Stem Cells and Ageing
1/44
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
Ageing
progressive loss of physiological integrity in the entire body which leads to impaired function and increased vulnerability to death
this deterioration is the primary risk factor for major human pathologies
Stem cell ageing
stem cells become exhausted and there is a loss of regeneration
What is a stem cell
a single cell that can replicate itself or differentiate into many cell types
Differentiation
the process by which cells become increasingly specialised to carry out specific functions in tissues and organs
Potency
the ability of stem cells to differentiate into specialised cell types.
potency varies between different types of stem cells
cells with the greatest potency can generate more cell types than those with lower potency
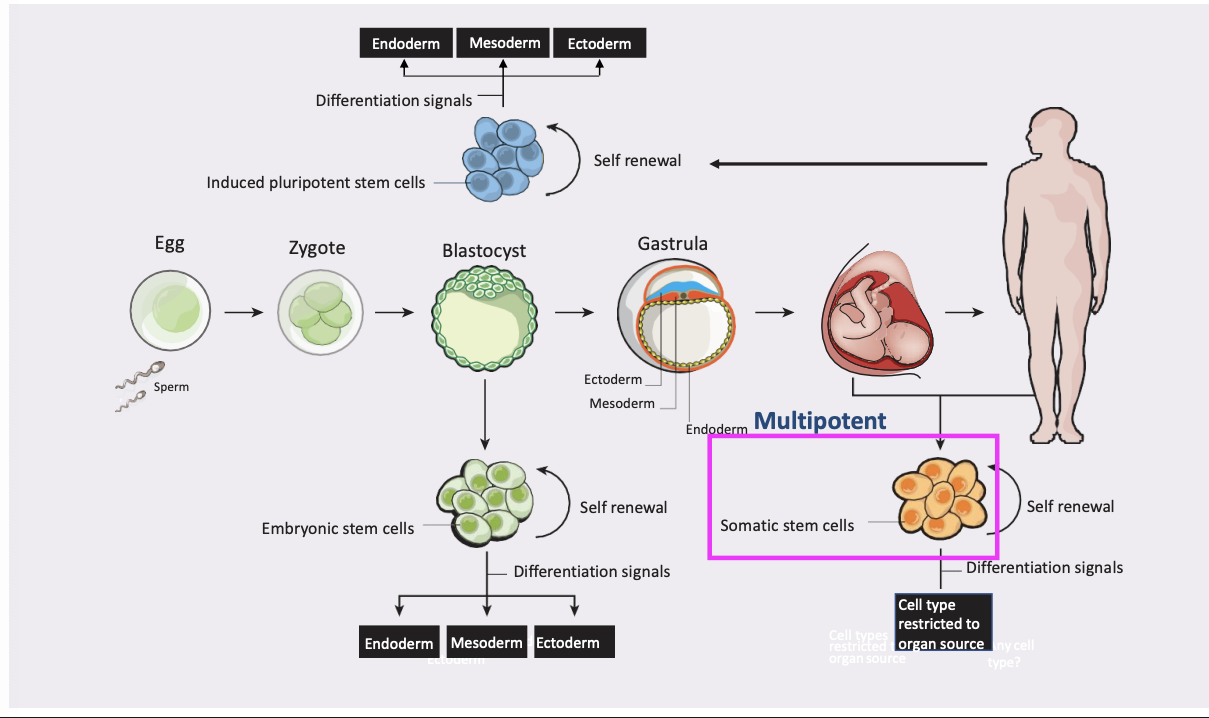
Totipotent cells
give rise to all cell types of the body as well as extra-embryonic cells (placenta)
can form a complete organism
Pluripotent stem cells
can give rise to all cell types of the body(but not the placenta)
CANNOT form a complete organism
Multipotent stem cells
multipotent stem cells develop into a limited number of cell types in a particular lineage
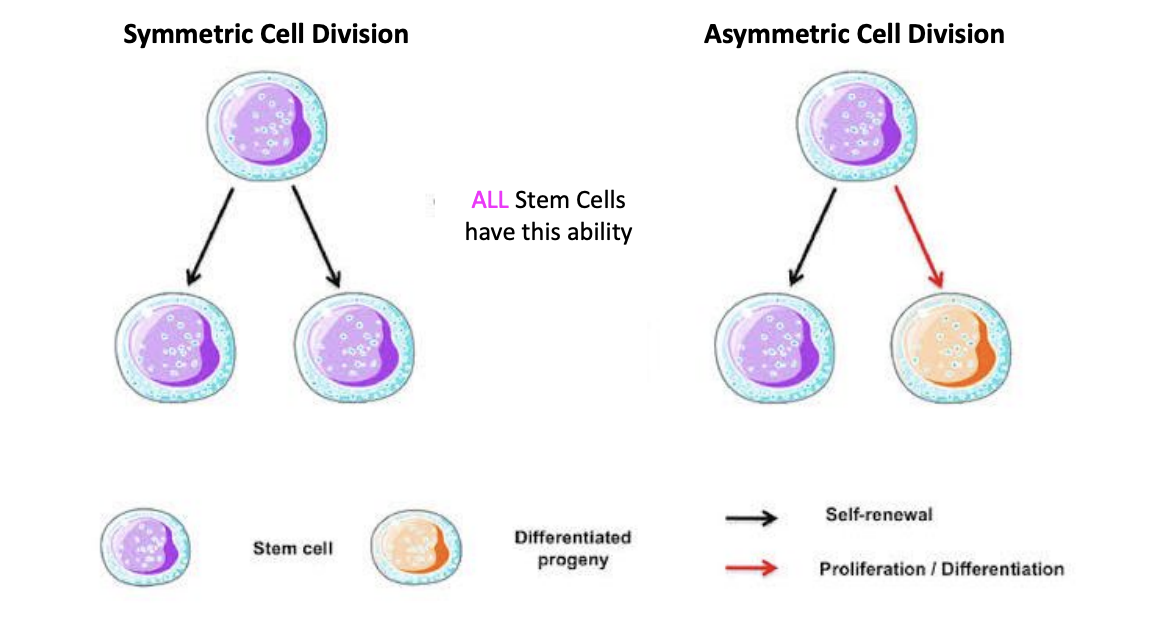
Symmetric and Asymmetric Cell Division
Symmetric
stem cell divides into two copies of itself
Asymmetric
stem cell divides into a copy of itself and a differentiated progeny
Embryonic Stem (ES) Cells
derived from the undifferentiated inner mass cells of a human embryo (extracted by using powerful scientific and medical tools)
Properties:
Pluripotent
Replicate indefinitely (immortal)
Embryonic stem cells as a tool
can become any cell type therefore if we control their differentiation we can create tissues for use in regenerative medicine (like forming new organs)
can direct stem cells to form damaged cells to replace them
useful for spinal cord injuries, type 1 diabetes, parkinson’s disease, amyotrophic lateral sclerosis, Alzheimer’s disease, stroke, burns, cancer, osteoarthritis, heart disease and etc
Ethical concerns of embryonic stem cell usage
to get embryonic cells you need a human embryo thus is this destroying a life and when does life begin?
Reprogramming somatic cells
proven in 2006 in mice and 2007 in humans
these cells are called induced pluripotent stem cells
Advantages of IPS cells
Properties:
pluripotent
replicate indefinitely
AND:
no embryo needed thus no ethical issues
there is potential for immuno-compatible regenrative medicine (patient specific IPS cells)
How human IPS cells are formed?
an adult cell is taken (normally skin or buccal cells)
add reprogramming factors which are genes active in ES cells
cell switches from adult cell to stem cell, changes in shape, gene expression and chromatic structure is observed
no resemble ES cells
can differentiate these cells into any other cell type
What can we do with IPS cells?
huge medical potential which is moving at such a rapid rate and it is impossible to review all applications it has been used for
at least 50 different diseases have been modelled with IPS cells
Cons of IPS cells
still a very new field thus still a lot to learn about the potential and use
can form tumours more than ES cells thus posing a major obstacle to stem-cell based regenerative medicine
genes we use to create iPSC also linked to cancer in one way or another
shown to illicit a greater immune response than ES cells
Stem cell continuum
Somatic/adult stem cells
Properties:
multipotent
replicate indefinitely (immortal)
AND:
found among differentiated cells in a tissue or organ
found in specific areas in each tissue (stem cell niche), various different niches all over the body
no embryo destroyed to make them - from adult tissues and can be patient specific
the stem cells that are responsible for stem cell ageing
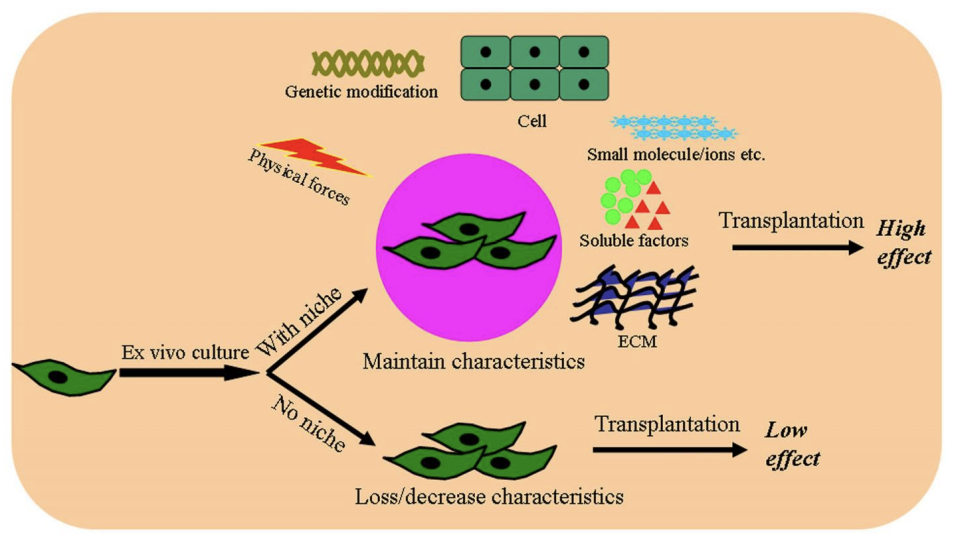
what else goes into a niche
the surrounding environment is crucial to their survival and ability to function
the niche is highly dynamic microenvironment adapts to physiological or diseased conditions
niche regulates how stem cells participate in tissue generation, maintenance and repair.
Prevents stem cell depletion - and stops overproduction of stem cells.
Adult stem cell niches - intestine
ISC - intestinal stem cell
present at the base of a glandular crypt
give rise to TA (transit-amplifying) cells - migrate upwards to replace cells on surface
from ISC in crypt to tip of villus is 3-5 days
Adult stem cell niches - bone marrow
bone marrow contains 2 stem cell niches
hematopoietic stem cells - give rise to all components of the blood and immune system (important part of a bone marrow transplant)
mesenchymal stem cells - make cartilage bone and fat
Bone marrow transplants
a well known form of stem cell therapy
hematopoietic stem are the major components in a bone marrow transplant - collect them from peripheral or cord blood stored umbilical cord blood
function: after high doses of chemotherapy or radiation, rescue the bone marrow damaged by treatment, restore immune function replace diseased or damaged marrow with new stem cells
Mesenchymal Stem Cells
found in bone marrow - also part of bone marrow transplants, cord blood or adipose tissue amongst others
can form a variety of tissue types - multipotent
modulate immune responses - mean less chance of rejection
can treat immune disease as well as used in tissue regeneration
Umbilical cord stem cells
a host of companies now sell storage of your new-born’s umbilical cord blood when needed can you can get blood out of cold storage
can be used to reconstitute bone marrow, to treat various blood cancers and forms of anaemia
is just as effective as bone marrow transplant but stem cells are younger
Limitations of umbilical cord stem cells
Not many stem cells in cord blood ( fewer than bone marrow) and not much material. One umbilical worth of blood often not sufficient for an adult
Irreplaceable
Only get haematopoietic stem cells – other stem cell types claimed by companies are unproven in therapeutic use.
Costly to store
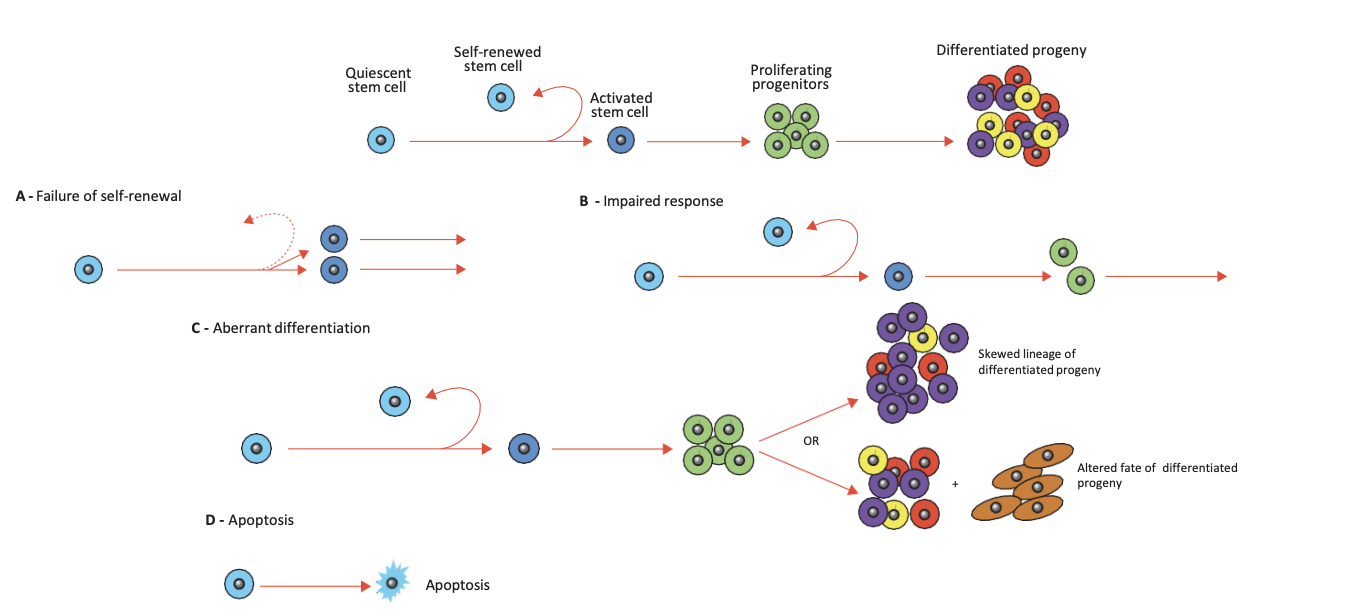
Stem Cell Hypothesis of Ageing
stem cells repair and replenish damaged tissues throughout life
these are adult stem cells in their various niches
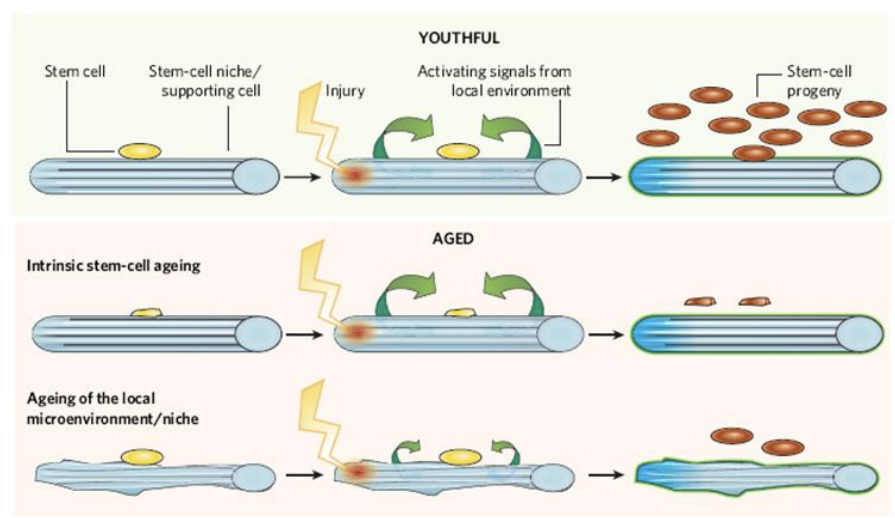
function of stem cells declines with age - undergo age-related damage
stem cells lose ability to self-renew and lose differentiation ability
stem cell theory of ageing postulates that aging is NOT a matter of the increase in deterioration of tissues - but a failure to replenish tissues due to a decreased number and decreased function of resident stem cells
Ageing of stem cells mirror ageing of other tissues –inflammatory responses, stress responses, and substantial alterations in the regulation of chromatin structure
Impact of ageing on adult stem cells
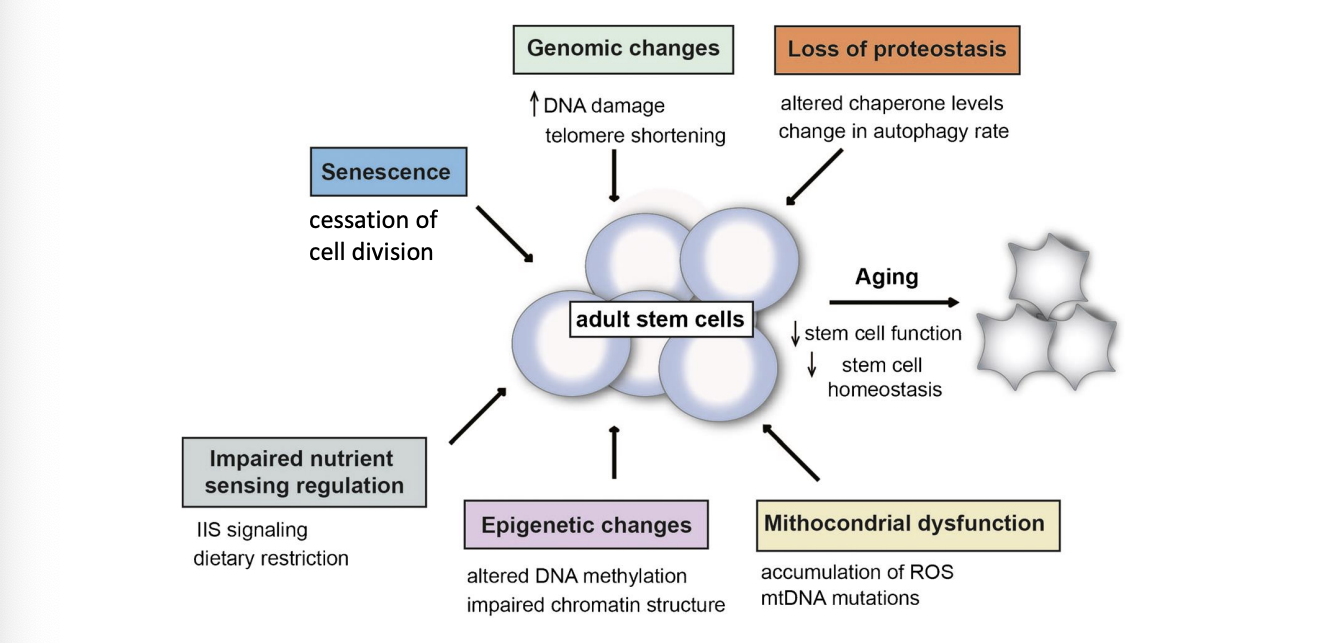
genetic and epigenetic changes in ageing stem cells
Many reports of unstable genomes in older stem cells. – DNA damage accumulates
Lines of evidence
Mice with defects in DNA damage repair display
some aspects of premature ageing
Enhancing DNA repair increases lifespan
Marker of DNA damage present in aged stem cells
Telomere shortening
Happens to all cells during DNA replication
Also happens to stem cells
Causes a stop in cell division- stop in self-renewal – stem cell failure
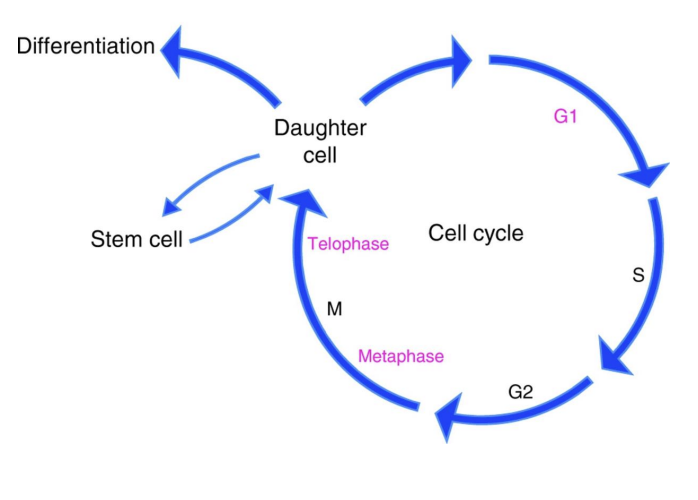
Cell cycle activity in stem cells with age
in aged mice HSCs have decreased cell cycle activity
Old HSCs undergo fewer cell divisions than young ones
Increase in factors that inhibit the cell cycle
ALSO - excessive proliferation is deleterious.
More the cells divide – the faster they age – this leads to premature exhaustion
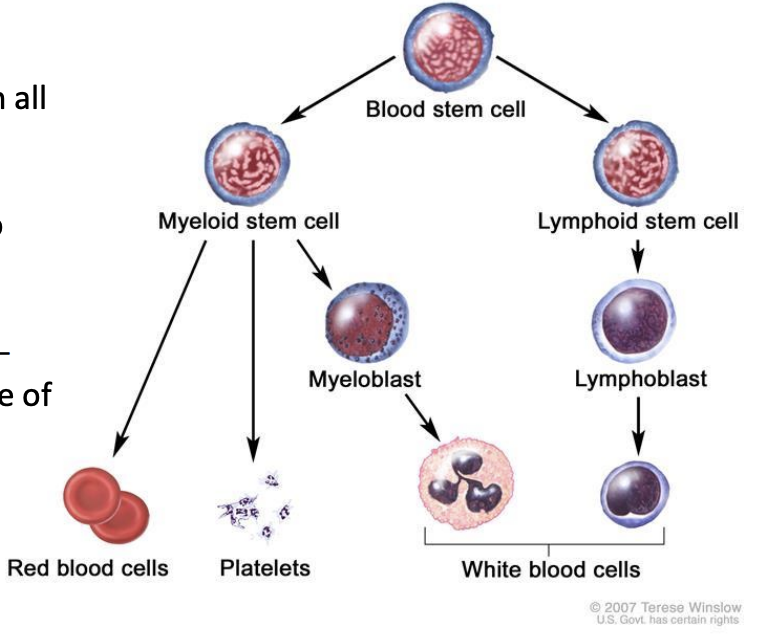
Ageing hematopoietic stem cells
Functional attrition of stem cells found in all adult stem cell compartments
Haematopoiesis declines with age due to exhausted stem cells.
Diminished production of immune cells— immuno-senescence.
Increased incidence of anaemia and myeloid malignancie
Ageing hematopoietic stem cells
Ageing Mesenchymal stem cells
isolating from bone marrow aspiration shows decline in MSC numbers with donor age.
older MSCs also show reduced proliferative capacity and reduced potential to form bone
enter cell cycle arrest (senescence)
Agening skin stem cells
Skin has different types of stem cells
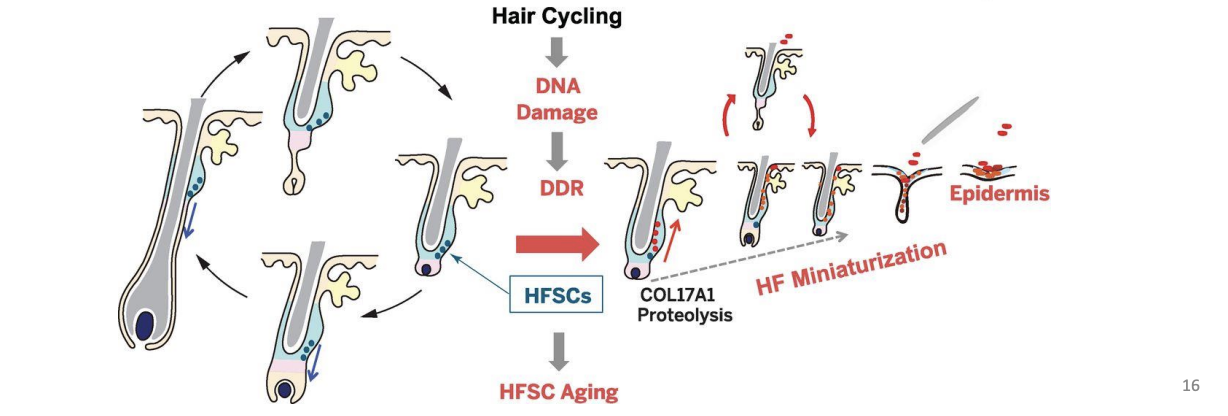
Hair follicle stem cells (HFSC) sustain hair growth
Epidermal stem cells replenish skin
Melanocyte stem cells generate pigment producing cells
Dramatic reduction in melanocyte stem cells numbers with age – visible effect in people
HFSC – DNA damage causes loss of stem cell and eventually loss of hair follicle entirely
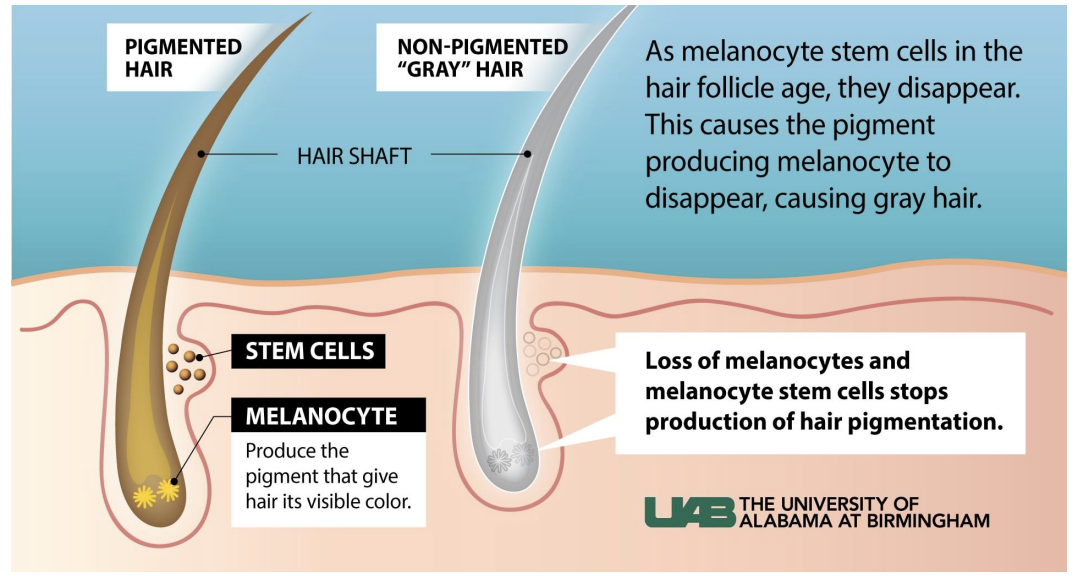
HSFC
Epidermal stem cells – reduced number with age - Impaired wound healing
HFSC – DNA damage causes loss of stem cell and eventually loss of hair follicle entirely
Ageing Skeletal Muscle Satellite (Stem) Cells
small population of quiescent stem cells
mobilised in response to injury
Age-related Changes:
Number of satellite cells decreases with age – loss of cell renewal
Regeneration potential on transplantation declines with age
Ability to replenish damaged muscles severely reduced
Recover from muscular injury is effected
Ageing neural stem cells
Adult neural stem cells (NSCs) present in some different brain regions - mediate local neurogenesis and brain functioning Ageing stem cells - decreased neurogenesis - advent of related ageing-associated disorders Reduction in neurons over time causes brain shrinkage - loss of efficacy
Hippocampus – major source of adult NSCs is crucial for memory and learning as well as ageing in general.
NB: neurons produced in hippocampus throughout adulthood, new studies prove this but still drop in quality of these new neurons with stem cell ageing
Ageing stem cells - decreased neurogenesis - advent of related ageing-associated disorders
Reduction in neurons over time causes brain shrinkage - loss of efficacy
Stem cell transplants as anti-ageing treatment
ageing stem cells causes deterioration of the body - transplanting young stem cells to cause recovery
progeria - abnormal rapid ageing, loss of muscle mass, difficulty moving, trembling
systemic effects cause by secreted factors from transplanted stem cells
Stem cell transplants - curing blindness
age-related macular degeneration: common form of blindness
deterioration of central part retina - responsible for focusing vision incurable
using embryonic stem cells - differentiated them into RPE (retinal pigment epithelium) - embed onto scaffold transplant them onto the retina - restores vision
in future will use IPS cells and remove ethically quadary
Stem cell transplants - slow brain ageing
neural stem cells injected (replacing those in hippocampus) which makes new neurons ageing slowed
NSCs release molecules called miRNAs - helped maintain a youthful status lost over time and with age
currently working on investigating in humans
Rejuvenating stem cells
Instead of replacing ageing stem cells can we rejuvenate them?
Hints from iPSC – have reprogrammed adult cells – give clues about resetting chronological age
Has been found that during iPSC reprogramming can reactivate telomerase (enzyme that extends telomeres)
BUT - emphasis of research in the field of reprogramming is not on reversing ageing. Reversal of the differentiation – attaining pluripotency is goal
In addition – we can make cells pluripotent – this is NOT reversing ageing – might be dangerous
Parabiosis
two animals share a common bloodstream
Current parabiosis research focuses on if adult stem cell rejuvenation occurs in a young environment
Old mice stronger, smarter and healthier. It even makes their fur shinier – Stem cells are rejuvenated
Question remains if it is de -ageing – or just restoring function to damaged tissues ?
ALSO – Paired young mice AGE. Stem cells have an aged molecular and functional state.
Systemic environment determines functional age – cost to this procedure