Chem final
1/62
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
63 Terms

exam 1

aqueous NaI (0.030 m), aqueous CoI2 (0.030 m), pure H2O, aqueous FeI3 (0.030 m), aqueous glucose (0.050 m)
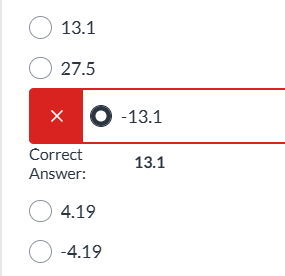
exam 1


exam 1

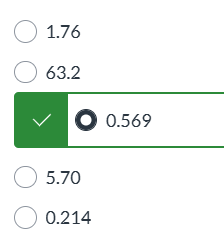
exam 1
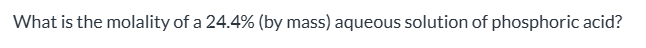
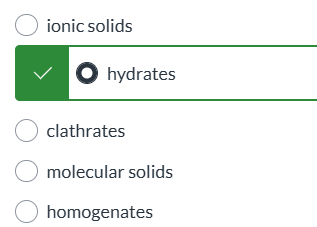
exam 1


0.552, exam 1

respiratory tissue is damaged by oxidation|| the urge to breathe is reduced and not enough CO2 is removed from the body|| No problems result from this situation|| the urge to breathe is increased and excessive CO2 is removed from the body|| hyperventilation results
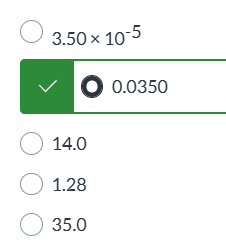
exam 1


aqueous glucose (0.050 m)|| aqueous CoI2 (0.030 m)|| pure H2O|| aqueous NaI (0.030 m)|| aqueous FeI3 (0.030 m)
exam 1


the density of the solution is 11 g/mL|| there are 11 g of sodium chloride in in 1.0 mL of this solution|| 100 g of the solution contains 11 g of sodium chloride|| 100 mL of the solution contains 11 g of sodium chloride|| the molality of the solution is 11
exam 1


exam 1
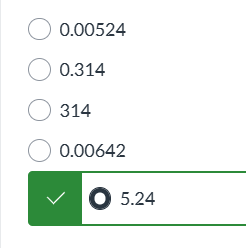

exam 1
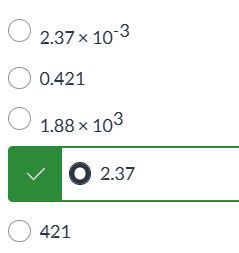

exam 1

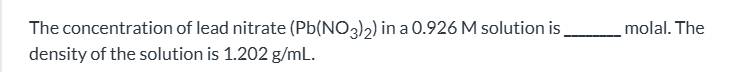
exam 1


exam 1


exam 1


exam 1
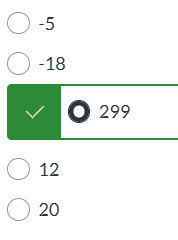

exam 1
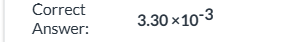
![<p><span>k is the reaction rate constant|| If [A] is doubled, the reaction rate will increase by a factor of 2.|| The reaction is first order in [A].|| The reaction is first order overall.|| The reaction is first order in [B].</span></p>](https://assets.knowt.com/user-attachments/14536134-e843-46cd-81f3-fef14aa2e3b7.png)
k is the reaction rate constant|| If [A] is doubled, the reaction rate will increase by a factor of 2.|| The reaction is first order in [A].|| The reaction is first order overall.|| The reaction is first order in [B].
exam 1


exam 1


exam 1


exam 1

![<p><span>the half-life of a first-order reaction does not depend on [A]0; the half-life of a second-order reaction does depend on [A]0|| the rate of both first-order and second-order reactions do not depend on reactant concentrations|| a first-order reaction can be catalyzed; a second-order reaction cannot be catalyzed|| the rate of a first-order reaction depends on reactant concentrations; the rate of a second-order reaction does not depend on reactant concentrations|| None of the above are true.</span></p>](https://assets.knowt.com/user-attachments/2f182056-7e8a-4b1a-98d8-51ff40c0daf8.png)
the half-life of a first-order reaction does not depend on [A]0; the half-life of a second-order reaction does depend on [A]0|| the rate of both first-order and second-order reactions do not depend on reactant concentrations|| a first-order reaction can be catalyzed; a second-order reaction cannot be catalyzed|| the rate of a first-order reaction depends on reactant concentrations; the rate of a second-order reaction does not depend on reactant concentrations|| None of the above are true.
exam 1

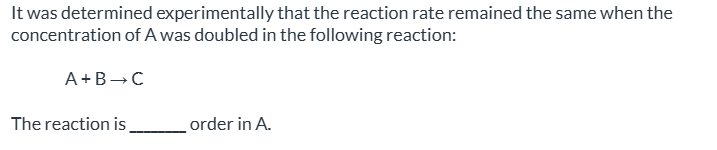
zero, exam 1

exam 1


exam 1


exam 1


exam 1


exam 1
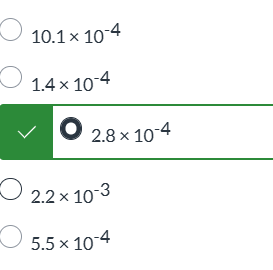
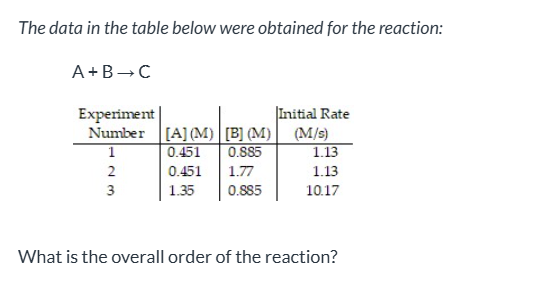
exam 1

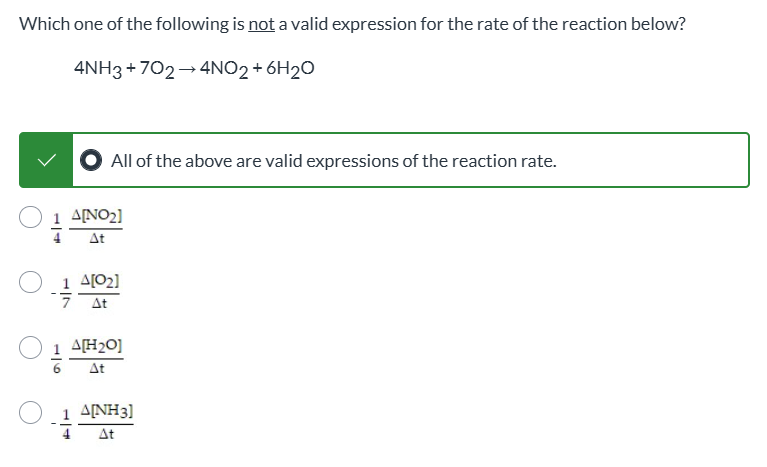
Not a question, just look and see since it’s impossible to screenshot multiple
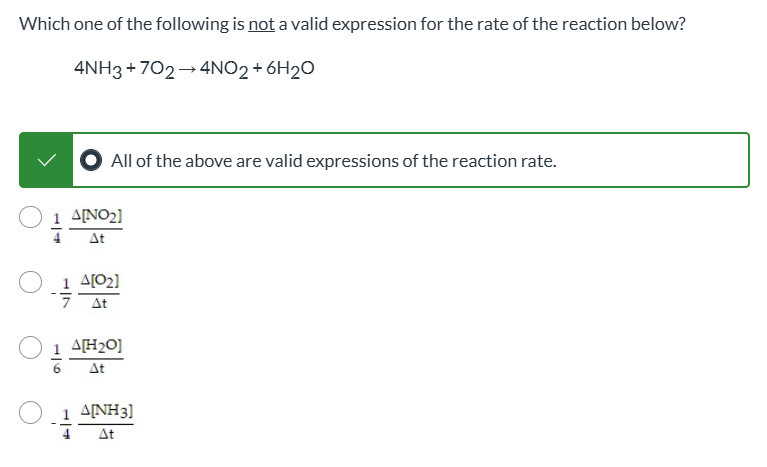

0.023 M NaF|| 0.015 M KF|| 0.030 M AgNO3|| 0.00750 M LiF|| AgF will have the same solubility in all solutions.||
exam 3


H3PO4, KH2PO4|| NaC2H3O2, HCl (C2H3O2- = acetate)|| KOH, HF|| RbOH, HBr||NH3, NH4Cl
exam 3


0.030 M LiF|| 0.023 M NaF|| 0.015 M KF|| Pure Water|| 0.0075 M AgNO3
exam 3
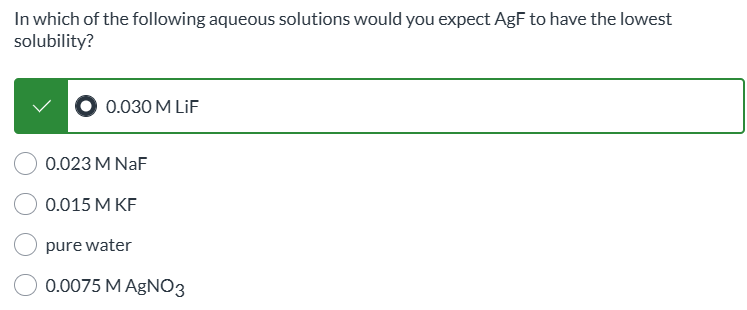

exam 3


exam 3


exam 3

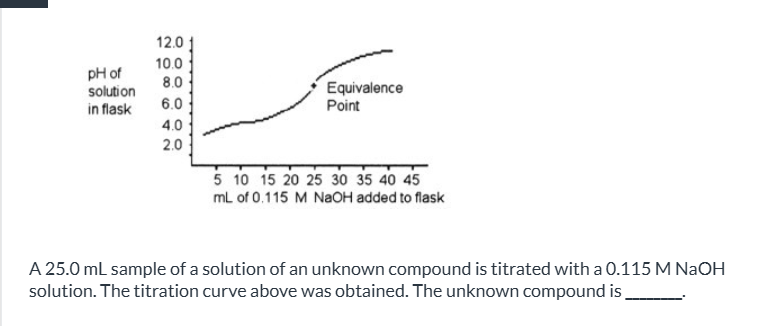
exam 3


1.15 M HF and 0.624 M NaF|| They are all buffer solutions and would all have the same capacity.|| 0.574 M HF and 0.312 M NaF|| 0.287 M HF and 0.156 M NaF|| 0.189 M HF and 0.103 M NaF
exam 3
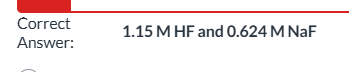

exam 3
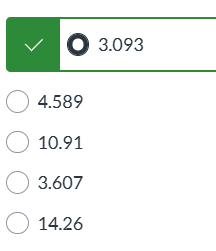

exam 3

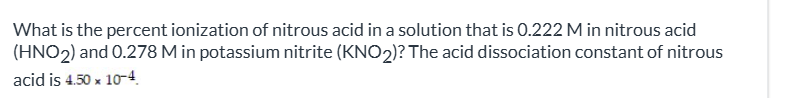
exam 3
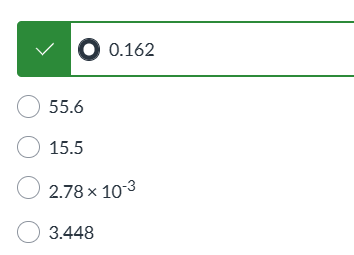

CuCl, HCl|| none of the above|| AgBr, HBr|| KI, HI|| Csl, HI||
exam 3


HC6H5O|| KOH|| NH3|| KNO3|| HNO3
exam 3


exam 3

exam 3

exam 3

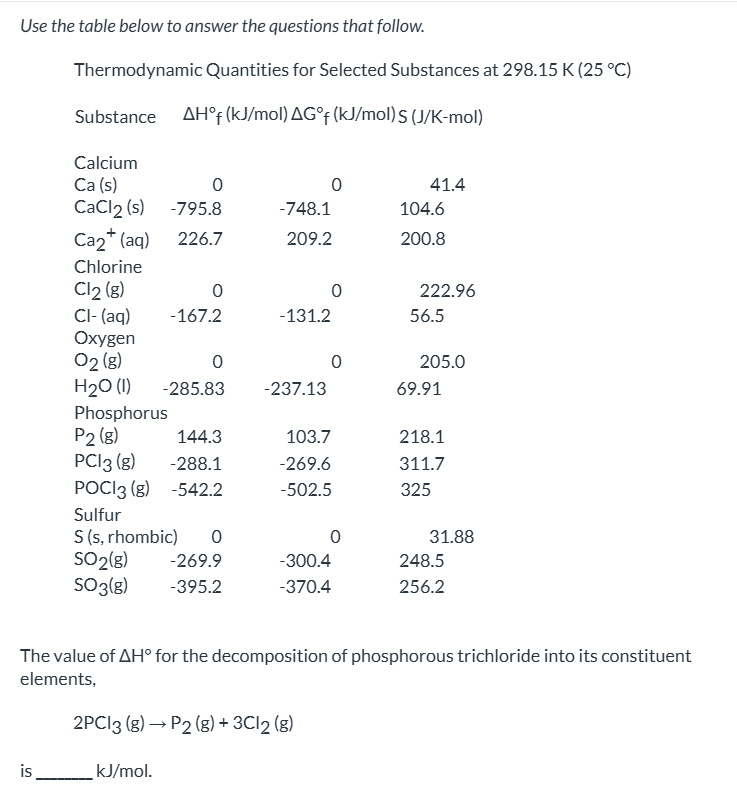
exam 3


exam 3

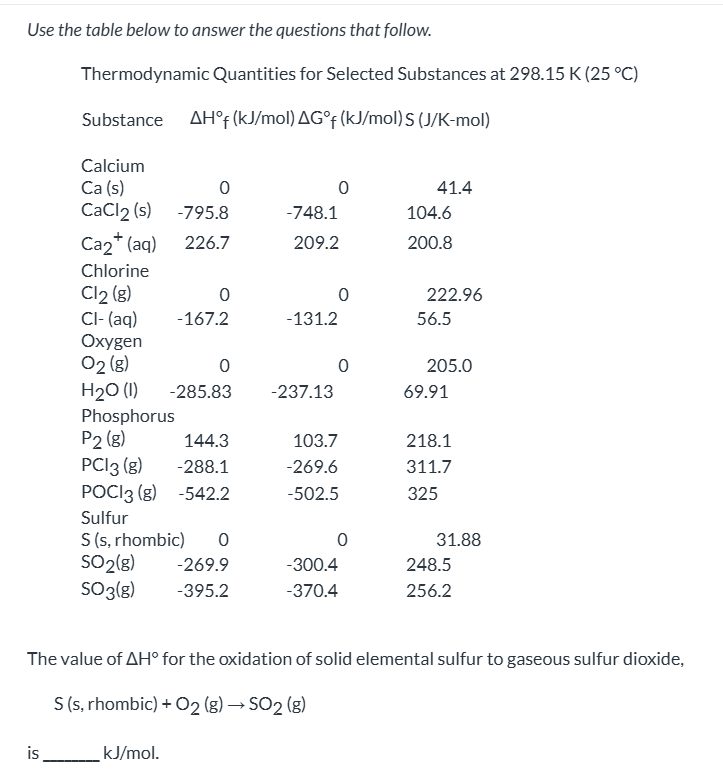
exam 3


no way to question, just look at answer and analyze why
exam 3
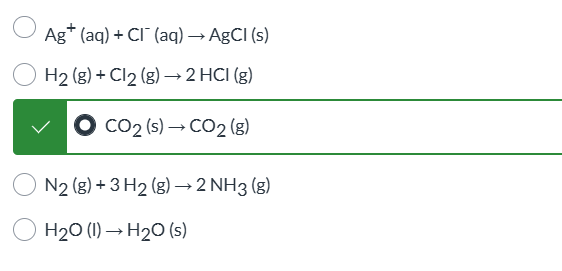

no way to question, just look at answer and analyze why
exam 3
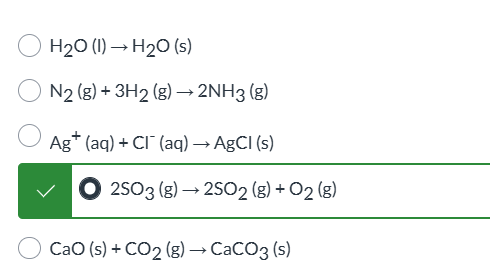

exam 3


exam 3
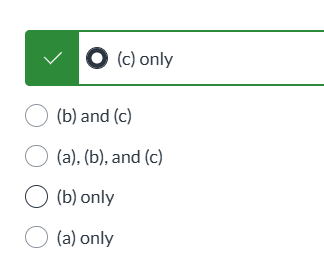
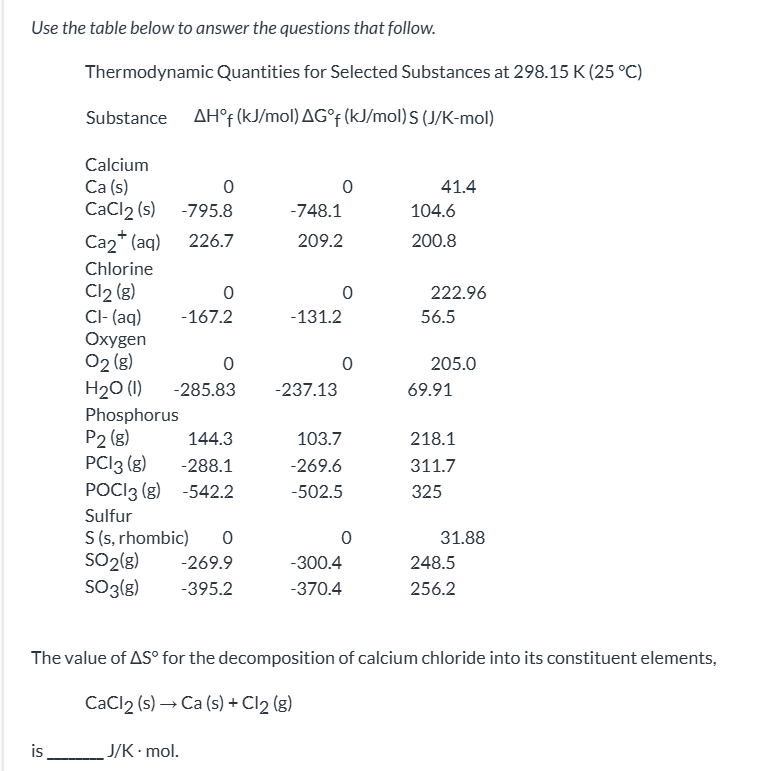
exam 3
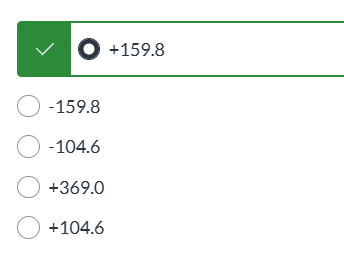

the forward process is spontaneous but the reverse process is not|| the forward and the reverse processes are both spontaneous|| both forward and reverse processes have stopped|| the process is not spontaneous in either direction|| the reverse process is spontaneous but the forward process is not
exam 3


exam 3
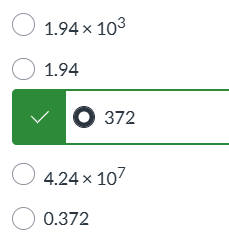

K = 1|| K > 1|| K=0|| more info needed|| K < 1
exam 3


exam 3

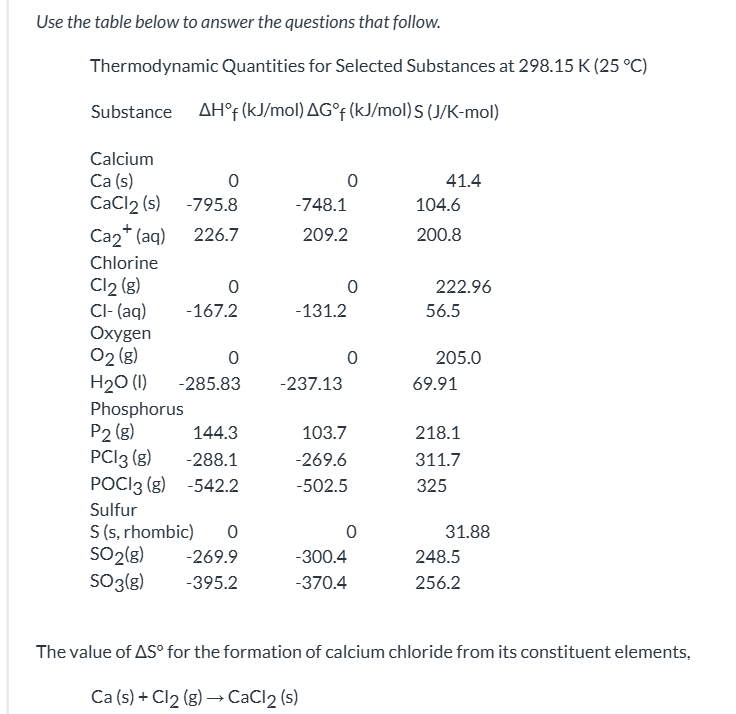
exam 3
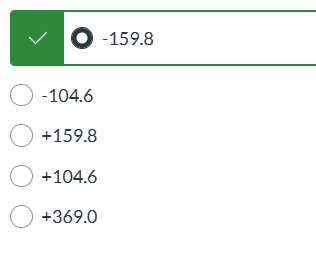

exam 3