low carbon - hydrogen economy, fossil fuels, and biomass (Q1)
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
different hydrogen production methods
steam reforming of methane
coal gasification
water electrolysis (green hydrogen)
biomass and waste food
steam reforming of methane (SMR)
most common method of producing hydrogen, but it relies on fossil fuels (natural gas)
reaction 1(steam reforming): CH4+H2O→CO+3H2
reaction 2 (water-gas shift): CO+H2O→CO2+H2
alternative (partial oxidation): CH4+0.5O2→CO+2H2
SMR: reaction 1(steam reforming)
CH4+H2O→CO+3H2
highly endothermic (+206.4 kJ/mol)
uses Ni catalyst
at 700°-1100°C
SMR: reaction 2 (water-gas shift)
CO+H2O→CO2+H2
mildly exothermic (-41.2 kJ/mol)
uses Fe/Cr oxide catalysts
SMR: Alternative (Partial Oxidation):
CH4+0.5O2→CO+2H2
exothermic (-35.7 kJ/mol)
alternative to steam reforming (autothermal reforming)
combines SMR, water-gas shift, and partial oxidation into single reactor
energy neutral - heat generated by partial oxidation matches heat required for steam reforming
Coal Gasification
coal undergoes devolatilisation to form gaseous mixture and solid char residue
it is then gasified into CO and H2
environmental issues: releases severe pollutants, including heavy metals (Mercury, Cadmium, Zinc), NOx, SOx, and poisonous CO
Coal gasification: reactions
partial oxidation of char: C+0.5O2→CO (exo)
steam reforming of C: C+H2O→CO+H2 (endo)
steam reforming of C: C+2H2O→CO2+2H2 (endo)
water-gas shift: CO+H2O→CO2+H2 (exo)
Water Electrolysis (Green Hydrogen)
splitting of water into hydrogen and oxygen using electric current
Reaction: 2H2O→2H2+O2
anode:
cathode:
Green opportunity: if electricity used to power electrolysis comes from renewable sources, hydrogen produced with no greenhouse gas emissions and no reliance on fossil fuels - truly sustainable hydrogen economy
Biomass and Waste Food
biomass = biological material that is carbon neutral (wood, crops, food waste, animal residues, industrial waste)
carbon neutral = absorbs CO2 when growing and releases it when burned
biological conversion (fermentation)
thermochemical (the carnol process)
biomass and waste food: biological conversion (fermentation)
microorganisms (e.g enzymes/archaea) convert glucose into ethanol
Equation: C6H12O6→2C2H5OH+2CO2
Biomass and waste food: thermochemical (the carnol process)
methane → methanol fuel
Step 1 (thermal decomp): CH4→C+2H2
Step 2 (methanol synthesis): CO2+3H2→CH3OH+H2O
overall:
2CO2+3CH4→2CH3OH+2H2O+3C
Hydrogen purification methods:
chemical methods
selective methanation
selective oxidation
physical methods
pressure swing adsorption (PSA)
palladium membrane separation
Selective methanation
uses Co, Fe, or Ru catalyst to convert CO back into methane
CO+3H2→CH4+H2O
Drawback: highly exo (-206.1 kJ/mol) and consumes valuable H2 just produced
Selective Oxidation
uses catalyst to selectively promote CO+0.5O2→CO2 over combustion of H2
Thermodynamics favour CO reaction at lower temps
Pressure Swing Adsorption (PSA)
removes impurities based on their Mr at high pressures
using beds of zeolites, silicia and carbons
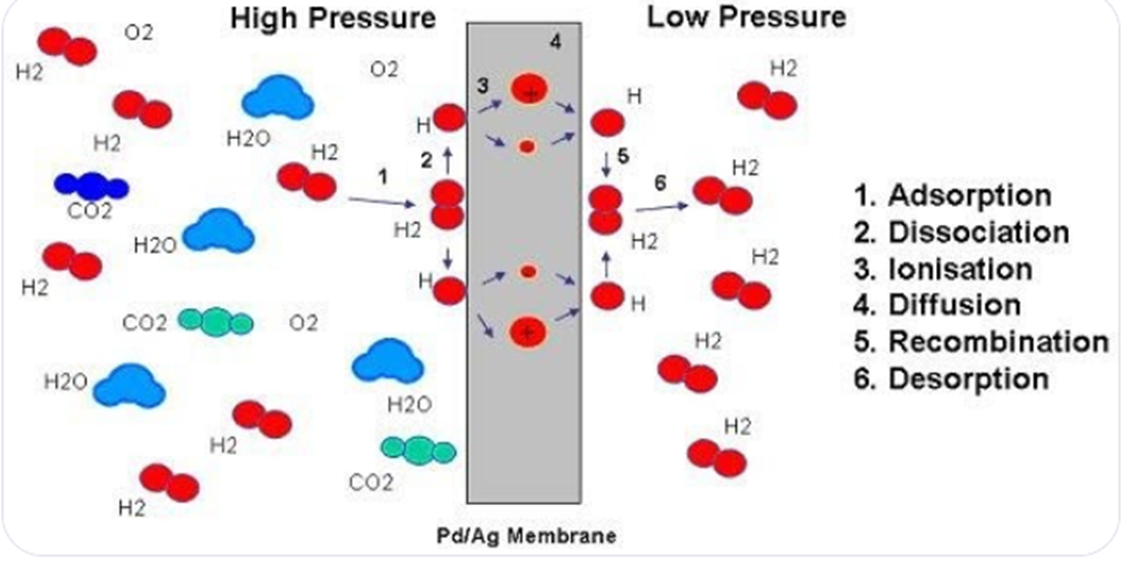
Palladium Membrane Separation
H2 molecules hit the Pd membrane
disassociate into monatomic hydrogen
diffuse through microscopic gases between metal atoms
recombine into H2 on the other side
+: yields 99.9999% pure H2
-: palladium is very expensive
why must hydrogen be purified?
Hydrogen produced via SMR or gasification contains 0.2% to 3% CO. CO poisons Platinum catalysts used in fuel cells, must be reduced to incredibly pure levels (1-10 ppm)
hydrogen storage issues
hydrogen has tiny molecular size and a negative Joule-Thompson coefficient
meaning leaks can easily escape and self-ignite
Physical hydrogen storage
stored as highly compressed gas in reinforced cylinders or cryogenic liquid hydrogen
liquid spills can cause severe explosive burns
hydrogen safety: protocols
Testing tanks/equipment rigorously to prevent leaks.
Installing extra safety valves.
Designing equipment to withstand shocks, vibrations, extreme temps.
Using H2/O2 leak detectors.
Keeping fuel cell supply lines physically separated from other equipment
Alternative fossil fuels and biofuels
the biodiesel cycle
shale gas (hydraulic fracturing/fracking)
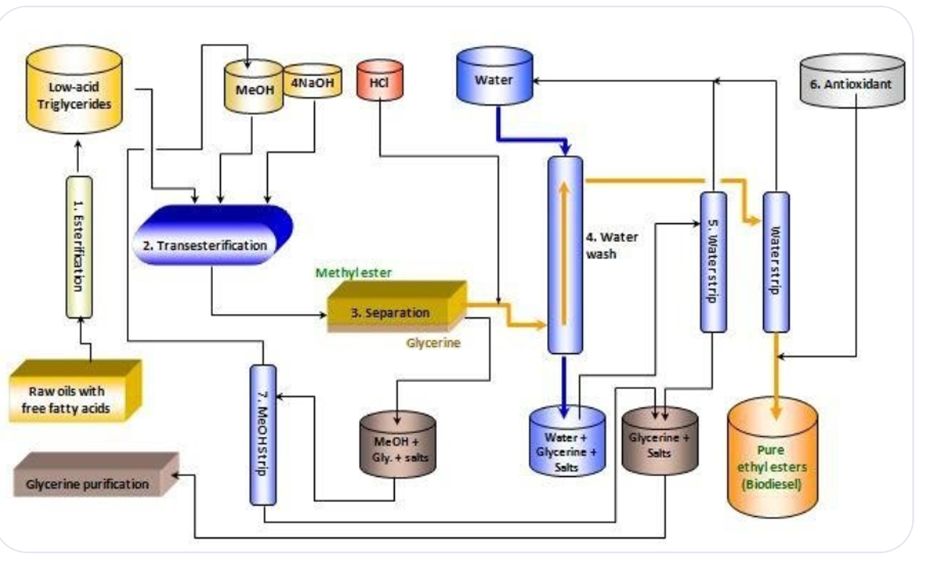
The biodiesel cycle
Reaction (Transesterification):
Triglyceride + 3 Alcohol ←> Alkyl esters + glycerol
conditions: requires catalyst (NaOH/KOH) and is heated to 160 °C
+: renewable, biodegradable, higher flashpoint (safer to store), higher lubricity prolongs engine life), significantly reduces Particulate Matter (40-60%) and Co (10-50%) compared to petroleum diesel
-: poor quality biofuel damages engines, SVO is too viscous to use directly, ‘food vs fuel’ supply conflicts
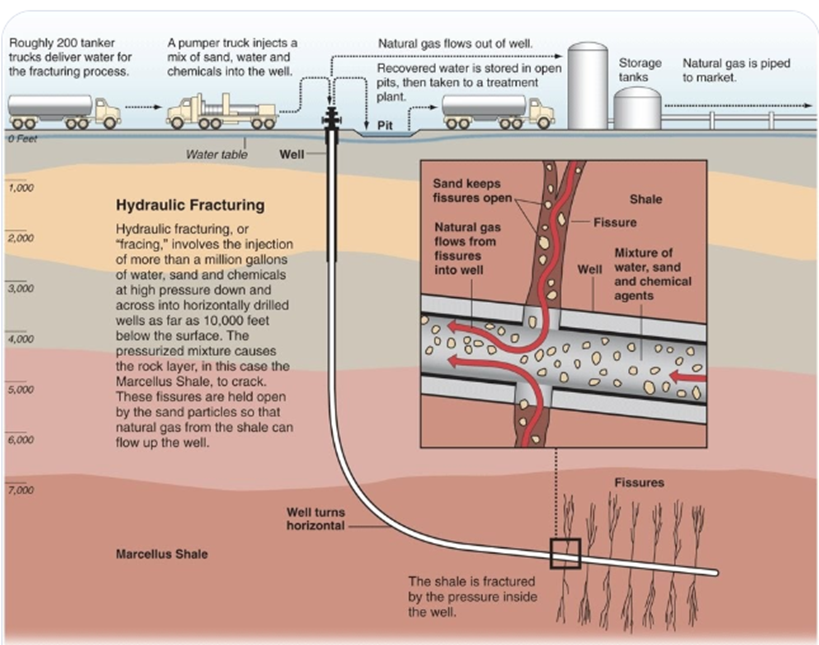
Shale gas (hydraulic fracturing/fracking)
natural gas trapped in impermeable shale rock deep underground
well is drilled horizontally
mixture of water, sand, chemicals pumped in at 1500 lbs per square inch to fracture rock and release gas
+: increases energy security, creates jobs, natural gas burns cleaner than coal/oil
-: requires lots of water, risks polluting freshwater aquifers with heavy metals/chemicals, high-pressure fracturing can trigger minor earthquakes
biodiesel cycle diagram
hydraulic fracturing
palladium membrane diagram