GCSE Chemistry: C10 - Organic reactions
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
What are alcohols in water?
Alcohols are soluble in water and form neutral solutions.
How does the solubility of an alcohol change with the number of carbon atoms?
As the number of carbon atoms increases, the solubility of an alcohol decreases.
Describe what happens in the reaction between ethanol and sodium
- in the reaction of the alcohol ethanol with sodium, we see bubbles of hydrogen gas being produced
- we have also made the product sodium ethoxide
ethanol + sodium ----->
ethanol + sodium ----> sodium ethoxide + hydrogen
Show the general word equation for the reaction of an alcohol with sodium
alcohol + sodium -> sodium (prefix of alcohol)oxide + hydrogen
methanol + sodium ---->
methanol + sodium ----> sodium methoxide + hydrogen
propanol + sodium ---->
propanol + sodium -----> sodium propoxide + hydrogen
butanol + sodium ---->
butanol + sodium ----> sodium butoxide + hydrogen
Name a good example of an oxidising agent
A good example of an oxidising agent is acidified potassium dichromate.
Describe what happens when an alcohol reacts with an oxidising agent
- when an alcohol reacts with an oxidising agent, they produce a carboxylic acid and water
Show the general word equation for the reaction of an alcohol with an oxidising agent
alcohol + oxidising agent -----> carboxylic acid + water
ethanol + oxidising agent
ethanol + oxidising agent ----> ethanoic acid + water
methanol + oxidising agent
methanol + oxidising agent ----> methanoic acid + water
propanol + oxidising agent
propanol + oxidising agent -----> propanoic acid + water
butanol + oxidising agent
butanol + oxidising agent ----> butanoic acid + water
Describe what happens in the combustion of an alcohol
- alcohols release energy when combusted
- combusting an alcohol in air produces carbon dioxide and water
Show the general word equation for the combustion of an alcohol
alcohol + oxygen ----> carbon dioxide + water
The carboxylic acids are a ....
The carboxylic acids are a homologous series.
This means that carboxylic acids all have the same functional group.
What is the functional group of carboxylic acids?
Carboxylic acids have the functional group COOH.
Methanoic acid
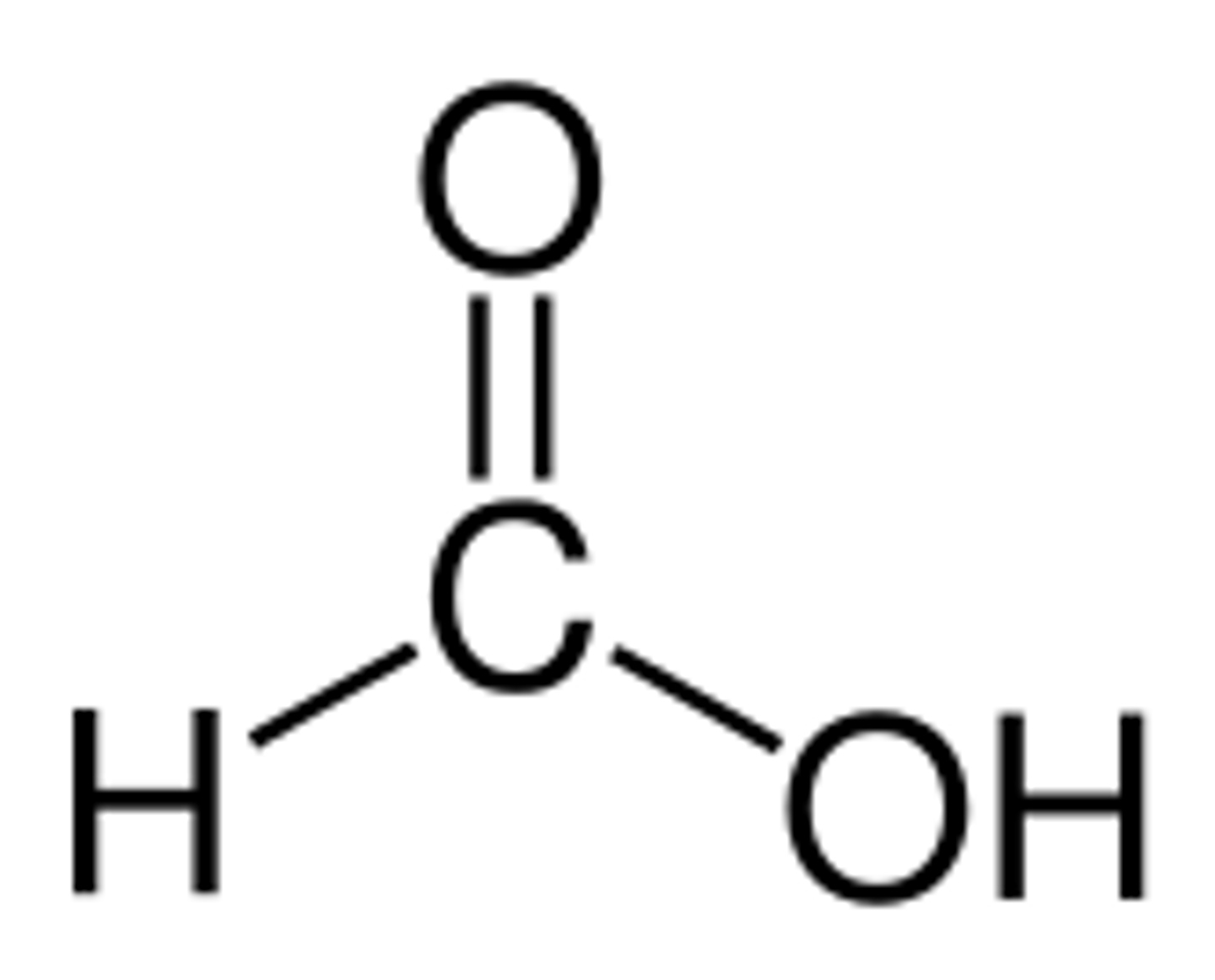
Structural formula of methanoic acid
HCOOH
Ethanoic acid
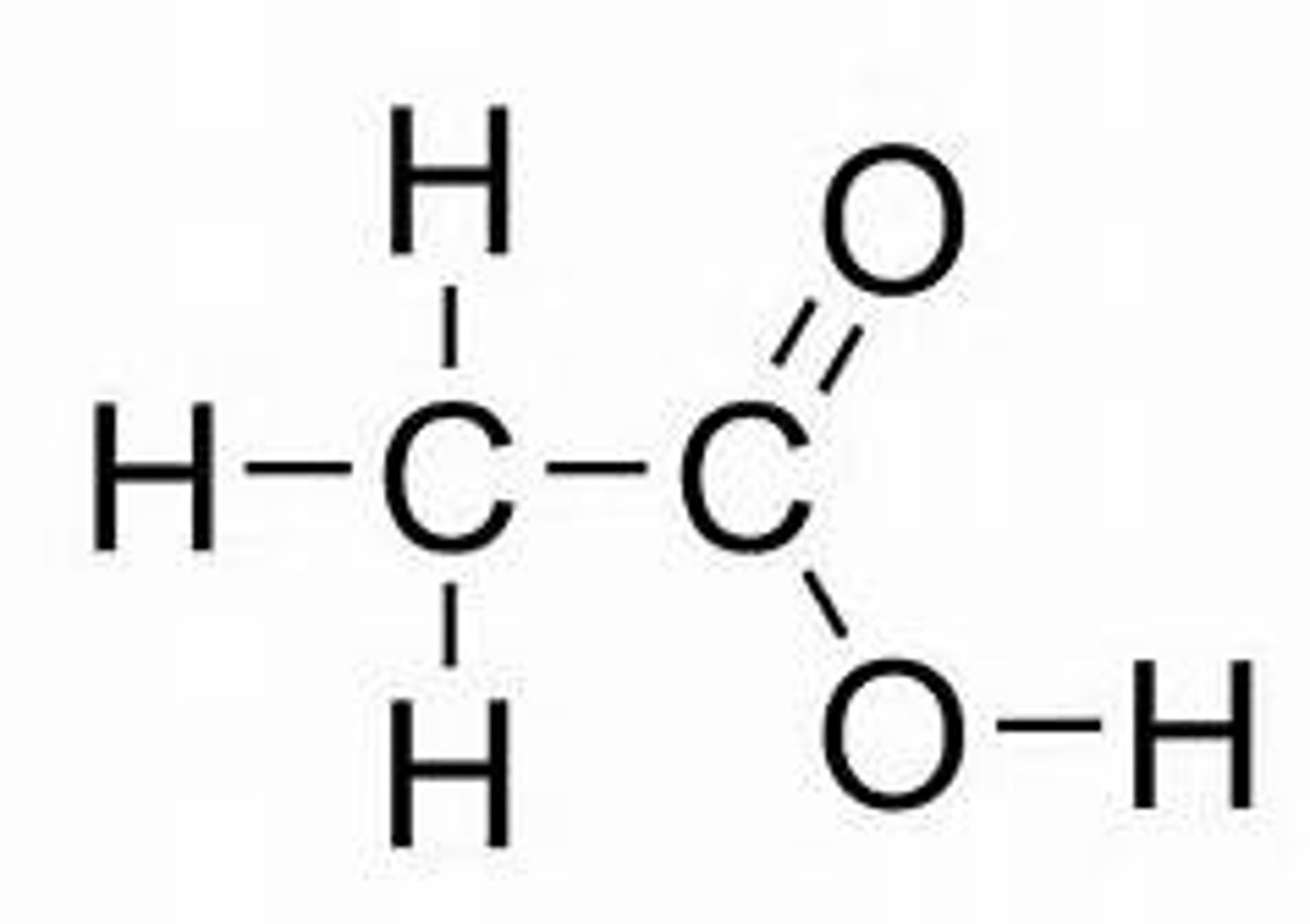
Structural formula of ethanoic acid
CH₃COOH
Propanoic acid
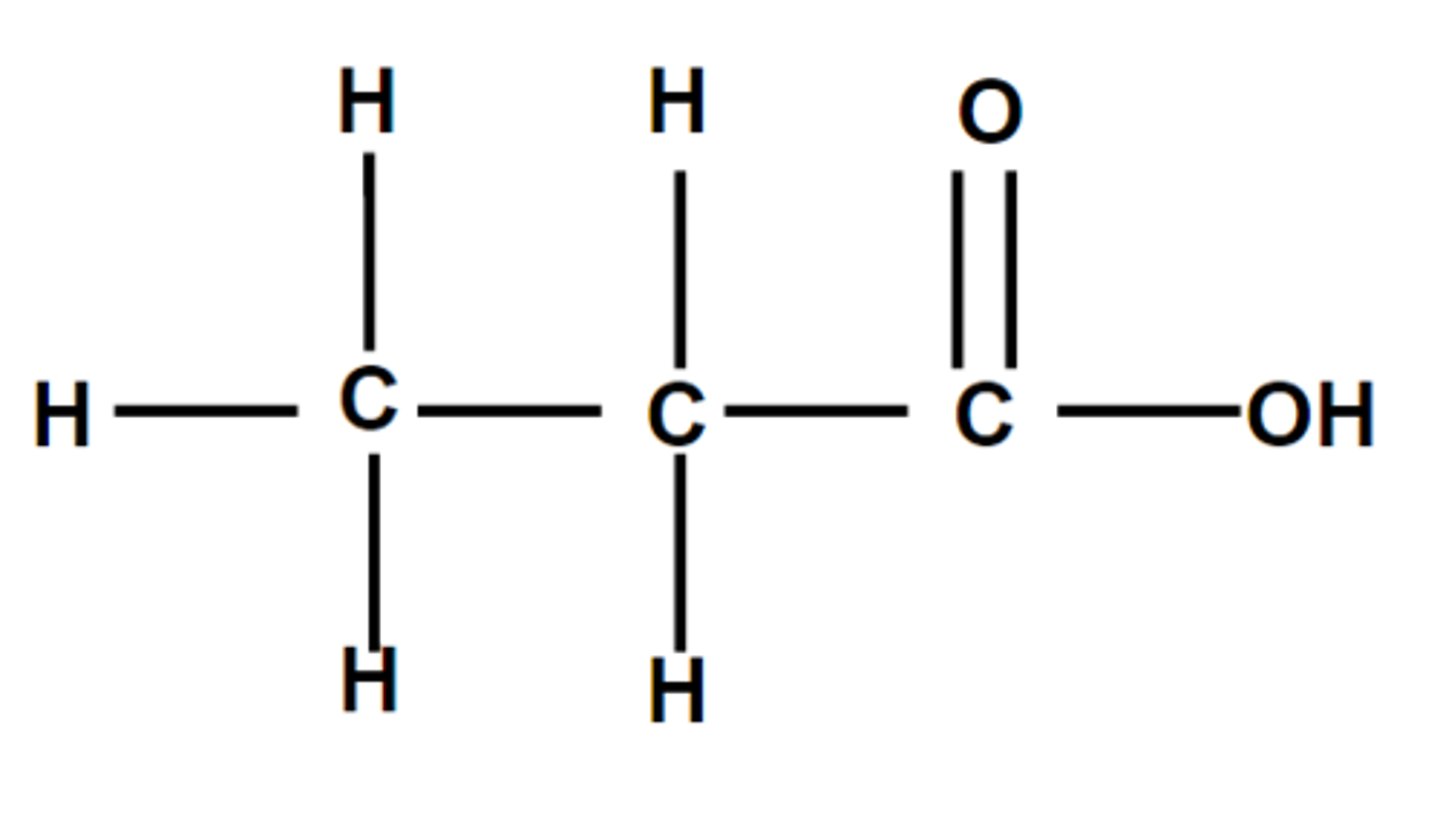
Structural formula of propanoic acid
CH₃CH₂COOH
Butanoic acid
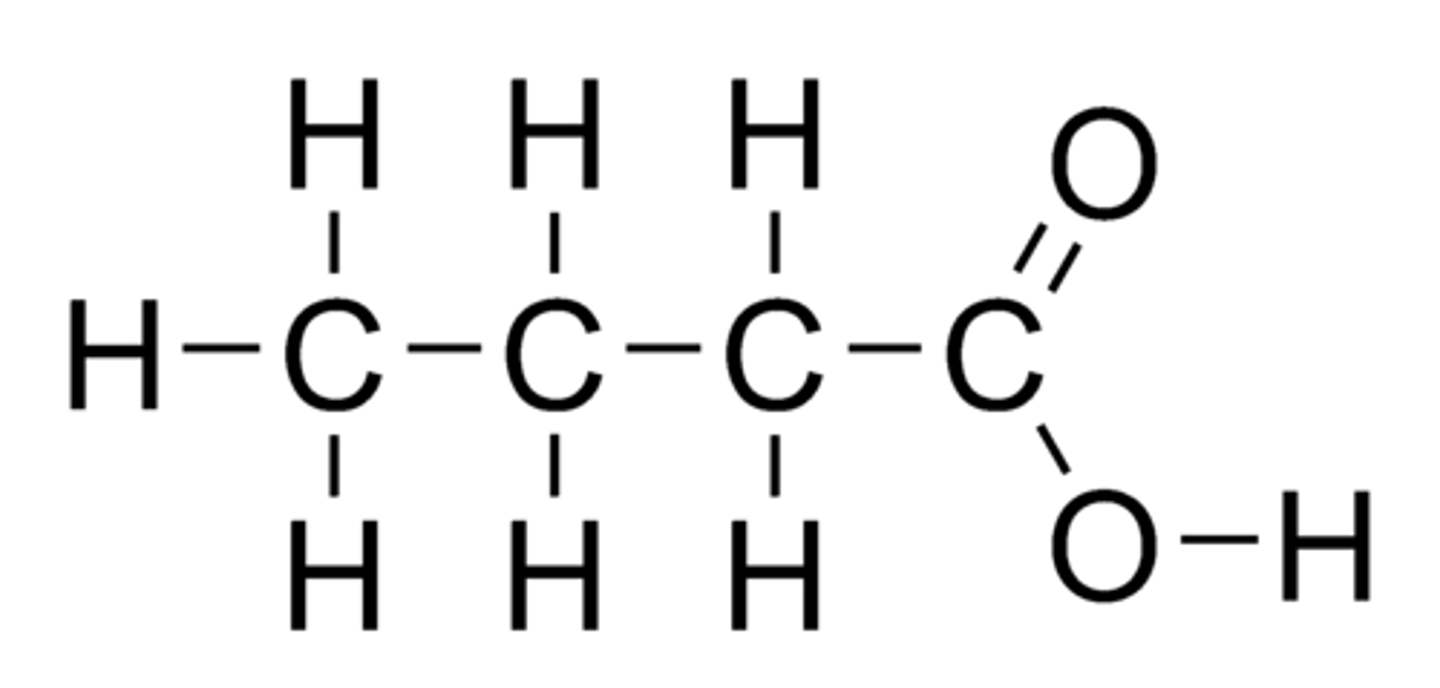
Structural formula of butanoic acid
CH₃CH₂CH₂COOH
In water, carboxylic acids are ...
In water, carboxylic acids are weak acids.
Describe what happens when the carboxylic acid ethanoic acid ionises
- when ethanoic acid ionises, the ethanoate ion and H⁺ hydrogen ions are produced
- this is a reversible reaction, so the ethanoate ion and H⁺ can recombine to form ethanoic acid
Ethanoate ion
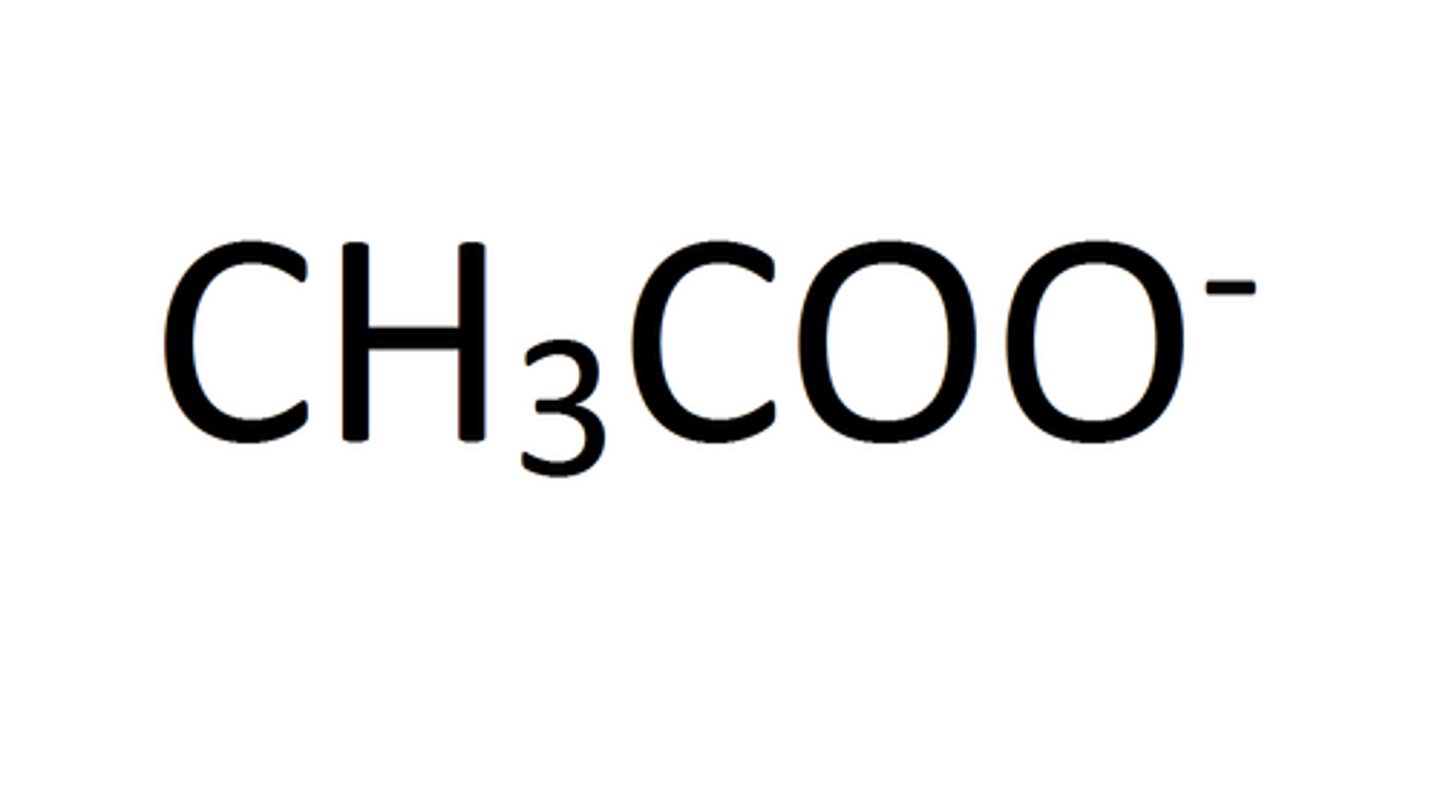
Hydrogen ion
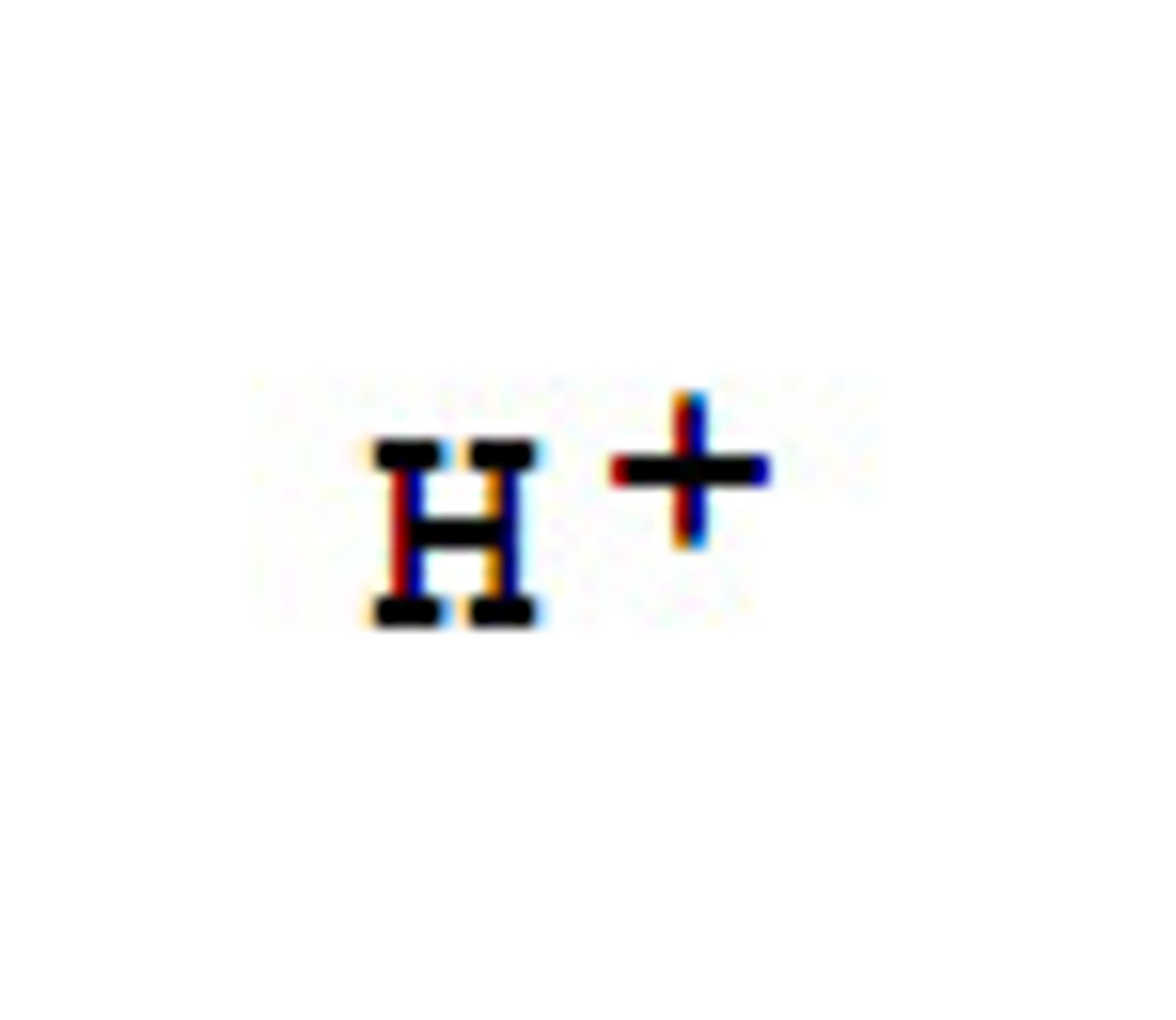
Why are carboxylic acids weak acids?
Because carboxylic acids only partially ionise in aqueous solution, they are weak acids.
What sort of pH do carboxylic acids have?
- as carboxylic acids are weak acids, this means that carboxylic acids have a higher pH than a stronger acid such as hydrochloric acid
- this is because, releasing H⁺ hydrogen ions in aqueous solution lowers the pH of an acid, but carboxylic acids only partially ionise as they are weak acids, and so this means that carboxylic acids have a high pH, as they release fewer H⁺ hydrogen ions in aqueous solution
Describe what happens in a reaction between ethanoic acid and sodium carbonate
- in the example of ethanoic acid reacting with sodium carbonate, we have made the salt sodium ethanoate, carbon dioxide gas and water
Show the general word equation for the reaction of a carboxylic acid with a metal carbonate
carboxylic acid +metal carbonate->salt +carbon dioxide+ water
The salt is ....
The salt is the positive ion from the metal carbonate, followed by the negative ion produced by the carboxylic acid.
Methanoic acid produces the negative ion
methanoate
Ethanoic acid produces the negative ion ....
ethanoate
Butanoic acid produces the negative ion ....
butanoate
Propanoic acid produces the negative ion ....
propanoate
Describe what happens in a reaction of a carboxylic acid with an alcohol
- When we react a carboxylic acid with an alcohol, we make a molecule called an ester, and water
What are esters and what are they used for?
Esters are really useful molecules, as they often have a pleasant smell, and so are used a lot in foods.
Give two properties of esters that make them suitable for use in perfumes
- esters have a pleasant smell
- esters are volatile
What is the name of the ester produced in the reaction between ethanoic acid and ethanol?
Ethyl ethanoate
Bonding diagrams to show: ethanoic acid + ethanol
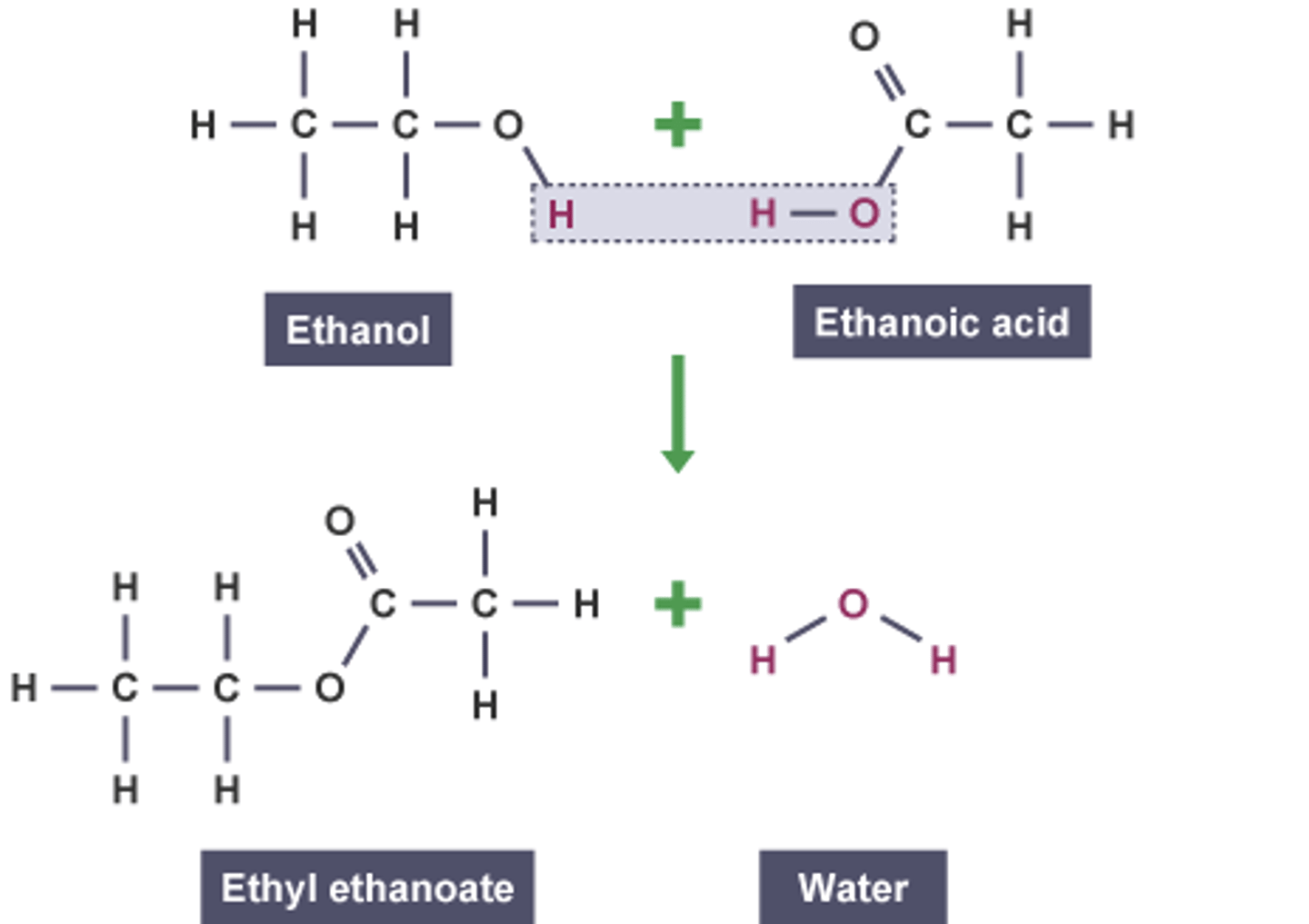
Ethyl ethanoate
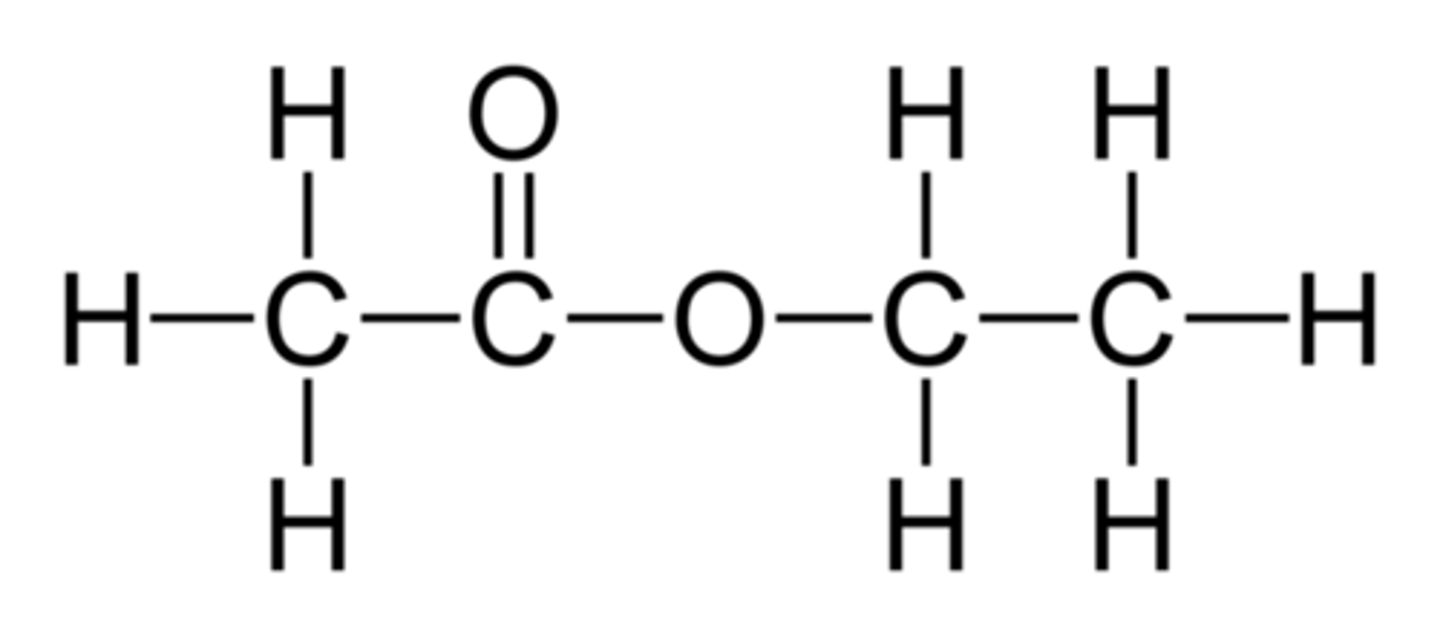
ethanoic acid + ethanol ----->
ethanoic acid + ethanol -----> ethyl ethanoate + water
In the reaction between ethanoic acid and ethanol, the ester ....
In the reaction between ethanoic acid and ethanol, the ester ethyl ethanoate is produced, as well as water.
What conditions are needed for the reaction between ethanoic acid and ethanol to produce ethyl ethanoate and water?
The reaction between ethanoic acid and ethanol requires sulfuric acid as a catalyst.
What is the functional group of an ester?
C=O-O
How is an ester named?
The first part of the name of an ester is the name of the alcohol but with the 'anol' suffix replaced by the suffix 'yl'.
The second part of the name of an ester is the negative ion produced by the carboxylic acid.
What does volatile mean?
Volatile means a substance evaporates into a gas easily.