BI1014 - redox reactions
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
oxidation
Loss of electrons (↑ oxidation number)
reduction
Gain of electrons (↓ oxidation number)
oxidising agent
- Gains electrons (is reduced)
- Causes oxidation
- Becomes more negative
reducing agent
- Loses electrons (is oxidised)
- Causes reduction
- Becomes more positive
general rules of oxidation numbers
1. Free elements = 0 (e.g. Na, Cl₂)
2. Monoatomic ions = their charge (e.g. Ca²⁺ = +2)
3. Molecular compounds:
- More electronegative element gets negative oxidation number
- Bonds between same element don't count
Total = 0 (or ion charge for polyatomic ions)
common assignments
- Hydrogen = +1
Exception: hydrides (e.g. LiAlH₄) = -1
- Oxygen = -2
Exception: F₂O = +2
group number rule
Max oxidation number = group number
Min oxidation number = group number − 8
anode
Reaction -> Oxidation
Charge -> Positive
Electron flow -> Out
Attracts -> Anions
cathode
Reaction -> Reduction
Charge -> Negative
Electron flow -> In
Attracts -> cations
standard electrode potentials
To calculate ΔE°:
- Identify half-reactions
- Place the more negative E° on top (to reverse)
- Add both E° values
If ΔE° > 0, the redox reaction is spontaneous
discharging
spontaneous redox (ΔE° > 0)
charging
reverse reaction with external voltage
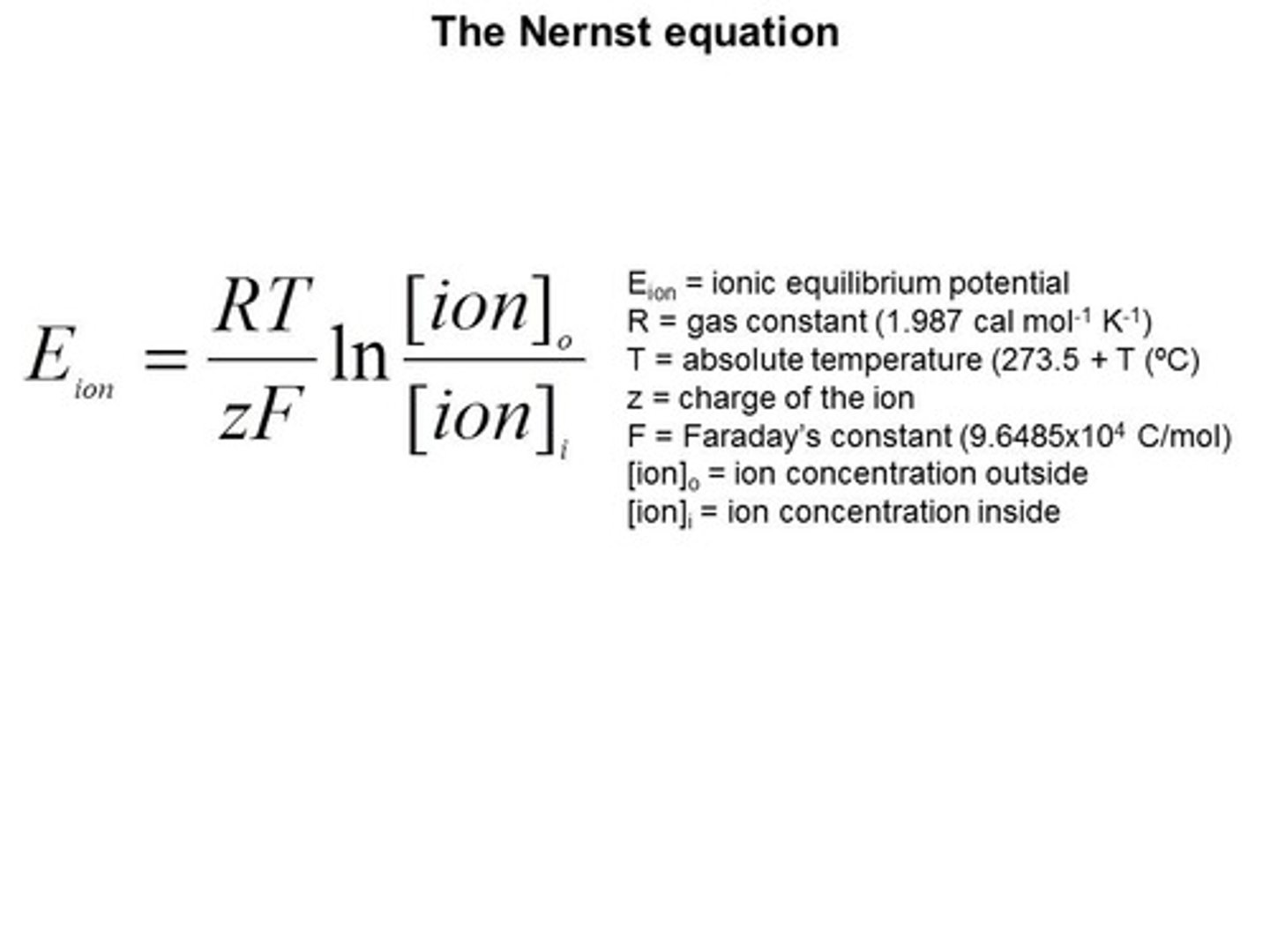
nernst equation
R = 8.314 J/mol·K
T = temperature (K)
n = number of electrons
F = 96,485 C/mol (Faraday constant)
Q = reaction quotient
types of electrode
1st kind
2nd kind
pH electrode
1st kind
Metal electrode in solution of its own ions
2nd kind
Inert metal (e.g. Pt) in contact with redox pair
pH electrode
Needs calibration due to unknown cell constant