HTHSCI 3PA2A - Module: Adaptive Immunity
1/106
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
107 Terms
The Immune System
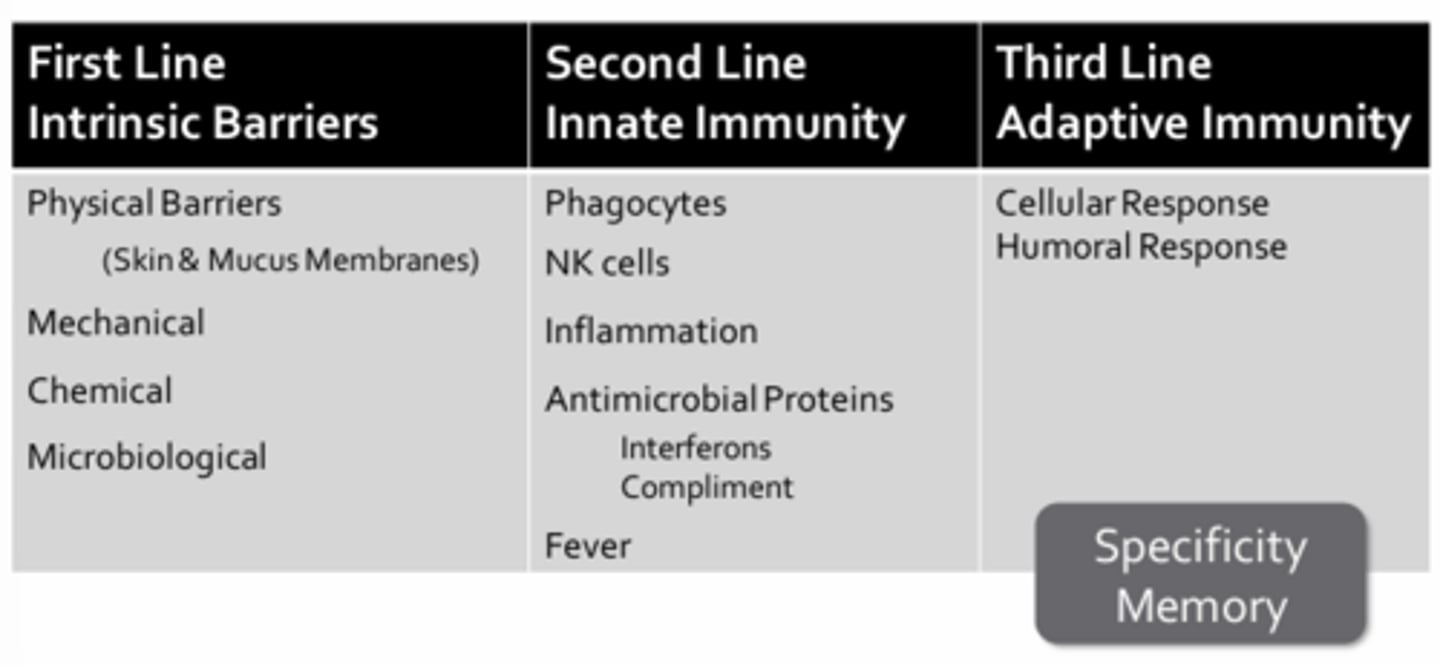
First Line of Defense:
- The physical barriers of the skin and mucus membranes
- Barriers of the mechanical, chemical, or microbiological kind
Second Line of Defense:
- The innate immune system
- Protection against a broad spectrum of pathogens through the use of surface receptors on phagocytic cells that recognize evolutionary conserved patterns unique to pathogens (ex. LPS = sugary lipids found in the outer cell wall or gram negative bacteria)
- The innate immune system consists of phagocytes, natural killer cells, the inflammatory response, antimicrobial proteins (ex. interferons, complement), and fever
Third Line of Defense:
- Adaptive immune response
- To eliminate pathogens that may have circumvented or overwhelmed the first two lines of defense
- To confer protection from new and emergent strains of pathogens
- A cellular arm involving targeted killing of infected or abnormal cells
- A humoral arm involving the production of soluble immunoglobulins (antibodies) that confer protection from specific pathogens through a variety of effector functions
Mechanical Barriers
- Expulsive forces (coughing, sneezing, defecation, urination) help to rid the body of pathogens before they can attach and breach the mucus membranes
- Ciliary beating helps to sweep pathogens from the upper and lower airways
- Tight junctions in epithelium prevent pathogens from slipping between cells to enter underlying host tissue
Chemical Barriers
- Low pH barriers (stomach, vaginal fluids, sebaceous fatty acids) prevent pathogenic bacterial colonization
- Proteolytic enzymes (lysozymes, pepsin in gut) breakdown pathogenic components
Microbiological Barriers
Commensal flora compete with pathogens for resources (space and nutrients)
Phagocytes
- Mainly consist of tissue resident and wandering macrophages
- Ingested pathogens occupy a phagosome which is then fused with a lysosome to produce a phagolysosome where digestion (breakdown) of the pathogen occurs
NK Cells
- Natural killer cells
- Do not phagocytose pathogens, rather they secrete toxic chemical or induce apoptosis in target cells
Inflammation
- A localized response to infection involving vasodilation, increased vascular permeability, and mobilization of phagocytes involving: Leukocytosis, margination, diapedesis, and chemotaxis
Antimicrobial Proteins
- Interferons released by infected cells help to prevent infection of neighboring cells by inducing neighboring cells to produce anti-viral proteins that block protein synthesis and degrade viral RNA
- Complement proteins help to enhance inflammation, promote phagocytosis, and cause cell lysis (membrane attack complex)
Fever
- A systemic response to widespread infection, resulting in an elevated body temp
- Initiated by release of pyrogens which include bacterial toxins and/or components (antigen-antibody complexes or substances released by phagocytes)
Adaptive Immunity
The hallmarks of the adaptive immune response include:
Specificity (self vs non-self):
- The ability o mount a response to a particular pathogen or foreign substance while being able to discriminate between self vs. non-self antigens
- The consequence of this specificity is that the B-cell may only be capable of mounting a response to one particular strain of pathogen without recognizing any of a number of closely related pathogens
Memory:
- The ability to recall past exposure to specific pathogens and provides the adaptive immune system with the ability to mount an even stronger response to a pathogen upon repeat exposure
Immunologic Memory
- Provides the basis for the protection offered by vaccination
- In a vaccinated individual, the immune response may be so quick and robust that they may not even exhibit signs or symptoms of infection
Innate vs. Adaptive Immunity

Innate Components
Broad Specificity:
- Pathogen-associated molecular patterns (PAMPs) are often located on structure that are essential for pathogen survival and accessible to host pattern-recognition receptors (PRRs) such as the Toll-like receptors (TLRs) which are present on phagocytes
- TLRs recognize PAMPs that are common to broad classes or organisms (ex. TLR-4 recognizes LPS which is a component of the outer cell wall of all gram negative bacteria)
Limited Repertoire:
- PRRs are encoded int he host germ line DNA, limiting the absolute number of PAMPs for which PRRs are produced
- If pathogens modify their PAMPs (ex. a bacterium changes the structure of its LPS), then the host PRRs (ex. TLR-4) will not mount a response
Immediate Response:
- Components are preformed and ready to respond to pathogens immediately upon binding PRRs
- The response may include increased phagocytosis, complement activation, and inflammation
Lacks Memory:
- Does not remember previous encounters with pathogens
- It will respond to infection with the same pathogen in the same manner it has done so in the past (i.e. same time-course and intensity)
Adaptive Components
Narrow Specificity:
- Both B-cell and T-cell receptors recognize very specific antigenic determinants (8-10 amino acids) that may be specific for only one particular strain of pathogen
- These antigenic determinates can be any protein on the pathogen and may not be essential for pathogen survival
Vast Repertoire:
- B-cell and T-cell receptors which recognize antigenic determinates are produced through somatic recombination (mixing, matching, and recombining) of host gene segments
- Results int he generation of T-cell and B-cell receptors that can detect virtually any antigenic determinant
Slow Response:
- Innated by antigen-presenting cells (APC) which travel from the site of infection to a nearby lymph node where immunocompetent cells of the adaptive immune system reside
- Process involves antigen presentation, differentiation and clonal expansion of T-cell and/or B0cells in a cellular or humoral response
- Process may take 3-5 days following initial infection
Memory:
- Hallmark of adaptive response
- Conferred by a subset of memory cells that are produced during each adaptive response
- These memory cells ensure that there is a large reservoir of differentiated cells ready to quell infections mediated by previously encountered pathogens
Origin of the Adaptive Response
Initiation of an adaptive response occurs in the secondary lymphoid tissues which include:
- Spleen
- MALT (mucosal associated lymphoid tissue)
- Lymph nodes
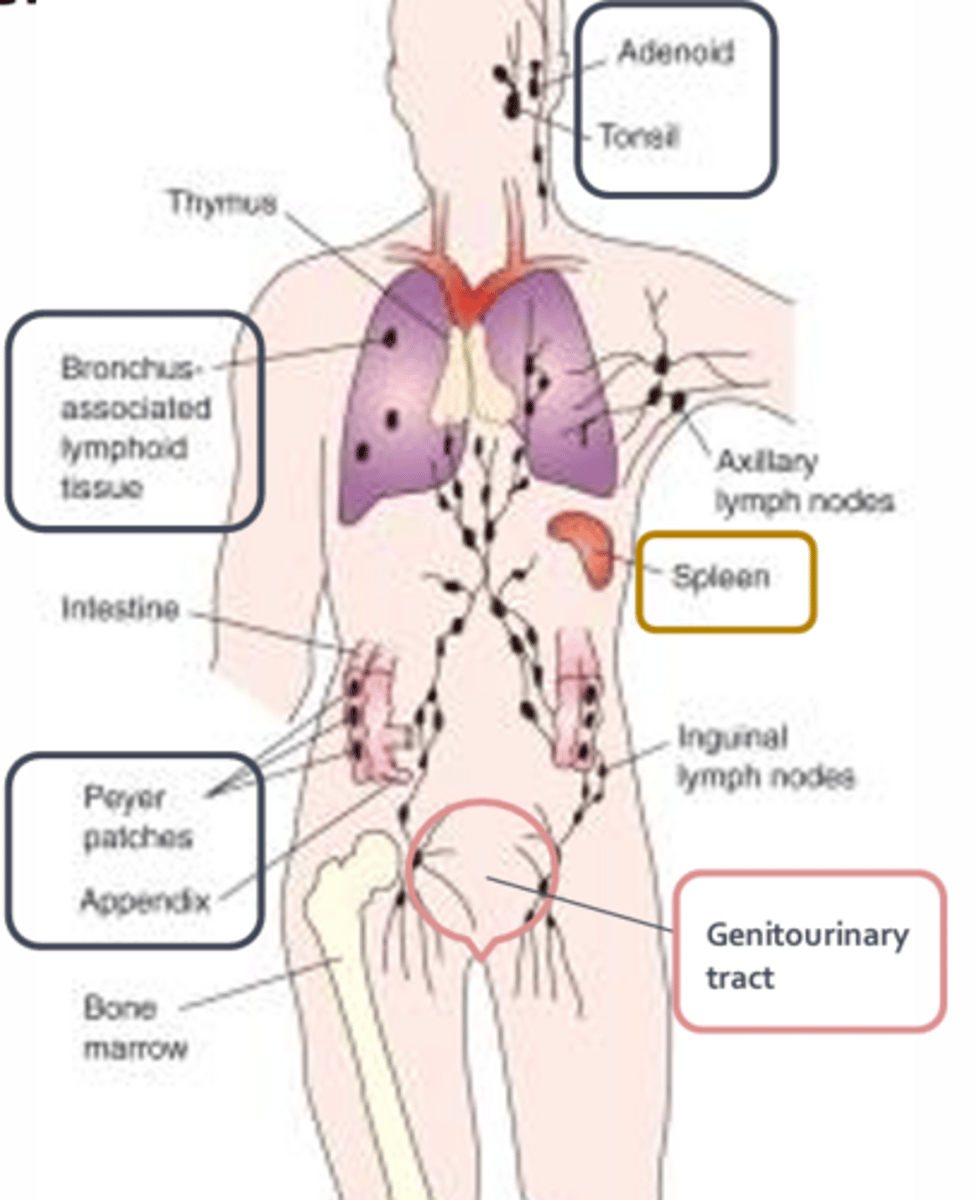
Spleen
- Acts to filter the blood of pathogens
MALT
- A loose aggregation of lymphoid tissue that is in a unique position to eliminate pathogens at mucosal surfaces before they spread to underlying tissue
- Includes adenoids, tonsils, Payer's patches of the gut, respiratory tract, GI tract, appendix
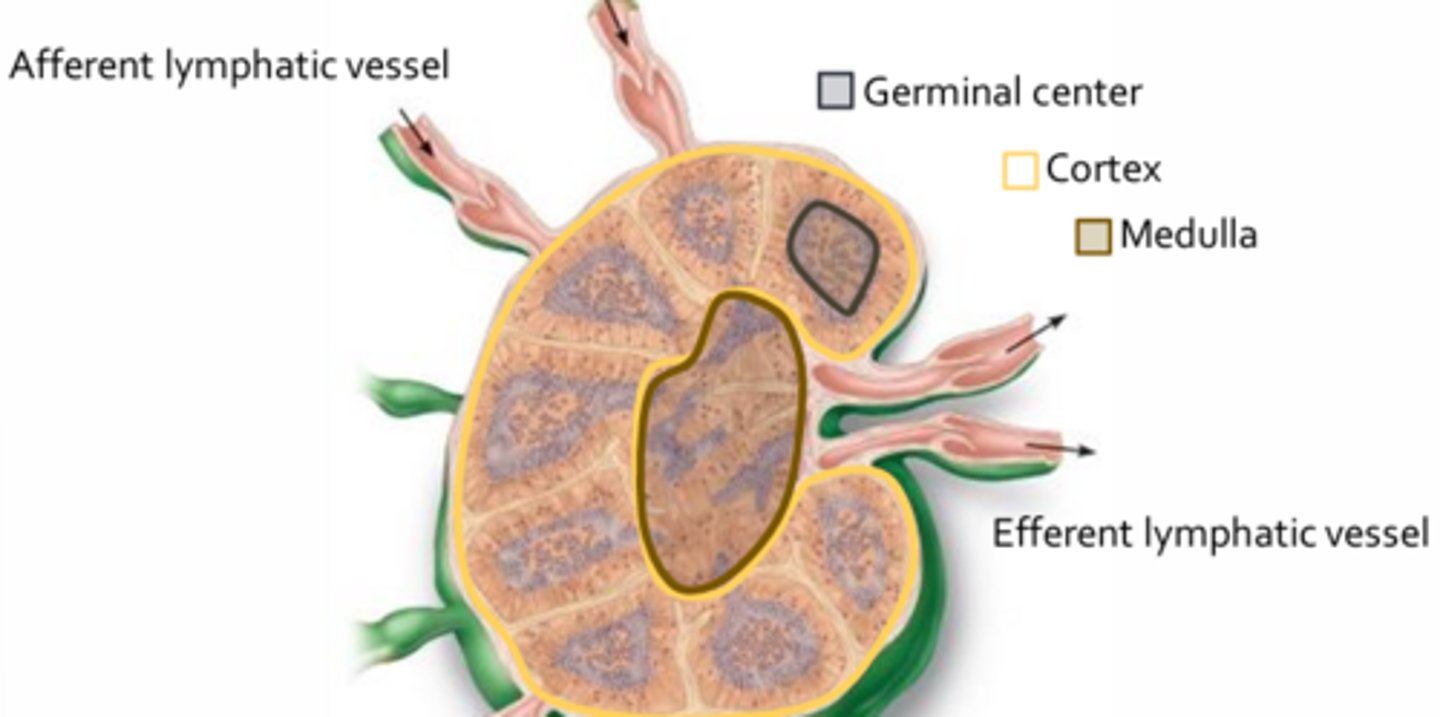
Lymph Nodes
- Provide immune surveillance of lymphatic fluid returning from the tissues
- During infection, lymph nodes may become swollen and can be detected visually or by palpation
- Extremely important in initiating and coordinating the adaptive immune response as they facilitate a rapid interaction amongst immune cells and between immune cells and pathogens
- Lymphatic fluids collected from tissues enter the lymph nodes via several afferent lymphatic vessels and leave by a limited number of efferent vessels
- Acts to slow the flow of lymph, thereby ensuring that resident T-cells and B-cells have adequate time to interact with incoming pathogens
- Contain macrophages and dendritic cells that have migrated from infected tissues (where innate and adaptive immune systems interact)
- Lymph nodes may contain actively proliferating B-cells, which are largely found within the germinal centers of the cortex
- These B-cells will differentiate into antibody-secreting plasma cells and migrate to the medulla or enter body tissues where they carry out their immune functions
Cells of Adaptive Immunity
- Adaptive immune response is mediated by B-cells and T-cells
- Activation of either cell type during an adaptive immune response can lead to damage to host tissues (so activities are tightly regulated)
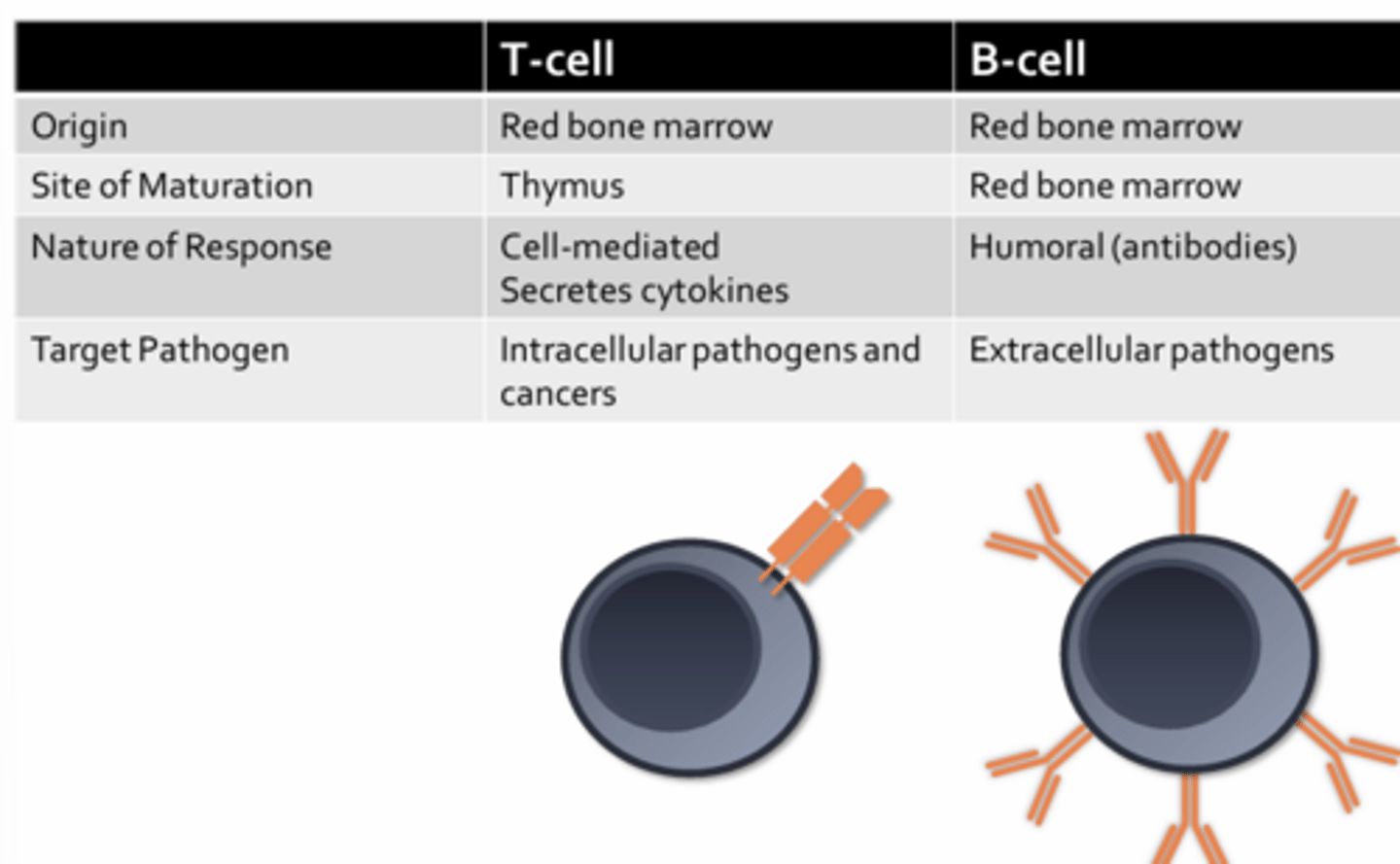
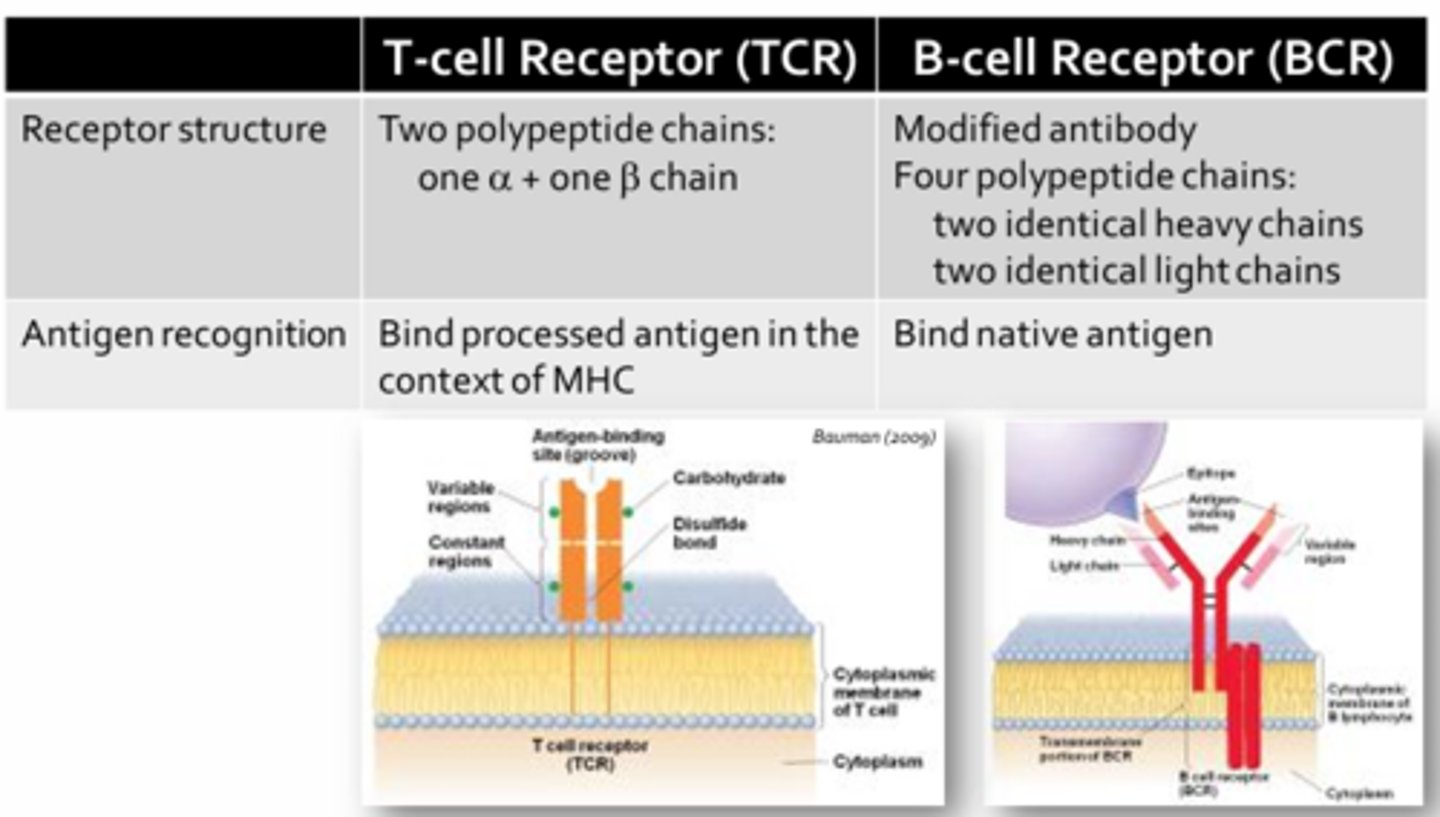
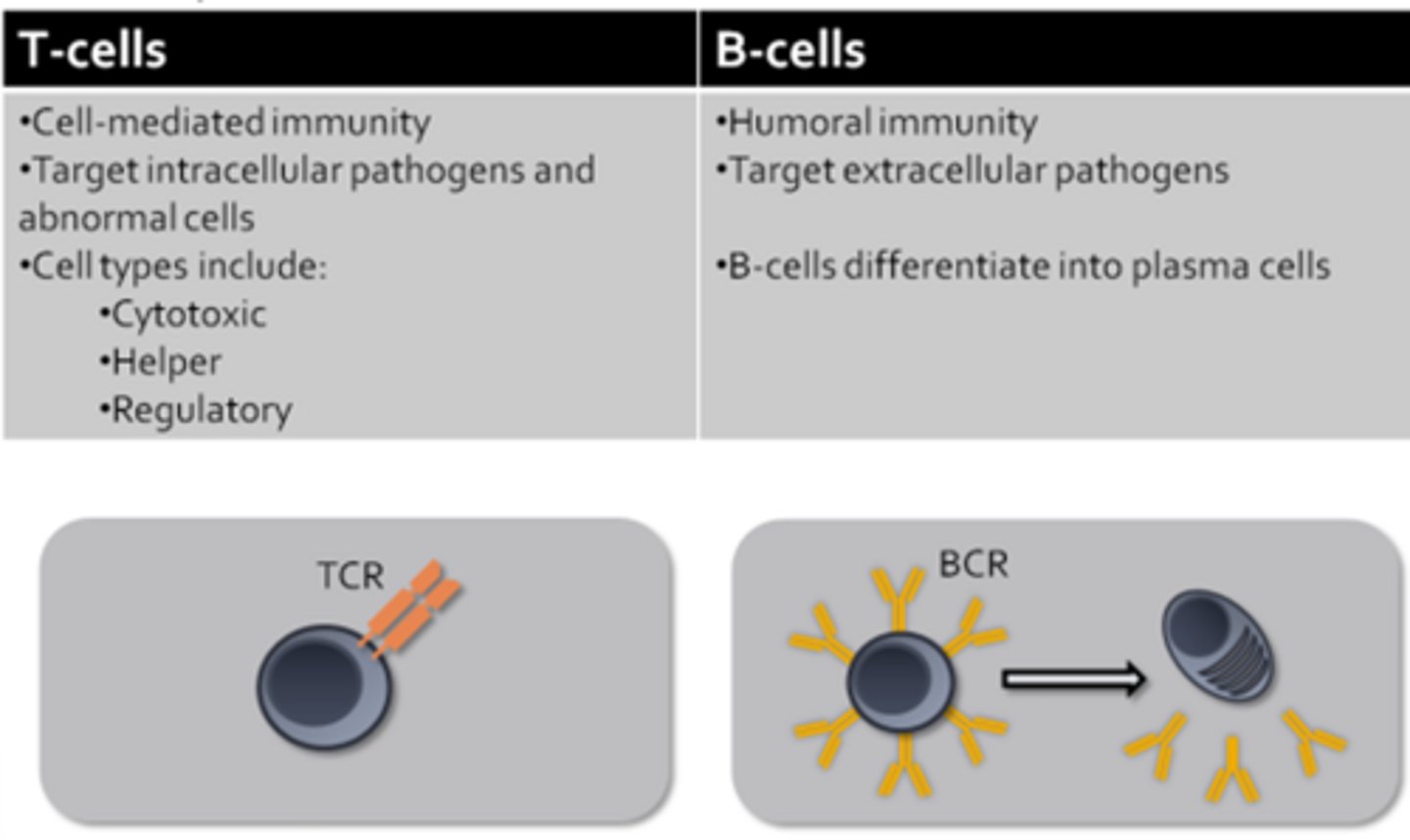
T-Cell & B-Cell Receptors
- Both T-cell and B-cell receptors are specific for only one antigen or pathogenic determinant (due to the fact that all the T-cell and B-cell receptors on a given T or B cell are identical)
- T-cells can only recognize antigens that have first been processed by specialized immune cells (i.e. antigen presenting cells = APCs)
- B-cells are capable of recognizing unprocessed or native antigens (no need for APCs before B-cells can become activated)
- The cells of the adaptive immune system must produce an astounding number of unique T-cell and B-cell receptors in order to confer protect over a trillion potential pathogenic determinants
APCs
- Process protein antigens by breaking them down into 12-18 amino acid segments that are then packaged and presented to T-cells via cell surface receptors termed MHC (major histocompatibility complex)
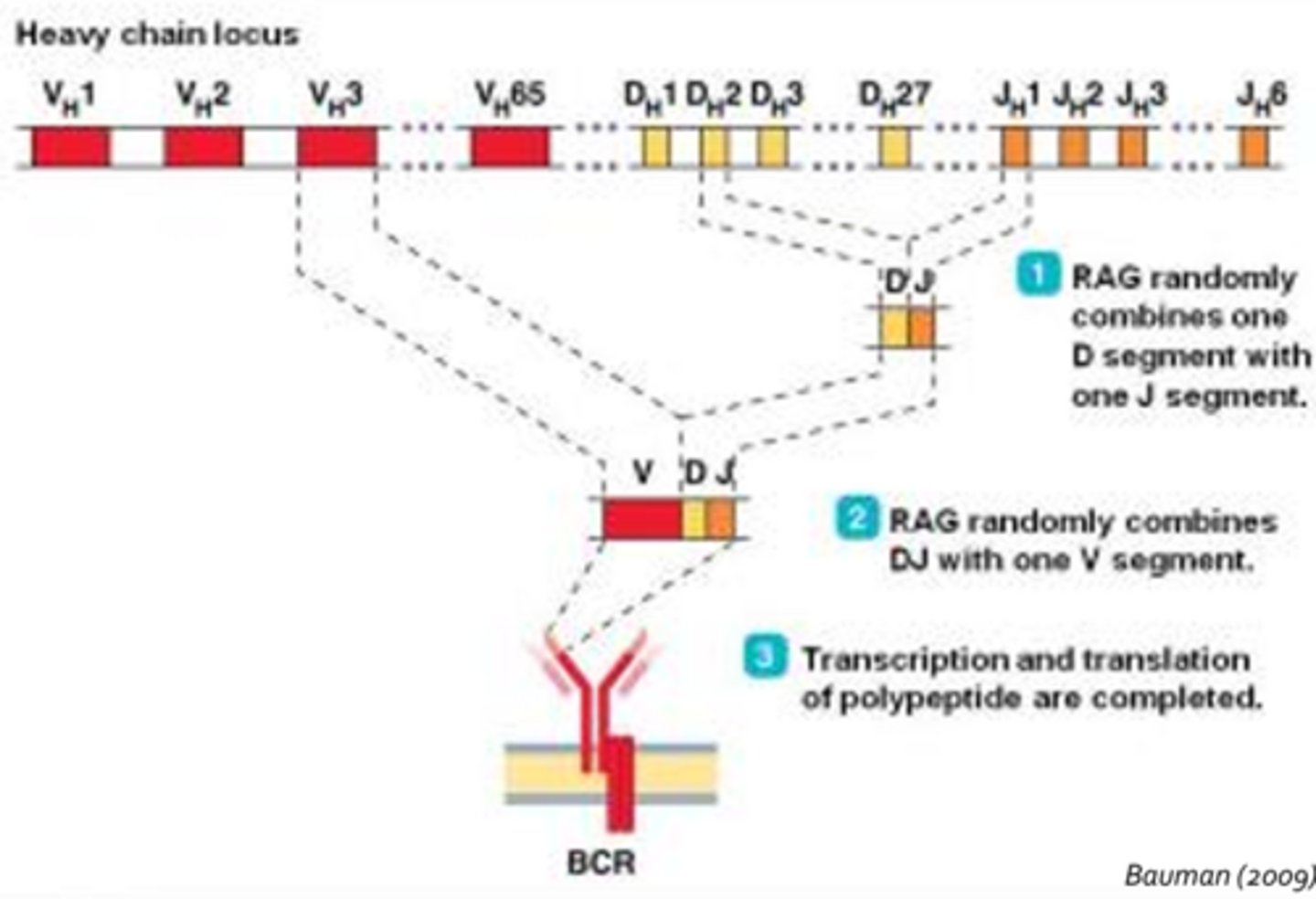
Somatic Recombination
- Random combination of DNA segments in T-cell and B-cell receptor gene regions to create a vast array of permutations
- B-Cells Receptors: Process involves random selection of on V segment, one D segment, and one J segment which are then joined
- T-Cells Receptors: V, D, and J gene segments are located on a different chromosome from that of the B-cell receptors
- Point mutations, deletions and insertions in these DNA segments are also incurred prior to transcription
- Our adaptive immune system creates a staggering array of unique T-cell and B-cell receptors
- However, all of this diversity can lead to the generation receptors that recognize self antigens, potentially causing autoimmune disorders
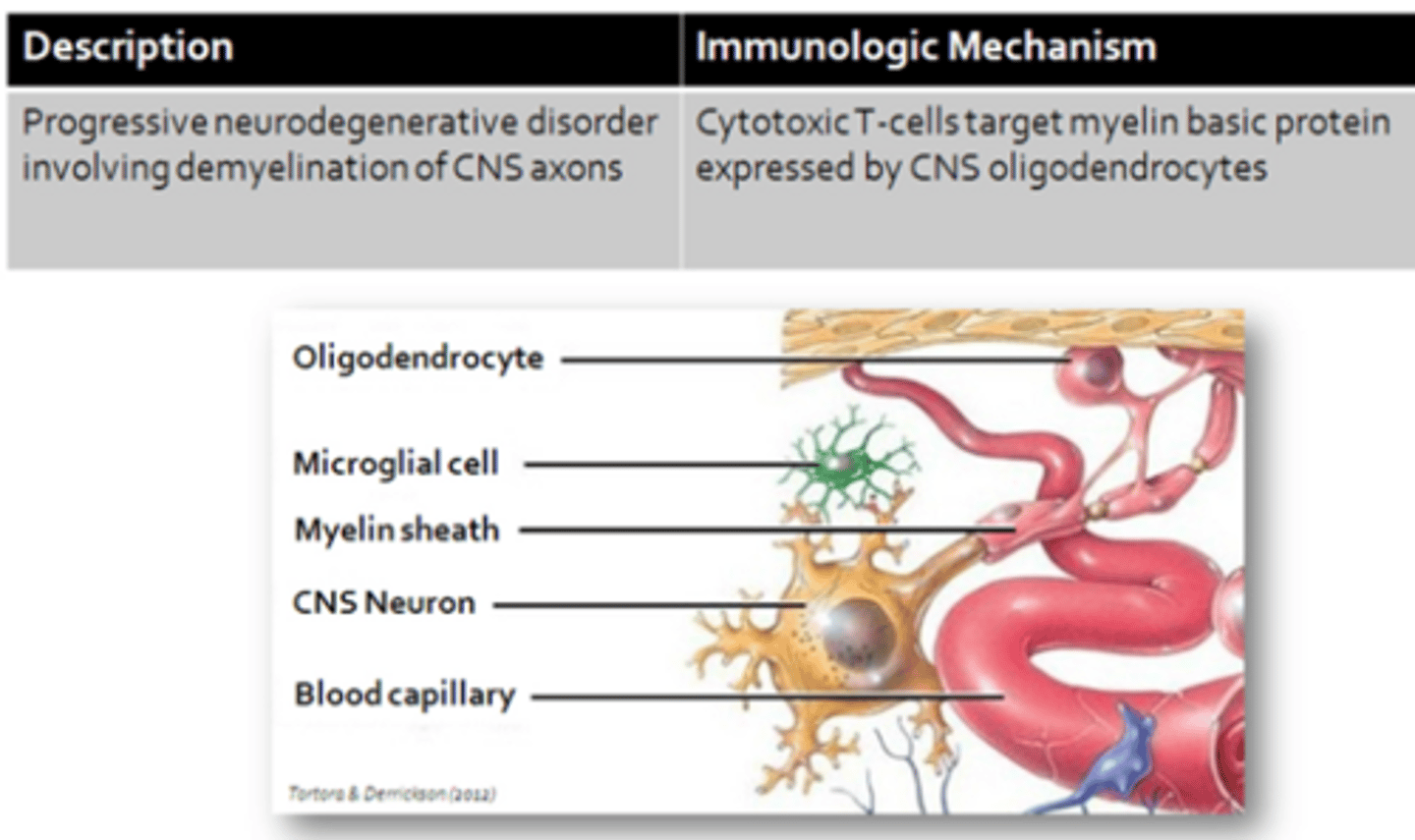
Cell-Mediated Autoimmune Disorders
- Multiple Sclerosis
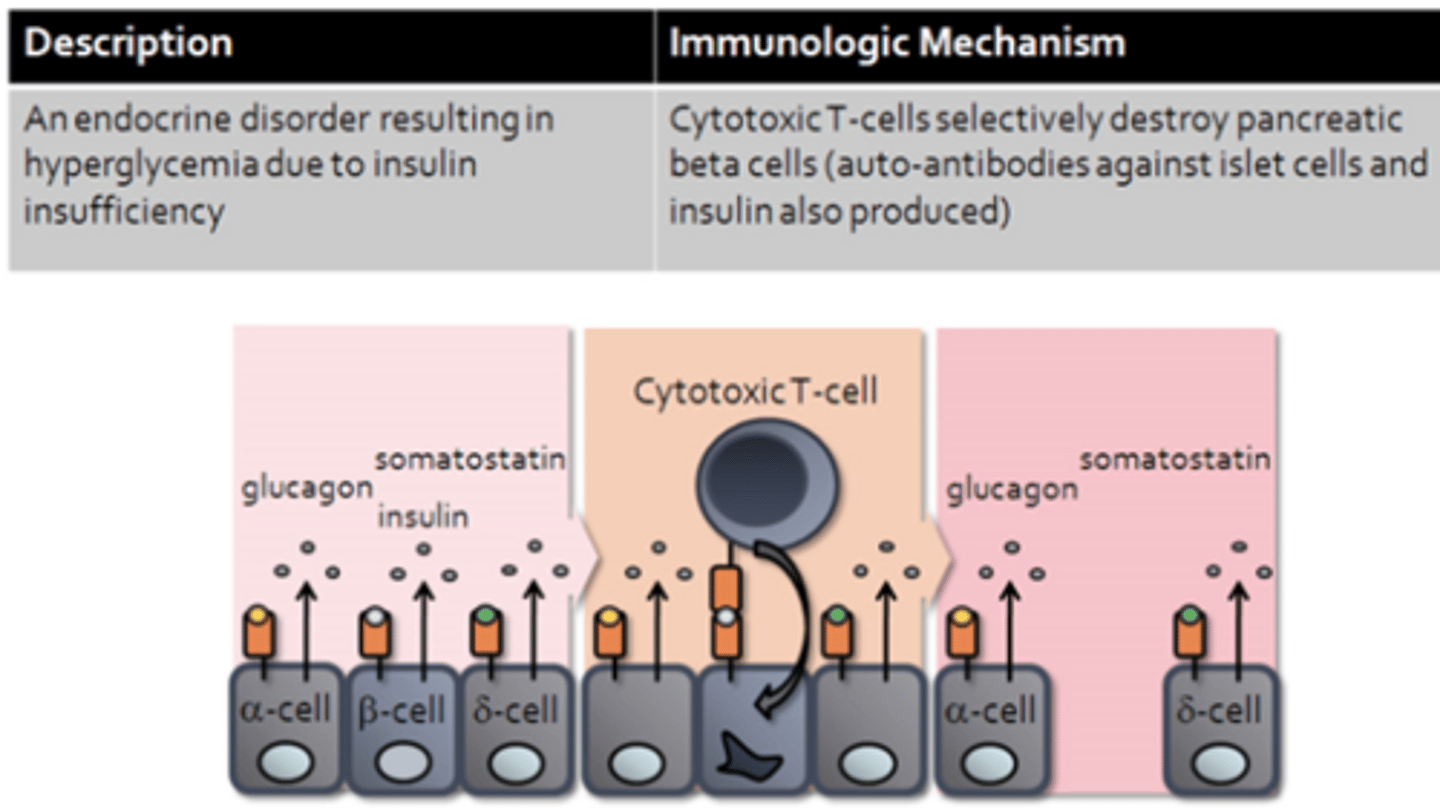
- Type I Diabetes Mellitus
Multiple Sclerosis (MS)
Type I Diabetes Mellitus
Antibody-Mediated Autoimmune Disorders
- Systemic Lupus Erythematosus
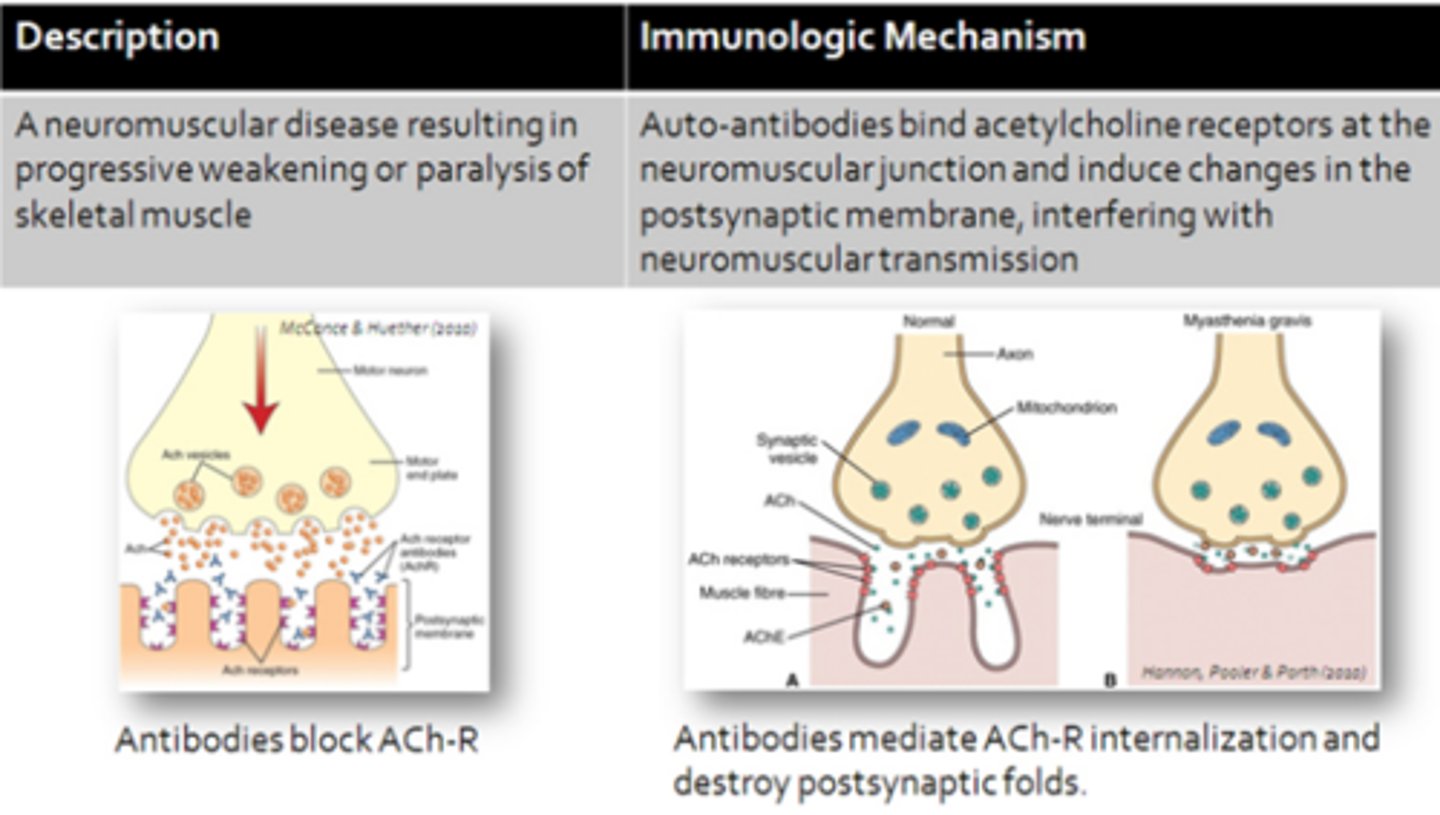
- Myasthenia Gravis
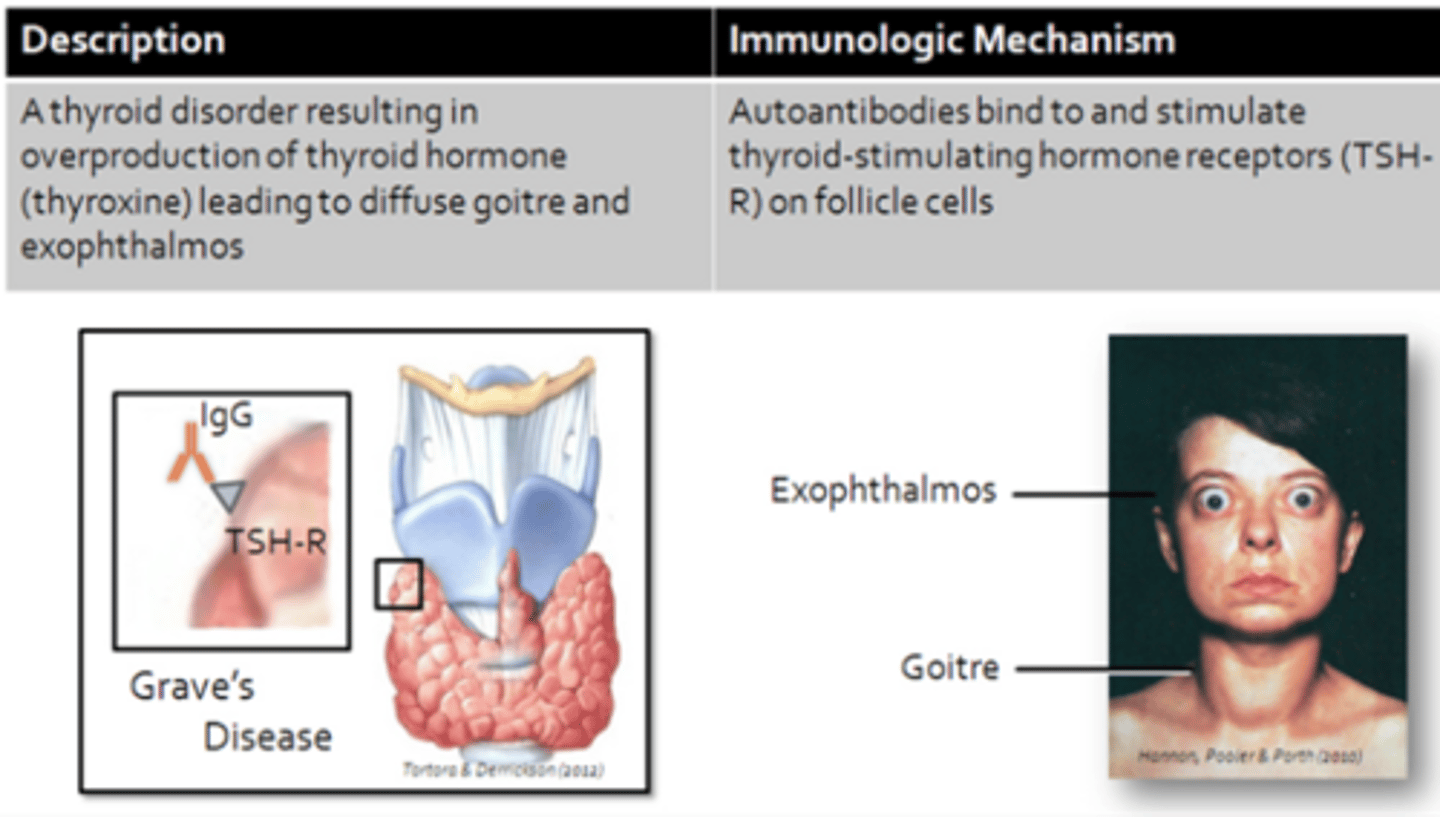
- Graves Disease
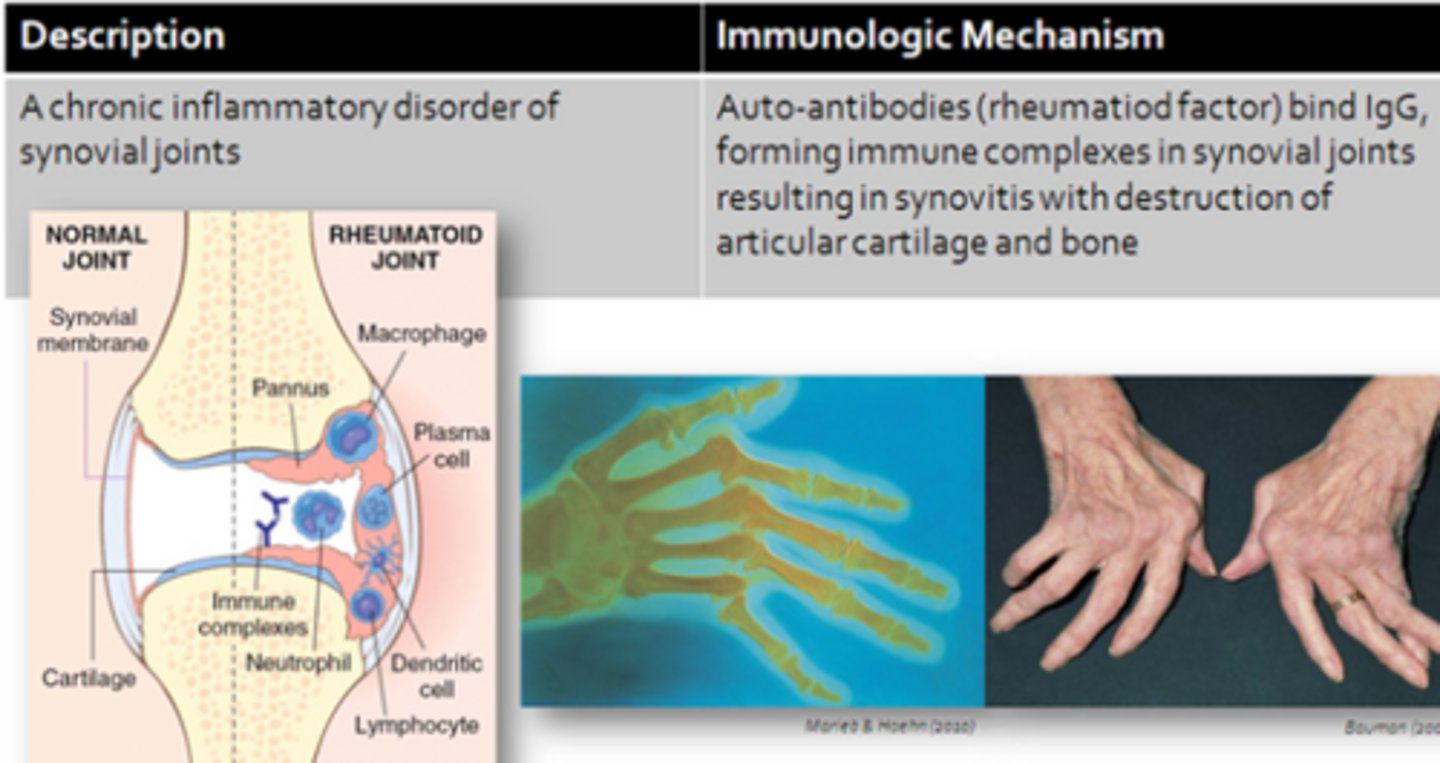
- Rheumatoid Arthritis
- Glomerulonephritis
Myasthenia Gravis (MG)
Graves Disease
Rheumatoid Arthritis
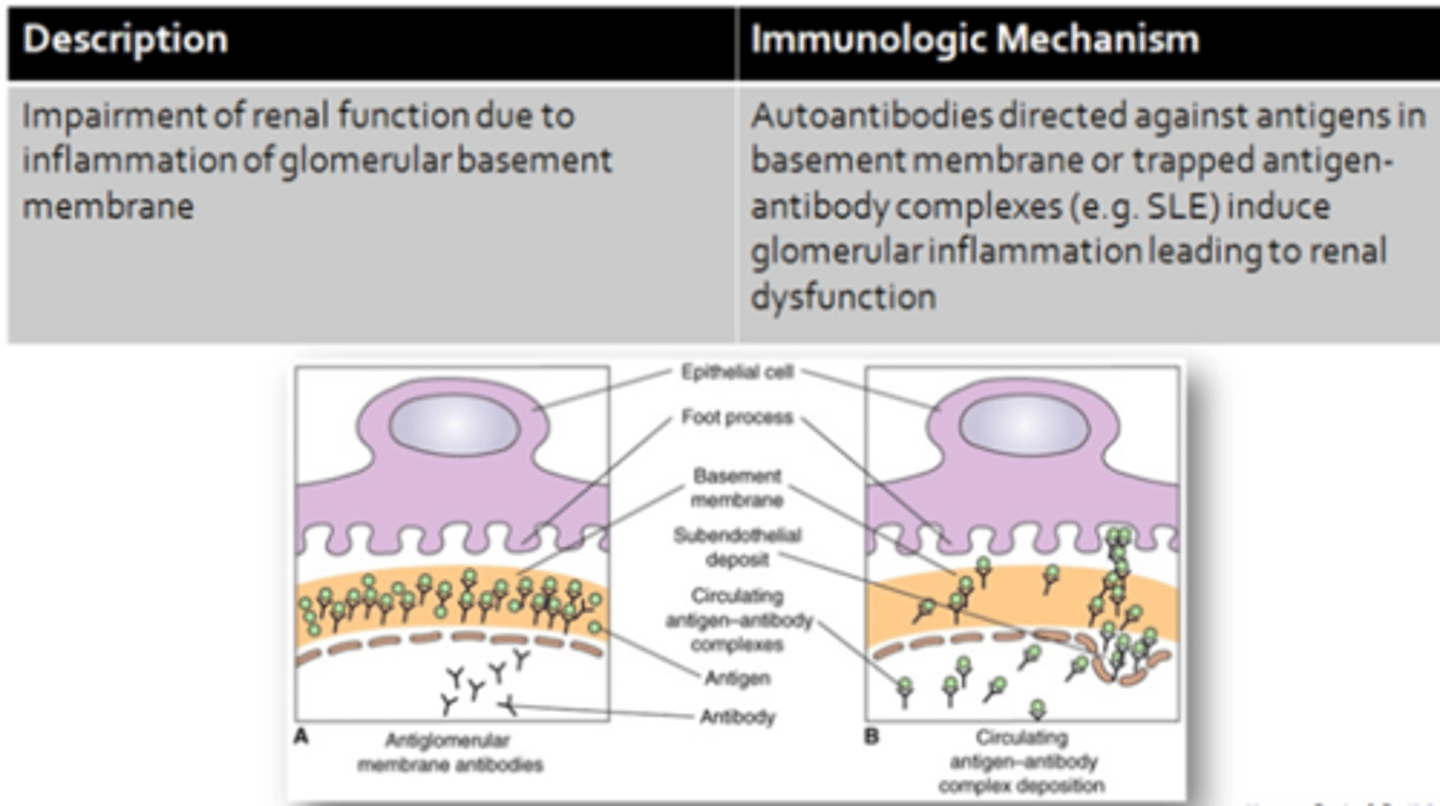
Glomerulonephritis
Systemic Lupus Erythematosus (SLE) - Autoimmune Disorders
- A chronic, complex, multisystem, inflammatory disease
- Slow to develop (10 yrs from detection of the first auto-antibody to diagnosis
- Inflammation involving multiple body systems due to the deposition of immune complexes within various organs throughout the body which may be affected concurrently or one-at-a-time
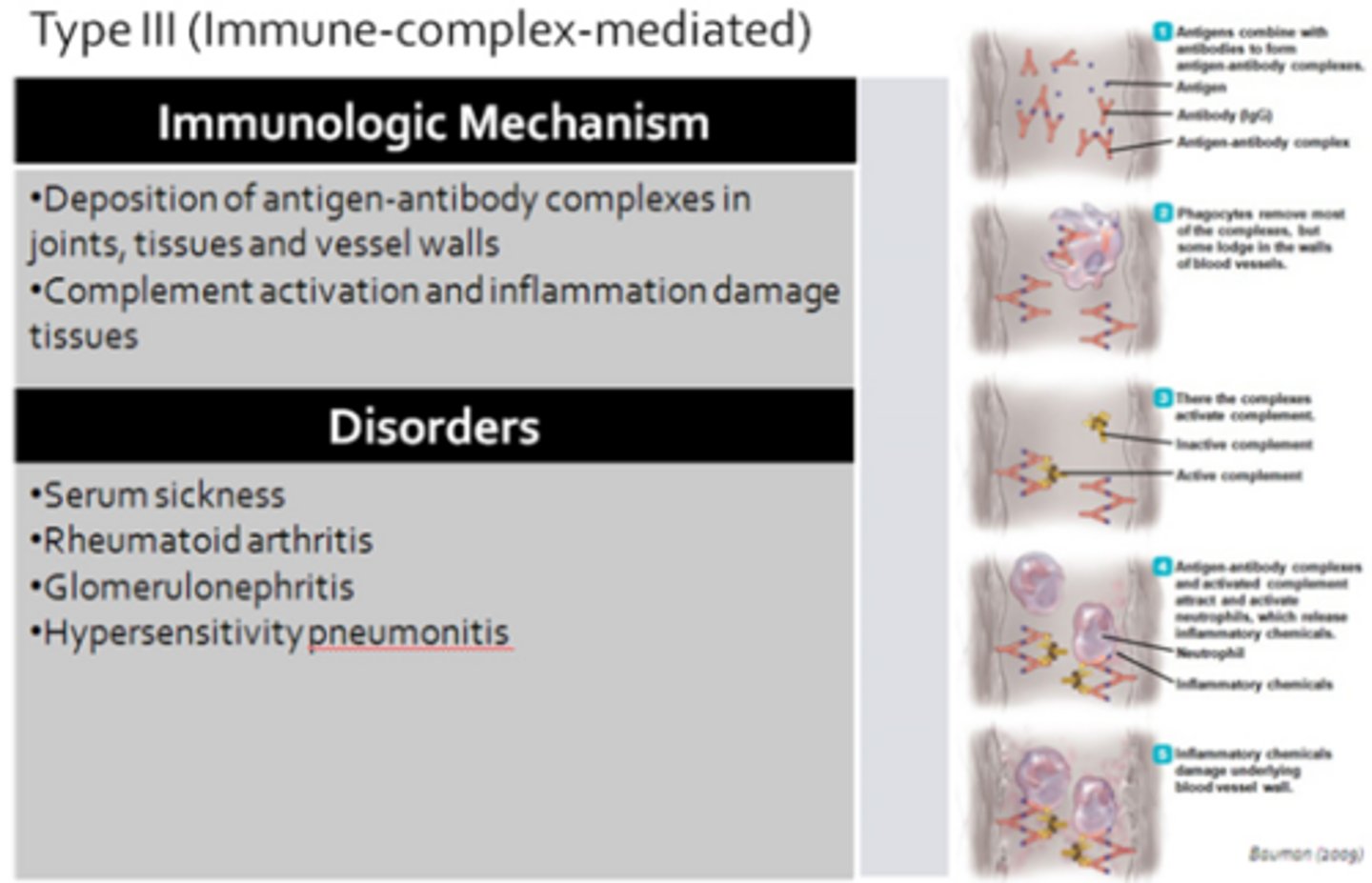
Type III Reaction
- Involving antigen-antibody immune complexes
Immunologic Mechanism - SLE
- Immune-complex-mediated (type III hypersensitivity reaction)
- Immune complexes are largely composed of IgG antibodies and nuclear antigens (DNA, histones)
- IgG can also complex with erythrocytes, coagulation factors, lymphocytes, and platelets
- Auto-antibodies commonly produced against nuclear proteins (nucleic acids, histones, ribonucleoproteins)
- Nuclear antigens often appear in circulation following cell damage associated with trauma, certain drugs (ex. hydralazine), and infections

Clinical Manifestations - SLE
- Arthritis (90%)
- Vasculitis and rash (70-80%)
- Renal disease (40-50%)
- Anemia (50%)
- Cardiovascular Disease (30-40%)
- Females more prone then males (10:1) = Estrogen may favour the development of SLE while androgens may be protective
- Exposure to UV radiation tends to exacerbate symptoms (due to release of nuclear antigens from the skin) = Causes butterfly rash to form, corresponding to areas of high UV exposure
Treatment - SLE
*No cure, symptoms management
NSAIDs:
- Reduce inflammation & pain
- Inhibiting cyclooxygenase enzymes responsible for generating prostaglandins
- Ex. Ibuprofen, ASA
Corticosteroids:
- For acute, active disease
- Reduce severity and duration of symptoms through an overall reduction of immune activity
Immunosuppressives:
- Including methotrexate (Apo-methotexate) and azathioprine (Imuran) to treat severe symptoms involving internal organs
- Interrupt lymphocyte replication by halting cell division resulting in decreased antibody production and decline in Lupus-related immune complexes
Antimalarials:
- Reduce antigen processing by APCs
- Ex. Hydroxychloroquine
- Reduce the need for corticosteroids
- Inhibit the early events of antigen processing and cytokine production by macrophages
- Reduces lymphocyte activation and subsequent autoantibody production
Biologics:
- BLyS inhibitor (belimumab) reduces B-cell survival in active disease (autoantibody titers; decreases immune complex formation and exacerbate SLE symptoms)
- Employ products from humans, animals, and microorganisms
- Often used as an add on to standard therapy
- Can cause butterfly rash
- Ex. Vaccines, blood, blood components (antibodies), recombinant proteins
IL-2 therapy:
- Promotes T-reg cell survival
- IL-2 is a cytokine that promotes the survival of regulatory T-helper cells (T-reg cells)
- Preventing inappropriate immune response by reducing the action of lymphocytes
- Cell population is often reduced in SLE
T-Cell Development
- T-cell development begins with positive selection or MHC restriction
- Only T-cells that express T-cell receptors capable of interacting with self MHC molecules present on thymic cells are allowed to develop
- If a T-cell cannot bind to MHC, then it cannot communicate with other immune cells and is completely useless (dead weight)
- T-cells unable to interact with MHC during its stint at thymic university, it will receive a strong signal to undergo apoptosis
- T-cells that survive positive selection now undergo negative selection or clonal deletion
- During this process, specialized thymic epithelial cells present self-antigens to developing T-cells in the context of MHC
- T-cells that bind strongly to self antigens undergo apoptosis within the thymus, while T-cells that do not bind self antigens are allowed to migrate to the secondary lymphoid tissues where they contribute to the working population of T-cells
- Ensures that immunocompetent lymphocytes are allowed to develop and form a self-tolerance known as central tolerance
- Regulatory T-cells = Function to suppress immune responses directed against self-antigens (when autoreactive T-cells escape negative selection)
- Anergy = When autoreactive T-cells may be inactivated
- Activation of T-cells requires additional signals from other immune cells, which are not present unless the immune system is actively employed in fending off an infection
- Peripheral tolerance = The development of regulatory T-cells and the induction of anergy (adaptive cells are regulated in the secondary or peripheral lymphoid tissues)
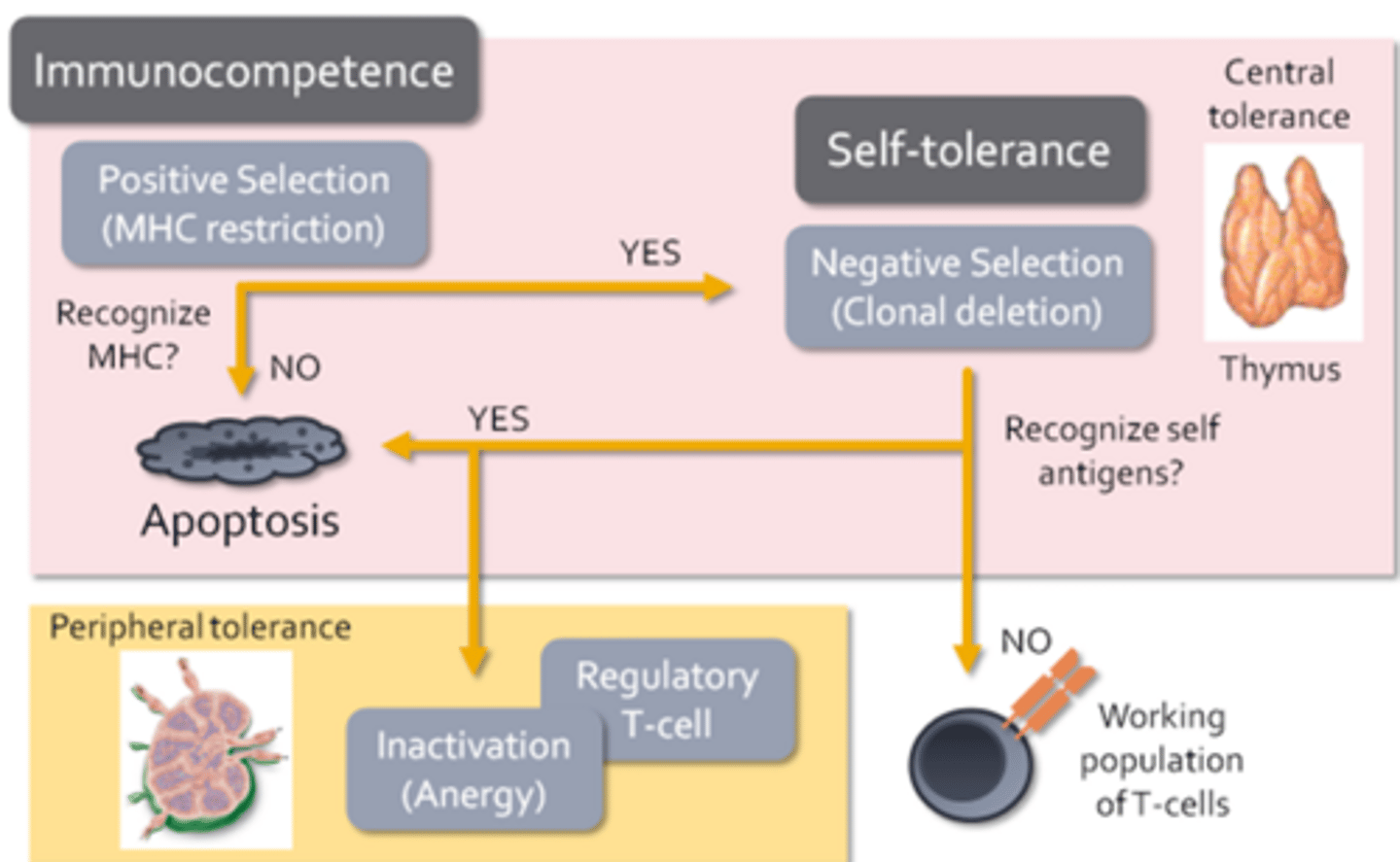
Thymus
- Where positive and negative selection occurs
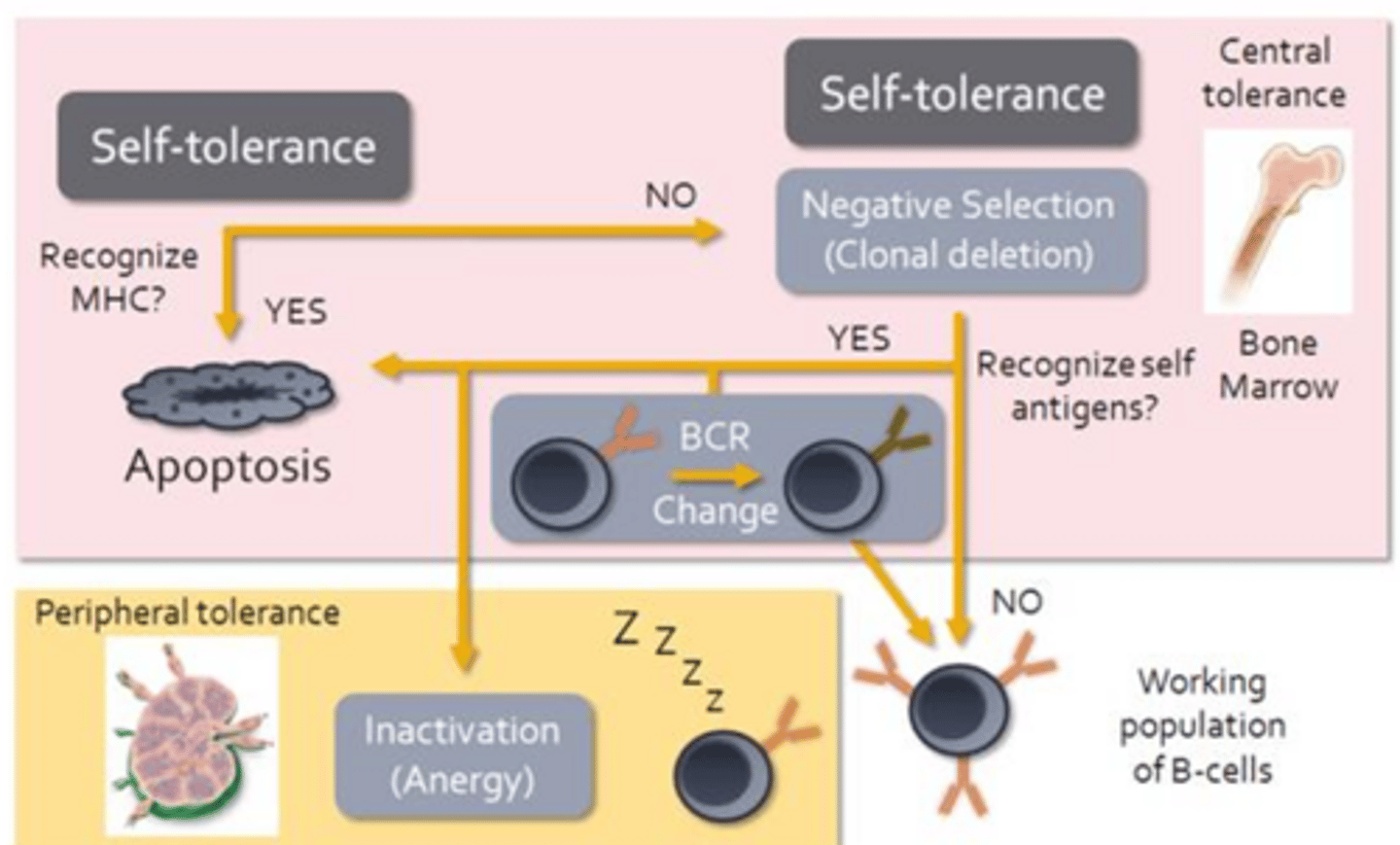
B-Cell Development
- Recognition of MHC by a B-cell receptor will result in apoptosis
- Recognition of MHC molecules would indicate that a B-cell is autoreactive
- While B-cells do undergo limited negative selection within the red bone marrow, autoreactive B-cells can be induced to change their BCR
- In effect they undergo a second round of somatic recombination
- If successful, these B-cells will contribute to the working population of B-cells
- Anergy appears to be the main mechanism responsible for preventing B-cell mediated autoimmune responses should autoreactive B-cells make their way out of the bone marrow
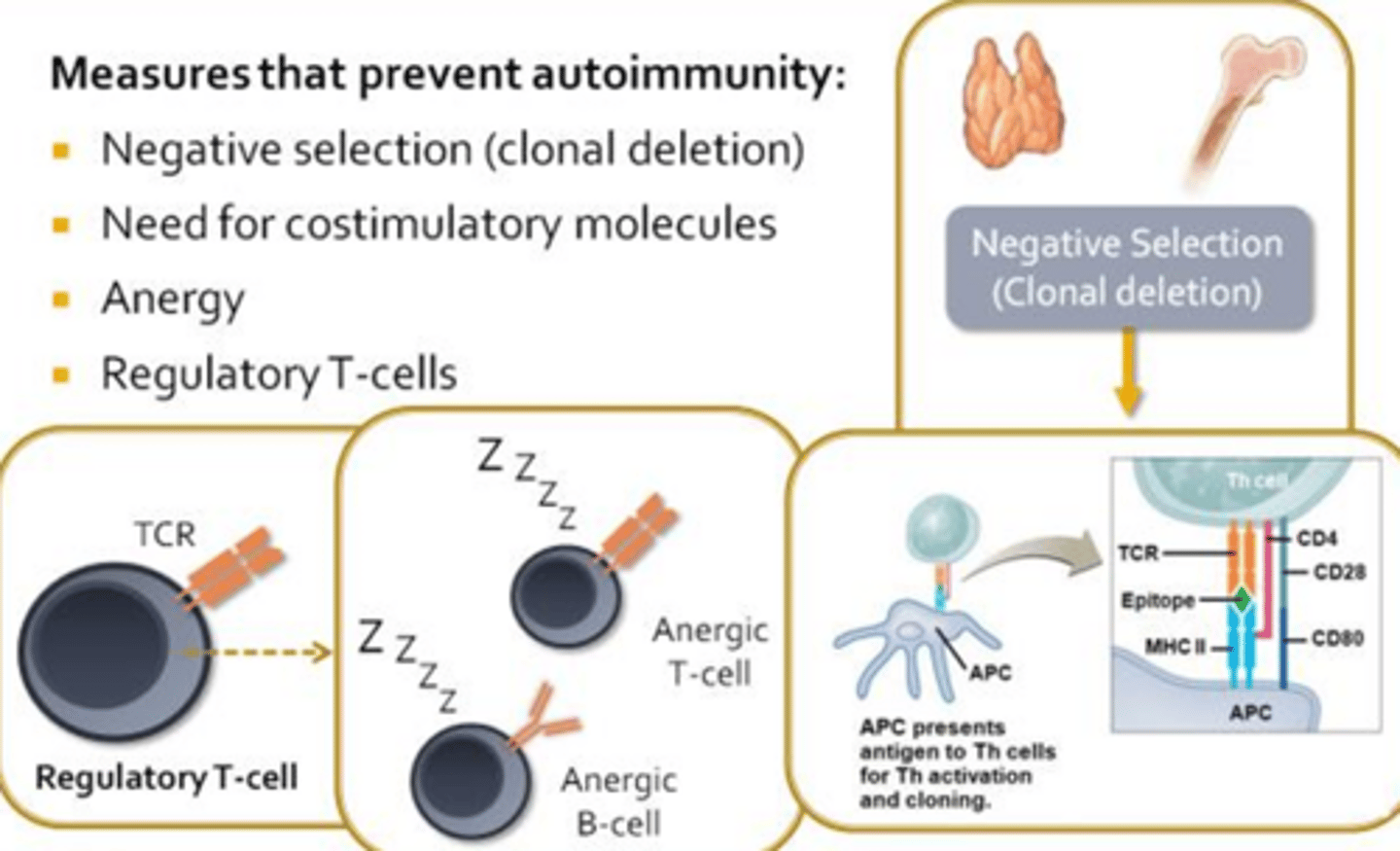
Measures that Prevent Autoimmunity
Negative selection (clonal deletion) -> Central Tolerance:
- Thymus and red bone marrow use negative selection leading to clonal deletion to ensure that autoreactive lymphocytes are eliminated before they become a problem
- Should autoreactive lymphocytes escape the primary lymphoid organs there are several check ins/safety measures
Need for costimulatory molecules -> Peripheral Tolerance:
- For T-cells and B-cells to become fully activated, antigen presenting cells must supply them with additional costimulatory molecules
- Antigen presenting cells only express these costimulatory molecules when they receive danger signals released by innate immune cells or damaged tissues during an on-going infection
- Controls humoral and cellular immunity
Anergy -> Peripheral Tolerance:
- If an antigen presenting cell does not express the costimulatory signals, its interactions with a T-cell or B-cell in the lymph node will invariable induce a state of anergy
- In some cases, anergic lymphocytes will undergo apoptosis and cease be a burden on the immune system
Regulatory T-cells -> Peripheral Tolerance
- Regulatory T-cells produced during thymic education, function to inhibit or suppress the activities of autoreactive lymphocytes, thereby contributing to peripheral tolerance and ensuring that autoimmune diseases do not arise
Causes of Autoimmunity
- Break-down of central and/or peripheral tolerance
- Molecular Mimicry (foreign antigens closely resemble self antigens)
- Appearance of new self-antigens
- Genetic predisposition
- Gender
Molecular Mimicry
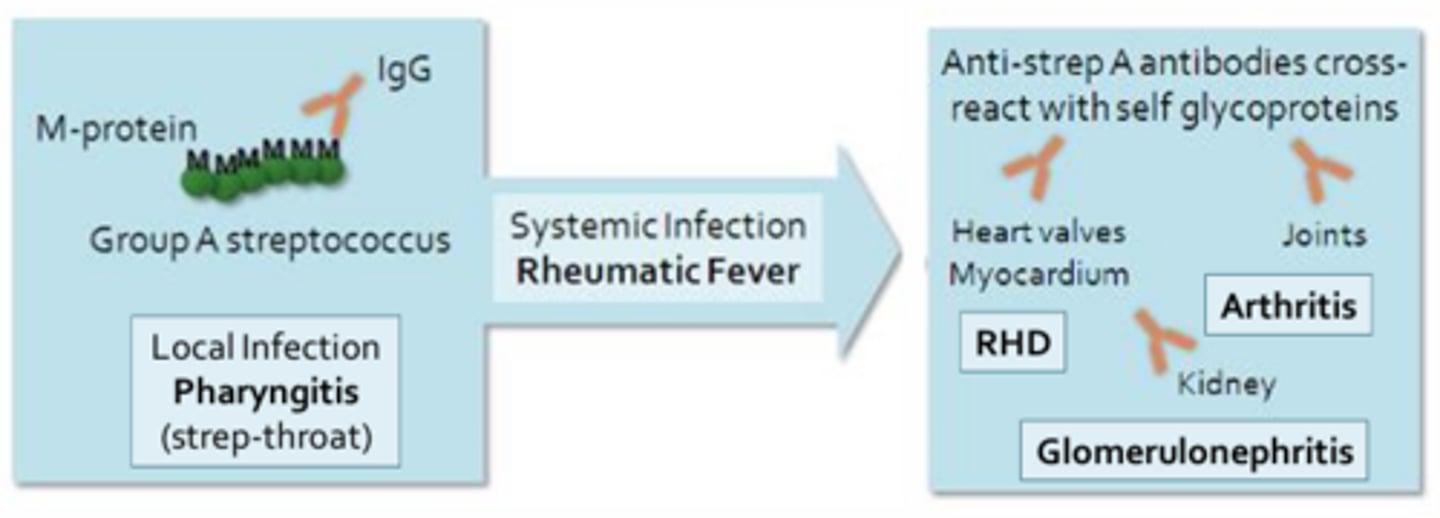
Ex. Rheumatic fever = Begins as a throat infection or pharyngitis mediated by Group A beta-hemolytic streptococci
- The antibodies produced during a strep infection target bacterial M-proteins and may cross-react with self-antigens expressed in the heart, resulting in long-lasting damage to the heat muscle and valves (rheumatic heart disease)
- Antibodies may also cross-react with self-antigens in the joints and kidneys, leading to arthritis or glomerulonephritis respectively
Appearance of New Self-Antigens
- Cancer: When mutations in host genes result in the expression of new proteins on cell surfaces
- Change in antigen structure (haptens): Self-antigens become altered by the attachment of small molecules (such as drugs or toxins) effectively rendering them foreign in the adaptive response
- Haptens = Incapable of evoking an immune response on their own
- Release of sequestered antigens due to tissue trauma: Spermatozoa during vasectomy or testicular trauma (either blunt force or penetrating); exposure of corneal or lens antigens following eye or contact lens trauma; and exposure of cardiac muscle following a myocardial infarction
- Predisposition to autoimmunity and hormonal factors (women more likely to develop autoimmune disorders then men due to hormones role in immune regulation)
Types of T-Cells
- Cluster of differentiation = CD
- CD molecules act as co-receptors during immune activations

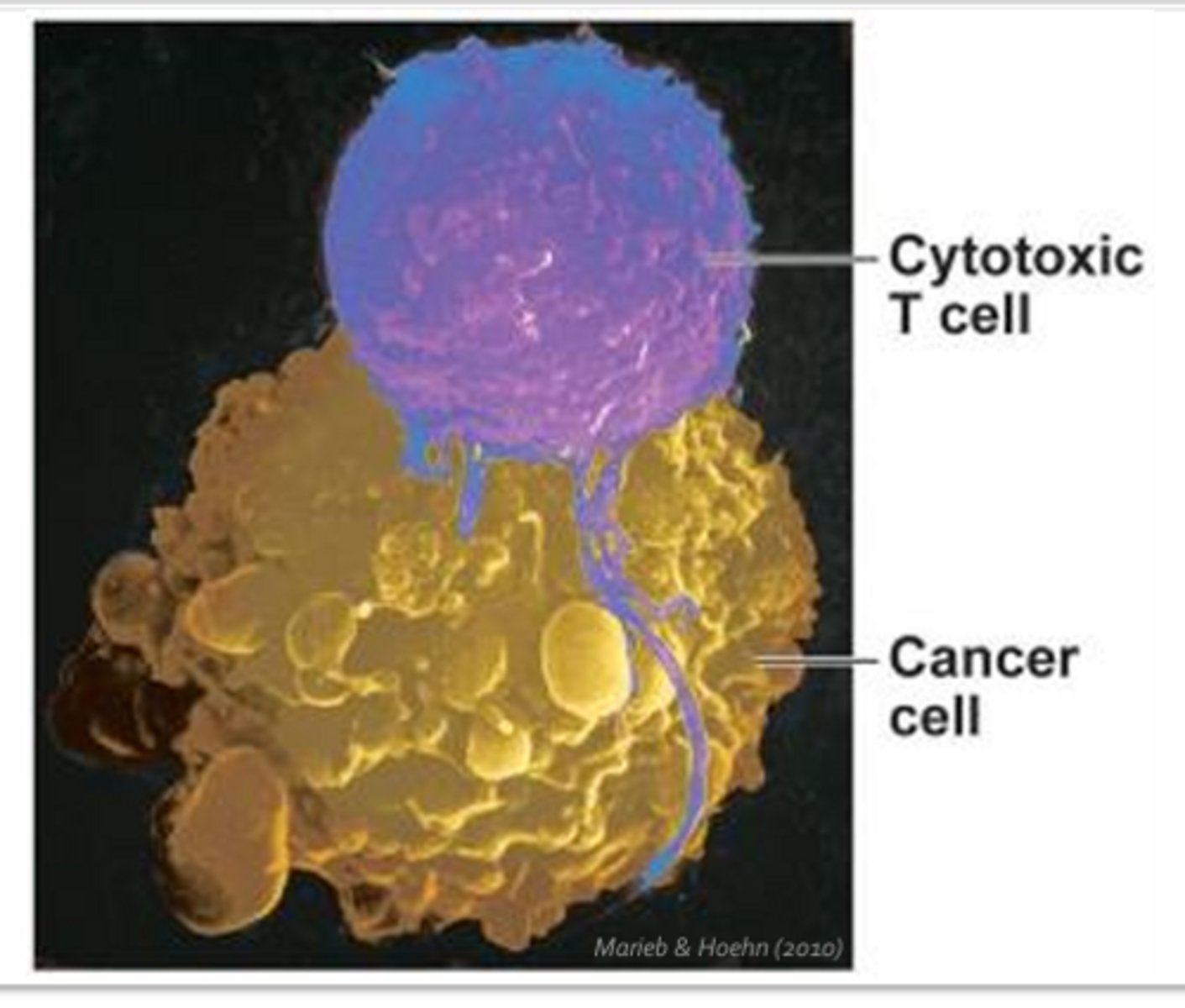
Cytotoxic T-Cell
- CD8+
- Responsible for mediating cellular immunity
- Undergo differentiation and clonal expansion within lymph nodes, they must then migrate to the tissues in order to deal with infected or abnormal host cells
- Once at the site of infection, cytotoxic T-cells will make direct cell-to-cell context with a target cell to induce apoptosis
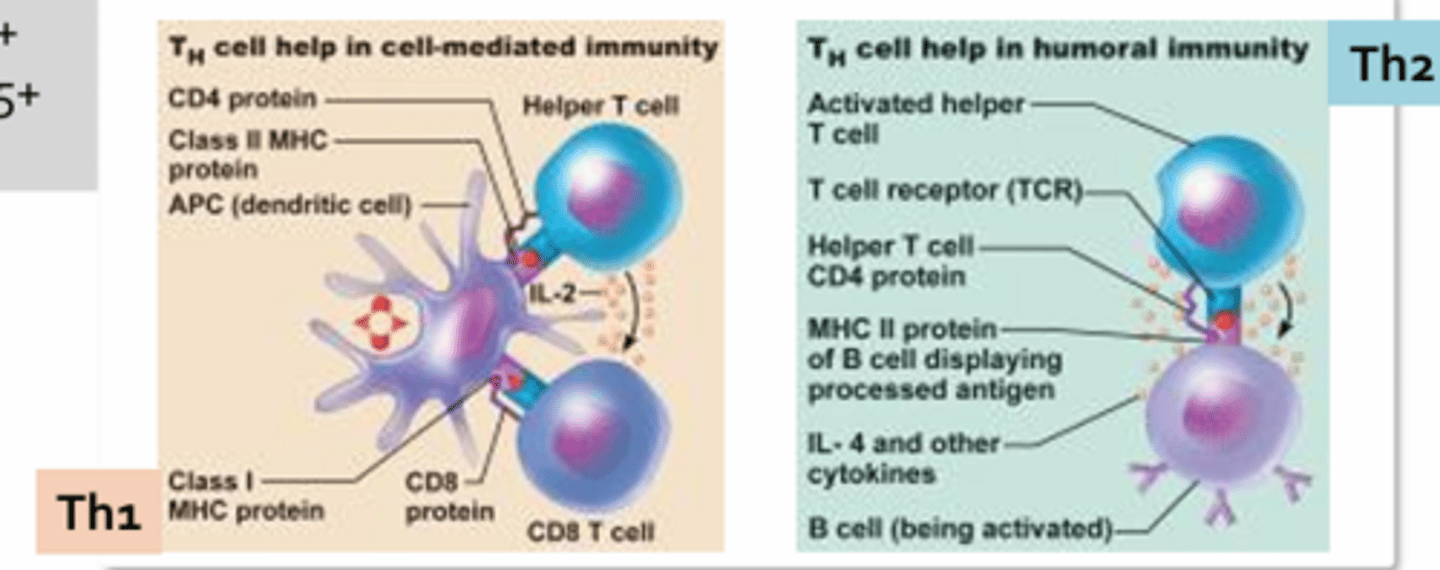
Helper T-Cell
- CD4+
- Aid in the induction of cellular and humoral response by providing essential signals that drive T-cell and B-cell differentiation and clonal expansion
- Without, would not be able to mount an effective adaptive immune response
- 3 subtypes: Th1, Th2, Th17
Th1:
- Largely secrete a chemical signal IL-2 which helps to fully activate cytotoxic T-cells
- Secrete interferon-gamma which helps to promote the differentiation of more T-helper cells into Th1 cells
Th2:
- Secrete IL-4 which provides the co-stimulatory signal necessary to fully active B-cells that have come into contact with foreign antigens
- IL-6 = Proinflammatory cytokine
Th17:
- Secrete Il-17 which activates neutrophils and plays a major role in fighting fungal infections
*Interaction between the B-cell and the helper T-cell is stabilized by CD4
Regulatory T-Cell
- CD4+ and CD25+
- Main function is to suppress adaptive immune responses
- Prevent autoimmune disease by inhibiting the activities of autoreactive immune cells
- Suppress immune responses through direct cell-to-cell contact or through the release of IL-10 which functions to inhibit macrophages, dendritic cells, and cells of the adaptive response
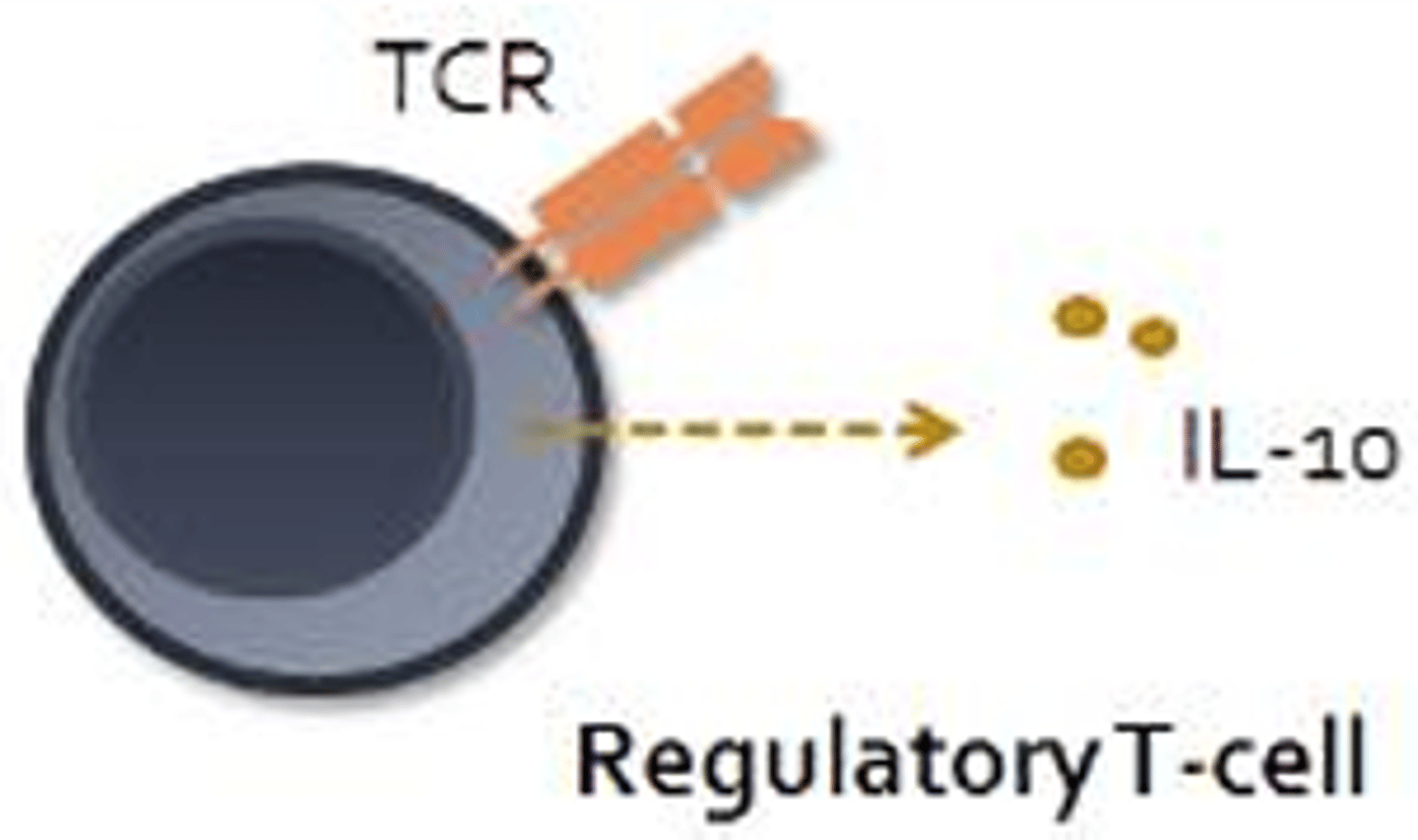
B-Cells
- Largely found in: Spleen; MALT; Lymph nodes (secondary lymphoid tissues)
- Abundant in the germinal centers of lymph nodes, where B-cells undergo rapid differentiation and proliferation following immune activation
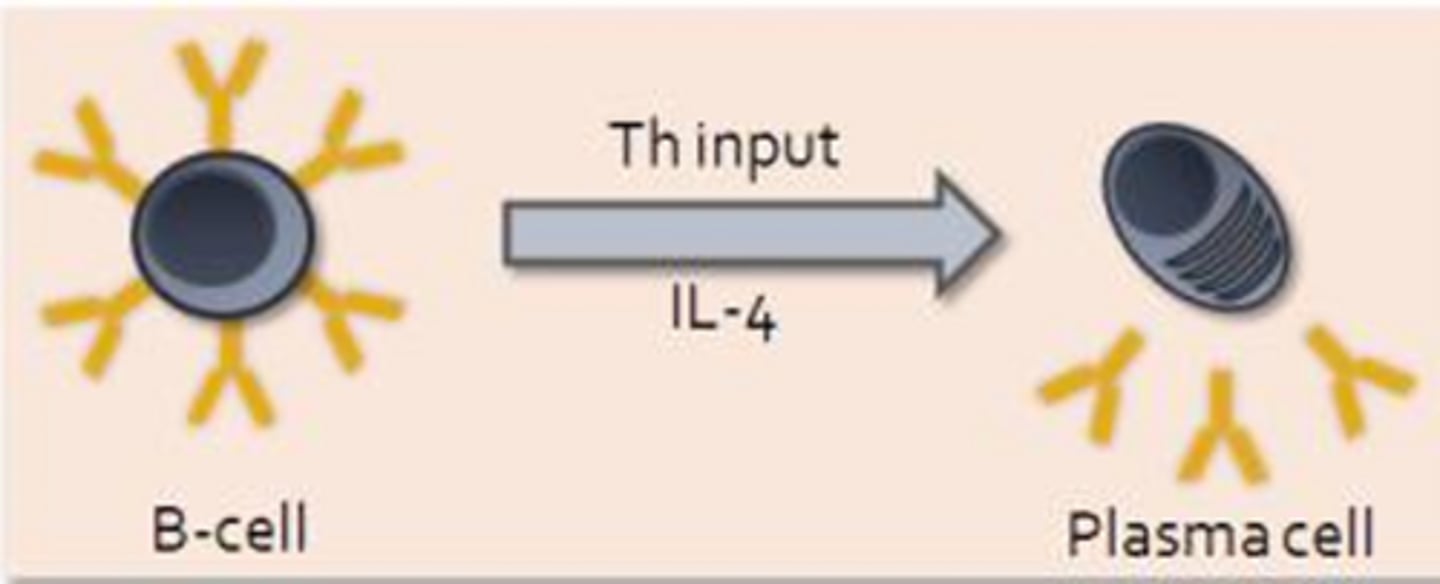
- Differentiate into plasma cells (B-cells with the aid of helper T-cell input and IL-4 will primarily differentiate into antibody secreting plasma cells)
- Plasma cells = Produce antibodies which compromise the body's humoral response (some plasma cells migrate and contribute to local antibody production)
- Secrete soluble antibodies (humoral)
- Attack from near or afar (Many plasma cells tend to stay within the secondary lymphoid tissues, where they can safely strike at pathogens from afar)
Antibody Functions
Neutralization:
- When antibodies bind toxins, they often inactivate it through direct or allosteric inhibition of the toxin's active site
- Antibodies may neutralize bacteria and viruses by binding key surface molecules that are necessary for bacterial adherence or viral fusion with host cells
Agglutination:
- Each antibody possesses at least 2 antigen binding sites, hence they are capable of binding more than one antigen resulting in clumping or agglutination
- Binding of soluble molecules, instead of cells will cause precipitates to form
Precipitation:
- Much easier for phagocytes to capture and engulf than freely moving antigens
Opsonization:
- Tagging antigens to help improve phagocytosis
Complement:
- Formation of a membrane attack complex or enhancement of inflammation by inducing histamine release from platelets, basophils, and/or mast cells or by acting as a chemotaxis agent for neutrophils
Fc Receptors:
- On the surface of mast cells, basophils, and eosinophils
- Capable of binding the stem region of antibodies (act as surface receptors)
- Results in degranulation and liberation of histamine, enhancing inflammation
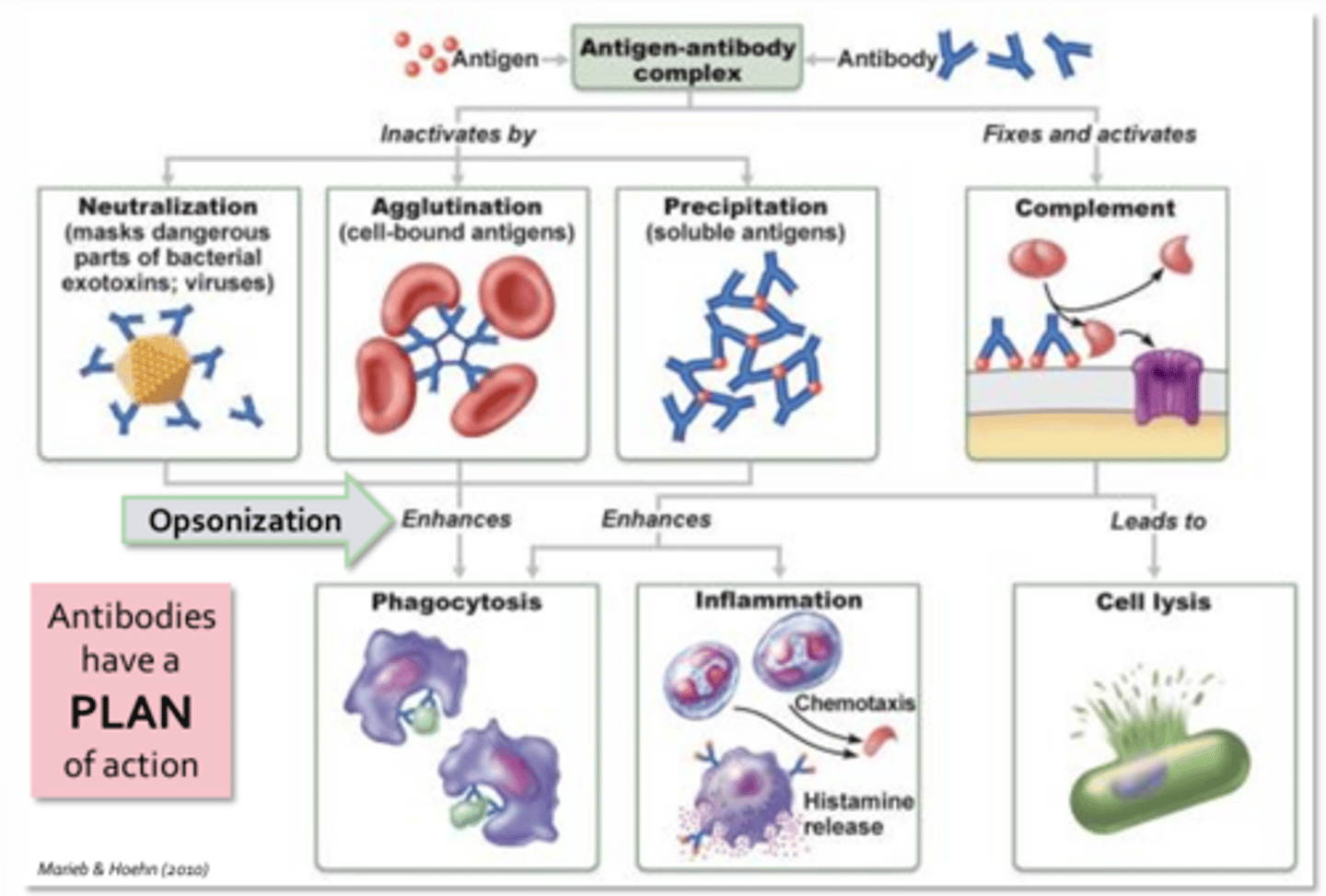
PLAN - Function of Antibodies
- Precipitation
- Lysis
- Agglutination
- Neutralization
Classes of Antibodies
- Ig = Immunoglobin
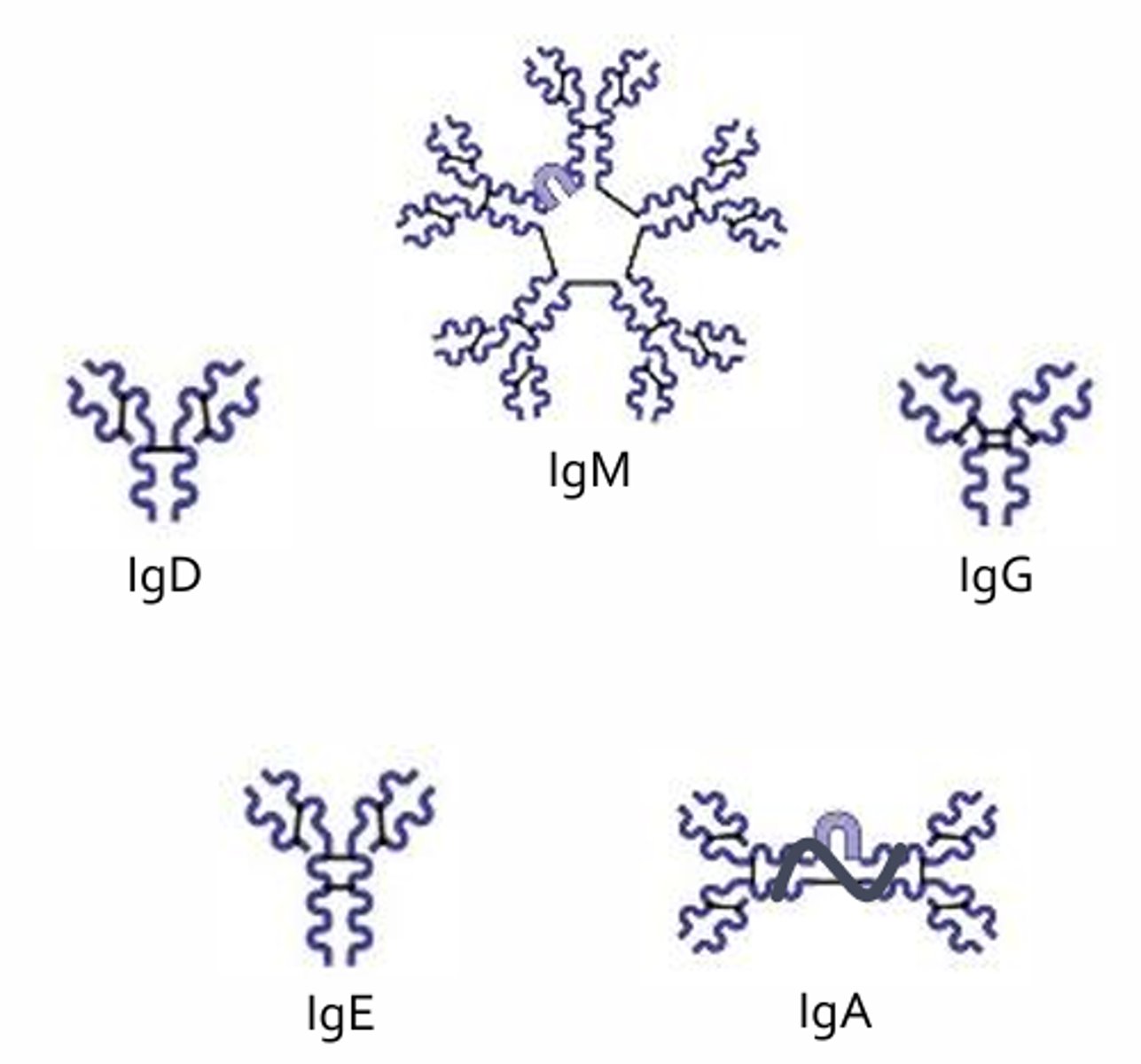
IgD
- Expressed on surface of B-cells
- Thought to participate in B-cell differentiation
- Loss has no appreciable consequences
IgM
- First antibody produced during an adaptive response
- Principle antibody class produced by the fetus
- Consists of 5 antibodies linked with a polypeptide (J chain)
Functions:
- Activate compliment
- Enhance agglutination
- Neutralize toxins, bacteria, and viruses
- Monomer expressed on surface of B-cells
IgG
- Most abundant serum antibody (80-85%)
- Longest lasting antibody (why vaccines work)
Functions:
- Complement activation
- Opsonization of pathogens
- Neutralization of toxin, bacteria, and viruses
- Capable of entering tissue from the bloodstream
- Actively transported across the placenta
IgE
- Implicated in host response to parasite and involved in allergic reactions
- Fc portion of antibody binds Fc-receptors on mast cells, basophils, and eosinophils
- Allergic reactions = Promotes mast cell and basophil degranulation resulting in histamine release
- Host defense against parasites = Activates eosinophils which release enzymes and mediators to combat parasites
IgA
- Found in mucosal secretions (milk, tears)
- Confers protection to GI, resp, urinary, and reproductive tracts
- Confers passive immunity to neonate via breast milk
- Most abundant form is a dimer linked by a J chain
- A secretory component protects antibody from degradation by enzymes found in mucosal surfaces
Functions:
- Agglutination
- neutralization of pathogens
Cells of Adaptive Immune System
Antigen Processing
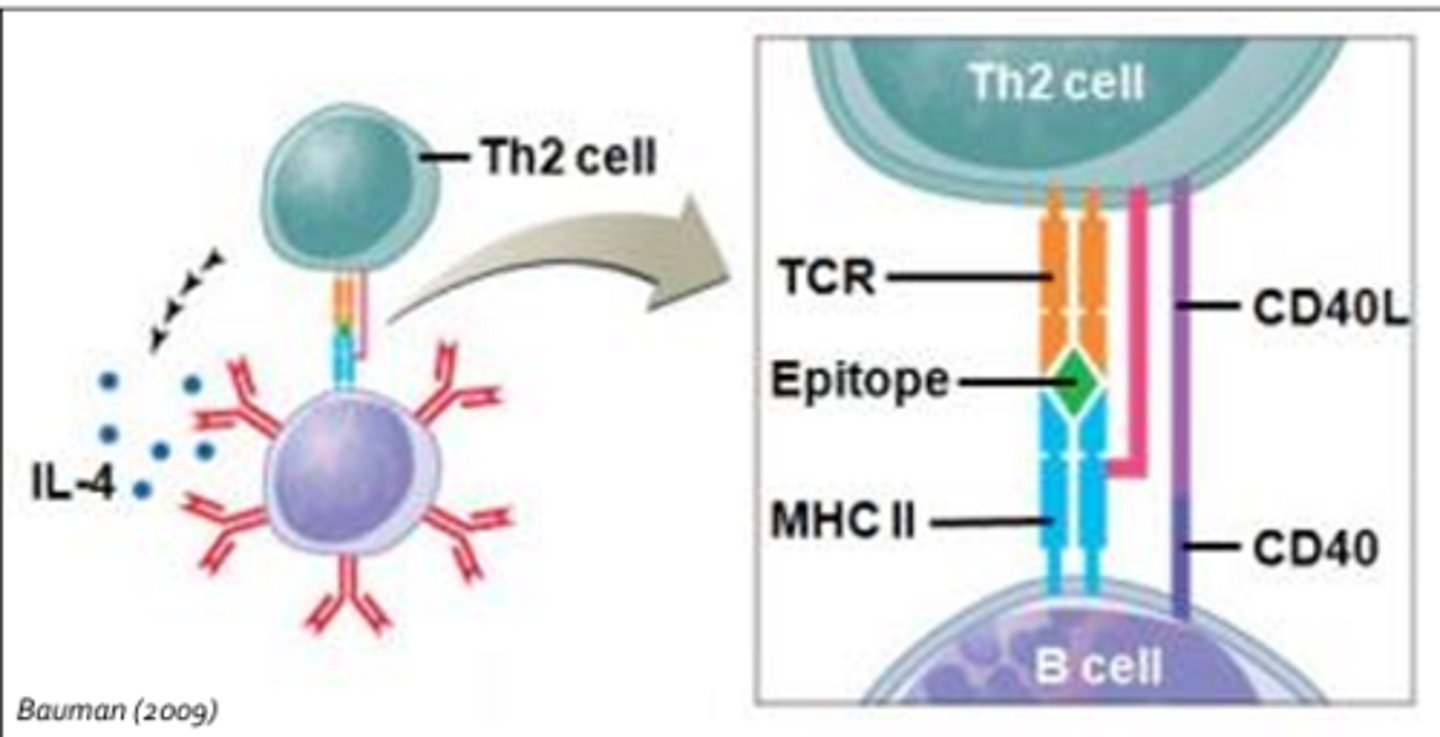
Antigen processing is performed by specialized cells including:
- Dendritic cells (located in skin and mucus membranes; once they encounter antigens they migrate to a nearby lymph node and present the antigen to T-cells and B-cells)
- Macrophages (begin as monocytes in circulation, once it enters tissues it rapidly enlarges and differentiates into a macrophage where it will carry out phagocytic activities)
- B-cells (capable of processing and presenting antigens to helper T-cells; B-cell mediated event = Antigen binds a B-cell receptor, is internalized, processed, and presented on a MHC molecule for recognition by a T-cell receptor)
Role of MHC
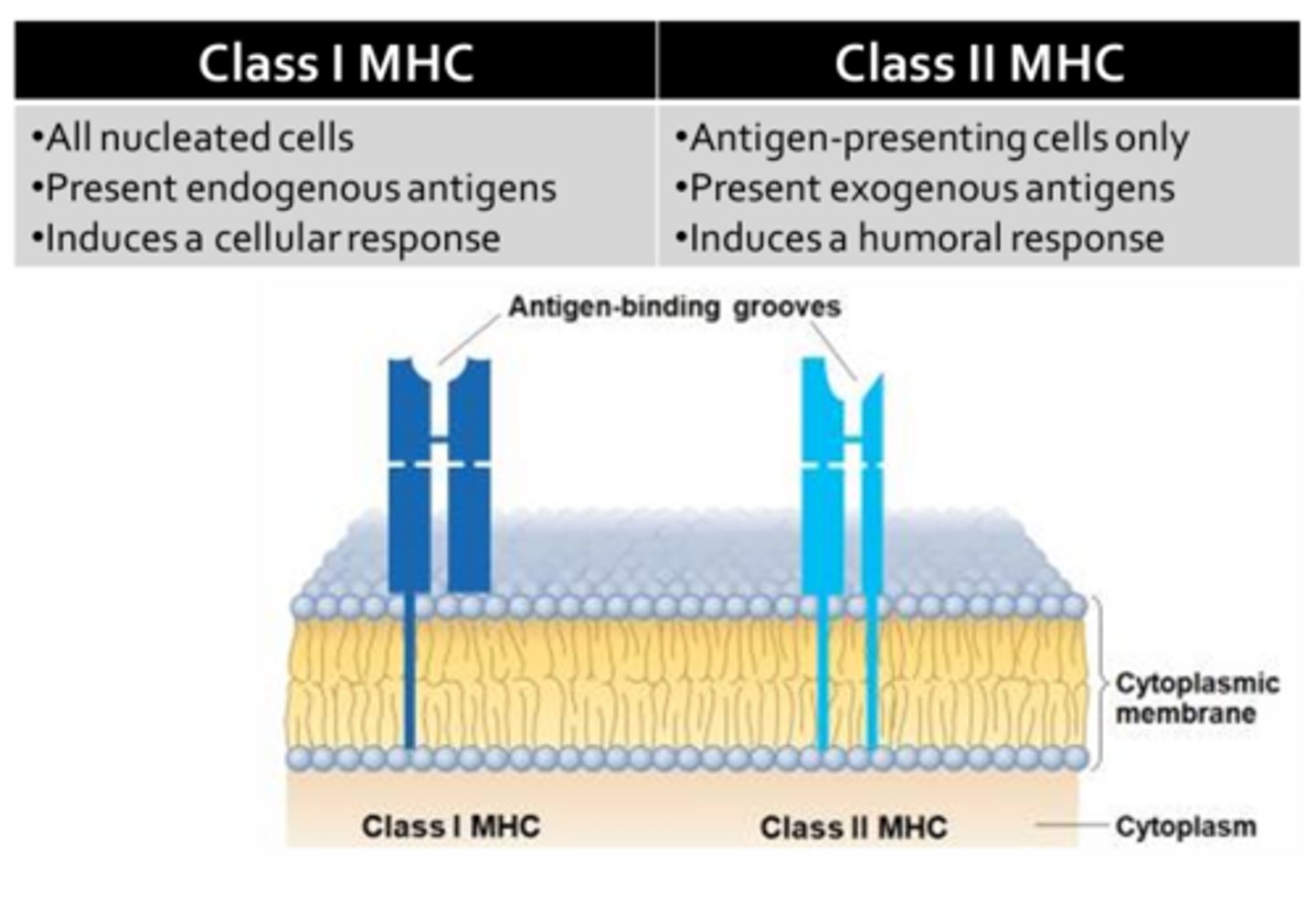
- Class I MHC: Cytotoxic T-cells (cellular; TCR, CD8) (found on virtually all nucleated cells of the body)
- Class II MHC: Plasma cell (humoral) (found only on professional APCs)
*All antigens, whether self or non-self must be presented to T-cells via MHC receptors
*Both antigen processing pathways are employed during infection with a pathogen
- Ex. Host response to Influenza virus = While the cellular arm targets influenza-infected host cells, thereby limiting viral replication, the humoral arm will result in production of anti-influenza antibodies that neutralize free viruses int he tissues and body fluids, thereby preventing the virus from entering and infecting other host cells
MHC Pathways
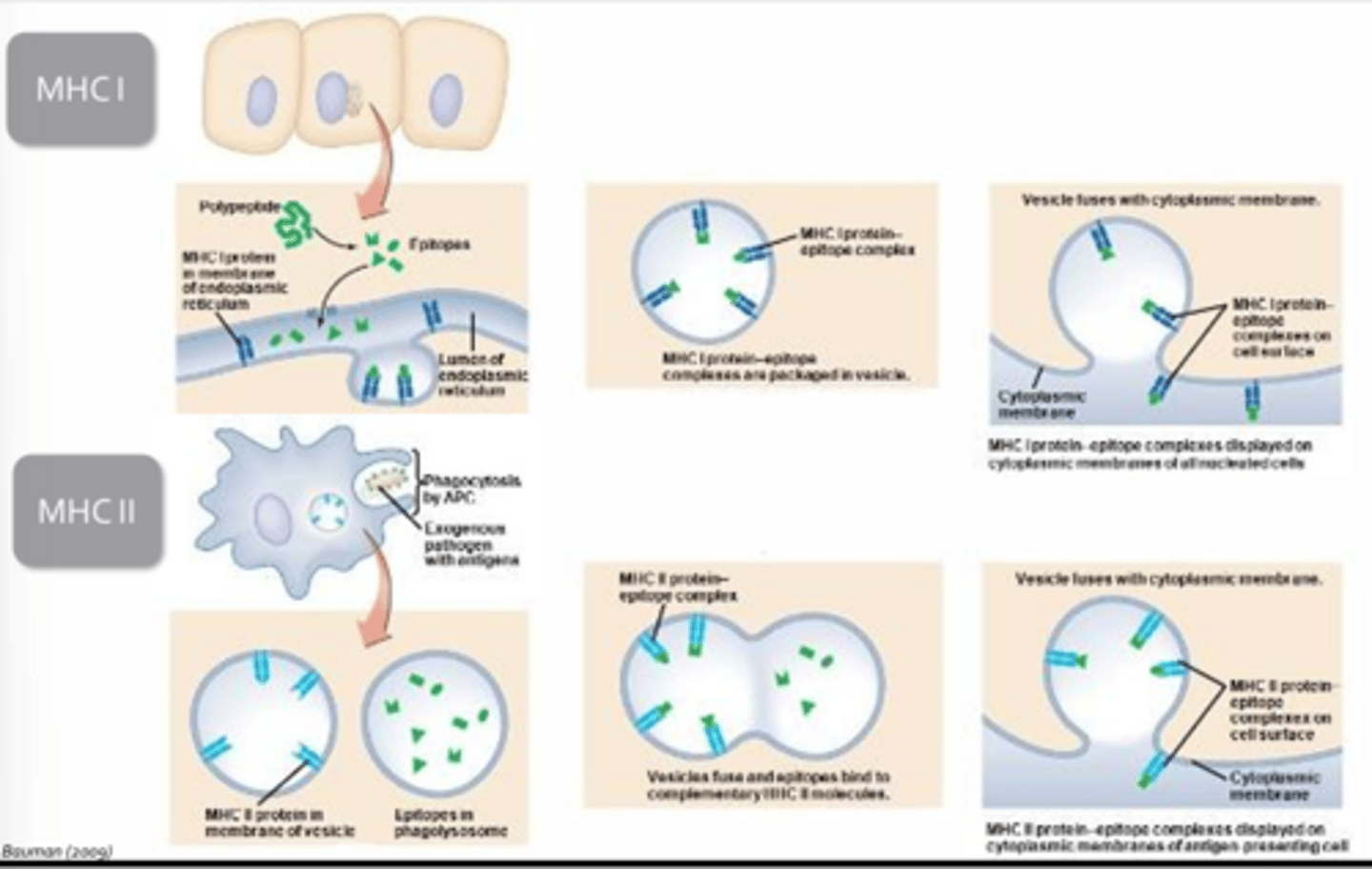
MHC 1:
- Involves processing of endogenous, cytoplasmic proteins which are then loaded onto MHC I within the endoplasmic reticulum
- The MCH-antigen complex is then packaged and transported to the plasmamembrane by vesicles
- Every nucleated cell in the body is constantly sampling and presenting fragments of cytoplasmic proteins via MHC 1
- Induces self-proteins, but viral proteins if cell is infected
MHC 2:
- Exclusive to professional antigen presenting cells
- Exogenous antigens are taken up by the cell through phagocytosis and processed by combining the resultant phagosome with a lysosome to create a phagolysosome
- Once the exogenous proteins have been digested into smaller fragments, the phagolysosome is fused with a vesicle containing MHC 2 molecules
- The antigenic fragments are then loaded onto MHC and sent to plasmamembrane for insertion
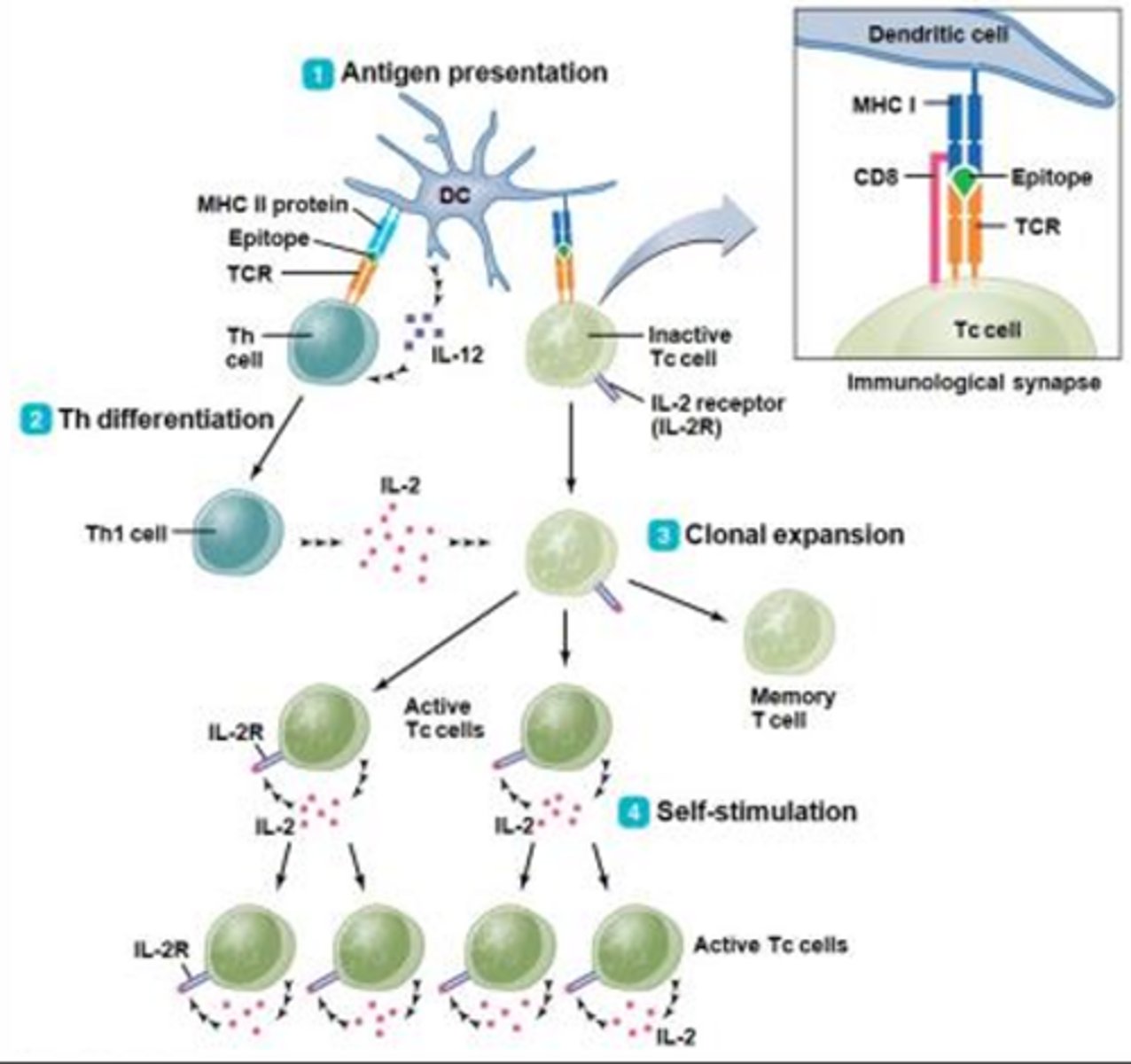
Cellular Immunity in 4 Steps
Step 1 = Antigen Presentation:
- Infected dendritic cells will present antigen via MHC I to cytotoxic T-cells
- The interaction between MHC I and the T-cell receptor is stabilized by CD8
- Signaling between the cytotoxic T-cell and the dendritic cell is not very strong, often requires enhancement from helper T-cells
Step 2 = Helper T-Cell Differentiation:
- For a helper T-cell to become activated, the dendritic cell must present antigen in the context of MHC II (dendritic cell must have engulfed or phagocytosed infected components of host cells at some point)
- When coupled, the helper T-cell will induce the dendritic cell to signal more vigorously to the cytotoxic T-cell, thereby ensuring that a cellular response is evoked
- Helper T-cells also provide aid in the form of cytokines, particularly Il-2 (must be initiated by the dendritic cell through release of IL-2, which is only produced by infected dendritic cells)
Stage 3 = Clonal Expansion of Cytotoxic T-Cells:
- Requires both the coupling of MHC I with the T-cell receptor and IL-2 from activated helper T-cells
- Once activated, cytotoxic T-cells will proliferate to form identical clones that will either become memory T-cells or more cytotoxic T-cells
Stage 4 = Self-Stimulation:
- As clonal expansion proceeds, many of the replicating cytotoxic T-cells will begin to secrete their own IL-2, thereby obviating any need for further dendritic or helpful T-cell support
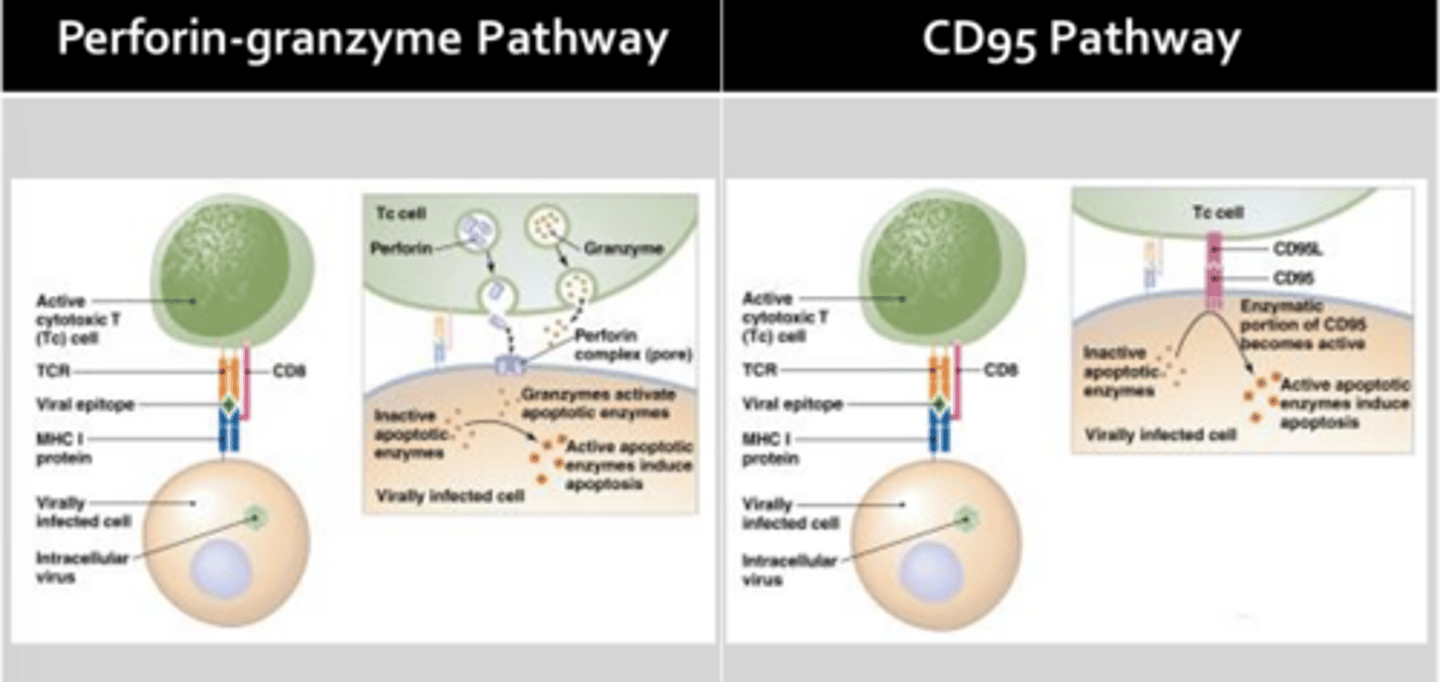
Paths of Cytotoxic T-Cell-Mediated Cellular Toxicity
Perforin-Granzyme Pathway:
- Involves the release of pore-forming proteins from cytotoxic T-cells (perforin) which create a channel through the target cell membrane
- Channel allows granzyme to enter the target cell where it activates apoptotic enzymes and also induces cell lysis by allowing water to enter the cell
CD95 Pathway:
- Involves activation of target cell CD95 receptors by CD95 ligand expressed on the surface of activated cytotoxic T-cells
- The intracellular portion of the CD95 receptor possesses enzymatic activity, that when engaged, will result in the activation of pro-apoptotic enzymes (cell commits molecular suicide)
- All cells in the body express CD95 and are potential targets for cell mediated immunity
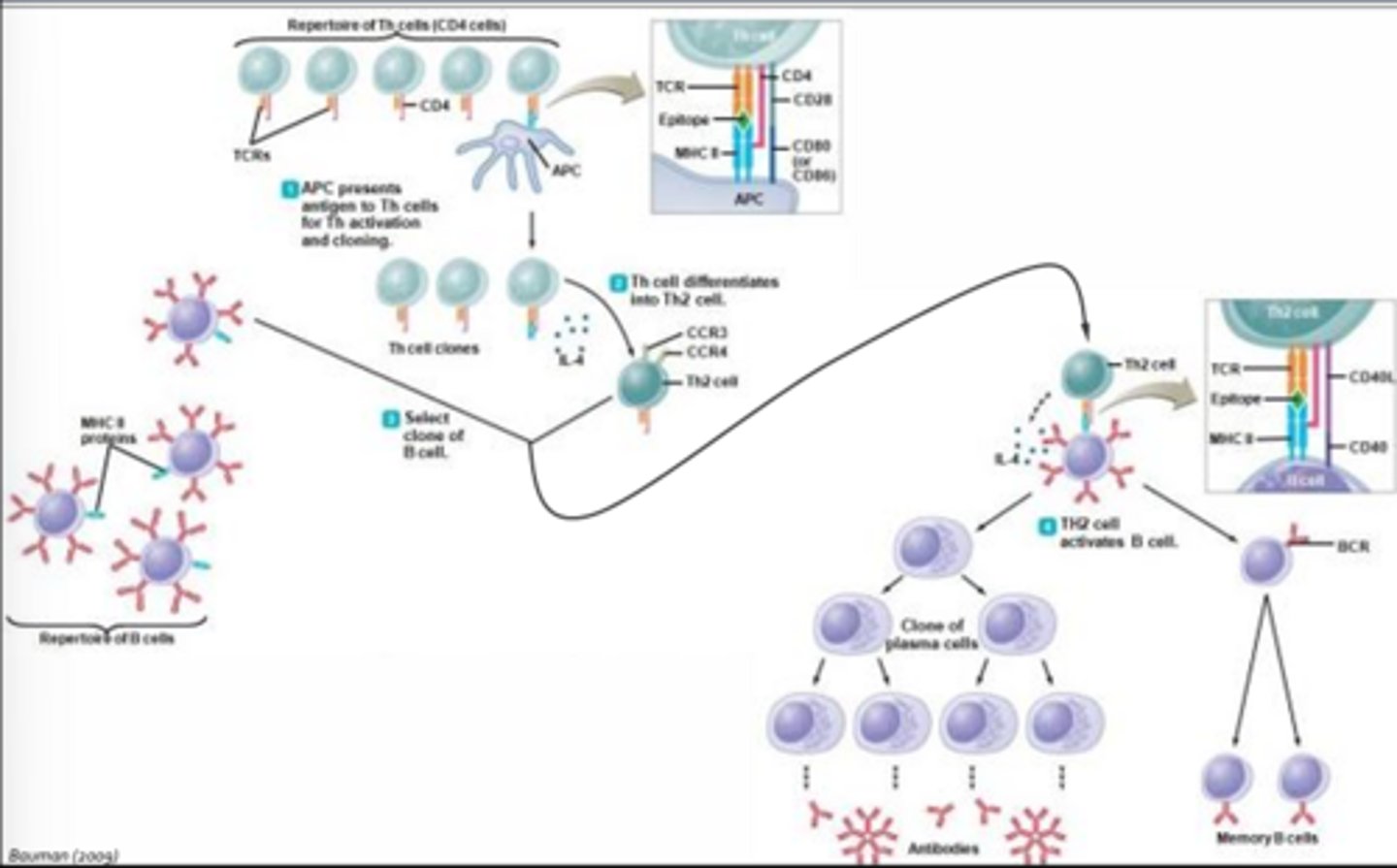
Humoral Response - T-Cell-Dependent
- Generated with or without help from T-cells
- T-cell dependent humoral responses involve the recruitment of antigen-specific helper T-cells that provide cytokines and other signals to help drive B-cell activation and differentiation
Step 1 = Antigen Presentation:
- After acquiring antigens in the skin or mucus membranes, dendritic cells will migrate to a local lymph node, where they interact with a helper T-cell
- Presentation of the antigen must occur in the context of MHC II to evoke a humoral response
- Interaction of the T-cell receptor with the antigen MHC complex is insufficient to activate the helper T-cell (costimulation is required)
- This is provided by interaction of CD80 on the dendritic cell with CD28 on the helper T-cell (only mature dendritic cells express CD80)
Step 2 = Differentiation of the Activated Helper T-cell into a Th2 Cell (requires Il-4):
- Il-4 is thought to be produced by cells of the innate immune system (i.e. mast cells), but will later be secreted by activated Th2 cells
Step 3 = Clonal Selection of B-cells:
- Only B-cells that have encountered and processed foreign antigen will become primed for antibody production
Step 4 = Activation of Primed B-cells which Require Input from Helper T-cells:
- Input is provided through interaction of the Th2 T-cell receptor with that of the antigen MHC complex expressed by B-cells (stabilized by CD4 on the helper T-cell)
- To fully activate a B-cell, co-stimulation is required in the form of CD40-ligand (CD40L) (expressed by the helper T-cell and Il-4)
- Most B-cells become plasma cells which are capable of secreting a lot of antibodies, bot only live for a few days to weeks
- Other B-cells will become memory cells which may reside in the lymph node for decades
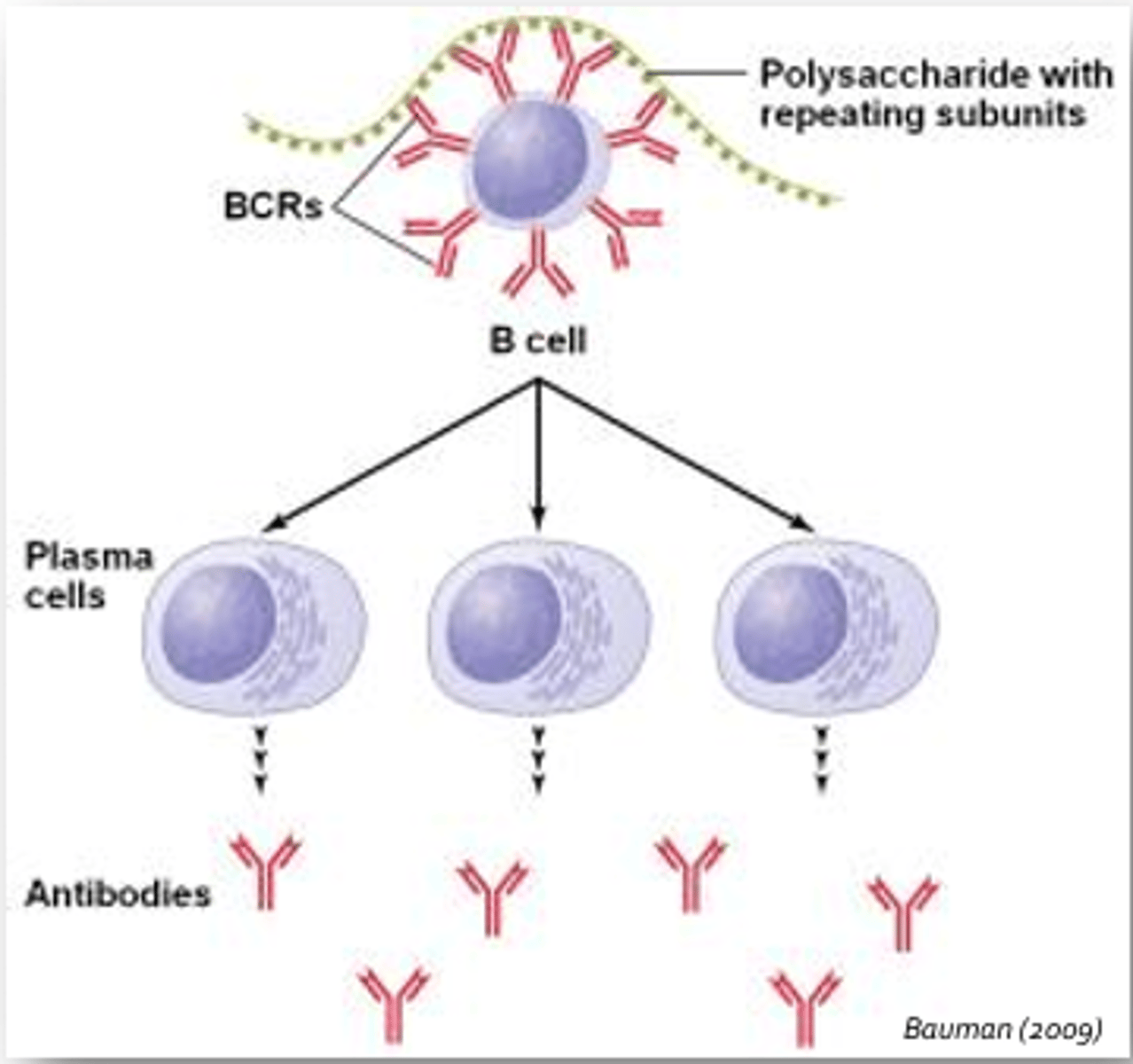
Humoral Response - T-Cell-Independent
- Occurs without help from T-cells
- Initiated by cross-linking of B-cell receptors by antigen
- Initiated by cross-linking of more than one B-cell receptor by repeating epitopes contained on evolutionarily conserved structure such as the capsule of the influenza virus, lipopolysaccharide expressed in the outer cell wall of gram-negative bacteria, bacterial flagella, and other viral capsids
- Less intense and not much immunologic memory; time course similar
Typical antigens include:
- Influenza virus capsule
- LPS of gram negative bacteria
- Bacterial flagella
- Other viral capsids
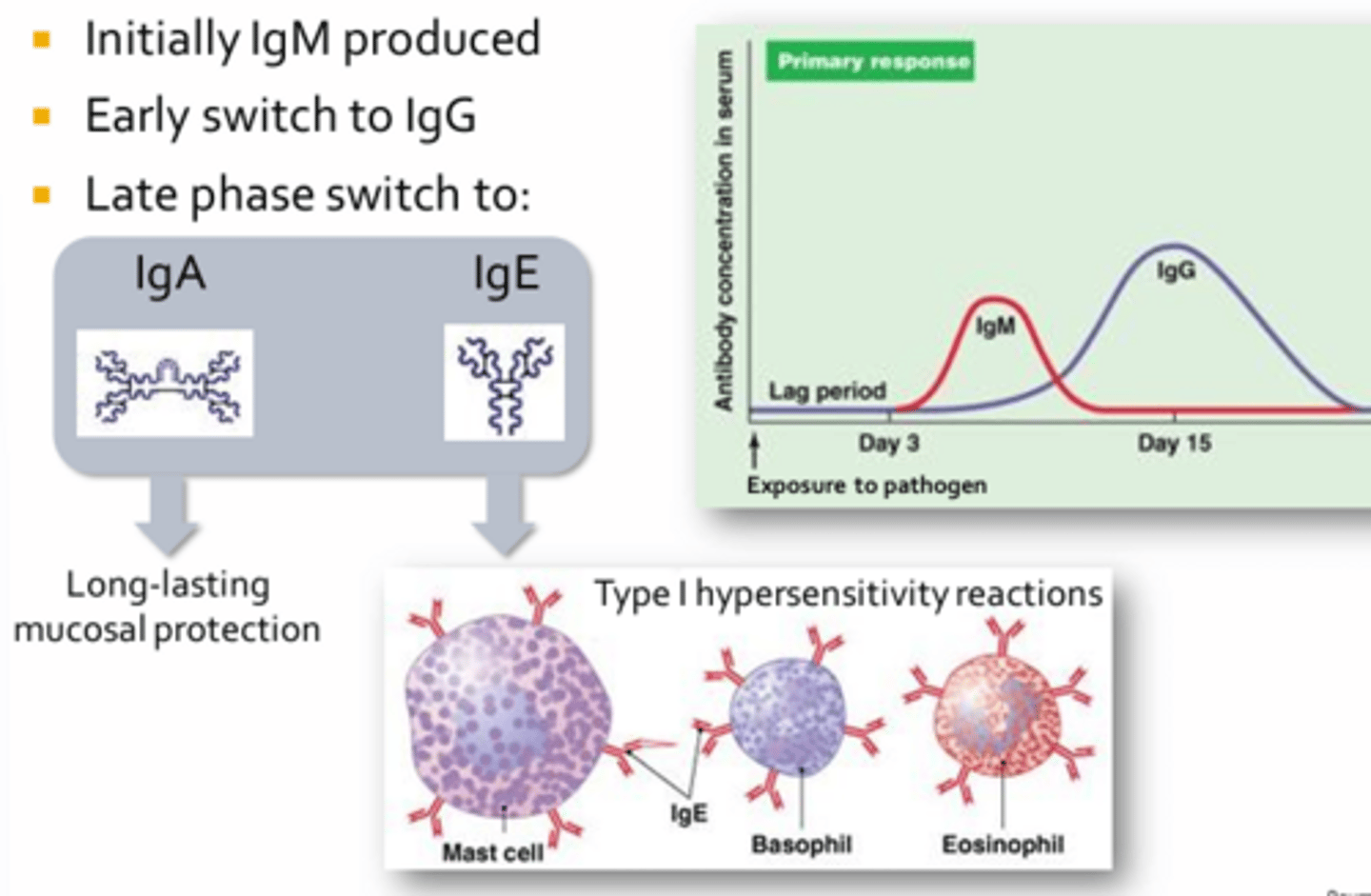
Class Switching
- Plasma cells will undergo class switching, where the type of antibody produced will change
- Largely involves an alteration in the stem or Fc region of an antibody
- All humoral responses begin with production of IgM antibodies which are largely restricted to the circulation and exhibit a broader binding specificity than other antibody classes
- Then, many plasma cells begin to secrete IgG
- These antibodies tend to have a greater specificity and affinity for antigens than IgM
- A small proportion of plasma cells will undergo a second class switch during the tail-end of the humoral response
- This class switch may result in the secretion of IgA antibodies that confer long-lasting protection to mucosal surfaces including the GI, resp, reproductive, and urinary tracts
- Late phase class switching may result in the production of IgE, which coats the surface of mast cells, basophils, and eosinophils (role in hypersensitivity reactions)
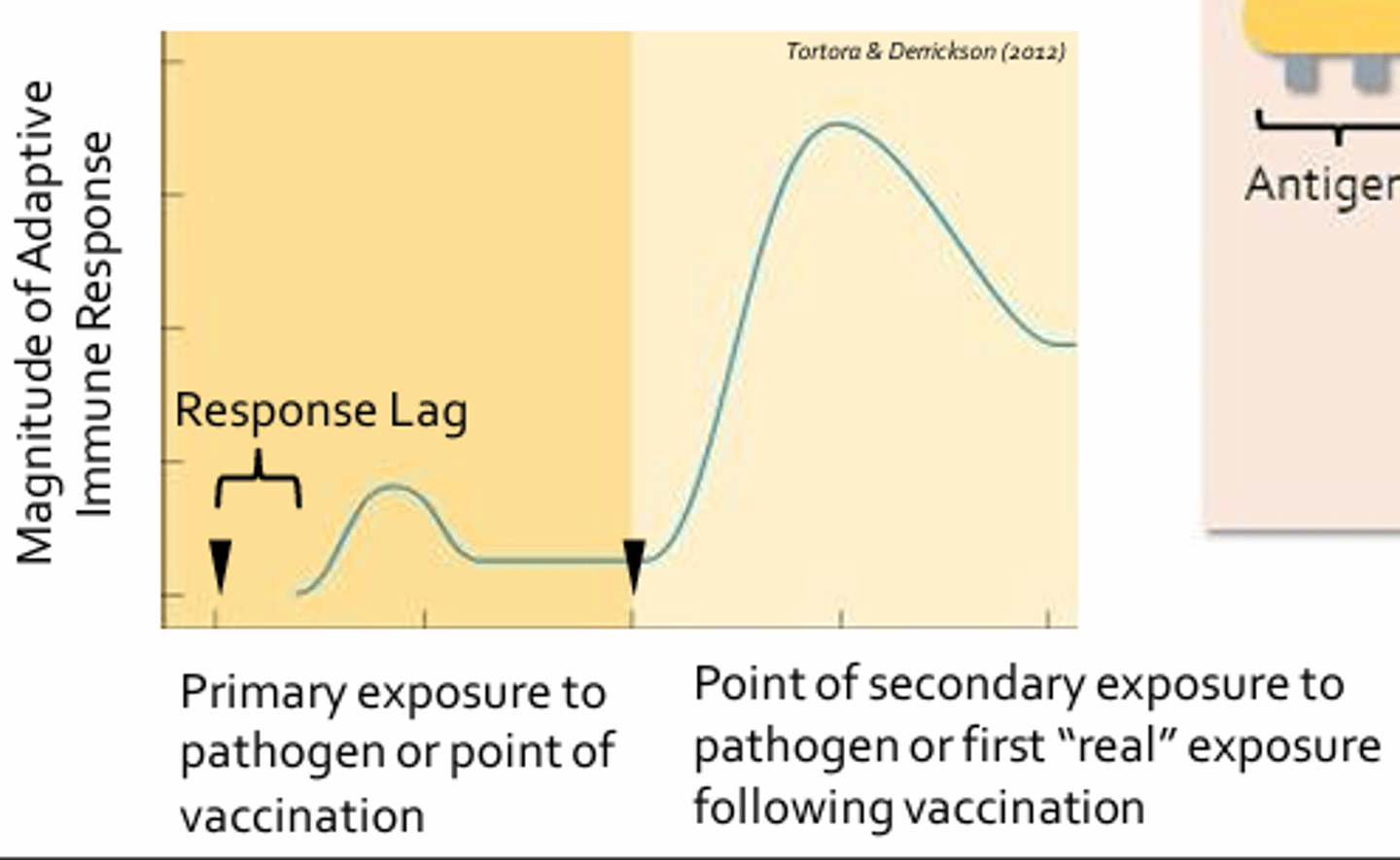
Immunologic Memory
- Hallmark of adaptive response
- Allows for quicker and more intense adaptive response upon repeat exposure to a pathogen
- Mechanism that confers protection to an individual following vaccination (basis for vaccination)
- Graph = Change in antibody concentration or titre measured in the peripheral blood of an individual following initial and secondary exposure to the same pathogen (primary and secondary responses)
- Initial exposure to a pathogen (or vaccine) results in an adaptive responses that takes time to reach full potency, indicated by the lag phase (usually 3-6 days)
- This is due to the fact that antigen-presenting cells must migrate from the site of infection to a nearby lymph node and that Th2 differentiation and clonal expansion of B-cells occurs (takes time)
- Subsequent exposure to the same pathogen (or the initial exposure to pathogen following vaccination) results in a response that is bigger, better, and faster (only 2-3 days to develop; due to the large pool of memory T-cells and B-cells generated during the initial response)
- Antibody titres may increase more than x2000 compared to the primary response and may clear the pathogen so quickly that an infected individual may not develop any signs or symptoms of infection
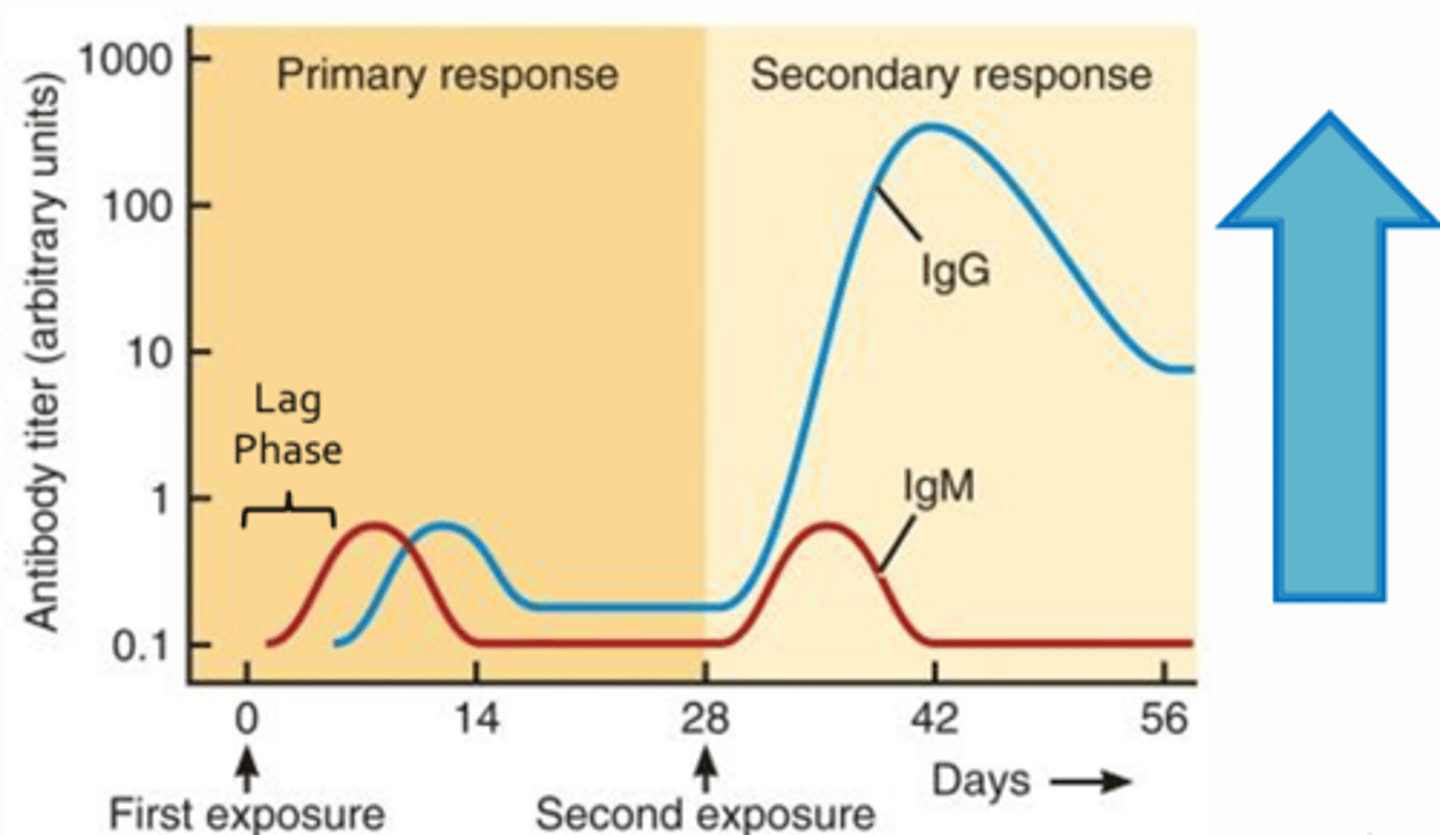
IgM vs. IgG
- The initial antibody produced during the primary response is IgM, while IgG production dominates the secondary response
- IgM is restricted to the circulation, while IgG can enter most tissues of the body and is able to cross the placenta
Hypersensitivity Reactions
- Exaggerated or inappropriate immune response to innocuous antigens
- The adaptive immune system over-reacts to harmless foreign antigens that are not capable of replicating or causing a an infectious disease
- Ex. Peanuts, shellfish, pollens, dust, mites, plant oils, metals, insect venoms, drugs
Types of Hypersensitivity Reactions
- Type I: Immediate/IgE-mediated
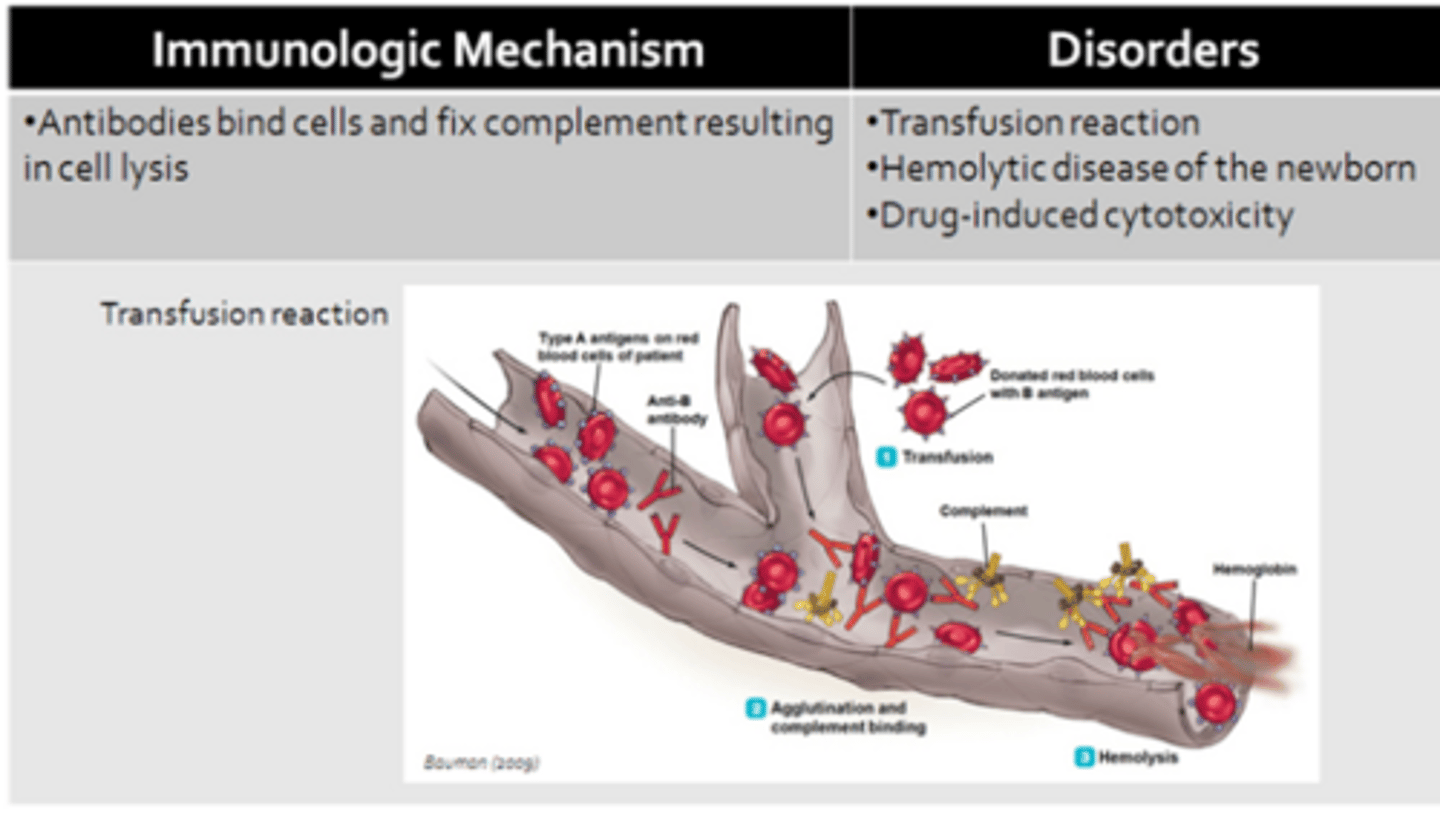
- Type II: Cytotoxic
- Type III: Immune-complex-mediated
- Type IV: Delayed/Cell-mediated
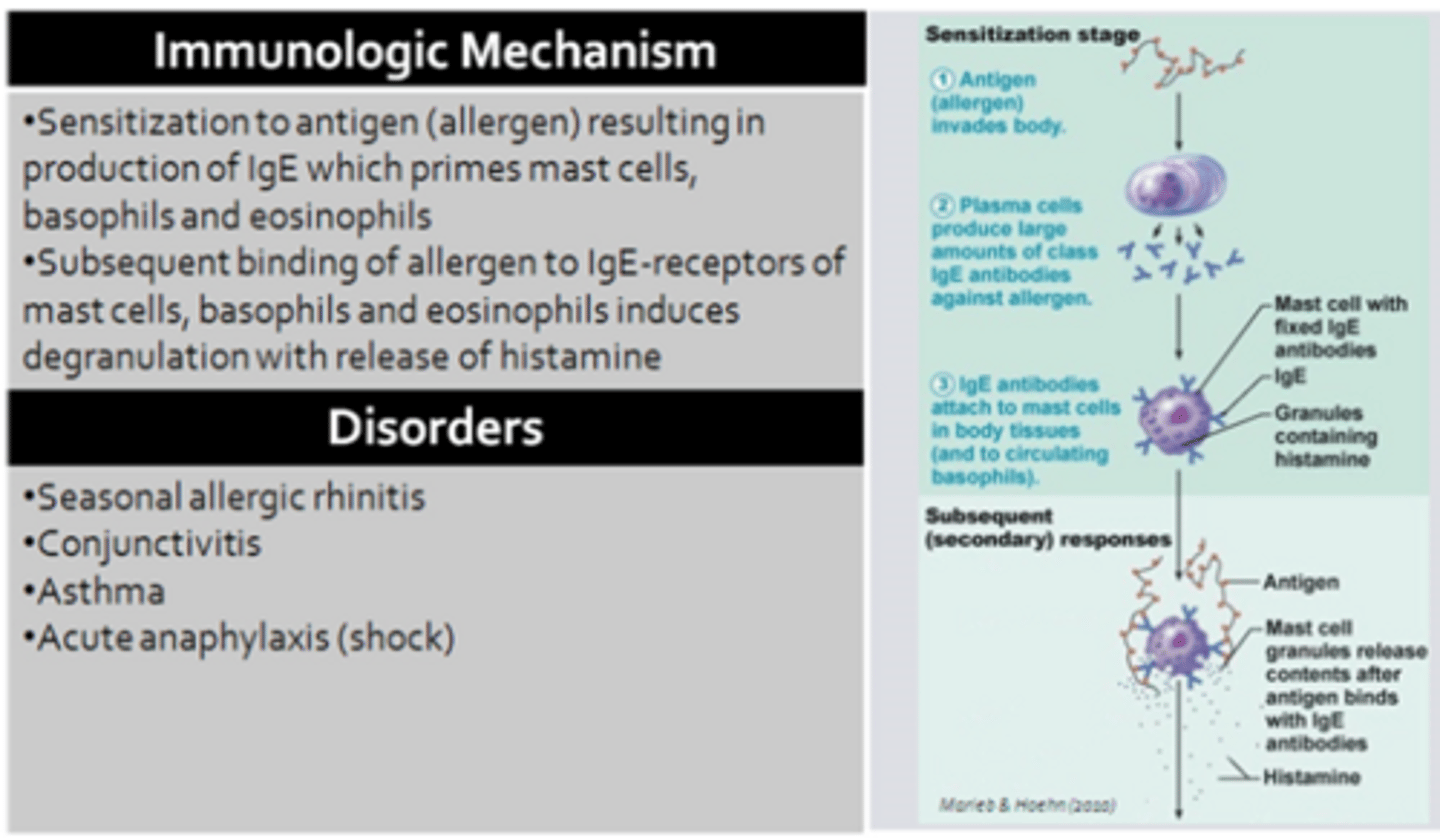
Type I: Immediate/IgE-Mediated
Type II: Cytotoxic
Type III: Immune-Complex-Mediated
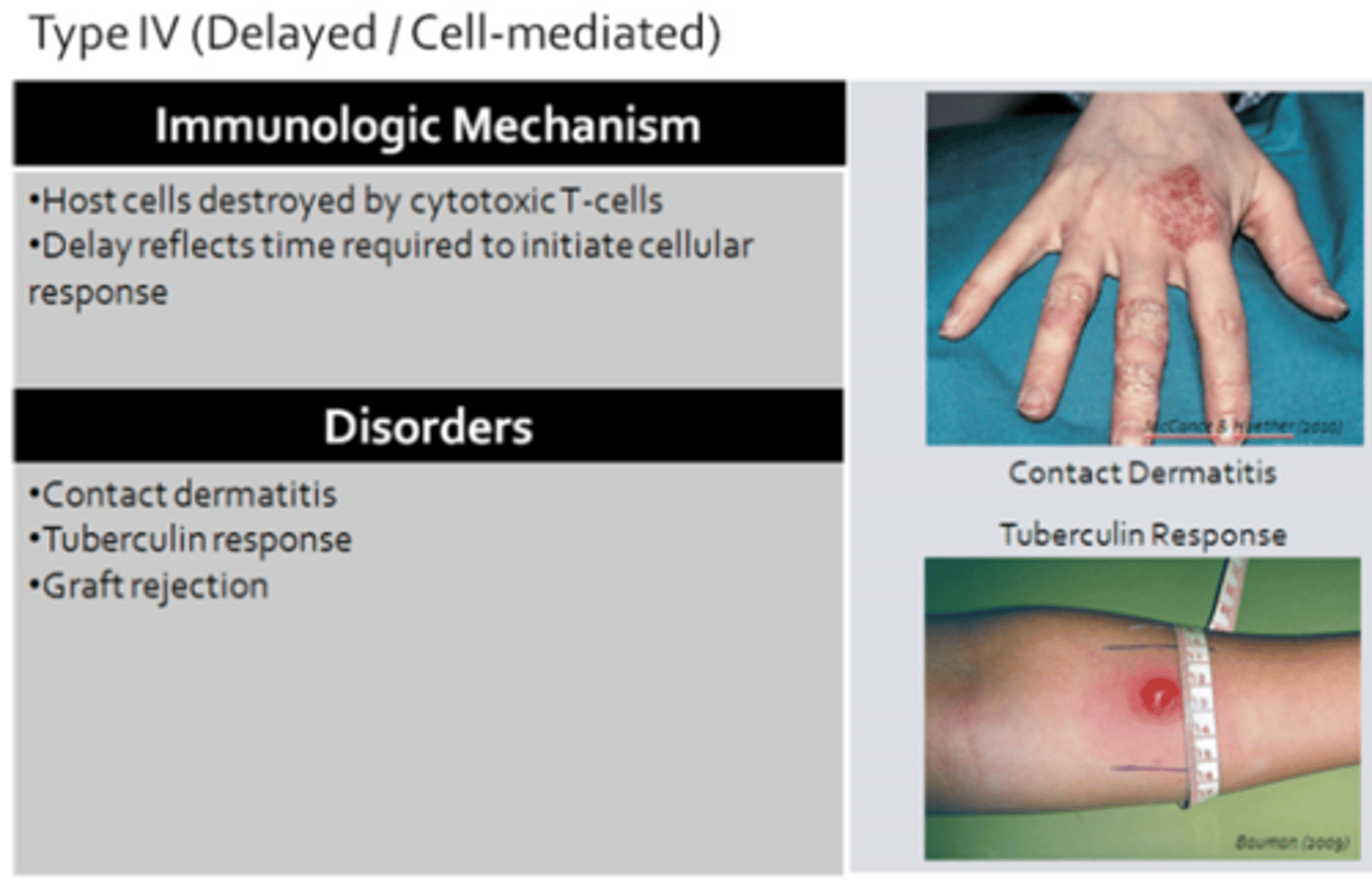
Type IV: Delayed/Cell-Mediated
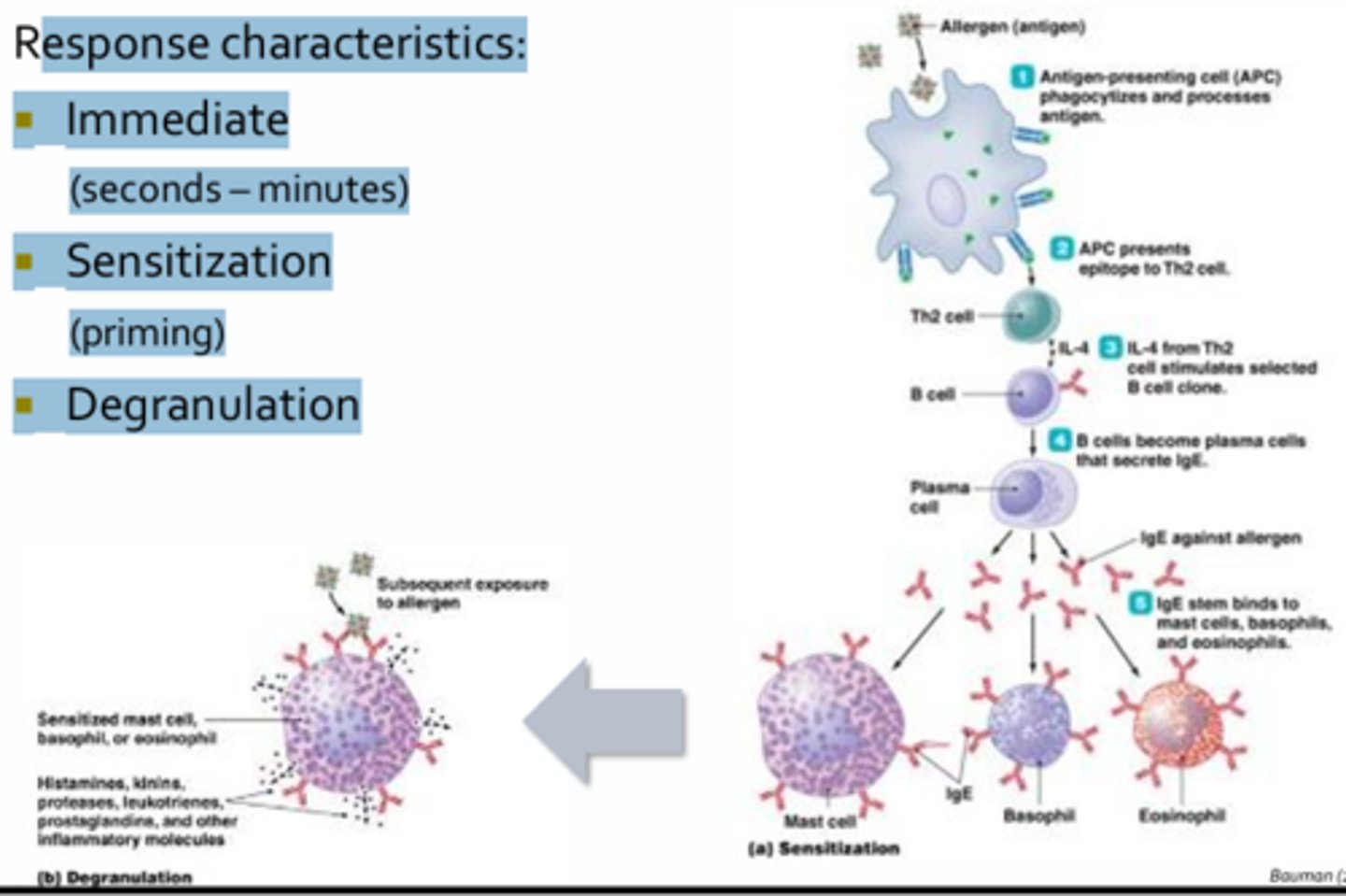
Type I Hypersensitivity
Response Characteristics:
- Immediate (seconds –minutes)
- Sensitization (priming): Naive T-cells and B-cells come in contact with an allergen that generates a humoral response predominated by the production of Ige antibodies
- Response it driven by Th2 helper T-cells which secrete cytokines such as IL-4
- In allergic individuals, atopics, IgE class switching occurs
- IgE antibodies end up coating the surface of mast cells, basophils, and eosinophils via high-affinity Fc receptors (priming these granulocytes)
- Some may become sensitized following a single exposure to an allergen, while sensitization in other may require several repeat exposures over a course of years
- Once an individual has been sensitized, subsequent exposure to the same allergen will result in cross-linking of IgE receptors on primed cells, resulting in immediate degradation
- Degranulation: Involves the release of preformed vesicles containing copious amounts of mediators (ex. histamine)
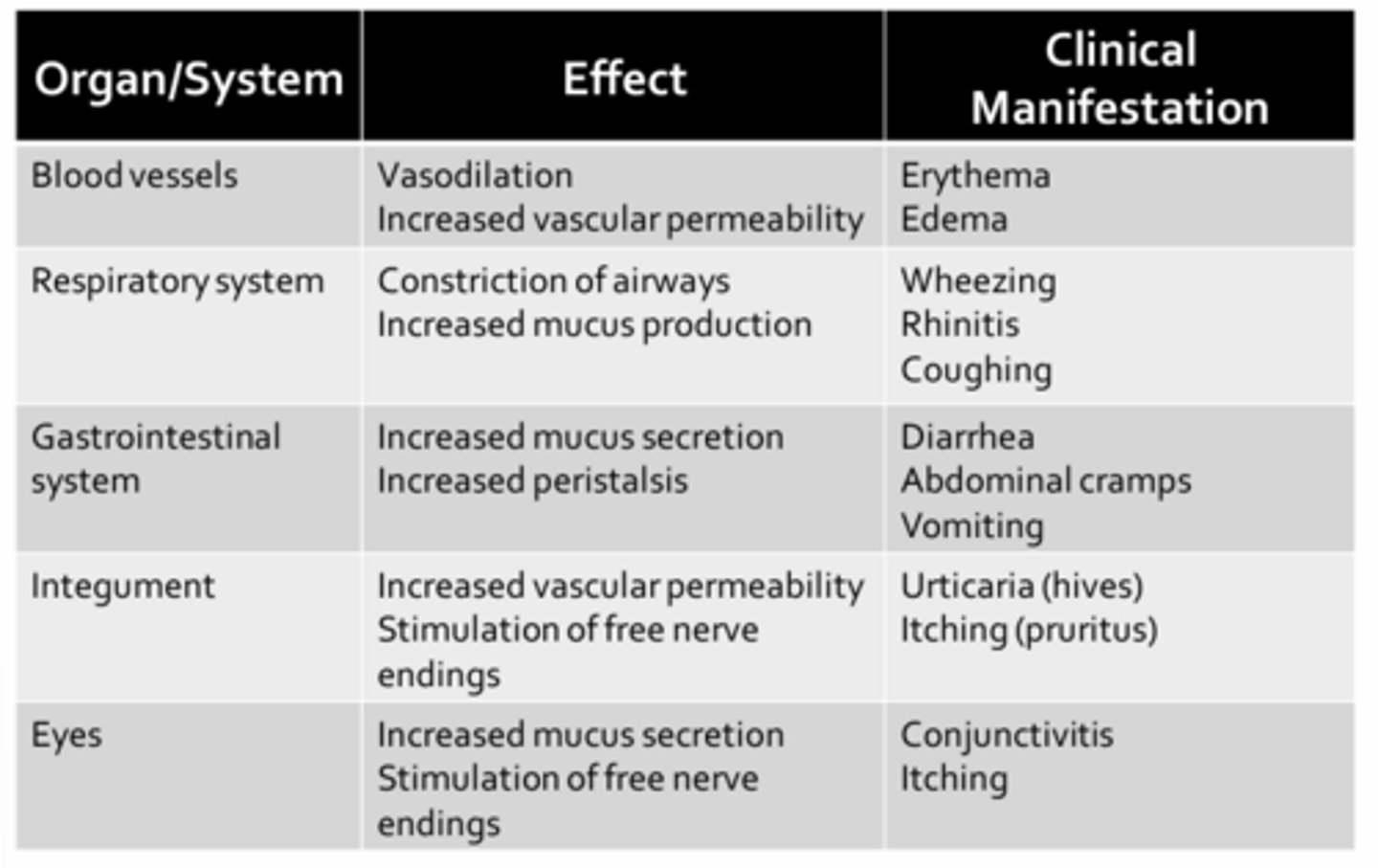
Effects of Histamine
- Promotes vasodilation and increases vascular permeability
- Tissues become red and swollen
- Constriction of airway smooth muscle contributes to wheeze
- Mucus secretions due to stimulation of goblet cells causes rhinitis in the upper airways or cough and congestion in the lower airways
- GI complications include diarrhea, abdominal cramps, vomiting due to intense stimulation of peristalsis
- Urticaria (hives) are red, raised areas on the skin that are accompanied by intense itching (pruritus) (due to increased vascular permeability and stimulation of free nerve endings, both mediated by histamine)
- Inflammation of the mucus membranes of the eye (conjunctivitis) may occur accompanied by itching
Anaphylactic Reactions
- Severe type I hypersensitivity reactions
- Due to massive systemic release of inflammatory mediators (histamine) from mast cells and basophils
- Triggers: Bee venom, shellfish, antibiotics, anesthetics (local), peanuts, vaccines, iodine, narcotics (morphine)
Complications of Anaphylaxis
Acute bronchospasm and swelling of airways due to histamine:
- Bronchospasm: Due to histamine-evoked airway smooth muscle contraction
- Airway swelling: Due to increased vascular permeability in the mucus membranes of the lower airway
- Laryngeal edema
- Bronchial mucus hypersecretion (due to histamine resulting in congestion)
- Impaired gas exchange = Acute anaphylaxis
System wide vasodilatation and increased vascular permeability (due to massive release of histamine from mast cells and basophils present in circulation):
- Expansion of vascular compartment (normal blood volumes are insufficient to maintain BP resulting in complete circulatory collapse or vascular shock)
- Massive reduction in blood pressure
- Vascular shock = Anaphylactic shock
Treatment of Anaphylaxis = Epinephrine
- Non-selective adrenergic agonist
- Emergency treatment choice
- Stimulates both alpha and beta adrenoreceptors to reverse effects of histamine
Improves gas exchange:
- Dilates airways (b2 receptors): Promotes airway relaxation, improving airway caliber and reducing airway obstruction
- Decreases mucus production
Restores blood pressure and tissue perfusion:
- Contracts vascular smooth muscle (a1) (increases total peripheral resistance)
- Decreases vascular permeability (a1) (retaining fluids within the vascular compartment)
- Improves myocardial contractility (b1) (direct effect on CO)
*All help to restore BP and tissue perfusion
Anaphylaxis Protocol
- Focus on resp function, CO, and tissue perfusion maintenance
- Patient in supine position with feet elevated (promote venous return)
- Give epinephrine (adrenaline): 1:1000 aqueous solution (max 0.5 mL per injection IM); Repeat twice if needed (same dosing at 10-15 minute intervals)
- Administer Diphenhydramine (Benadryl) during severe anaphylactic reactions: Max 50 mg IM; No repeats (antihistamine that blocks H1 receptors)
- Provide oxygen: Improve blood oxygen saturation if airways were compromised
- Establish intravenous line: To help improve BP
- Monitor and document vital signs frequently
Prophylactic Measures
1. Avoidance: Food challenge, Skin test, IgE titres
2. Antihistamines
3. Corticosteroids
4. Desensitization: Produce blocking antibodies; Increase Treg population
Avoidance Measures
- Skin-prick tests are the preferred method since oral challenge may induce an anaphylactic response
- Lab tests measure serum IgE levels are not as sensitive
- Prior sensitization to an antigen will result in a typical wheal and flare reaction
- Multiple antigens can be tested at once
Antihistamine Measures
- Diphenhydramine (Benadryl) = Selectively block H1 receptors
- Best used in a prophylactic manner, as they do not reverse binding of histamine to H1 receptors
Corticosteroid Measures
- Ex. Prednisone
- Used to suppress immune responses, thereby limiting the degree of sensitizing IgE production and dampen inflammation
- Asthmatics are often prescribed a combination corticosteroid and bronchodilator such as Advair which contains the glucocorticoid fluticasone and a long-acting beta 2 agonist (salmeterol) to help prevent airway constriction
Desensitization Measures
- Lengthy process involving injection of minute amounts of antigen with a slow increase in concentration over a long period of time
- The immunologic mechanism of allergic desensitization is thought to involve either production of blocking antibodies (act to neutralize or coat antigens before they can bind mast cell or by stimulating the expansion of regulatory T-cell populations that suppress IgE production)
PACE (peanut allergen immunotherapy clarifying the evidence) Report:
- Reviewed the safety and efficacy of oral immunotherapy vs. allergen avoidance or placebo for peanut allergy
- Study affirmed that oral immunotherapy is effective in inducing desensitization, current desensitization protocol were also associated with an increased risk and frequency of anaphylaxis and epinephrine use during the build-up and maintenance phase
- Future studies should focus on quality of life and reducing the frequency of adverse events
Dietary Allergens in Infancy
- Waiting to give infants allergens can deprive their immune system of stimuli necessary to develop tolerance against these innocuous antigens
Canadian Pediatric Society recommendations:
- Do not restrict maternal diet in pregnancy and during lactation (no evidence that avoiding milk, egg, peanut, or other allergens during pregnancy helps to prevent allergy)
- Breastfeed exclusively for the first six months (total duration may be more protective than exclusive breastfeeding)
- Breastfeeding exclusively for at least 3-4 months decreases the incidence of eczema and wheeze in the first 2 years of life
- Choose a hydrolyzed cow's milk-based formula (more effective than partially hydrolyzed cow's milk in preventing atopic dermatitis)
- Do not delay the introduction of any specific solid food beyond six months of age (could actually increase the risk of developing a food allergy; does not prevent atopic disease)
- Randomized trial reported a lower frequency of peanut allergy and modulated immune responses to peanut allergens among children at high risk for allergy with early introduction of peanuts (4-11 months)
- Infants with severe eczema, egg allergy, or both, has an increased risk of developing an allergy to peanuts at 60 months of age if they avoid ingesting peanuts before 11 months of age
- Prevalence of peanut allergies at 60 months in the avoidance group was 35%, while those in the early introduction group was 10%
Acquired Immunodeficiency Syndrome (AIDS)
- Due to infection with HIV
- HIV primarily targets CD4+ T-cells resulting in a deficiency in adaptive response
- A syndrome
- Clinical signs late in disease, until untreated HIV infection has resulted in a loss of at least 2/3 of the bodies helper T-cell population
- The dramatic loss of helper T-cells during the course of HIV infection often results in a reduction in cellular immunity first (can cause emergencies such as Kaposi's sarcoma in AIDS patients)
- Death from rare cancers and opportunistic infections
- Mechanism of action of various antiretroviral medications (ART)
- Following the breakdown of both branches of immunity, AIDS patients often end up succumbing to opportunistic infections such as pneumocystis pneumonia, resulting in death
Malignancies

Opportunistic Infections
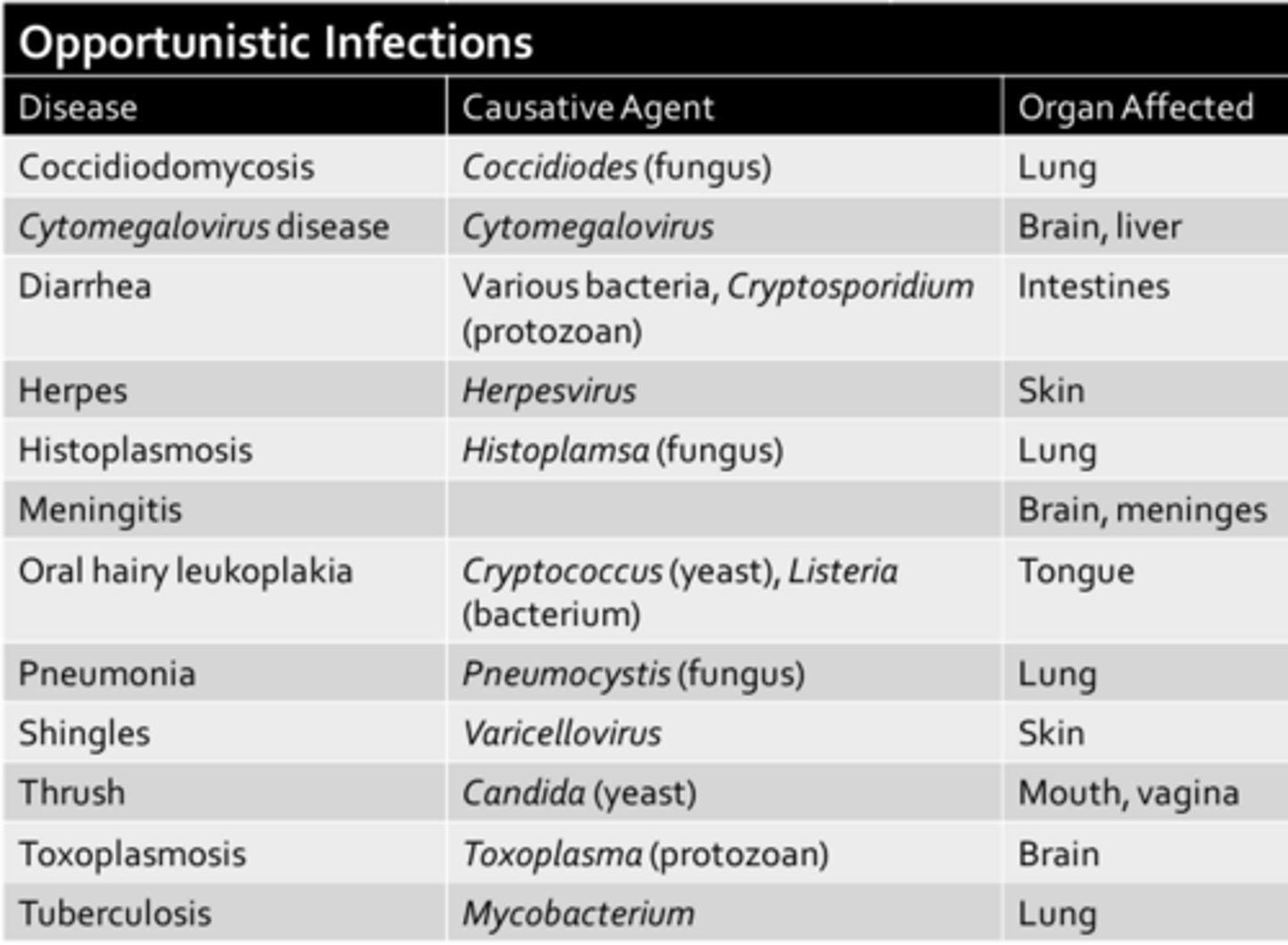
AIDS
- Acquired immunodeficiency syndrome
- Caused by infection with the human immunodeficiency virus
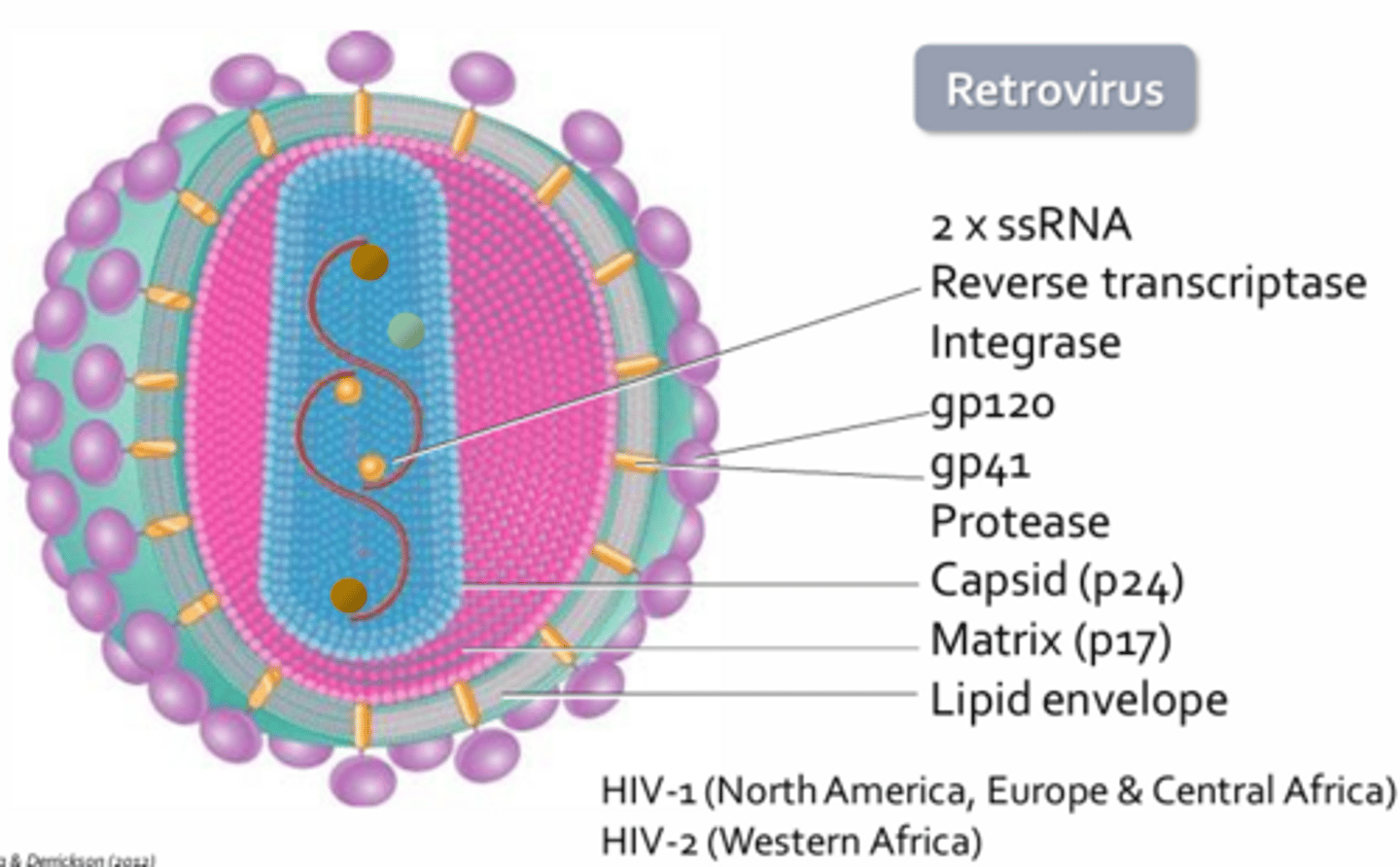
Structure of HIV
- HIV = Lentivirus = Retroviruses
- HIVs genome is present on 2 single stranded RNA molecules
- Surface glycoproteins gp120 and gp41 which aid in viral attachment and entry into host cell respectively
- Protease, processes viral proteins produced during replication to allow for final viral assembly
- Other proteins make up the caspid and matrix of the virus which help to protect the nucleoside core
- Lipid bilayer acts like a cloaking device to hide the virion from host immune cells
HIV-1:
- Most prevalent in North America, Europe, and Central Africa
- Replicates more quickly than HIV-2
HIV-2:
- Predominantly found in West Africa
Viral Replication of HIV #1
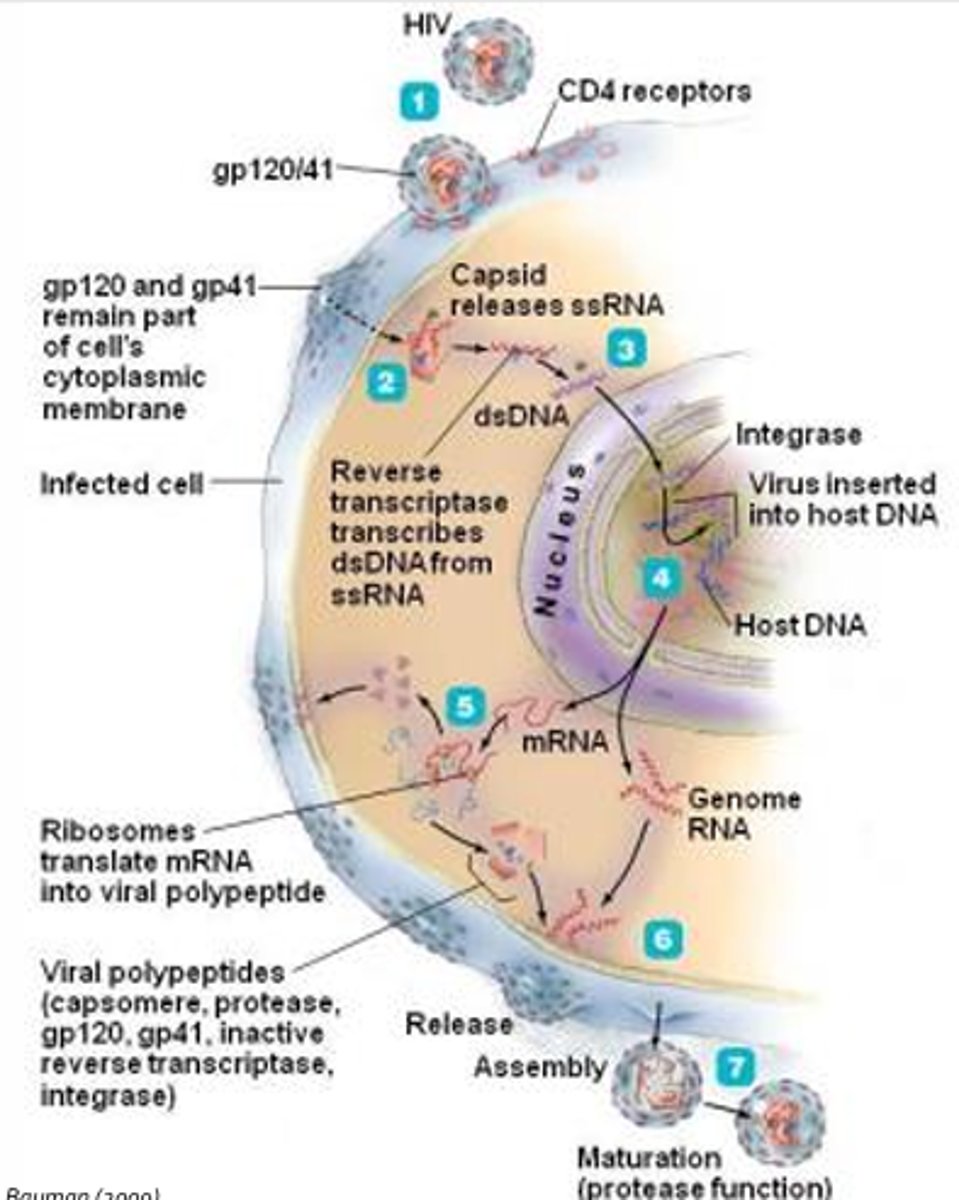
1. Attachment (of virus to a host cell; attachment is mediated by the interaction between viral gp120 and host cell CD4 receptors) (HIV preferentially infects helper T-cells, macrophages, and dendritic cells which all express CD4 to various extents)
2. Entry: Gp120 must bind a chemokine receptor such as CXCR4 or CCR5 found on target cells (acts to remove gp120 allowing gp41 to mediate fusion of the viral envelope with the host cell plasma membrane; gp41 will remain in the host cell membrane where it can promote cell fusion to spread infection)
3. Synthesis of viral DNA: Reverse transcriptase will make a single-stranded copy of DNA from single-stranded viral RNA to create a hybrid molecule consisting of one single0strnaded RNA molecule and on single-stranded DNA molecule (RNA is then degraded by reverse transcriptase, leaving a single-stranded DNA molecule)
4. Integration: One of the components packaged within the viral capsid, helps to insert double-stranded proviral DNA into the host cell genome (occurs 72 hrs following infection; once integrated the provirus will be part of the host cell genome for life and may remain dormant for years)
Viral Replication of HIV #2
5. Synthesis of viral RNA and polypeptides: Messenger RNA is used to synthesize viral polypeptides that consist of capsomeres, gp120, gp41, reverse transcriptase and integrase (these polypeptides need to be cleaved by viral protease to create functional viral proteins)
6. Release: Two-molecules of single-stranded viral RNA, and viral polypeptides are released from the host cell through budding (reverse transcriptase is then inactive and the viral capsid proteins are not assembled, rendering the virus non-infective)
7. Assembly: HIV becomes virulent (viral protease cleaves a polypeptide containing reverse transcriptase and capsomeres or capsid proteins; both activate reverse transcriptase and allows for final assembly of the virus within the envelope)
- Internal capsid only forms once the virion has been released
*HIV is unable to replicate outside of a host cell, needs host machinery to produce copies of itself
Reverse Transcriptase
- Synthesizes a second copy of DNA to form a double-stranded DNA molecule (i.e. provirus = Integrated into the host cell genome)
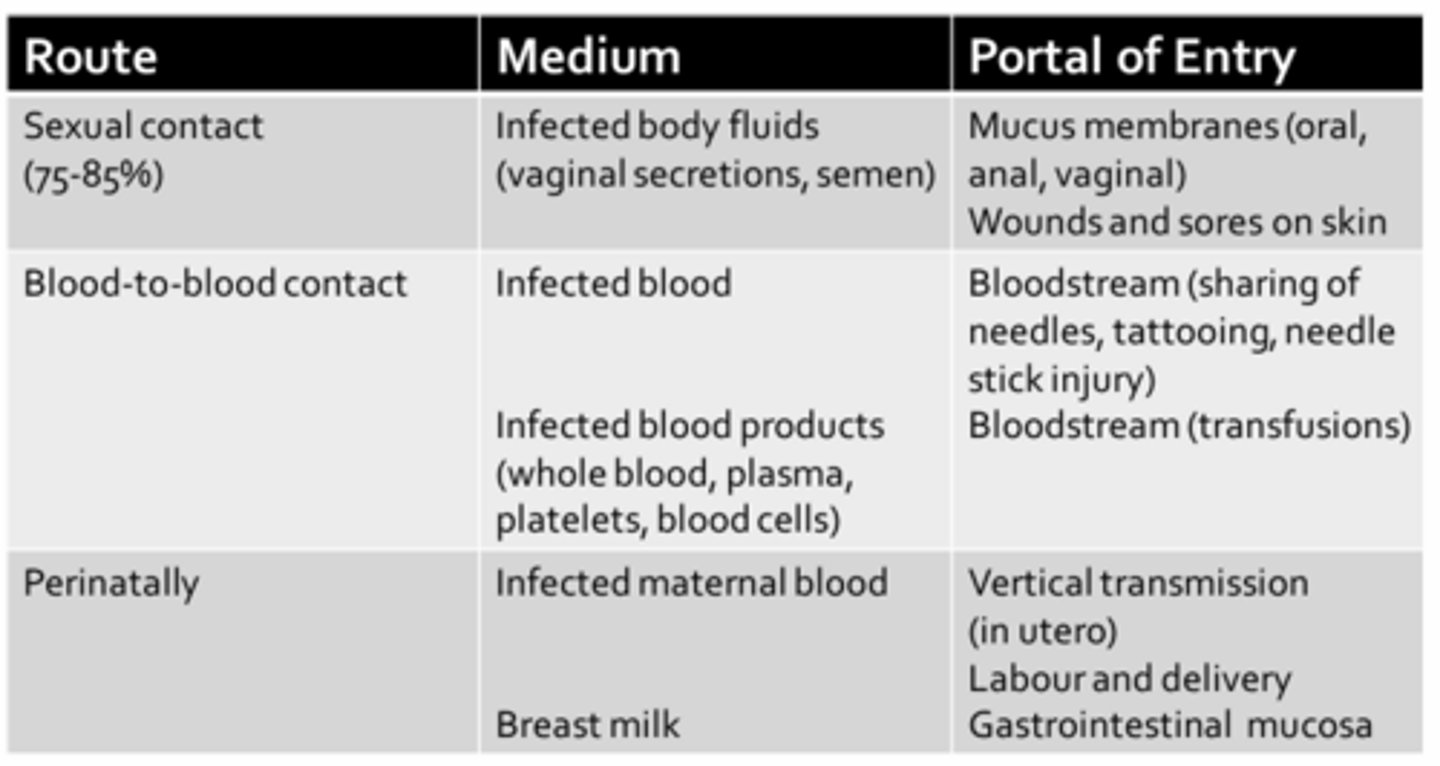
Transmission of HIV
- Sexual contact due to contact of mucus membranes with infected bodily fluids through various sexual activities (i.e. anal/oral sex)
- Individuals with others sexually transmitted diseases, both ulcerative and non-ulcerative, are at increased risk for HIV infection (due to open wounds)
- HIV is principally found in blood
- Prior to 1985, when routine screening of blood products for HIV was not performed, 70-85% of hemophiliacs receiving regular transfusions for clotting factors became infected with HIV
- Early infection is often asymptotic, but highly contagious
Phases of HIV Infection
1. Primary Infection
2. Latency
3. AIDS
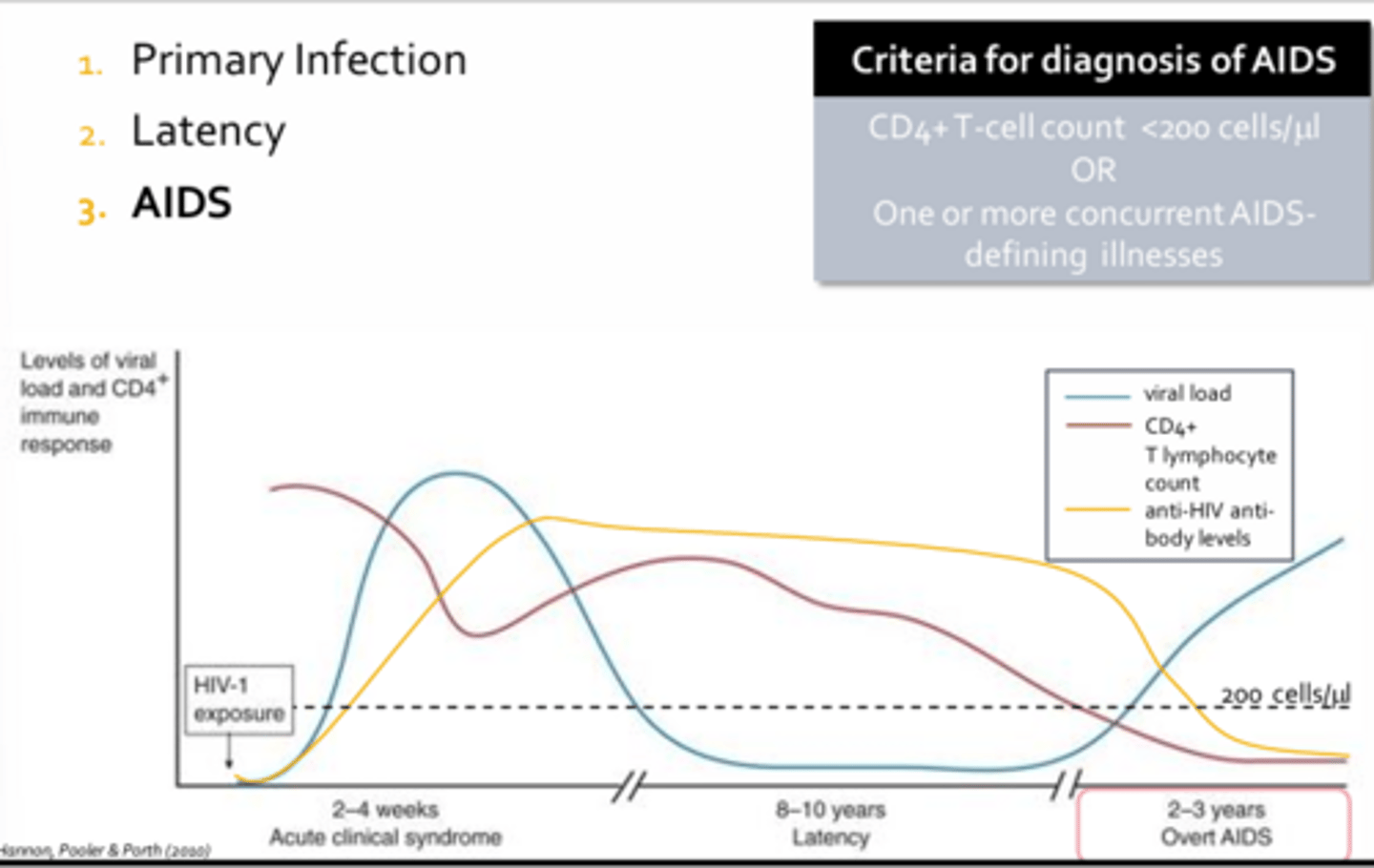
Diagnosis of AIDS
- Diagnosis is conferred when an individuals CD4+ T-cell count falls below 200 cells per micro-litre or if they present with one or more AIDS defining illnesses
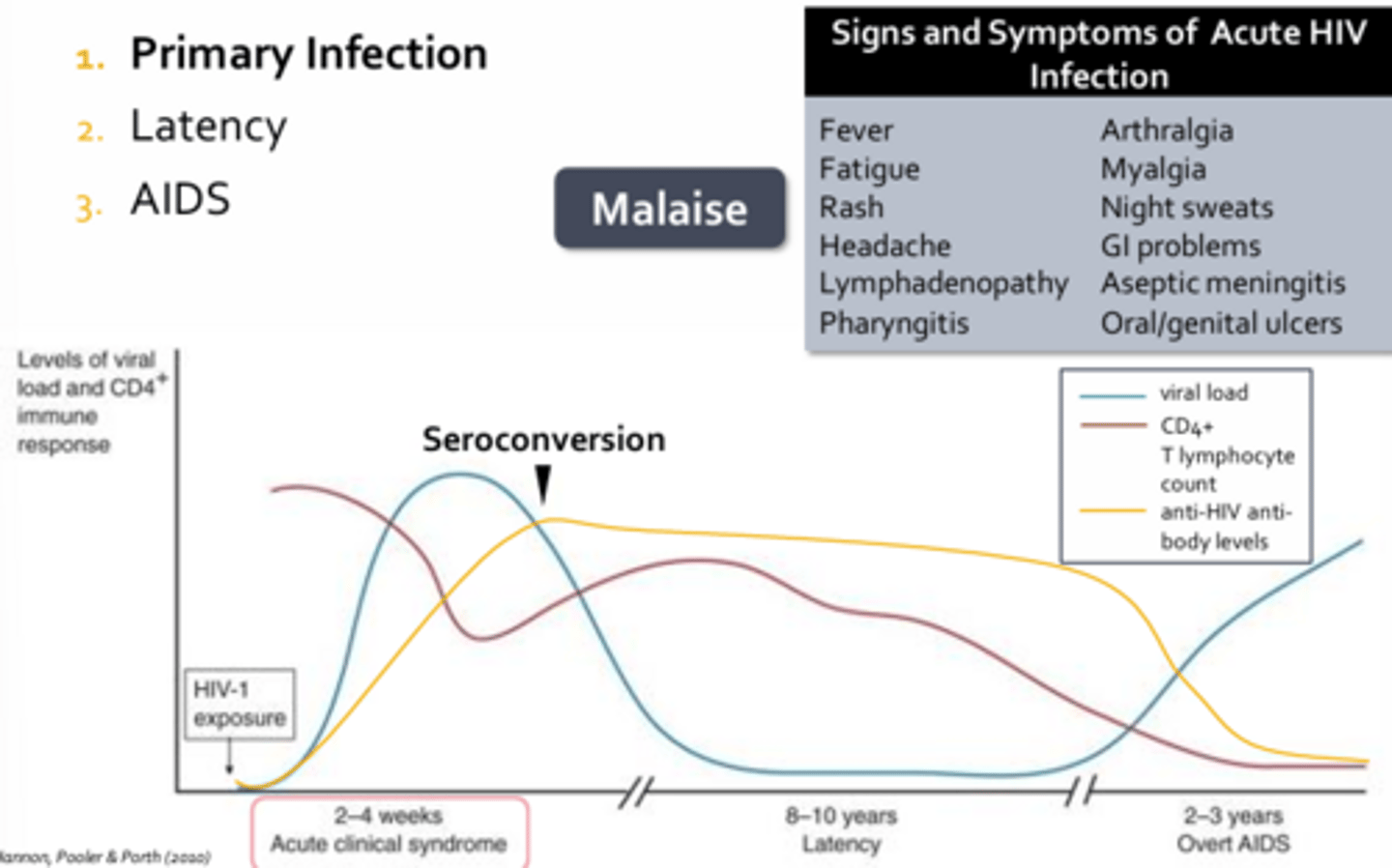
Primary Infection - HIV
- Characterized by fever, myalgia, fatigue, headaches, malaise
- Occur one to four weeks following infection and may last up to ten days
- There is a burst of viral replication resulting in a dramatic increase in viral load and a precipitous drop in the CD4+ positive T-cell count
- As the adaptive responses, cellular and humoral, finally get underway, viral loads will begin to plummet while CD4+ T-cell numbers will begin to recover (recovery is neither complete nor sufficient to keep HIV at rest)
- Seroconversion usually occurs one to three months following infection
- Characterized by detectable levels of anti-HIV antibodies
- Most common point-of-care rapid lab test used to confirm HIV infection detects the presence of anti-HIV antibodies against the viral capsid protein (p24)- Seroconversion usually occurs one to three months following infection
- Characterized by detectable levels of anti-HIV antibodies
- Most common point-of-care rapid lab test used to confirm HIV infection detects the presence of anti-HIV antibodies against the viral capsid protein (p24)
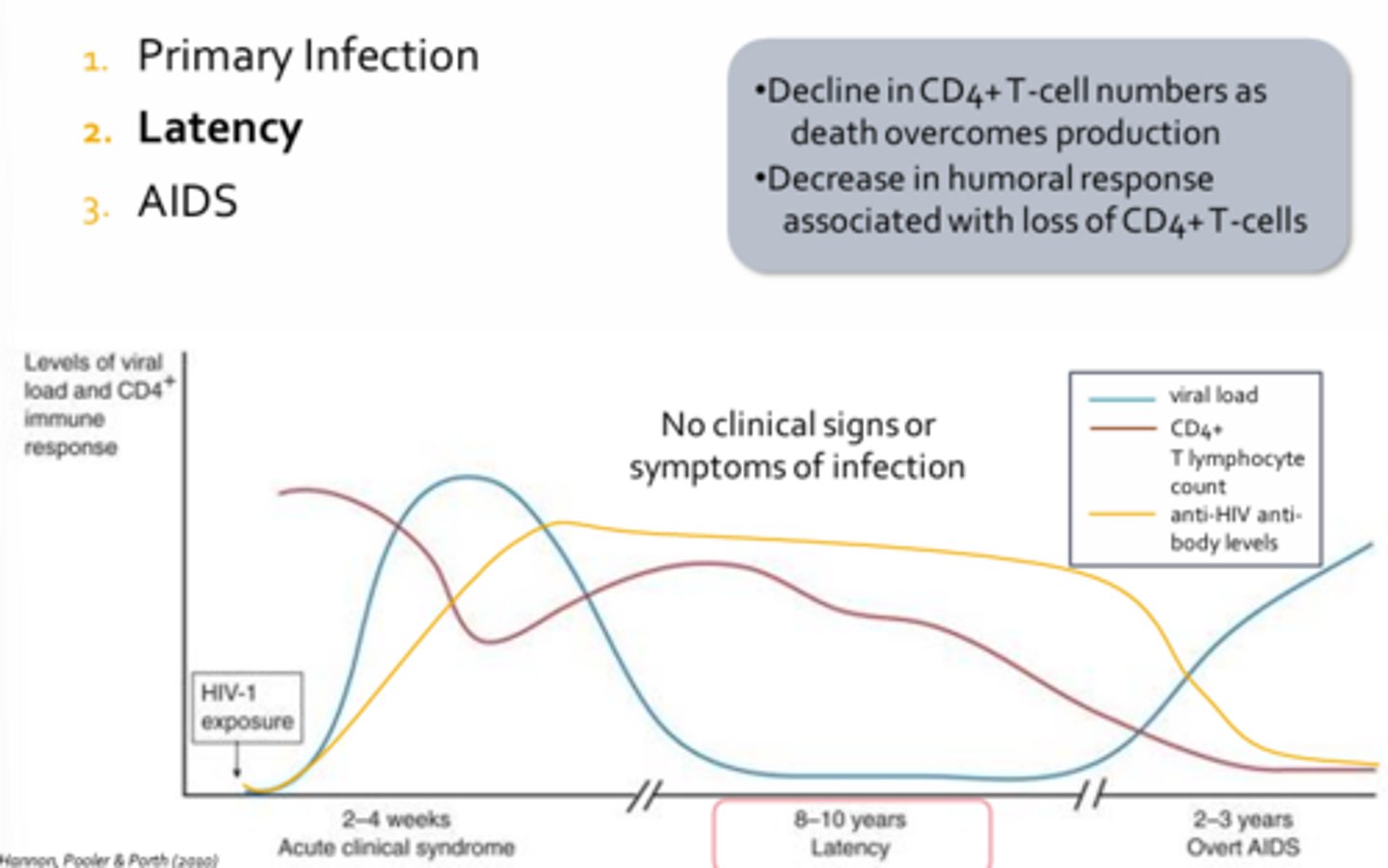
Latency - HIV
- Last 8-10 years in an untreated individual
- Decline in CD4+ T-cell numbers as death overcomes production
- Decrease in humoral response associated with loss of CD4+ T-cells
- Rate of anti-HIV antibodies production declines
- Patient doe snot exhibit any clinical manifestations
AIDS - HIV
- Patient manifestations to diagnosis of AIDS
- Without therapy, HIV production will precede unabated, resulting in death from opportunistic infections or rare cancers within 2-3 years
- Many remain asymptomatic until their CD4+ counts drop below diagnosis threshold
Treatment of HIV
- There is no cure, meds prolong patient life
5 Classes of antiretroviral medications:
1. Nucleoside/nucleotide analogs
2. Reverse transcriptase inhibitors
3. Protease inhibitors
4. Entry inhibitors
5. Integrase inhibitors

HAART Therapy
- HAART = Highly active anti-retroviral therapy
- To treat HIV efficiently, aggressive therapy combining at least 3 anti-retrovirals from at least 2 different drug classes (can result in undetectable viral loads and an improvement in CD4+ T-cell numbers to extend patients lifespan)