31)
Pi + Glucose → G6P and H2O (endergonic: +14)
Indirect coupling
Two consecutive rxns occuring on two different enzymes, with one driving the next. PPi is the common intermediate.
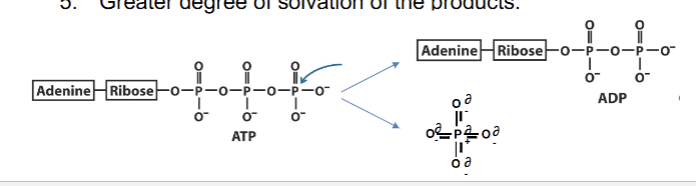
ATP as an energy source
Paradoxically stable in solution, despite the high repulsion generated from the 3 phosphate groups and their negative charges. High free energy upon breaking of the phosphoanhydride bond due to:
Reduction of electrostatic repulsion
Resonance stabilization of products (Pi)
More products than reactants (entropy increases)
Ionized, more stabilization of products
Greater solvation (water able to interact/coat the products better than reactants)
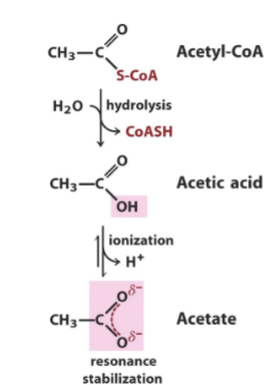
Acetyl-CoA as an energy source
Thioester bond that can undergo hydrolysis, forming Acetic acid. This can then undergo ionization, generating acetate.
Formation of resonance stabilized products
More products than reactants (favouring entropy)
Ionized, more stabiliziation of products
NAD(H)as an energy source
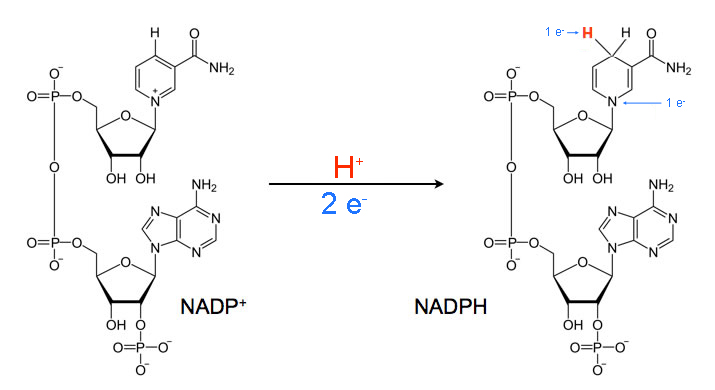
The oxidized form (NAD+) used in catabolism rxns by dehydrogenases to accept 2 electrons (through a hydride ion). Contains an adenosine connected to a nicotinamide = dinucleotide. Derived from vitamin B3 (niacin)
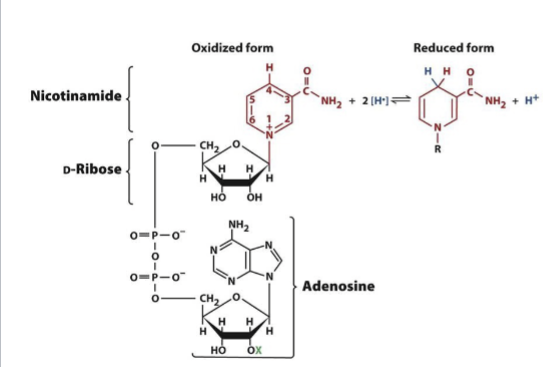
Hydride ion
Hydrogen with 2 electrons attached to it, giving it an overall charge of -1. How electrons are transported in most reactions.
NADP(H) as an energy source
Same molecule as NAD, just contains a phosphate on the 2’ Carbon. The oxidized form (NADP+) is used in anabolic pathways.
Cofactors/Coenzymes
Convert inactive apoenzymes into active holoenzymes. Not used up in the rxn (as they are associated with the enzyme = catalyzed). Can be ions, cosubstrates (loosely bound = ATP, NAD+) or prosthetic groups (tightly bound = FAD)
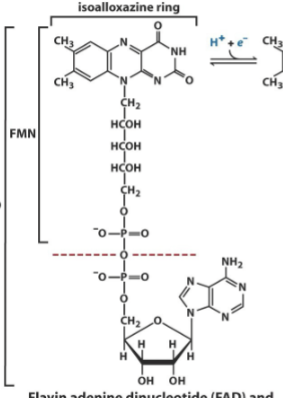
FAD(H/H2) as an energy source
Contains an adenine nucleotide connected to a isoalloxazine ring nucleotide = dinucleotide. Able to be reduced two seperate times by the gaining of hydride ions, on the isoalloxazine ring portion.
CO2
A fully oxidized carbon molecule (+4 oxidation state), the end result electron transfers and the release of energy in biological systems
Hydrocarbons/Fatty acids
A fully reduced carbon molecule (-2 oxidation state), the beginning of electron transfers and where energy is stored in biological systems.
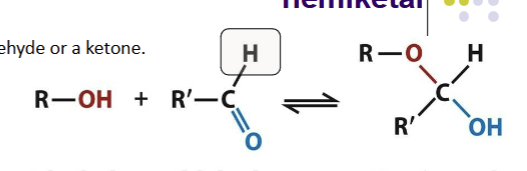
Hemiacetal formation
The addition of an alcohol to the carbon of an aldehyde will generate this; changes geometry from planar to tetrahedral and forms a reducing end. Happens when linear aldose sugars circularize and form rings
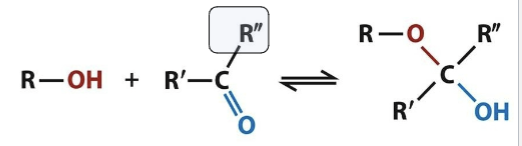
Hemiketal formation
The addition of an alcohol to the carbon of a ketone will generate this; changes geometry from planar to tetrahedral and forms a reducing end. Happens when linear aldose sugars circularize and form rings
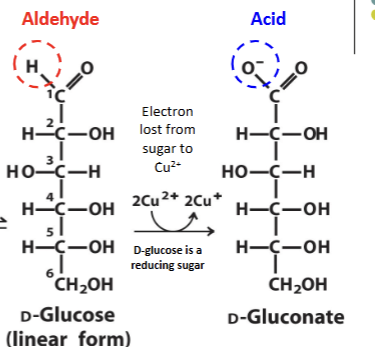
Sugars as reducing agents
Aldose groups can be oxidized in solutions of Cu2+ or Fe3+ to form a carboxyl group at carbon 1 (aldehyde to an acid). This can only occur in the linear form.
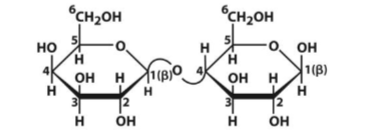
Lactose
Disaccharide of Gal and Glc. Connected via (β 1 → 4) glycosidic bond = reducing sugar due to reducing end at Glc (mutarotating). Both pyranose rings. Broken down by lactase.
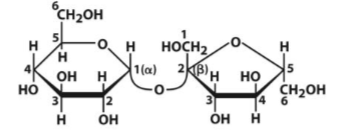
Sucrose
Disaccharide of Glc and Fru. Connected via (α 1 ↔ 2 ) glycosidic bond = non reducing sugar. 1 pyranose and 1 furanose ring
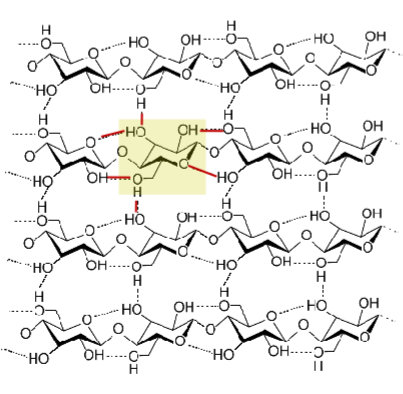
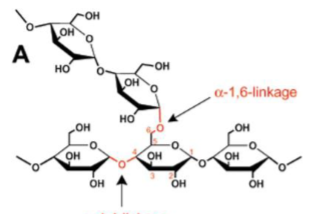
Cellulose
Unbranched, linear chain of Glc, connected via (β 1 → 4) linking, with 1800 rotation between each Glc. Chains which form line up side by side in a secondary structure similar to a beta sheet, which satisfies all H bond requirements without water. H bonds formed within and between chains. Animals unable to digest this bc we cannot break up a (β 1 → 4) bond. Digested by fungi that contain cellulase
Starch
Made up of two separate polymers, both only Glc containing (for Glc storage in plants): Amylose (unbranched) and Amylopectin (branched). Polymers intertwined with one another and form helices with the help of water (mix of single and double helicies). Many non-reducing ends, only one reducing end. Striated appearance.
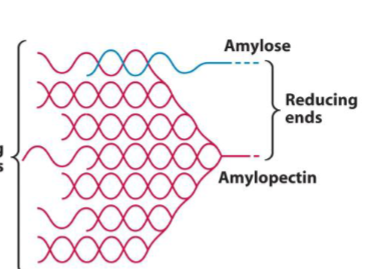
Glycogen
Very highly branched (more so than starch) Glc storage homopolymer for animals. Primarily (α 1 → 4) but branching occurs every 8-14 residues where an (α 1 → 6) links together two chains. Forms roughly spherical structures with a protein in the middle. Many non-reducing ends, protein attaches to the single reducing end (no more reducing?)
Combustion of wood
1 uncontrollable step, where O2 is an electron acceptor and heat/light are released in the breaking of linear cellulose bonds. Exothermic. Contrast to biological catabolism steps
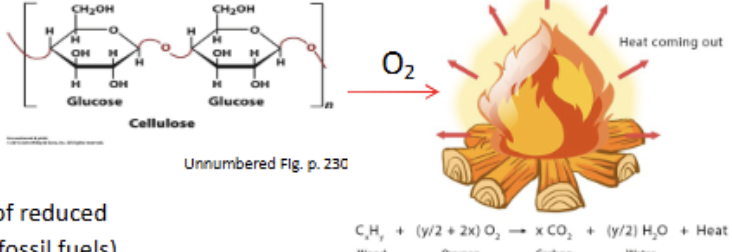
Biological catabolism
Many small, controlled steps that release energy in a specific fashion.
Glycolysis
Oldest cellular pathway, predates photosynthesis (no oxygen involved). The breakdown of glucose into energy for the cell. Anaerobic, 10 reactions. Converts 1 Glucose into 2 pyruvate, 2 ATP and 2 NADH. Consumes 2 ATP in the process, but generates 4 total. Oxidative process.
Glycolysis rxn 1
PHOSPHORYLATION: Glucose + ATP → G6P and ADP. Rate limiting step, irreversible (highly exergonic) and regulated. Traps glucose in the cell due to (-) phosphate charge. Catalyzed by Hexokinase.
Glycolysis rxn 2
ISOMERIZATION: G6P ↔ F6P. Reversible (not exergonic). Catalyzed by phosphoglucose isomerase; changes the aldehyde to a ketone (glc to fruc).
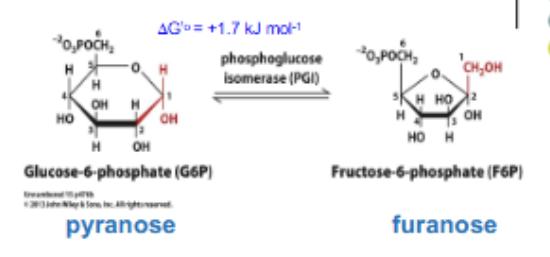
Glycolysis rxn 3
PHOSPHORYLATION: F6P + ATP → F-1,6-BP + ADP. The comitted step, as it is unique to the glycolysis pathway. Rate limiting, irreversible (highly exergonic). Products of this rxn really can only be used for glycloysis (F6P still has other pathways). Catalyzed by PFK-1
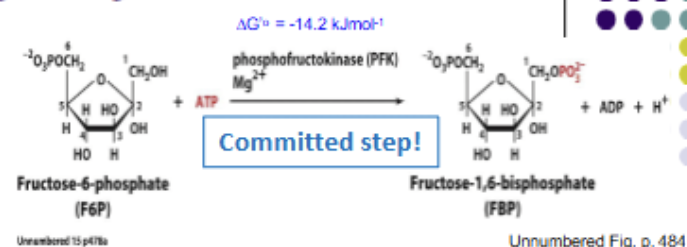
Glycolysis rxn 4
CLEAVAGE: F-1,6-BP ↔ DHAP + GAP (isomers). Requires H2O (as an energy source) for aldolase to lyse the hexose into 2 triose’s.
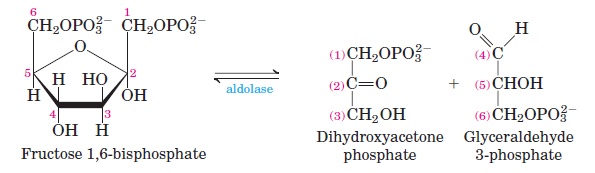
Glycolysis rxn 5
ISOMERIZATION: DHAP ↔ GAP. Isomerization, catalyzed by triose phosphate isomerase. Low cellular concentration of GAP pulls this rxn forward in vivo.
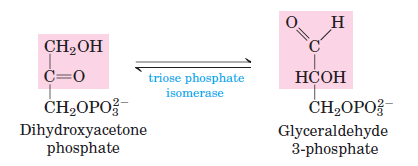
Glycolysis rxn 6
OXIDATION/DEHYDROGENTION: NAD+ + Pi + GAP ↔ 1,3-BPG + NADH. Catalyzed by GAPDH (glyceraldehyde -3P dehydrogenase). Forms a high energy intermediate (1,3-BPG) and NAD+ is the electron acceptor. GAP oxidized

What makes 1,3-BPG a high energy intermediate
Due to repulsion of the negatively charged phosphate groups, which are only seperated by 1 carbon. Took energy investment to put a free Pi onto GAP, which was paired with NAD+ reduction.
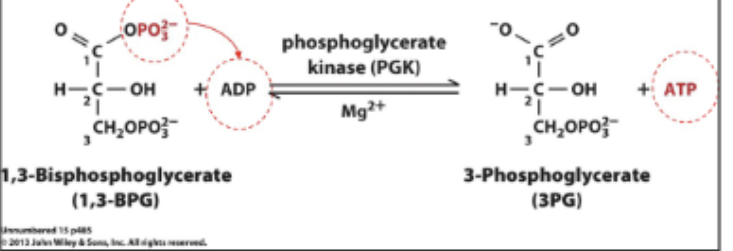
Glycolysis rxn 7
SLP: 1,3-BPG + ADP ↔ ATP + 3-Phosphoglycerate (3PG). Catalyzed by phosphoglycerate kinase.
Glycolysis rxn 8
MOLECULAR REARRANGEMENT: 3PG ↔ 2-Phosphoglycerate (2PG). Not an isomerization because an intermediate is involved (A different phosphate is put on the 2 spot, causing the 3 P to leave; not the same P). Catalyzed by phosphoglycerate mutase
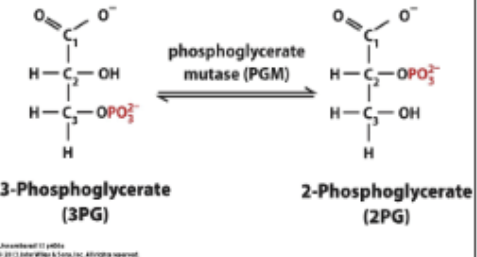
2PG considerations ***
Due to the closeness of the negativley charged Phosphate and the carboxylic acid group.
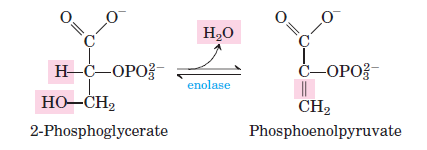
Glycolysis rxn 9
LYASE: 2PG ↔ Phosphoenolpyruvate (PEP) + H2O. Dehydration rxn catalyzed by enolase that results in the production of a high energy intermediate, which was powered by 1,3 BPG.
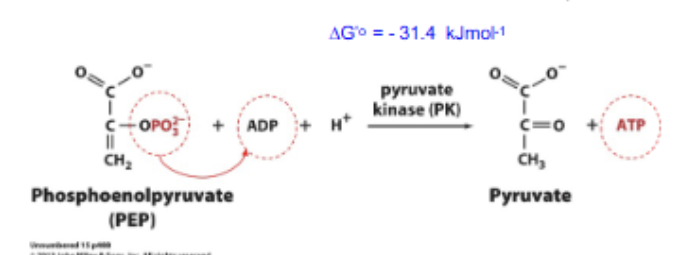
Glycolysis rxn 10
SLP: PEP + ADP + H+ → ATP + Pyruvate. Highly exergonic, catalyzed by pyruvate kinase.
Glycolysis step 4,5 and 6
∆G ~ 0, since all rxns are reversible and the products/reactant ratio is relatively even. Therefore, these rxns only proceed due to high concentrations of reactants (glucose) or low concentrations of products (pyruvate).
How does glycolysis generate ATP, even when intracellular ATP concentration is high?
Able to do so continuously due to substrate level phosphorylation, where high energy intermediates are used to hold on to the energy as a placeholder (such as 1,3-BPG and PEP).
NAD+/NADH balance impact on glycolysis
NAD+ must be regenerated in order for another cycle of glycolysis to begin. Luckily this happens very fast, and so there is only a tiny amount of NAD+ needed, compared with the amount of glucose.
Gluconeogenesis
The reverse process of glycolysis, bypasses the irreversible steps of glycolysis (1,3 and 10). Uses 11 enzymes instead of 10, 4 of which are unique. Does not produce energy, consumes 6 ATP and 2 NADH. Reductive process.
Gluconeogenesis rxn 10 bypass
Pyruvate + ATP + CO2 → Oxaloacetate + ADP + Pi. Catalyzed by pyruvate carboxylase. Exergonic. Only in the mitochondria (for humans)
Oxaloacetate + GTP → PEP + CO2 + GDP. Catalyzed by PEPCK (phosphoenolpyruvate carboxykinase) = master regulator for gluconeogenesis. Highly exergonic. In the mitochondria and cytosol (for humans)
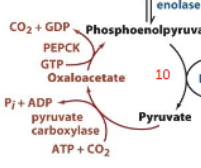
Oxaloacetate intermediate
For both the kreb’s cycle and gluconeogenesis
Gluconeogenesis rxn 3 bypass
Gluconeogenesis rxn 1 bypass