Chapter 8- Water and Minerals
1/143
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
144 Terms
Minerals
Naturally occurring, inorganic, homogeneous substances
Minerals are categorized under
micro nutrients
Major minerals are needed in how much
needed greater than 100 milligrams/day
Trace minerals
needed less than 100 milligrams.day
Shortfall nutrients
•Calcium, potassium, magnesium, and iron (important but underconsumed)
_ is the most indispensable nutrient
water
Water is ___ % of an adult’s weight
60%
•Water content of brain and muscle is ___ %
75 to 80%
Water content of bones is ___ %
25%
Water is a __for minerals, vitamins, amino acids, glucose, and other small molecules
solvent
Water is active in many ___
chemical reaction
Water cleanses ___ of wastes
tissues and blood
Water acts as ___ around joints and organs
acts as a lubricant and shock absorber
Water is a primary component in
amniotic fluid: protects fetus during growth
Water aids in
regulating the body’s temperature
Why is water indispensable?
It has many functions
*Carries nutrients
•Solvent for minerals, vitamins, amino acids, glucose, and other small molecules
•Active in many chemical reactions
•Cleanses the tissues and blood of wastes
•Acts as a lubricant and shock absorber around joints and organs
•Primary component in amniotic fluid
•Aids in regulating the body’s temperature
Water Balance/Equillibrium is influenced by
•Dehydration
•Water intoxication
Body water varies
weight/pounds
____ govern water intake
Thirst and satiety
•Hypothalamus signals
the pituitary gland that instructs the kidneys
•Hypothalamus can prevent
the loss of fluids .
Dehydration consequences
building of body heat
sweating halts
body can go in distress
Symptoms of dehydration
headaches, fatigue, forgetfulness, confusion,
Caffeine is a ___
Diuretic
Diuretic
increases urine excretion
T/F: Water intoxication is unrare
False
Consequences of water intoxication
Diluted blood: impedes blood from functioning, death
What is considered mild dehydration?
(Loss of <5% Body Weight)
What is considered severe dehydration
(Loss of >5% Body Weight)
Effects of mild dehydration
Thirst |
Sudden weight loss |
Dry, cool skin |
Dry mouth, throat, body linings |
Rapid pulse; low blood pressure |
Lack of energy; weakness |
Impaired kidney function |
Reduced quantity of urine; concentrated, dark yellow or amber-colored urine |
Headache; reduced mental clarity |
Diminished muscular work and athletic performance |
Fever or increased internal temperature |
Fainting and delirium
Effects of Severe Dehydration
Pale or shriveled skin |
Bluish lips and fingertips |
Confusion; disorientation |
Rapid, shallow breathing |
Weak, rapid, irregular pulse |
Thickening of blood |
Scant urine; brown-colored urine |
Shock; seizures |
Coma; death
Effects of Chronic Low Fluid Intake
Cardiac arrest (heart attack) and other heart problems |
Constipation |
Dental disease |
Gallstones |
Glaucoma (elevated pressure in the eye) |
Hypertension |
Kidney stones |
Pregnancy/childbirth problems |
Stroke |
Urinary tract infections |
Water needs vary based on
eating habits, temp of environment, activity levels
DRI for water:
Men: 13 cups/day
Women: 9 cups/day
metabolic water
Water generated in the tissues during the chemical breakdown of the energy-yielding nutrients in foods
Factors that increase fluid needs
•Alcohol consumption
•Cold weather
•Dietary fiber
•Diseases that disturb water balance, such as diabetes and kidney diseases
•Forced-air environments, such as airplanes and sealed buildings
•Heated environments
•High altitude
•Hot weather, high humidity
•Increased protein, salt, or sugar intakes
•Ketosis
•Medications (diuretics)
•Physical activity
•Pregnancy and breastfeeding (see Chapter 13)
•Prolonged diarrhea, vomiting, or fever
•Surgery, blood loss, or burns
•Very young or old age
Water in Foods and Beverages and Percentages
Percentage | Foods and Beverages |
100% | Water, diet soft drinks, seltzer (unflavored), plain tea |
95–99% | Sugar-free gelatin dessert, clear broth, Chinese cabbage, celery, cucumber, lettuce, summer squash, black coffee |
90–94% | Sports drinks, grapefruit, fresh strawberries, broccoli, tomatoes |
80–89% | Sugar-sweetened soft drinks, milk, yogurt, egg white, fruit juices, low-fat cottage cheese, cooked oatmeal, fresh apple, carrot |
60–79% | Low-calorie mayonnaise, instant pudding, banana, shrimp, lean steak, pork chop, baked potato, cooked rice |
40–59% | Diet margarine, sausage, chicken, macaroni and cheese |
20–39% | Bread, cake, cheddar cheese, bagel |
10–19% | Butter, margarine, regular mayonnaise |
5–9% | Peanut butter, popcorn |
1–4% | Ready-to-eat cereals, pretzels |
0% | Cooking oils, meat fats, shortening, white sugar |
•All beverages meet the body’s fluid needs except
•alcohol
All drinking water originates from
surface water or groundwater
•Types of water
Hard water and soft water
•Hard water contains
calcium and magnesium
•Soft water contains
sodium which may aggravate hypertension
•Bottled water impacts the environment how?
Bottle water generates increased waste which can derive from the same water as tap water
Public water systems points
•Risk of contamination increases
•Purifying equipment can remove many hazards
Water follows __
salt and electrolytes
•Fluid and electrolyte balance:•Rapid fluid shift causes
an imbalance
•Acid-base balance:When dissolved in watery body fluids, some of the major minerals
give rise to acids (H, or hydrogen, ions) and others to bases (OH ions)
The body’s proteins and some of its mineral salts help prevent changes in the acid-base balance of its fluids by serving as
buffers
buffers
molecules that gather up or release H ions as needed to maintain the correct pH
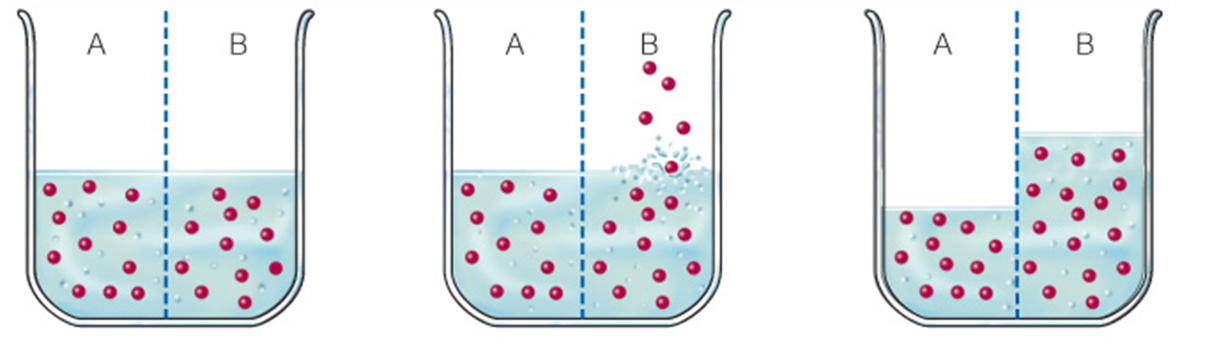
How Electrolytes Govern Water Flow
With equal numbers of dissolved particles on both sides of a water-permeable divider, water levels remain equal.
Now additional particles are added to increase the concentration on side B. Particles cannot flow across the divider. In the case of a cell, the divider (cell membrane) partitions fluids inside and outside the cell
Water can flow both ways across the divider but tends to move from side A to side B, where the concentration of dissolved particles is greater. The volume of water increases on side B, and the particle concentrations on sides A and B become equal
Major Minerals need requirement(also called macrominerals)
•The need for each of these is greater than 100 milligrams per day, in some cases far greater
Types of Major Minerals
•Calcium
•Chloride
•Magnesium
•Phosphorus
•Potassium
•Sodium
•Sulfate
Which mineral is the most abundant in the body?
Calcium
Calcium is stored in
teeth and bones
Bones are
an active living tissue
Blood travel in
in capillaries throughout the bone, bringing nutrients to the cells that maintain the bone’s structure and carrying away waste materials from those cells
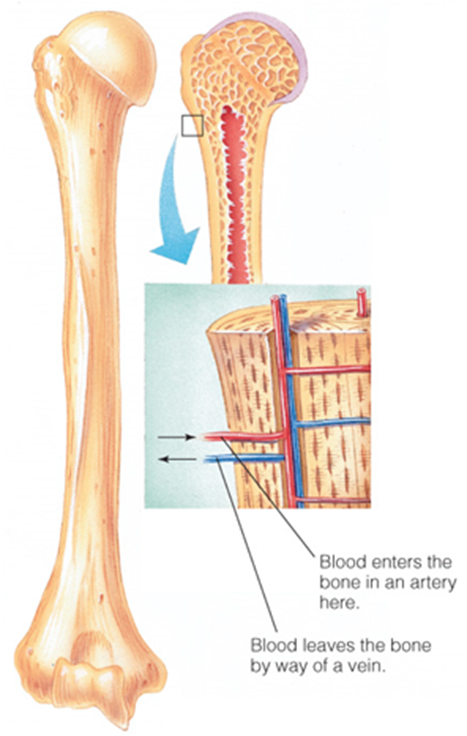
Blood in the bones ___ as instructed by hormones.
picks up and deposits minerals
Bone derives its structural strength from
the lacy network of crystals that lie along its lines of stress.
. If minerals are withdrawn to cover deficits elsewhere in the body, the bone will
will grow weak and ultimately will bend or crumble.
Hydroxyapatite
•The chief crystal of bone and teeth, formed from calcium and phosphorus
hydroxyapatite crystals invade
the collagen and gradually lend more and more rigidity to a youngster’s maturing bones until they are able to support the weight they will have to carry.
hydroxyapatite crystals form on a collagen matrix to
create the dentin that gives strength to the teeth
The inner layer of dentin is
bonelike material that forms on a protein (collagen) matrix.
. The outer layer of enamel is harder than
bone
One percent of the body’s calcium is in
in the fluids
•Roles of Calcium
•Regulates the transport of ions
•Helps maintain normal blood pressure
•Plays an essential role in the clotting of blood
•Is essential for muscle contraction
•Activates cellular enzymes
___ is tightly controlled
blood calcium
The crystals of enamel may become even harder when exposed to
to the trace mineral fluoride.
•Organ systems that respond to an increased need
•Intestines, kidneys, and bones
Skeleton serves as a
calcium bank
Intestinal lining can substantially increase its absorption when
when the body needs more calcium
•Bone loss
Inevitable consequence of
Aging
Peak bone mass age
12-30
At what age does bone loss occur
35+
Insufficient calcium savings cause
Osteopenia, Osteoporosis
Osteopenia:
A condition of low bone mass that often progresses to osteoporosis
Osteoporosis
Reduction of the bone mass of older people in which the bones become porous and fragile
DRI calcium
D R I: Adults: 1,000 milligrams/day (men and women, 19–50 years; men, 51–70 years)
1,200 milligrams/day (women, 51–70 years; men and women, >70 years)
Tolerable Upper Intake Level of Calcium
Adults: 2,500 milligrams/day (19–50 years) and 2,000 milligrams/day (>50 years)
Chief Functions of Calcium
: Mineralization of bones and teeth; muscle contraction and relaxation, nerve functioning, blood clotting
Deficiency of calcium
Stunted growth and weak bones in children; bone loss (osteoporosis) in adults
Toxicity of calcium
Elevated blood calcium; constipation; interference with absorption of other minerals; increased risk of kidney stone formation
Which mineral is the second most abundant
Phosphorus
•Roles of Phosphorus
•Phosphorous salts are critical buffers
•Essential for growth and renewal of tissues
•Phosphorous compounds carry, store, and release energy during metabolism of energy nutrients
•Phosphorous compounds assist in extracting energy from nutrients
•Phospholipids are the principal components of cell membranes
•Present in some proteins
Are deficiences of phosphorus likely?
No
•Excess phosphorus in the blood are indicators of
heart and kidney diseases
DRI of Phosphorus
D R I: Adults: 700 milligrams/day
UL for phosphorus
Tolerable Upper Intake Level: Adults (19–70 years): 4,000 milligrams/day
Chief Functions of Phosphorus
: Mineralization of bones and teeth; part of phospholipids, important in genetic material, energy metabolism, and buffering systems
Deficiency of Phosphorus
: Muscular weakness, bone pain
Toxicity of phosphorus
Calcification of soft tissues, particularly the kidneys
Magnesium is in
•Over half of it is in the bones
•Roles of Magnesium
•Cofactor for hundreds of enzymes
•Needed for the release and use of energy from the energy-yielding nutrients
•Part of the cellular protein-making machinery
•Critical to nerve transmission, muscle contraction, and heart function
•Works with calcium for proper functioning of the muscles
•Deficiency of Magnesium
•Causes diabetes, heart failure, hypertension, inflammation, and stroke
•Toxicity of Magnesium
•Causes diabetes, heart failure, hypertension, inflammation, and stroke
•Symptoms include diarrhea, acid-base imbalance, and dehydration
Magnesium DRI
D R I: Men (19–30 years): 400 milligrams/day and Women (19–30 years): 310 milligrams/day
Tolerable Upper Intake Level: Adults:
350 milligrams/day
Sodium Roles
•Major part of fluid and electrolyte balance
•Helps maintain acid-base balance
•Deficiency of sodium
•Results in hyponatremia
•The condition of having too little sodium in the blood
•Dietary Guidelines for Americans, 2015–2020 (Sodium)
▶Consume less than 2,300 milligrams per day of sodium (ages 14 years and older)
▶Further reductions to 1,500 milligrams of sodium may produce greater benefits in people with hypertension or prehypertension