LAB 6: Classification and identification of hydrocarbons
1/8
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
9 Terms
Purpose of experiment
1) Investigate the physical properties, solubility, and density of some hydrocarbons.
2) Compare the chemical reactivity of an alkane, an alkene and an aromatic compound.
3) Identify an unknown
Experiment steps;
1) test the water solubility of hydrocarbons
2) test the solubility of hydrocarbons in ligroin
3) perform bromine test
4) perform potassium permanganate test
5) perform the sulfuric acid
Saturated molecules
Molecules that only have carbon-carbon single bond.
alkane
unsaturated
molecule that either has one double or triple bond
alkenes and alkynes
Aromatic compound
Is a carbon chain that is a closed loop.
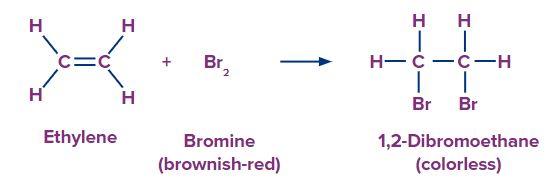
Bromine test reaction
- Saturated substances will not react.
- unsaturated substance will react and change the br2 colorless.
- cyclohexane react rapidly when light or heat is applied.
Aromatic react with bromine in presence of a catalyst iron or aluminum chloride.
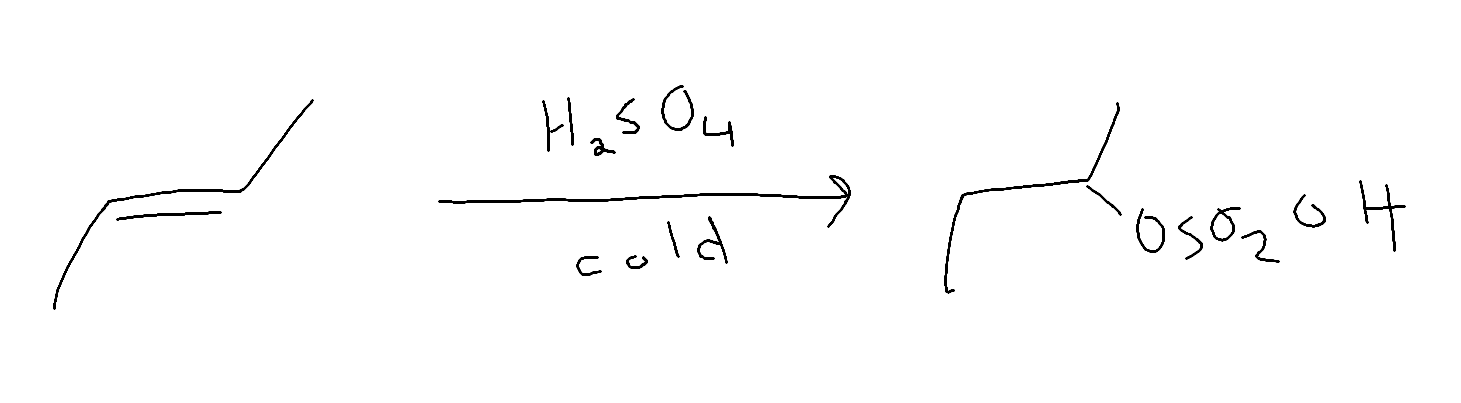
Sulfuric acid test
- Alkanes will not react
- alkenes reat with cold concentration
- alkynes react slow and need a catalyst (HgSO4)
- Aromatics does not react
addition reaction
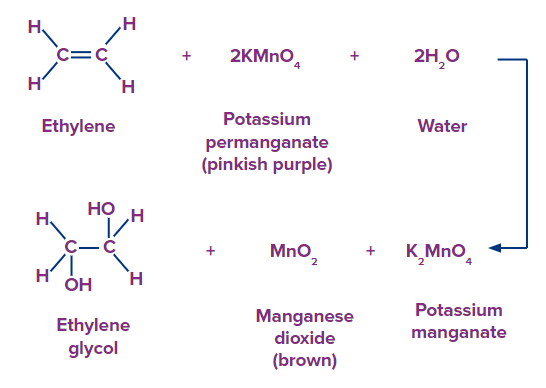
Reaction with potassium permanganate
- Alkanes and aromatic are unreactive
- Alkynes react
- alkene react and produce glycol.
When there is an oil spill in the ocean, an oil-slick develops on the surface of the water and spreads quickly. Why does this happen?
Oil and oil products are mixtures of hydrocarbons when spilled on water, spread quickly along surface because they are insoluble in water