substance use disorder
1/89
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
90 Terms
person first language
Stigmatizing language | Non stigmatizing language | Reason: |
Addict User Drug abuser Junkie Alcoholic Drunk | Person with substance use disorder Patient Person in recovery Person who previously used drugs | Person first language The patient “has” a problem instead of the person “is” a problem Terms avoid negative connotation, punitive nature, or blame. |
Habit | Substance use disorder Drug addiction | Inaccurately describing the disease Undermines the seriousness of disease |
Abuse | Use Misuse | Negative connotation Judgment |
Opioid substitution therapy Medication assisted therapy | Opioid agonist therapy Medication for substance use disorder Addiction medication | Avoid the misconception that medications only substitute one drug for another or that medication is only supplemental to better treatments |
Clean | Testing negative Abstinent, in remission | Only use clinically accurate terms (even if your patient uses slang) |
Dirty | Testing positive Person who uses drugs | Only use clinically accurate terms (even if your patient uses slang) |
DSM V diagnostic criteria chart

DSM V diagnostic mild score
2-3
DSM V diagnostic moderate score
4-5
DSM V diagnostic severe score
6+
physical dependance
the body’s reaction to sustained exposure to a drug
physical and observable withdrawal symptoms
this process can be painful and consuming
psychological dependance
•Sustained mental need for the drug or substance
•Can occur with essentially any substance
•Hardwiring of the brain- we develop attachments or a need for the substance
•May last longer than a physical dependance
SUD is a chronic condition
SUD involves lapses and relapses
SUD is estimated to be 40-60% genetic
SUD responds to appropriate treatment
defined by persistent use despite harmful consequences
brain area involved in SUD
cortex - helps us make good choices
amygdala - rules our emotions
midbrain - controls our reward system
pathophysiology of SUD
Repeated substance use overstimulates dopamine reward pathways.
Brain begins prioritizing substance use over normal survival/reward activities.
Tolerance develops over time.
Withdrawal occurs when the substance is removed and the body is no longer in equilibrium.
why withdrawal happen
The body adapts to chronic substance exposure. When the substance is suddenly stopped, the body becomes overactive in the opposite direction.
alcohol withdrawal syndroms
severe alcohol withdrawal can be associated with alcohol withdrawal delirium (delirium tremens)
if unmanaged, this can result in severe seizures and death
<5% of alcohol withdrawal cases
benzodiazepine withdrawal syndrome
Benzodiazepine withdrawal can result in seizures that can be life threatening
High doses and chronic use increases the risk
More prevalent when the drug is quickly reversed
other withdrawal syndrome
Other withdrawal syndromes rarely result in death
Of note: any prolonged situation where the body is deprived of a necessary element can result in death (i.e. prolonged dehydration)
opioid system affects what
pain
thirst
mood
hunger
medications for opioid use disorder (MOUD)
methadone
buprenorphine
naltrexone
evidence based benefits of MOUD
•Improve survival
•Increase functioning and sustainability in treatment
•Decrease opioid use and criminal activity associated with Opioid Use Disorder
•Increase ability to gain and maintain employment
•Improve birth outcomes
naltrexone’s role in MOUD (compared to others)
Naltrexone is considered MOUD and can be effective for patients
However:
Lower treatment retention (detox schedule)
Decreases tolerance- adherence is really important
Lest robust evidence for primary outcomes
NOT recommended to begin in pregnancy. Only methadone and buprenorphine are first line and improve birth outcomes
methadone MOA and half life
full agonist
half life: 22-48 hours
methadone forms
dissolvable tablets or liquid
methadone BBW
abuse
respiratory depression
QT prolongation
cytochrome P450 interaction
methadone regulation
heavily regulated: only dispensed at a registered opioid treatment center (OTP)
methadone dose: starting and target
day 1: 5-10 mg with no or low opioid tolerance
day 1 maximum: 50 mg
typical target: 60 mg/day or higher associated with greater retention 80-120 mg/day (some require higher doses)
•Dosing is once daily, usually starts at 20-30mg based on tolerance then is titrated
methadone common adverse events
•constipation, lightheadedness, dizziness, sedation, nausea, vomiting, sweating
methadone rare adverse events
ECG abnormalities
psychosis
pruritis
sexual dysfunction or decreased libido
amenorrhea
weight gain
edema
seizures
hypotension
methadone tests/considerations
liver function tests (LFT)
EKG (QTc)
pregnancy test for those of child-bearing potential
serum concentrations
interactions: CYP3A4 and 2C9
methadone hepatic impairment
lower starting doses
methadone renal impairment
lower starting doses
methadone adolescents
not FDA approved, can be provided to adolescents under special circumstances
buprenorphine MOA
partial agonist
can be combined with naloxone, an antagonist
buprenorphine half life
oral: 24-48 hours
injection: 43-60 days
buprenorphine dosage form
Available in buccal film, sublingual tablet, and sublingual film, patch, implant, injection
New reports the protentional for the severe tooth decay with orally dissolving buprenorphine. More than 300 case reports. Benefit still outweighs risk.
buprenorphine BBW
addiction
REMS
respiratory depression
buprenorphine considerations and when to start
•Need to consider what type of opioid has been used (synthetic, long acting, etc.)
•Wait until the patient is past withdrawal or until they are clearly in withdrawal
buprenorphine buccal, sublingual dosing: initiation, targe, max
initiation: 2-12 mg total on first day
typical target: 16 mg/day
maximum: 24 mg/day
*Doses up to 32mg/day have been studied but have not been demonstrated to provide any clinical advantage. Can consider up to 16mg on day 1 in people using fentanyl.
buprenorphine injectable dosing: monthly start and maintenance
•Monthly start: two doses of 300mg
•Monthly maintenance: 100mg*
buprenorphine injectable dosing: weekly start and maintenance
•Weekly start: 16 mg + 8mg in 3 days
•Weekly maintenance: 16-32 mg titrated as needed
buprenorphine common adverse effects
sedation
constipation
nausea
headache
hyperhidrosis
oral hypoesthesia
glossodynia
oral mucosal erythema
buprenorphine rare adverse effects
hepatitis
respiratory depression
serotonin syndrome
buprenorphine test/considerations
liver function tests
UDS will show buprenorphine up to 12 months after discontinuation after chronic injectable buprenorphine
injectables must be administered by professional
buprenorphine hepatic impairment
•combo products not recommended in moderate to severe impairment
•For single products: lower starting doses in severe impairment
buprenorphine renal impairment
no adjustments
buprenorphine adolescents
no FDA approved, but some studies show efficacy in 16 years and up
naltrexone MOA
full antagonist
naltrexone BBW
none
naltrexone dosage form
•Available in oral tablets an LA IM Injection
•HOWEVER oral tablets have not shown any benefit over placebo due to adherence issues
naltrexone half life
•IM injection has a peak in 2 hours (first peak) and 2-3 days (second peak) Duration of 4 weeks
•Oral half life is about 4-13 hours
naltrexone requirement
patient NEED to be opioid free for at least 7-10 days
can precipitate overdose with nonadherence
naltrexone dosing
Oral: 50 mg daily
Injection: 380 mg monthly
naltrexone common adverse effects
nausea
vomiting
headache
low energy
anxiety
depression
rash
decreased alertness
injection site reactions
naltrexone rare side effects
hepatotoxicity
naltrexone monitor
liver function tests
naltrexone what can you not do
cannot treat acute or chronic pain with opioids while on this
naltrexone hepatic impairment
caution in severe impairment, but weigh risk and benefit
naltrexone renal impairment
use with caution in moderate to severe impairment
extensive renal clearance
naltrexone adolescents
not studied
alcohol consumption mechanism
•Ethanol increases inhibitory (GABA) and decreases excitatory (glutamate) neurotransmission in many parts of brain
•Activates opioid receptors to release dopamine and serotonin
chronic alcohol use
Regular alcohol use will result in increased tolerance
To maintain homeostasis, the body will adapt to the effects of alcohol
This can cause or worsen withdrawal affects when stopping use
Can be more sensitive to alcohol in new environments
chronic alcohol use can lead to increased risk with
dilated cardiomyopathy
arrhythmias
stroke
cancers
pancreatitis
chronic alcohol complications
Liver disease
Steatosis → steatohepatitis → fibrosis → cirrhosis
Wernicke Korsakoff Syndrome
Thiamine deficiency (B1)
Type of encephalopathy
first line for alcohol use disorder
naltrexone
naltrexone for alcohol use disorder
•Typically used first line due to dosing, cost, and flexible start
•Is also a treatment for opioid use disorder
•A lot of evidence for efficacy
LAI and oral, but mostly LAI
naltrexone side effects - alcohol use
•LAI- injection site including the more serious abscess, necrosis, cellulitis and hematoma
•Oral- nausea, headache, dizziness (but tends to go away with continued use)
disulfiram
•Causes undesirable effects to alcohol (dose dependent)
•Recommended to be used under supervision
•No alcohol for minimum of 12 hours prior to start (really need 2 days abstinence)
•CYP substrate and inhibitor
•Has been used off label for cocaine use disorder
•Counsel on what products have alcohol: toothpaste, mouthwash, some tonics and mixers
disulfiram contraindication
patients with coronary artery disease, psychosis
disulfiram dosing
250-500 mg/day oral
disulfiram common side effects
abnormal taste
headache
sedation
allergic dermatitis
impotence
disulfiram rare side effects
hepatotoxicity
optic neuritis
disulfiram monitoring
liver function tests
CBC
electrolytes
cardiac function
acamprosate how long to take
•Takes 5-8 days for full effect
acamprosate
•Appears to work through its GABA-ergic activity
•Takes 5-8 days for full effect
•Used after naltrexone or can be used to supplement Naltrexone
•Patient must be fully abstinent and already gone through withdrawal
•Studies show some consistent efficacy
acamprosate contraindications
severe renal dysfunction
acamprosate dosing
666mg oral TID
acamprosate common side effects
diarrhea
acamprosate rare side effects
attempted and completed suicides
acamprosate monitoring
renal function
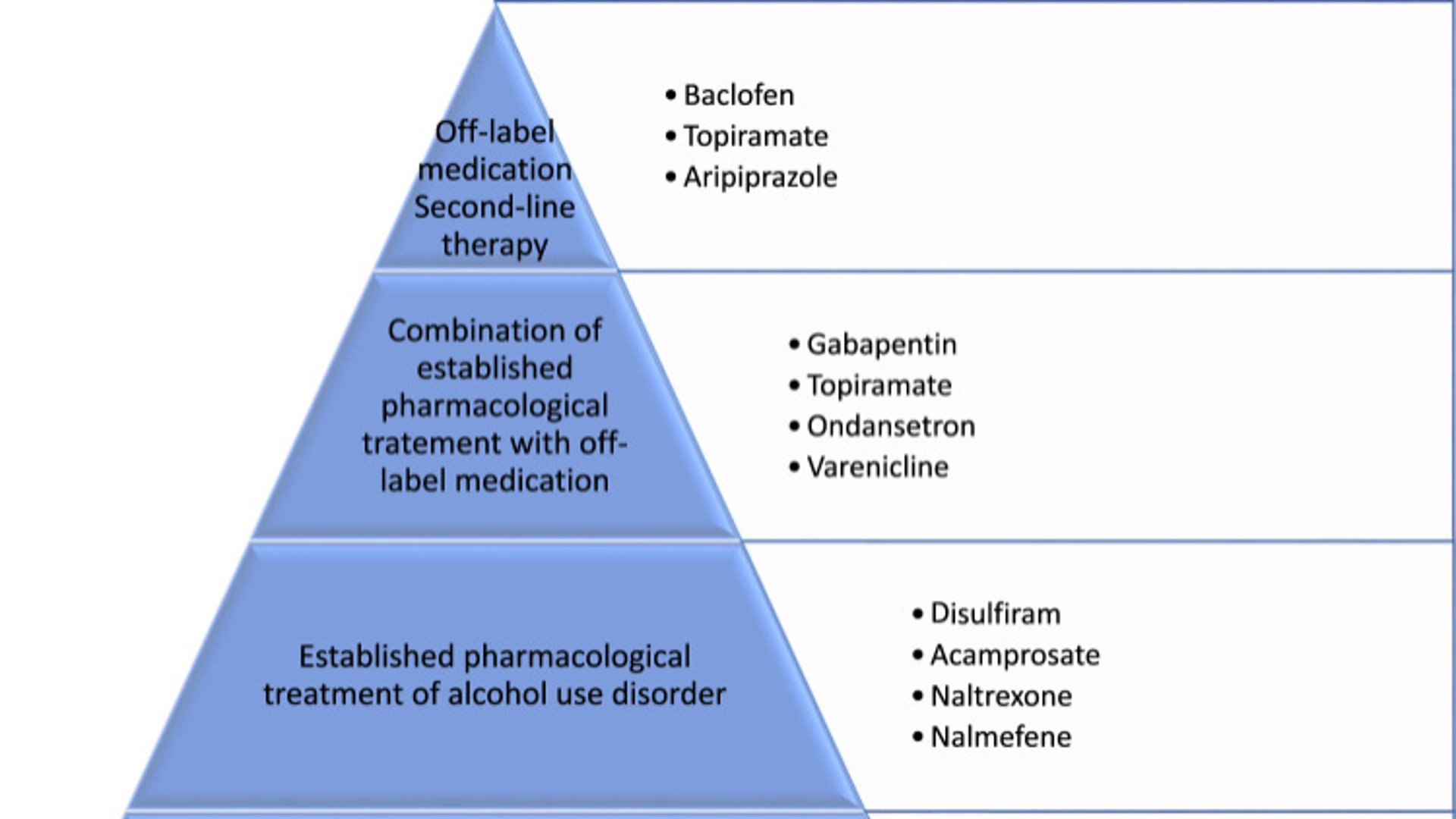
alcohol use disorder drug chart
stimulant use
•Stimulants increase the amount of monoamines in the brain including dopamine, norepinephrine, and serotonin. It also inhibits reuptake of monoamines and inhibits monoamine oxidase.
•Increased extracellular monoamine levels impact our reward pathways and addiction potential
•Causes increased motor activity, increased alertness, increased energy, decreased appetite, a heightened sense of well-being and euphoria.
chronic stimulant use
•Neuropsychologic impairment
•Exacerbated mental health conditions
•Malnutrition
•Weight loss
•Insomnia
•Pruritus
•Risk of seizures, arrythmias, stroke, heart attack
methamphetamine medications
bupropion and naltrexone
bupropion
mirtazapine
cocaine use disorder medications
topiramate
naltrexone
disulfiram
modafinil
dopamine agonists (Adderall)
TA-CD vaccine
evidence based treatments in stimUD
contingency management
cognitive behavioral therapy
peer support
matrix model
discontinuing medications for substance use disorder
different for all patients
patient centered care model
lifestyle modifications - like any other chronic condition
prescription monitoring program
also known as PMP or PDMP
electronic database that tracks controlled substances by state
can be combined with other states
typically tracks the medication, date prescribed, date recieved, quantity, day supply, prescriber and dispenser
what does PMP not required to include
inpatient services
including day surgery clinic
pharmacies operated by the department of corrections
veterinarians
substance use treatment programs such as opioid treatment programs
needle and syringe exchange programs
•Goal: minimize infection transmission risks by supplying sterile equipment and other support services at little or no cost
•Many programs also provide information on accessing treatment and other health care services
needle and syringe exchange program do not…
•Many studies have clearly shown that needle/syringe exchange programs are effective in reducing infectious disease transmission and do not increase rates of community drug use
drug testing strips
•Example: Fentanyl testing strips are used to identify the presence of fentanyl in unregulated drugs
•Increased awareness = decreased overdose risk
•Others: Xylazine testing strips, Rohypnol test strips
•Used in patients using ANY illicit substance due to contamination
nalaxone and nalmefene
3 types of intranasal naloxone
Brand name Narcan
Naloxone 4 mg per dose
Generic Naloxone nasal spray
Naloxone 4 mg per dose
Brand name Kloxxado
Naloxone 8 mg per dose
1 type of intranasal nalmefene
Brand name Opvee
Nalmefene 2.7 mg per dose
flumazenil
Noncompetitive antagonist of the benzodiazepine (BZD) receptor
Use of flumazenil is highly controversial due to ability to cause seizures in withdrawal
“only reverse if you caused it”
Really need to have a good patient history to accurately assess risk vs benefit
If overdose is combined with opioids- Treat the opioid overdose!
Not first line in BZD overdose
Supportive care: oxygen, monitoring, glucose levels, etc.