Shapes of molecules and intermoleculr forces
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
What does molecular shape depend on?
Number and arrangement of electron pairs surrounding central atom
Bonding pairs = forming covalent pairs with other atoms
Lone pairs =. Not involved in bonding and remain on central atom
What is the electron pair repulsion theory?
Molecule adopts a shape that minimises repulsion between electron pairs due to negative charge
Electron pairs repel eachother
Lone pairs cause more repulsion than bonding pairs because they are closer
Electron pairs arrange themselves as far apart as possible to minimise repulsion
What order does repulsion decrease in?
Lone pair - lone pair > lone pair - bonding pair > bonding pair - bonding pair
Consequently , lone pairs occupy more space, affecting molecules shape when compressing bond angles
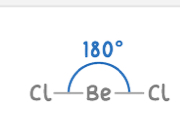
Linear shape
2 electron pairs
2 bonding pairs, 0 lone pairs
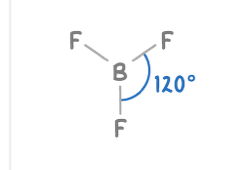
Trigonal planar
3 electron pairs
3 bonding pairs , 0 lone pairs
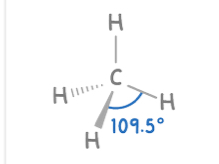
Tetrahedral
4 bonding pairs
109.5
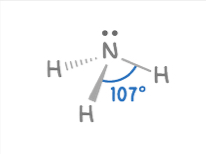
Trigonal Pydramidal
4 electron pairs
3 bonding, 1 lone pairs
Bent / linear
2 bonding pairs, 2 lone pairs
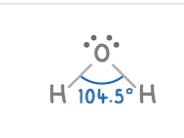
Trigonal bipyramidal:
5 electron pairs
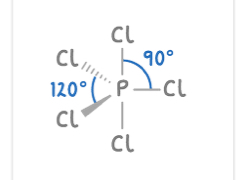
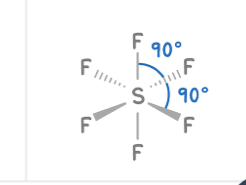
Octahedral
6 bonding pairs
Shapes of species with multiple bonds:
Treat each multiple bond as if it is a single electron pair
What is electronegativity?
Ability of atoms to attract shared electron pair in a covalent bond towards itself
Fluorine is most electronegative , then oxygen , nitrogen and chlorine
What is Pauling scale
Electronegativity measured on Pauling scale, higher value = more electronegative
What factors affect electronegativity?
Atomic radius = smaller atom are more electronegative because their electrons are closer t nucleus, stonger electrostatic attraction
Nuclear charge = higher positive charge in nucleus increases strength of electrostatic attraction between nucleus and electrons
Shielding = electrons in inner shells weaken electrostatic attraction between nucleus and outer electrons
Trends in electronegativity across a period
Increase
Nuclear charge increases, pulls electronics closer and decreases atomic radius + results in stronger electrostatic attraction between nucleus and outer shells
Trends in electronegativity down a group
Decreases
Down a group, nuclear charge increases, atomic radius increases more, number of inner shell electron shielding also increases
Increased distance between nucleus and outer electrons + greater shielding weakens electrostatic atttraction
What are polar bonds and how are they formed?
In a covalent bond between atoms with different electronegativity els, bonding electrons are more attracted to electronegative atom - the unequal sharing makes bond polar
Polar bonds have permanent dipole (seperation of positive and negative charges within polar molecule, resulting from uneven distribution of electrons)
More electronegative atom has negative charge and less electronegative atom has positive charge, bigger difference in en, more polar the bond
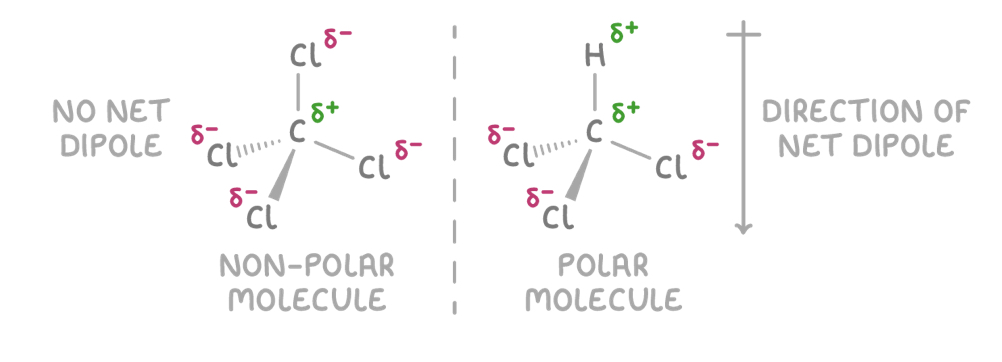
How is the polarity of a molecule dependant on how its polar bonds are arrange in 3D?
Charge is unevenly distributed in polar molecules
CCL4 = non- polar = symmetric arrangement causes dipoles to cancel out, even though it has polar bonds
CHCL3 = polar = asymmetric arrangemtn doesn’t allow dipoles to cancel out, lead to a net dipole
Difference in electronegativity between C-Cl and C-H causes dipoles to not fully cancel out
How does electronegativity predict bond type?
En difference is 0, like diatomic molecules - H2, O2 = pure covalent bond as electrons shared equally
En difference increases, bond is more polar covalent e,g H-Cl
En difference is very large, bond is ionic = more en atom takes bonding electrons from less en atom NaCl
What are intermolecular forces? + 3 types
Attractive forces that exits between molecules, these are weaker than covalent bonds that hold atoms together within molecules
Induced dipole dipole forces (london forces)
Permanent dipole - dipole
Hydrogen bonding
Summarise characteristics of the 3 intermolecular forces
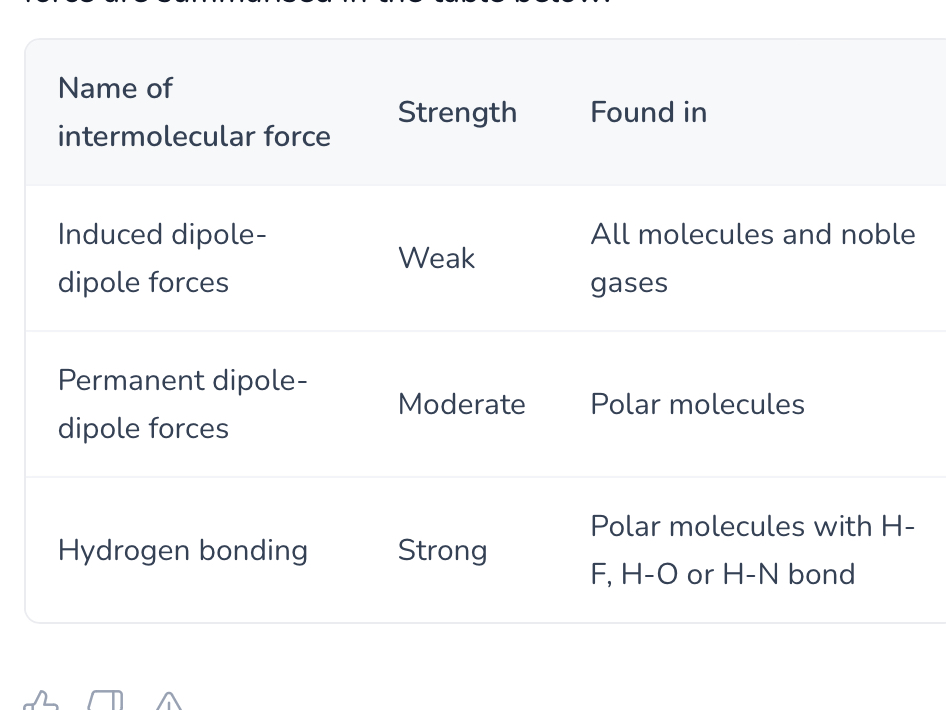
What are london forces and how do they occur?
Present between all atoms and molecules
Electrons in atom constantly move, at one point there are more electrons on one side than other = temporary dipole
This temporary dipole induces an opposite dipole in a neighbouring atom, cause weak electrostatic attraction between atoms
Induced dipole induce further dipoles in nearby particles
Dipoles constantly forming and disappearing as electrons move, overall effect is net attraction between atoms
What factors affect the strength of induced dipole dipole forces?
Size - larger atoms and molecule have more electrons that can become polarised, stronger temporary dipoles
SA = larger SA has stronger induced dipole dipole forces as more electron aloud is exposed for interactions
So stronger london forces = higher BP
Can london forces be used to hold molecules together in a lattice structure?
Yes
Solid iodine = molecules held together by covalent bonds
I2 molecules attracted to eachother by london forces, form a molecular lattice
London forces also explain existence of noble gas liquids / solids, as it allows them to condense into liquid and solids at very low temps
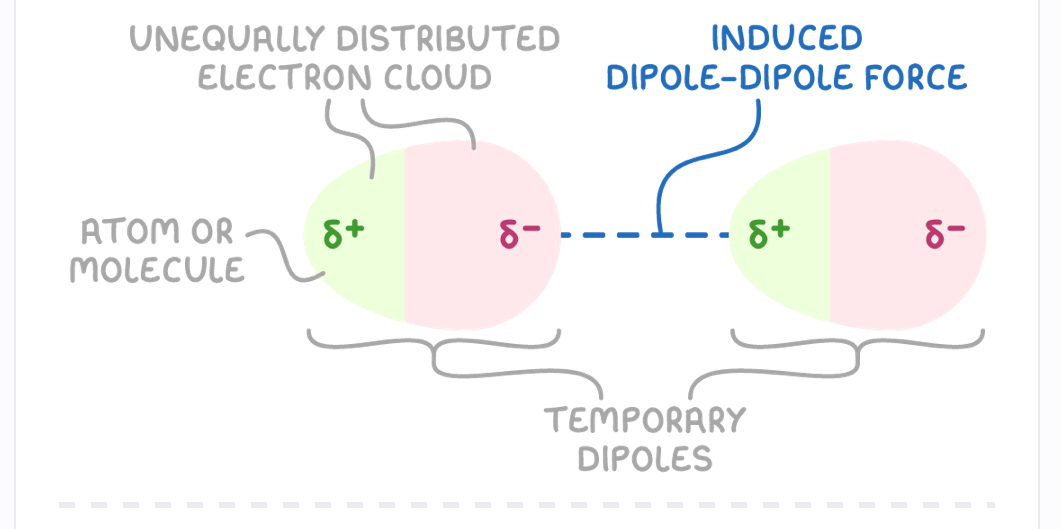
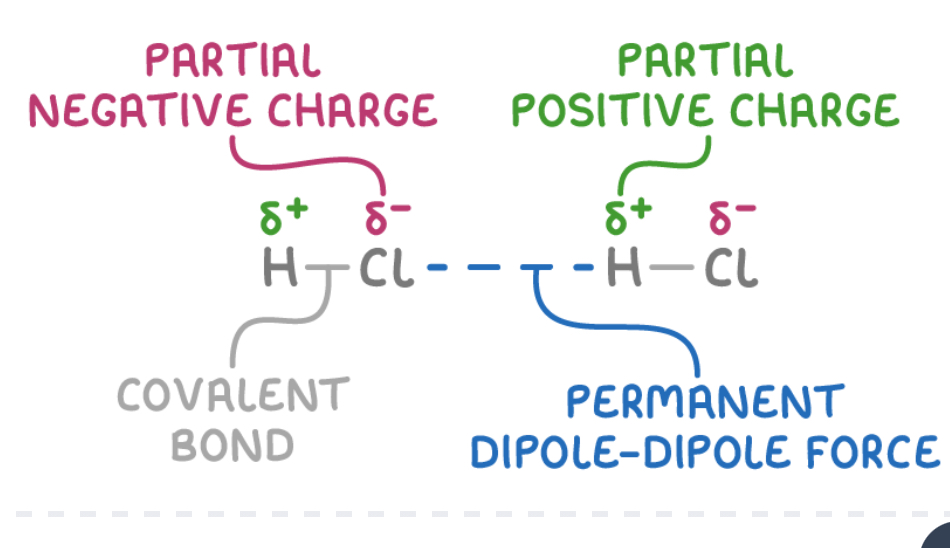
What are permanent dipoles and how do they occur?
Electrostatic attractions between partial positive end of polar molecule and partial negative end of another
H-Cl bond is polar due to greater electronegativity of chlorine
H atom has a partial positive charge and Cl has partial negative
HCL molecules align so the delta positive charge on H is attracted to delta negative chlorine
Methanal, CH2O has higher boiling point than ethane C2H6 - explain using intermolecular forces
Methanal is polar due to carbonyl group C=O (has permanent dipole bond and london forces)
Ethane is non polar , only has london forces
More enegry required to overcome stronger intermolecular forces in Methanal, lead to higher BP
What is hydrogen bonding and its requirements?
Type of permanent dipole dipole force that occurs when hydrogen is bonded to highly electronegative element fluorine, oxygen or nitrogen
Molecule must contain a H atom bonded to F O N
Must be a lone pair of electrons on F O N to interact with H
How do hydrogen bonds form?
H-F, H-O, H-N bonds are highly polar due to large electronegativity difference between H and elements - leads to partial positive charge on H and partial negative on FON atom
Small size of H allows it to get close to lone pair of electron on adjacent FON
Lone pairs on FON are regions of high electron density (high partial negative charge)
Positively charged H is strongly attracted to negatively charged lone pair, form H bond between molecules
How to draw hydrogen bond?
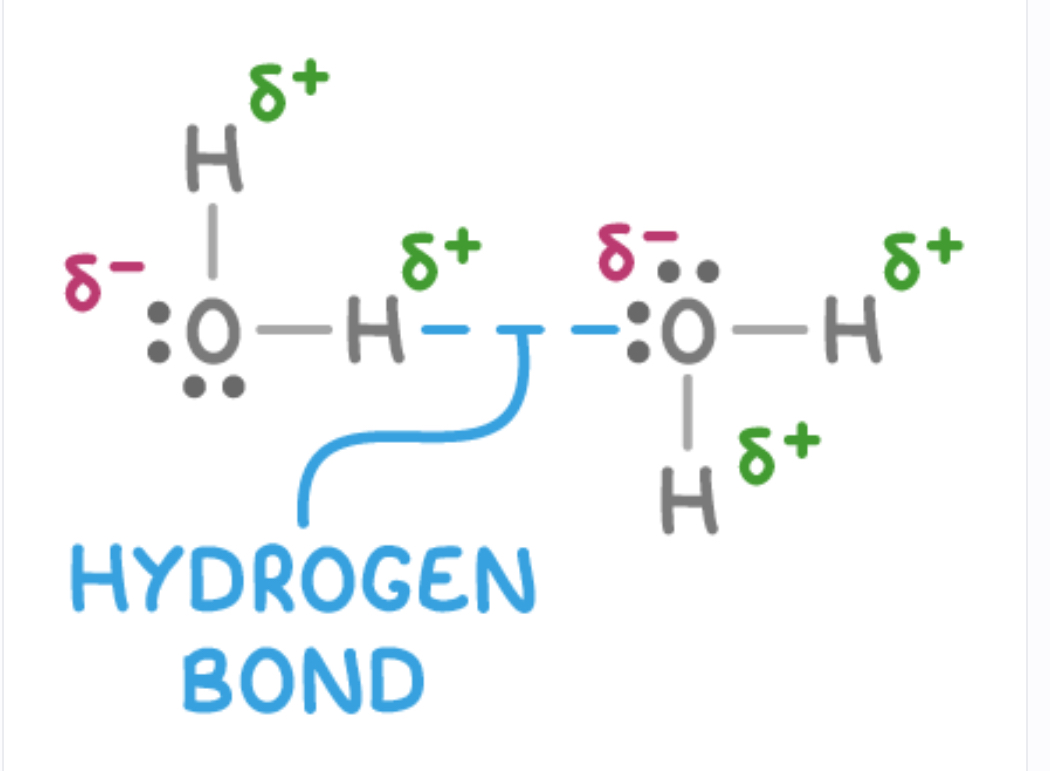
How does hydrogen bonding impact properties of substance?
Greater solubility in water = substances that form h bonds in water (ethanol) are soluble
Higher MP / BP = extra energy needed to overcome strong H bond forces
How does hydrogen bonding explain the anomalous properties of water and ice?
Ice is less dense than water = in ice, water molecules arranged in a 3D lattice held by H bonds, when melting, these H bonds break H bonds are relatively longer than covalent bonds, so ice is less dense
Water and ice have high MP / BP = compared to other molecules of similar size, due to H bonds which require more energy to break
How do intermolecular forces explain properties of simple molecular substances?
Low MP / BP = weak intermolecular forces dont need a lot of energy to overcome, often liquid/gas at room temp
Solubility in water = polar molecules interact with water molecules, non polar molecules (only have london forces) are insoluble
Electrical conductivity = covalent compounds do not conduct electricity, regardless of polarity