Diffraction Laws
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
Bragg’s law
Condition for constructive interference is given by 2dsinθ=nλ where n is an integer.
Most common applications to x-rays, neutrons, electrons, which have a wavelength comparable to interatomic spacing ∼1A˚=10−10m.
Bragg’s law assumes specular reflection (equivalent angles of incidence and reflection) of waves from lattice planes.
Laue condition for diffraction
Laue condition for diffraction is Q=G, where Q is the scattering vector, Q=k’−k.
Laue approach assumes the crystal is made of identical (sets of) atoms at lattice sites R that scatter waves elastically.
Constructive interference will occur if Q⋅R=2π×integer for all lattice vectors R, and is only satisfied for all Q if the Laue condition is satisfied.
Positioning of Q relative to the scattering planes
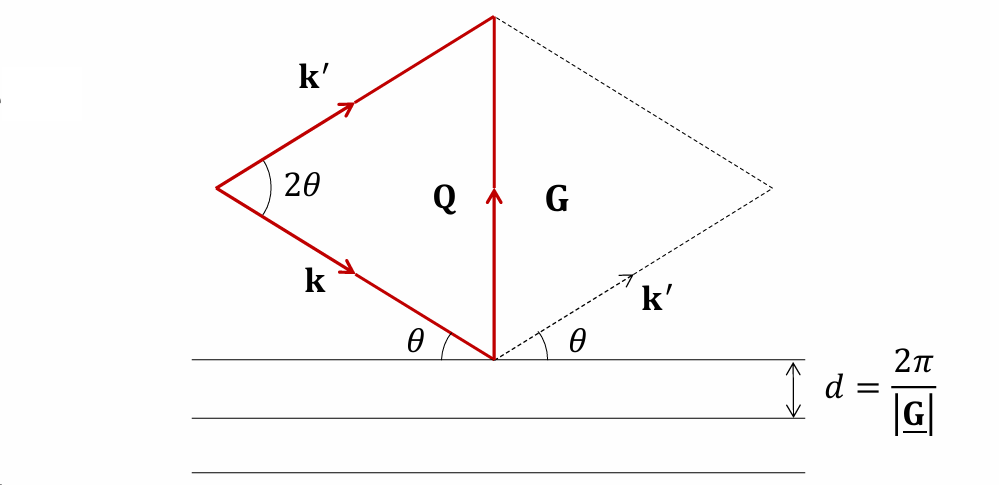
When the Laue condition is satisfied, Q is normal to the scattering planes.
Ewald sphere
For a incident beam of certain wavelength 𝜆 on a crystal, the magnitude of k (and k′) is fixed and therefore all possible scattering vectors Q lie on the surface of a sphere, the Ewald sphere, of radius ∣k∣.
Scattering amplitude F(Q)
Is the Fourier transform of the electron density function n(r). Experimentally, only intensity I=∣F∣2 can be measured, so information about phase is lost.
F(Q)=∫solidn(r)e−iQ⋅rdV
Structure factor SG
If the Laue condition is satisfied, all the points separated by lattice vector R scatter constructively, so F(Q)=NSG where N is the number of unit cells in a solid.
Atomic form factor fj(Q)
Is the amplitude scattered by the atom, given by the Fourier transform of the electron density of an individual atom, taken over a unit cell.
fj(Q)=∫cellnj(ρj)e−iG⋅ρdV
Atomic form factor value for Q limits
As ∣Q∣→0, we find fj=Z, the atomic number.
As ∣Q∣→∞, we find fj=0.
Powder diffraction
A common methods for determining material structures, using x-rays.
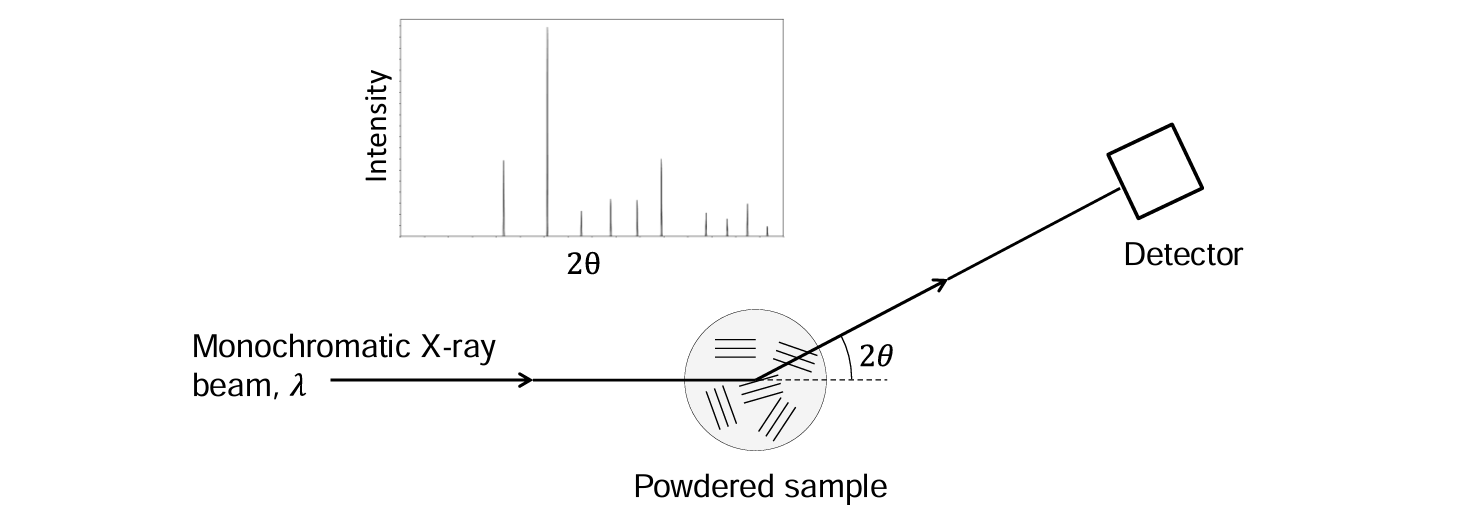
Diffraction leads to Debye-Scherrer rings of intensity at specific values of 2θ.
Determining structure from powder diffraction
Rearrange Bragg’s law and the distance between the planes, written d=a/h2+k2+l2 to give an equation in terms of sin2θ.
Eliminate unknown lattice parameter a by taking ratios from the nth to the 1st peak, giving sin2θn/sin2θ1=R.
Calculate the (h2+k2+l2) ratios for all allowed ℎ𝑘𝑙 reflections for the two potential structures, and then compare to the experimental sin2𝜃 ratios.
Absence/presence of an extra R determines which structure it is.
Intensities from powder diffraction
Variation of the structure factor and the form factor lead to peaks having different scattered intensities for a material.
Why is powder diffraction useful?
All incident angles of the incoming waves to the Bragg planes are effectively sampled in a single experiment.
Powdered (or polycrystalline) samples are generally easier to prepare than single-crystal samples.