Paper 1 errors
1/42
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
What type of compound of magnesium can be used as an “antacid” and why
insoluble strong base - Mg(OH)2
the mols that you calculate enthalpy change of neutralisation are..
mols of H2O formed
A graph of ln(k) is plotted against 1 / T for a reaction.
(k = rate constant, T = temperature in K.)
The gradient has the numerical value of –16 000.
What is the activation energy, in kJ mol–1, for this reaction?
A +1.92
B +133
C +1920
D +133 000
B. ea = gradient / -8.314
turn to kj = / 1000
Define the term lattice enthalpy
The amount of energy released when one mole of ionic lattice is formed from its gaseous ions
at RTP, ionic lattice is almost always
solid
Explain using collision theory, how the gradient of conc / time decreases over time
concentration decreases, so collisions are less frequent
In an equilibrium, the total gaseous mols on each side must..
be equal
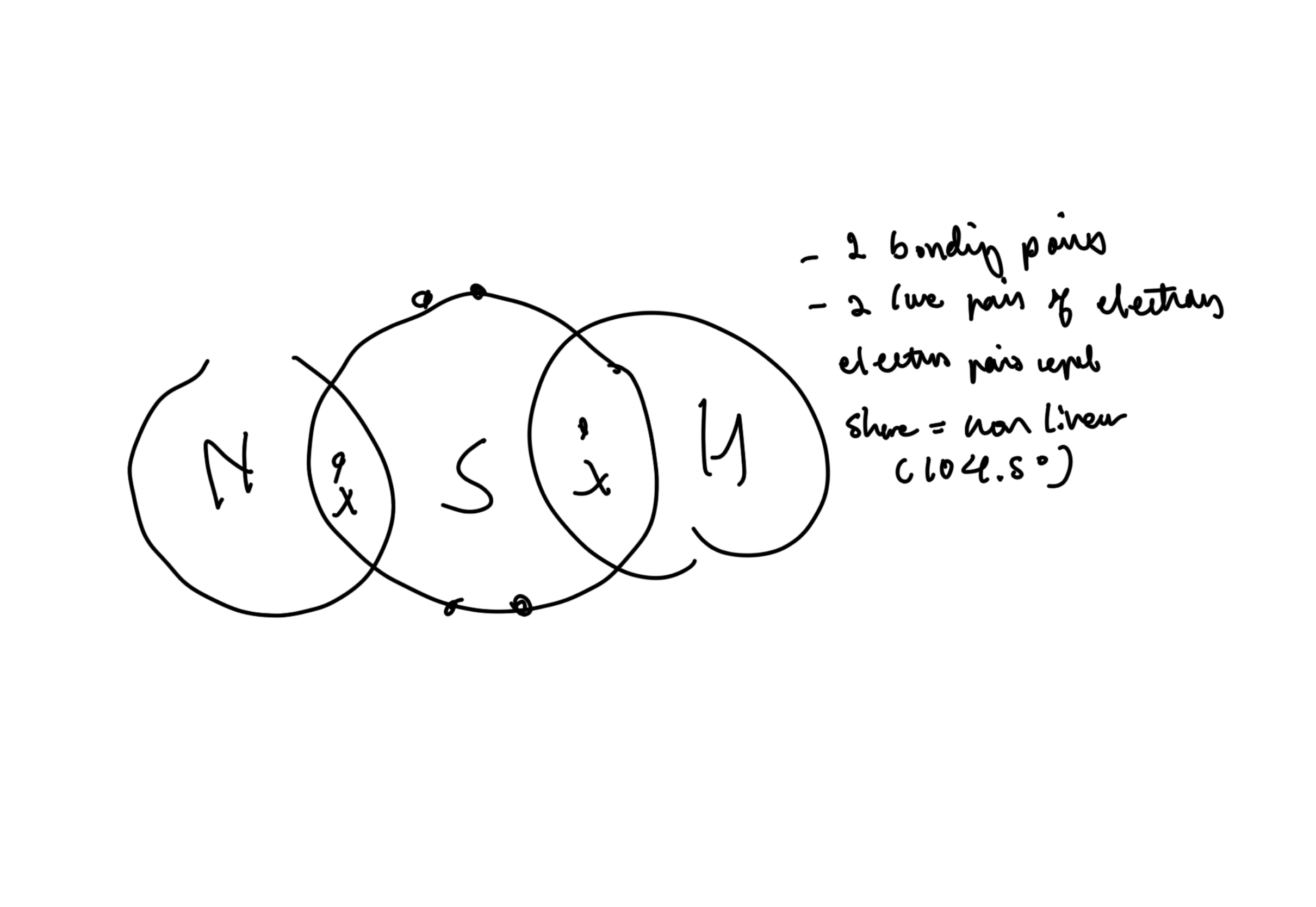
Why ar the bond angles in SO3 120? (1)
3 bonding REGIONS, no lone electron pairs
Why do SO2 and SO3 have polar bonds, but only SO2 has polar molecules (2)
Polar bonds because difference in electronegativity between S and O
Only SO2 has polar molecules due to lone pair of electrons, assymetrial molecule and non-linear / charges not dispersed evenly, and dipoles don’t cancel
State the approximation that means that you can form [H+][A-] into [H+]2
[H+] = [A-]
[H+] / disassociation from water is negligible
Sea water contains aqueous bromide ions.
Chlorine is used to extract bromine from sea water.
Construct the ionic equation for this reaction and explain why chlorine is suitable for this extraction of bromine but iodine is not. (2)
Cl2 + 2Br- → Br2 + 2Cl-
Chlorine is a stronger oxidising agent than bromine, but bromine is a stronger oxidising agent than iodine.
Explain P4s and S8s low boiling points
P4 and S8 have the lowest boiling point because they are simple molecular structure and strong covalent bonding. Only have weak induced dipole-dipole forces between them, which are easy to break. S8 has a higher melting point because it has more electrons, therefore London forces are stronger, and require more energy to break
‘Standard electrode” refers to…
Element’s half-cell compared to H2
Standard electrode potential definition
The voltage of half cell measured against standard hydrogen electrode under standard conditions
how to get from Ba(OH)2 to Ba(Cl)2
acid base reaction
Ba(OH)2 + 2HCl → 2H2O + Ba(Cl)1
explain, using ligand substitution, how haemoglobin transports oxygen around the body
lone pair from O is donated to / forms coordinate / dative bonds with Fe2+
O can reversibly substitute with / be replaced by H2O and CO2
Which compound has polar molecules and why
HCN
BCl3
CO2
C2F4
HCN because it is assymetrical and uneven distribution of charge
percentage dissacociation of methanoic acid 0.015 mol dm3, Ka = 1.6×10-4
[H+] = Root of Ka x [HA] = 1.549×10-3
percentage dissociatoin = ans / 0/015 × 100
Which substances have induced dipole-dipole interactions in their solid state and why
C2H6, H2O, Si
All but silicone, because silicone is a giant covalent lattice, which do not have london forces. Onlty simple molecular compounds do
If you hae a diatomic atom being atomised, what do you need to do to the enthalpy change
x 2
why is lattice enthalpy always negative
amount of energy released when gaseous elements form one mole of ionic lattice structure. bonds being formed is always exothermic
When a reaction is exothermic, and has less moles of gas on product side:
increase temperature, increase pressure; why is it dificult to predict how the amount of product changes at equillbrium (3)
forward reaction is exothermic, so if temperature is increased, equillibrium will shift to left / reactants side, decreasing yield of product
product side has less moles, so increasing pressure will shift equillibrium to right side, increasing yield of products
difficult to predict relative contributions with opposing factors
ideal gas equation and units
pV= nRT
pressure = pa
V = m3
n = moles
R = gas constant (8.314)
T = temp, kelvin
relationship between m3, dm3, and cm3
m3 =
1000000 cm3 (1×106)
1000 m3 (1×103)
A student reacts an excess of magnesium with 25.0 cm3 of 0.500 mol dm–3 hydrochloric acid, HCl . The student also reacts an excess of magnesium with 25.0 cm3 of 0.500 mol dm–3 ethanoic acid, CH3COOH.
Explain why these two reactions of magnesium produce the same volume of gas but at different rates. (3)
same moles of [H+] produced by the end
HCl is strong acid and fully dissociates, CH3COOH is a weak acid, and only partially dissociates
greater H+ concentration in HCl, so more frequent collisions, increasing reaction rate
A buffer solution is prepared by dissolving 3.39 g of potassium hydroxide in 250 cm3 of 0.376 mol dm–3 butanoic acid. This buffer solution has a pH of 5.07 at 25 °C.
Calculate the acid dissociation constant, Ka, of butanoic acid at 25 °C.
Assume that the volume of the solution remains constant at 250 cm3 when the potassium hydroxide is dissolved. (4)
1.5(3) x 10–5
Colour of MnO4- and Mn2+
clear and pink
Describe the precipitation reaction of Copper and potassium iodide
2Cu2+ + 4I- → 2CuI + I2
Forms white precipitate of CuIm from blue solution
An acidified solution containing Cr2O7 2– ions reacts with vanadium(III) ions in a redox reaction to form a solution containing Cr3+ ions and VO2 + ions. Construct the overall equation
2H+ + Cr2O72- + 3V3+ → 2Cr3+ + H2O + 3VO2+
Explain Aluminium’s high melting point
Al: Giant metal lattice structure and metallic bonding. strong electrostatic attraction between positive metal ions and delocalised electrons. a lot of energy needed to break bonds
Explain Si’s very high melting point
Si: giant covalent structure, strong covalent bonds between atoms, between each pair of shared electrons and nuclei of atoms. each Si atom forms 4 bonds with other Si atoms.
Electron affinity definition
The enthalpy change when one mol of gaseous atoms / ions gains an electron to decrease their charge by 1
[Cr(NH3)6]3+ colour
purple solution
[Cr(OH)6]3- colour
dark green solution
Draw a graph of the melting points of Al to Ar
Al, then Si a lot higher
all lower than al:
S higher than P
Cl lower than P
Ar higher than Cl
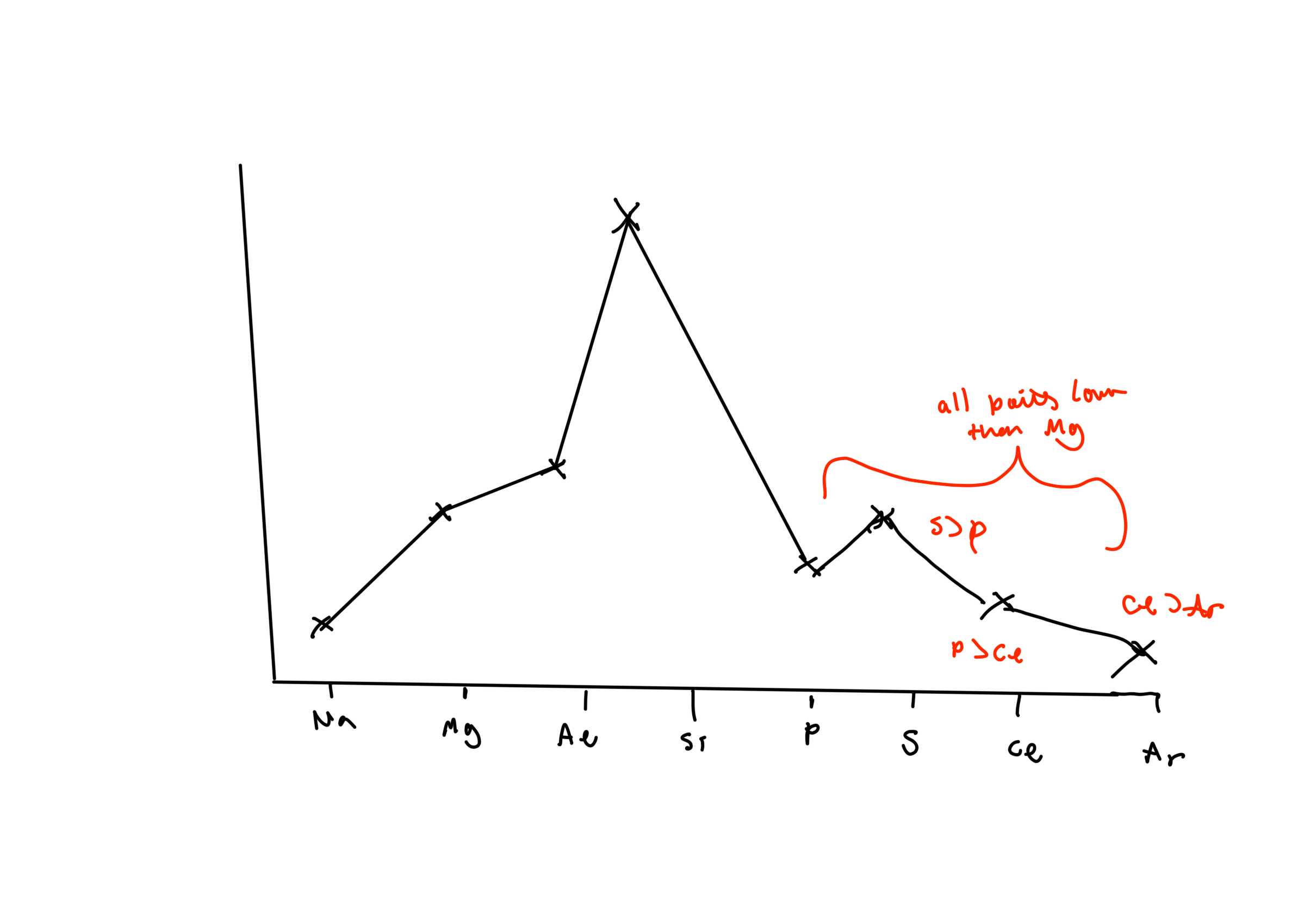
Explain why silicone has a higher melting point than aluminium (3)
Aluminium has giant metallic structure and metallic bonding
Si has giant covalent structure and strong covalent bonds
More energy required to break covalent bonds in giant covalent structure than metallic bonds
Write what observations are seen when Br2 reacts with I-
No reaction or colour change, solution remains orange
Draw and explain molecular shape of H2S
KP=8,13
H2O (g) + Co(g) → H2(g) + CO2 (g)
Equal number of moles of steam and carbon monoxide at start
At equilibrium, H2 and CO2 both have partial pressure of 211kPa
Calculate partial pressures of H2O and CO at equilibrium
Both equal 74
Draw a Boltzmann distribution curve showing how platinum catalyser increases rate of reaction
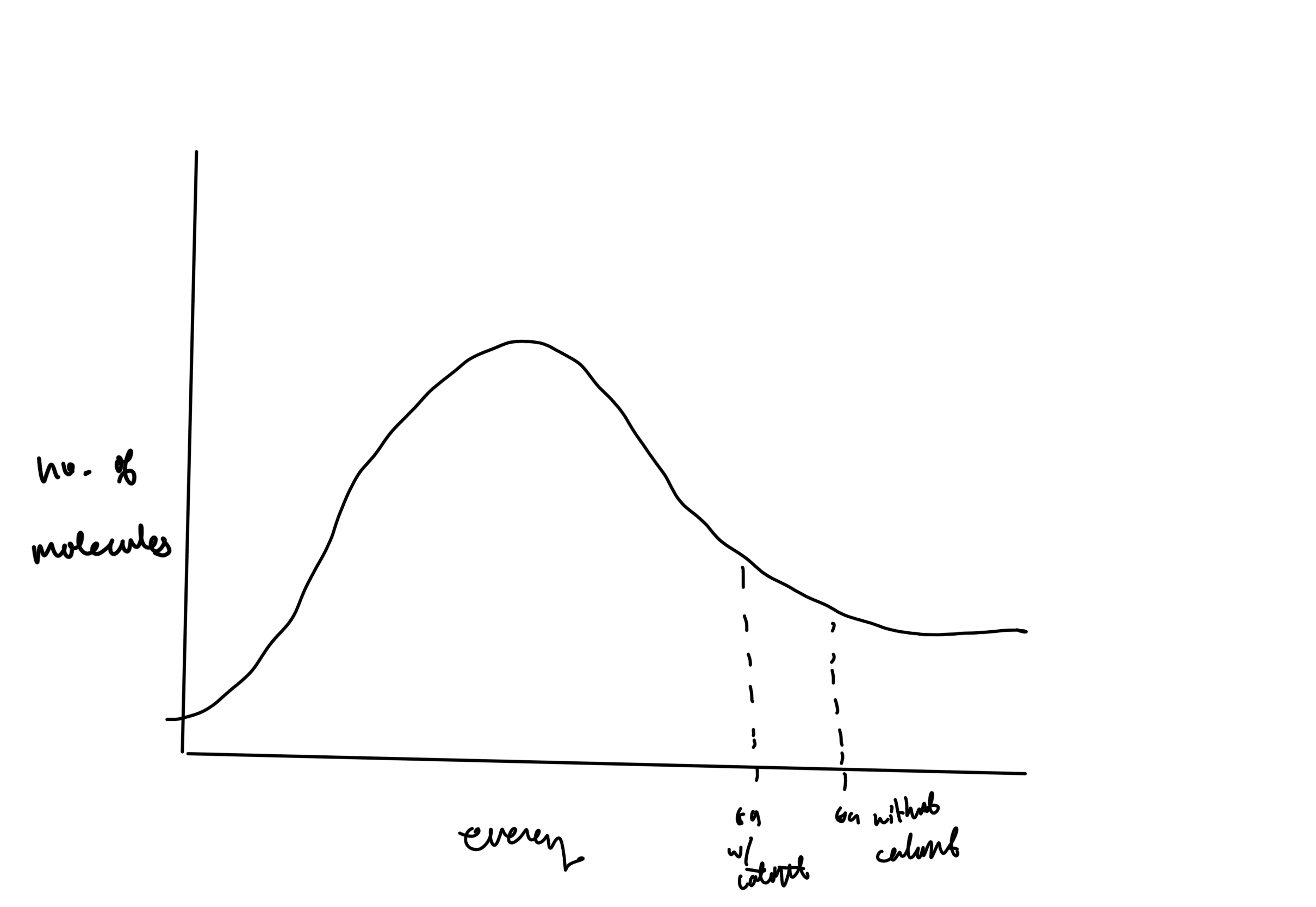
if gradient is negative on Arrhenius equation, Ea is
Positive
Explain why buffer solution resists change in pH when a small amount of alkali is added, using general equations
OH- reacts with H+
HA → (reversible) H+ + A-
Equilibrium shifts right to oppose the change of H+ concentration decreasing
Minimising change in HA / A-
Explain the relative reactivites of Halo-alkane precipitates and ammonia
Ag-Cl: dissolves in dilute ammonia
Ag-Br: dissolves in high concentration ammonia
Ag-I: does not dissolve in ammonia