NMR
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
1- Drug chemical shift and exchangeability
What does NMR reveal about drug products?
Which atoms
How many atoms
What are each atom connected to
Purity
What happens to proton spins in the absence of a magnetic field vs when an external magnetic field (B₀) is applied?
They spin with randomly oriented axes and do not precess at a defined frequency.
They begin to precess around the direction of B₀, tracing a cone‑shaped path.
What is the relationship between precessional frequency (ν) and magnetic field strength (B₀)?
ν ∝ B0
What is the Larmor frequency.
Frequency at which protons precess in a magnetic field
Why is precessional frequency important in MRI?
MRI uses radio frequency pulses tuned to the Larmor frequency to excite protons and generate measurable signals.
What physical interaction causes proton precession in a magnetic field?
The interaction between the proton’s magnetic moment and the external magnetic field B₀.
What happens to proton spin states when B₀ = 0 vs when B₀ is applied?
Spins are randomly oriented with no energy difference between states and no population imbalance.
They split into Nα (low energy) and Nβ (high energy) states.
What determines the energy gap between Nα and Nβ?
ΔE = hν
The gap increases with magnetic field strength B₀.
What is the Boltzmann relationship between Nα and Nβ?
At room temperature, Nα ≈ Nβ.
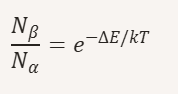
Why does increasing B₀ improve NMR/MRI sensitivity?
Larger B₀ → larger ΔE → greater population difference → more protons absorb RF → stronger signal.
Which spin state is lower in energy: Nα or Nβ?
Nα (aligned with the magnetic field).
What does the FID represent in NMR?
The Free Induction Decay: ¹H signal intensity vs time after excitation.
It is a complex time‑domain signal containing overlapping frequencies, so difficult to interpret directly
What does the Fourier Transform do in FT‑NMR?
Converts the time‑domain FID into a frequency‑domain spectrum.
Why is the frequency‑domain NMR spectrum easier to interpret?
It displays distinct peaks at chemical shift frequencies corresponding to different proton environments.
Why is FT‑NMR preferred over continuous‑wave NMR?
It collects all frequencies at once, is faster, and improves signal‑to‑noise through averaging.
What type of data does the Fourier Transform produce from the FID?
A ¹H NMR spectrum showing signal intensity vs frequency (chemical shift).
What does the x‑axis of a proton NMR spectrum represent?
Chemical shift δ (ppm), running from downfield (deshielded) to upfield (shielded).
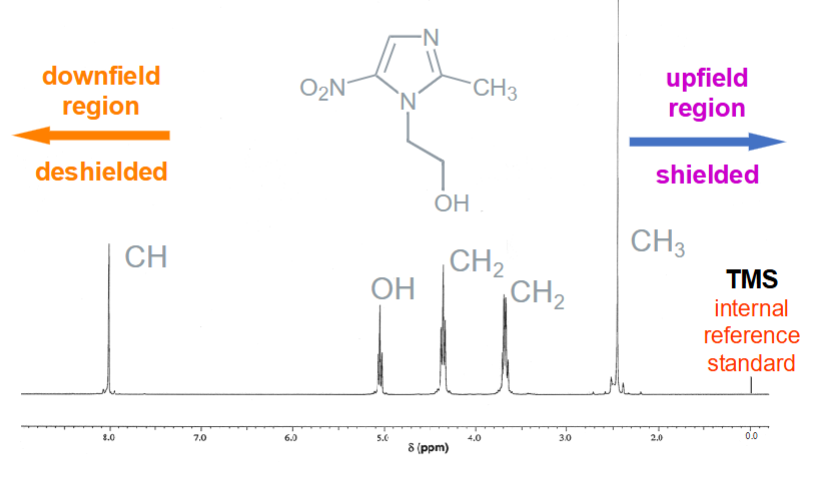
What types of protons appear downfield (left side, high ppm) vs upfield (right side, low ppm)?
Deshielded protons near electron egative atoms or electron‑withdrawing groups.
Shielded protons in electron‑rich environments, typically alkyl groups.
What is the chemical shift of the internal reference standard TMS?
0 ppm, used as the reference for all ¹H NMR chemical shifts.
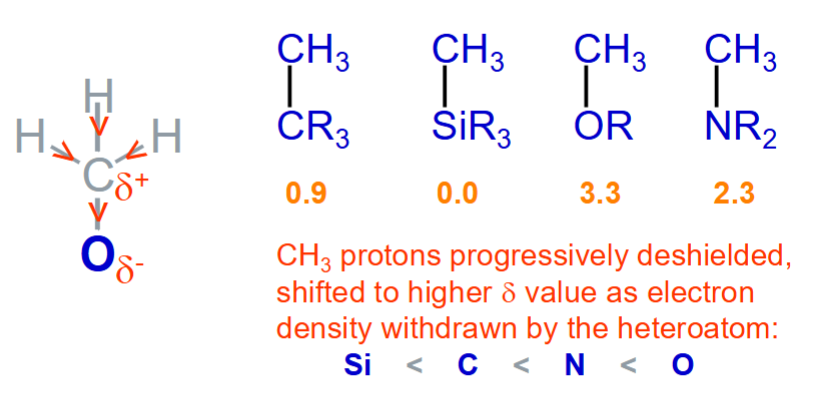
How does an electron‑withdrawing group (e.g., NO₂) affect nearby proton chemical shifts vs an increased electron density around a proton affect its NMR signal?
It deshields nearby protons, shifting their signals downfield.
It shields the proton, shifting its signal upfield.
What does chemical shift represent in NMR?
The position of a ¹H or group of protons in the NMR region relative to a reference compound (TMS at 0 ppm).
What determines a proton’s resonance frequency in NMR?
Its magnetic environment, which affects how strongly it feels the external field
What causes shielding and deshieding in NMR?
Shieding - local electron density around a proton reduces the effective magnetic field it experiences.
Appears upfield (lower δ values, closer to 0 ppm).
Electronegative atoms or electron‑withdrawing groups pull electron density away from the proton.
Appears downfield (higher δ values, further from TMS).
What is the shielding/deshielding trend for common X–H bonds?
C–H (most shielded) > N–H > O–H (most deshielded).
What is the delta scale?
The scale is relative so the 1H peaks in aspirin appear
at the same δ value on all NMR machines but better resolution at higher operating frequency
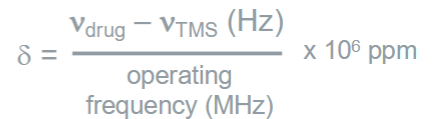
How many Hz correspond ppm on a MHz spectrometer?

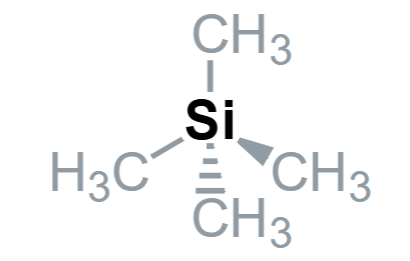
What are the advantages of TMS having 12 identical protons, inert, volatile, soluble and absorption?
12 identical protons - give only one absorption in the 1H NMR
Inert - can be added ~ 0.5% to drug samples
Volatile - evaporate off to recover drug sample
Absorption - occurs at 0 ppm away from most other protons
Soluble - In organic solvents (CDCl3, d6-DMSO)
Alternatively, use solvent reference peak
Why are deuterated solvents needed?
Ordinary solvents contain ¹H, which produce huge proton signals and swamp the sample’s peaks.
Deuterated solvents replace ¹H with ²H, which is invisible in ¹H NMR as it resonates at a different frequency
They’re not 100% deuterated, so small amounts of residual ¹H produce peaks.

What are the residual ¹H peak of different solvents?
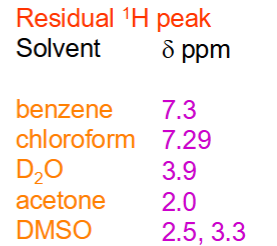
What causes the 3.3 ppm peak in DMSO‑d₆?
Moisture (HOD) in the sample.
What additional function do deuterated solvents provide besides reducing ¹H signals?
They provide a lock signal to stabilise the magnetic field during acquisition.
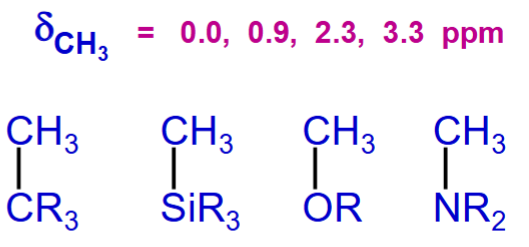
Match the given chemical shifts for the methyl protons with each functional group where R is the same each time
What is the D2O (deuterium) shake?
To detect the absence or presence of labile protons
-OH, -NH, -SH, -CO2H, -NH2, -CONH2
Labile protons exchange with other protons or deuterium
Labile protons are attached to atoms more electronegative than carbon and therefore display acidity
What is the slowest functional group to exchange with D2O and how would you make it faster?
-NH
Add acid/base catalysts - addition of deuterium Cl- to form or deuteroxide can speed up the exchange.
Increase temperature - heating the sample increases the exchange, encouraging the breaking of intramolecular H bonds that shield the amide proton.
Add denaturing agents - using high concs of guanidinium chloride (GdnHCl) or urea will denature proteins, exposing buried amide protons to the solvent.
Use misible solvents - utilizing co-solvents like DMSO-d6 can help disrupt hydrogen bonds.
Increase concentration - ensuring a large excess of
drives the equilibrium forward
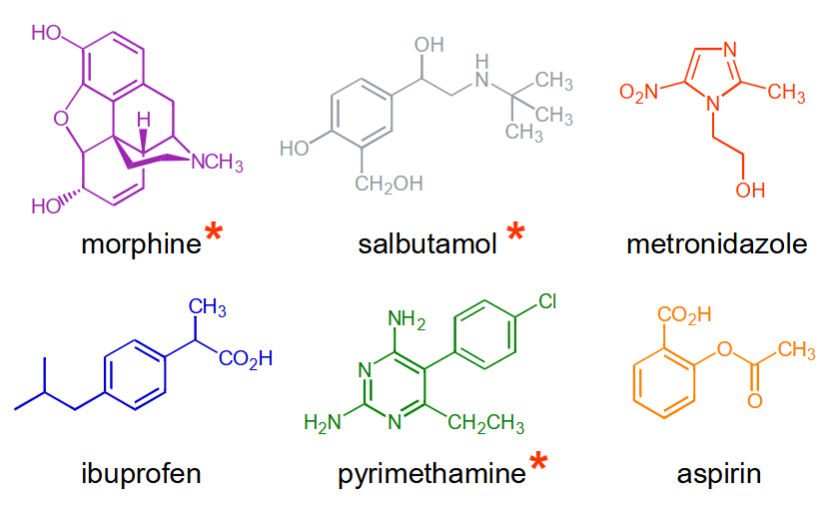
How many contain an even number of D2O exchangeable protons?
2 - Integration and multiplicity