Optics
1/24
Earn XP
Description and Tags
MST1
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
definition of light. How is vision possible?
light is electromagnetic radiation that we can see
vision is made possible by photons (packets of energy) being absorbed by photoreceptor cells of the eye
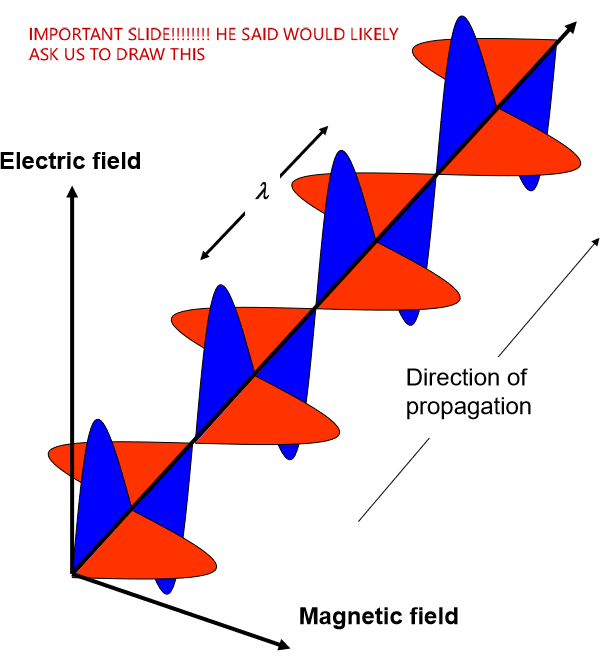
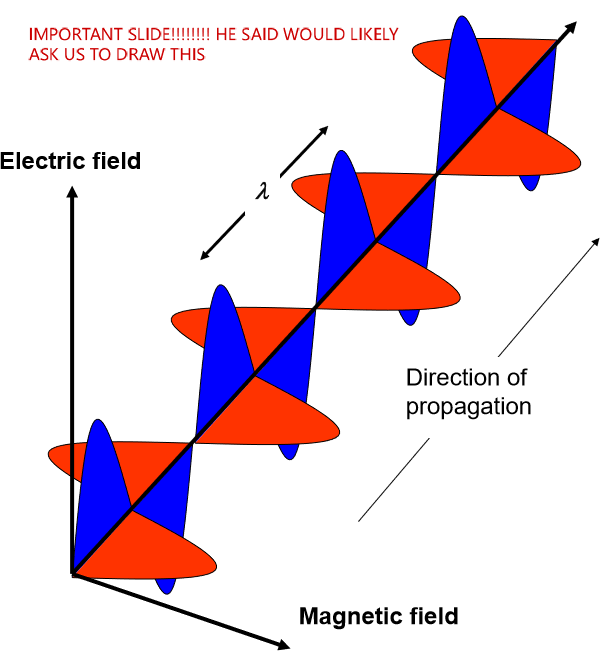
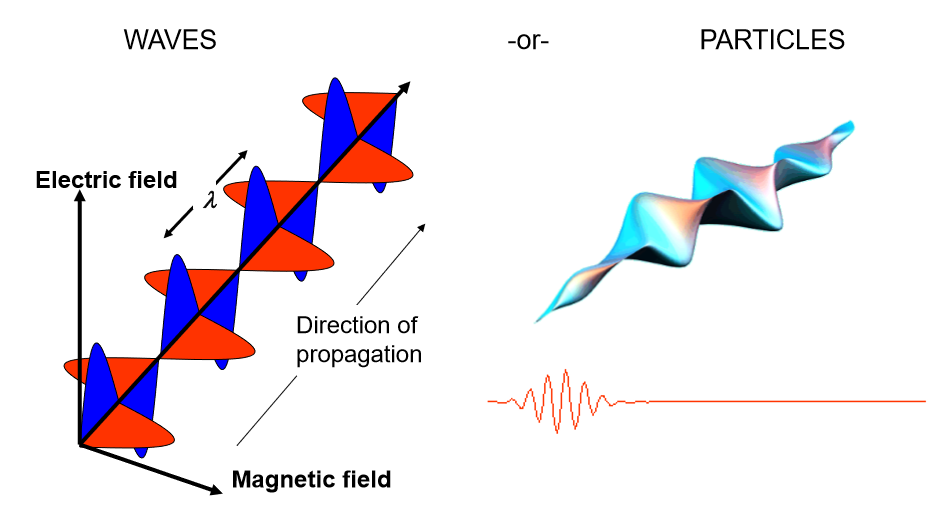
what kind of wave is light. Understand how to draw this
transverse electromagnetic wave (there is both a magnetic and an electric field). Light is part of the electromagnetic radiation spectrum that our eyes can detect!
understand the quantisation of energy
Light transfers energy in separate packets called photons, not as a continuous flow, and each photon’s energy depends on its frequency (E=hf)
how does energy of each quantum vary with wavelength?
as wavelength increases, energy INCREASES!! (and frequency)
inversely proportional!
shorter wavelength → higher energy
longer wavelength → lower energy
Example:
blue/UV light = shorter wavelength = higher energy
red light = longer wavelength = lower energy
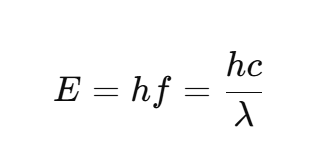
range of spectra that can be produced by artificial light sources
Artificial light sources can produce very broad continuous spectra, uneven/spiky spectra, or very narrow single-wavelength spectra
examples:
Tungsten bulb: broad spectrum, with a lot of energy in the infrared.
Fluorescent lights / LEDs: can have uneven or spiky spectra, even if they still look white.
Lasers: very narrow spectrum, close to a single wavelength.
Describe the propagation of light wave as a combination of electric and magnetic field components
time-varying electric fields (E) and magnetic fields (B) generate and re-enforce one another, underlying the propagation of electromagnetic radiation
Magnetic flux changing with time —> change the electric field
Electric flux changing with time —> change the magnetic field
they influence one another
Maxwell’s equations describe this relationship
How to calculate diopters?
100 divided by power to get the answer in cm
e.g. a 5D lens has a 20 cm focal length.
how to calculate frequency?
f = c/λ
f = frequency
c = speed of light = 3×108 m/s
λ = wavelength
3× 108 is the speed of light in a vacuum, but this travels much shower through media
frequency and wavelength are INVERTED! (as frequency increases, wavelength decreases)
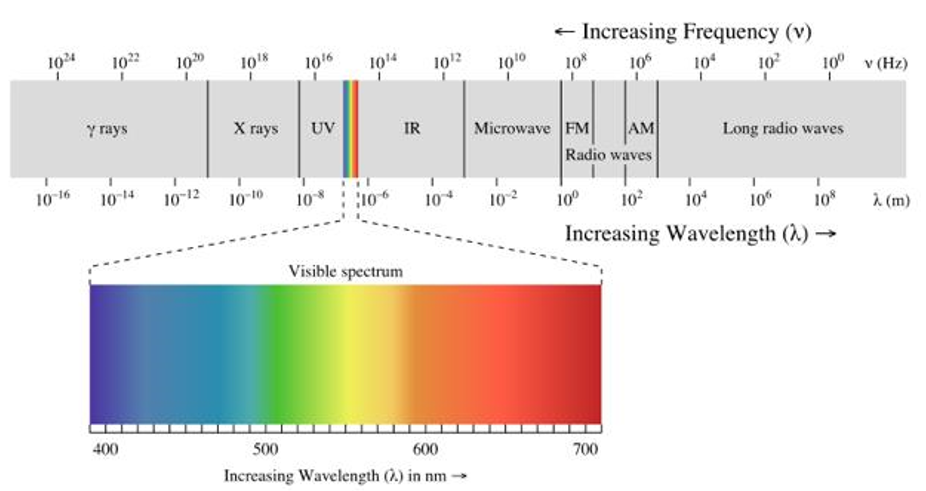
what is the wavelength of the visible spectrum
About 380 to 700 nanometres (nm)
red = largest wavelength
blue = shortest wavelength
why do we usually refer to wavelength rather than frequency when speaking about light
because frequency extremely high
around 5 million-fold greater than FM radio (which we refer to in frequency)
660 nm light has a frequency of 5 × 108 MHz
What is the relationship between wavelength (λ), frequency (Hz) and energy (in eV)
as wavelength increases, frequency and energy decrease
frequency is proportional to the energy of each photon!
E = hf = hc/λ
what changes with refractive media: wavelength, speed, frequency and energy
wavelength changes with refractive media
speed changes with refractive media
frequency does not change with refractive media
energy stays constant in refractive media as well (because frequency defines energy)
what is the dual nature of light?
Light shows both wave-like and particle-like behaviour.
light travels like a wave and interacts with matter like a particle!
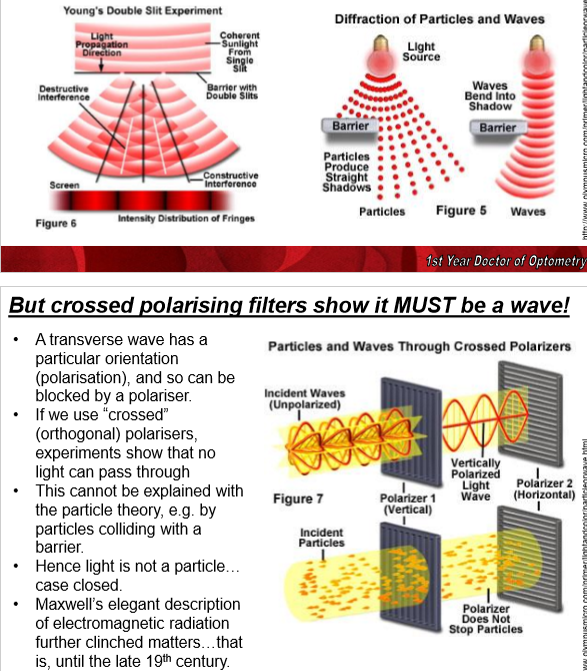
What experiments support light behaving as a wave?
Double-slit experiment
in the double-slit experiment, light passing through 2 slits forms an interference pattern of bright and dark pands (like ripples on a pond), which is a wavelike property (particles would produce straight shadows)
Crossed polarising filters
With crossed polarisers, the second polariser blocks the light because light has an orientation (polarisation), showing it behaves like a transverse wave. If they were particles, these would not be blocked!
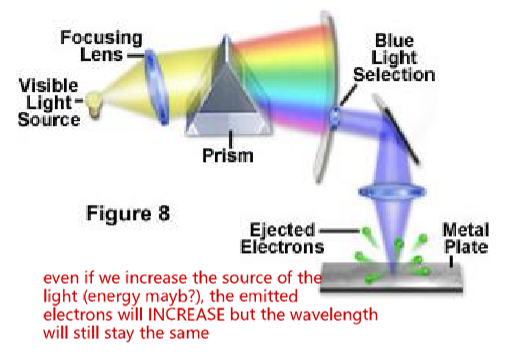
What experiment supports light as a particle?
The photoelectric effect
this experiment showed that electron ENERGY depended on wavelength (y), BUT not intensity of incident light
if we increase the intensity of the light, more electrons will be emitted but the energy and wavelength will stay constant
Blue or UV (higher f) light emits electrons (higher energy), but red did not
This is not consistent with wave behaviour of light
if light was acting as a wave, increasing its intensity should increase the energy delivered to each photon
energy calculation. Can you calculate energy with c and λ?
E = hf
h = Planck’s constant (6.626×1034 Js)
this calculation underlies the photoelectric effect!
E = hf = hc/λ
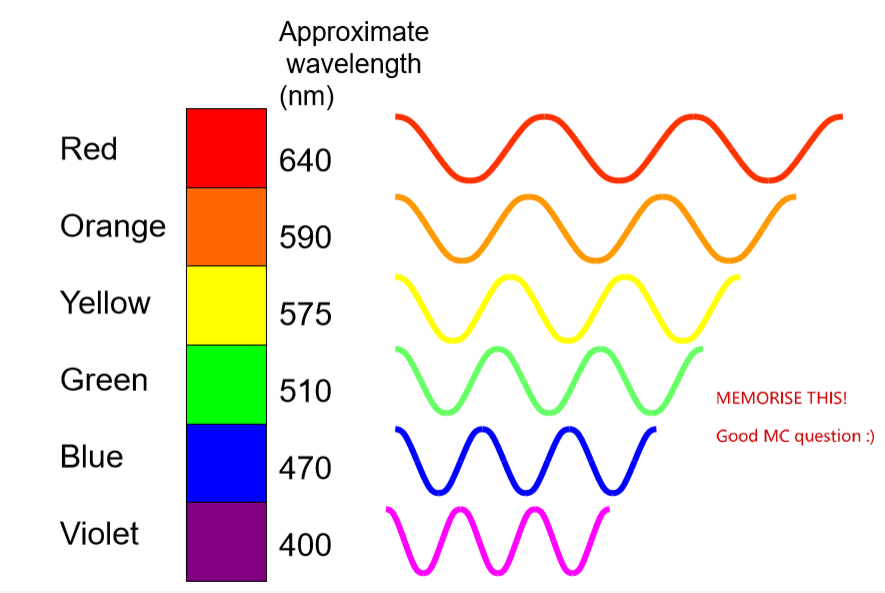
what are the approximate wavelength (nm) of colours of light
(red, orange, yellow, green, blue, violet)
Red: 640 nm
Orange: 590 nm
Yellow: 575 nm
Green: 510 nm
Blue: 470 nm
Violet: 400 nm
The sensitivity of our eyes to different wavelengths is described by what? What colours do our eyes have maximum sensitivity to?
The photopic sensitivity curve (cone-mediated, bright light)
We are most sensitive to colours in the middle of the visible spectrum (i.e. yellow-green)
Describe (and know how to draw) the photopic sensitivity curve and the scotopic sensitivity curve? What numbers do they peak at?
sensitivity to wavelengths during the day: photopic sensitivity curve (cone-mediated, bright light) - peaks (λmax) ~ 555 nm
L cones peak at ~565 nm and M cones at ~545 nm. S cones are not invited to the party…
sensitivity to wavelengths at night: scotopic sensitivity curve (rod-mediated, dim light) - peaks (λmax) ~ 507 nm (just rods)
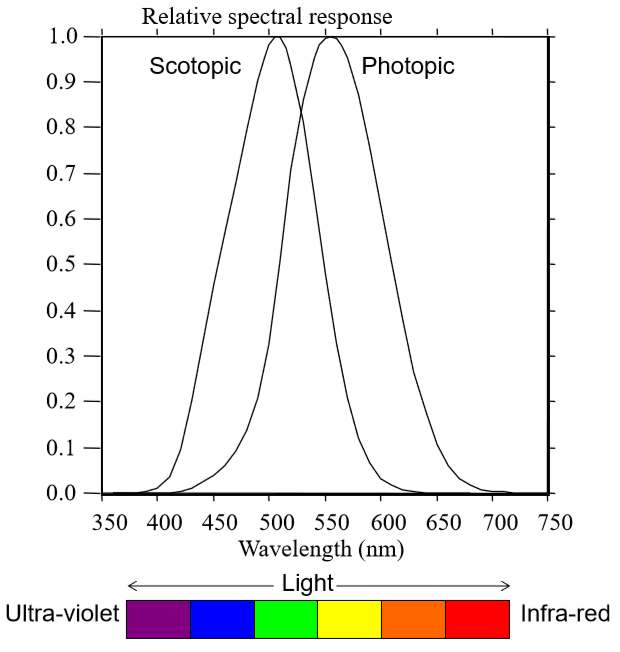
what does our peak photopic sensitivity coincide with?
Our peak photopic sensitivity falls near the peak of the solar emission spectrum.
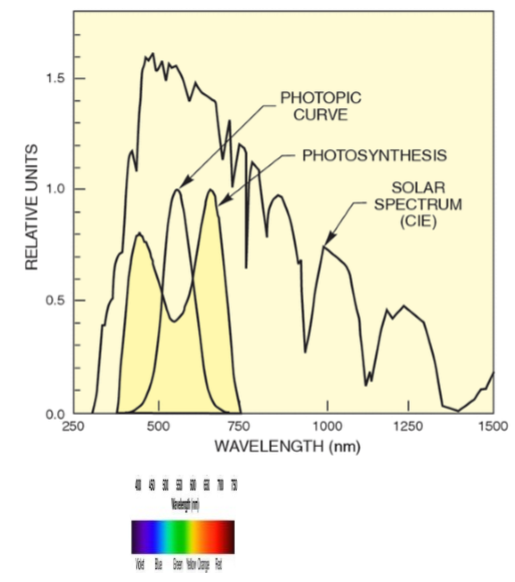
Why are tungsten bulbs hot?
Because most of it’s energy goes into the IR part of the spectrum (very inefficient)! Want most in our visible light spectrum
What is white light? What does it mean that very different light spectra can all appear white?
Light can appear white if all 3 cone types are stimulated (even in an unbalanced manner!) - although not always the case, but you do not need to be very precise.
Different mixes of wavelengths can still give the visual system a “white” signal
What are the 3 main parts of understanding vision? (reductionism)
Vision can be broken into three main parts:
the stimulus (light),
the interpreter (the brain)
the receptor (the eye)
How did eyes likely develop?
Eyes likely evolved step by step, from simple light-sensitive cells that detected light/dark, to pits that improved protection/reducing unwanted glare (limpet) then modified to allow directional sensitivity, and later to lens-containing image-forming eyes.
problem with the pit:
risk of becoming blocked!
Mutation —> adaptation: transparent membrane to cover pits
Mutation —> adaptation: membranes that are thicker in the middle (this focuses LIGHT intensity) * mutation: encapsulate the lens
vertebrate vs compou