A Level Chemistry Paper 4
1/44
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
Define enthalpy change of solution. [1]
enthalpy change when one mole of a solute AND dissolves in water to form a solution of infinite dilution
Suggest the trend in the magnitude of the lattice energies of the Group 1 iodides, LiI, NaI, KI.
Explain your answer. [2]
cationic) charge density decreases Li+ to K+ [1]
so lattice energies become less negative / less exothermic AND because less attraction between ions [1]
The reaction of I- ions with persulfate ions, S2O82-, can be catalysed by Fe3+ ions.
2I- + S2O82- → I2 + 2SO42-
Write equations to show how Fe3+ catalyses this reaction. [2]
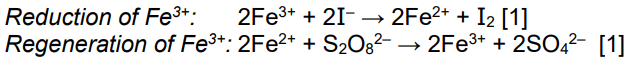
TiO2+(aq) is a colourless ion.
Suggest why. [2]
Ti is in +4 oxidation state so no d electrons / d0 OR Ti in TiO2+ has no d electrons / d0 [1]
cannot absorb photons / light in visible spectrum OR no wavelength / frequency absorbed in visible spectrum [1]
The TiO2+ ion forms when TiO2 reacts with an excess of sulfuric acid. TiO2+ can be reduced by zinc metal in acidic conditions to form a purple solution containing Ti3+(aq).
Write an ionic equation for the reduction of TiO2+ by zinc metal in acidic conditions. [1]
2TiO2+ 4H+ + Zn → 2Ti3+ + 2H2O + Zn2+
Nitrophenol can be produced from benzene in a five-step synthesis route. Outline this multi-step synthesis reaction. [10]
benzene → nitrobenzene (concentrated HNO3 + H2SO4 @ 25 — 60oC)
nitrophenol → phenylamine (hot Sn, concentrated HCl followed by NaOH)
phenylamine → diazonium salt (HNO2, <10oC)
diazonium salt → phenol (warm with H2O)
phenol → nitrophenol (dilute HNO3 at room temperature)
![<p>State the two types of reaction that occur when <strong>J</strong> reacts with NaOH(aq). <strong>[2]</strong></p>](https://assets.knowt.com/user-attachments/258a58ba-49a1-492a-b3ef-74faddaf69de.png)
State the two types of reaction that occur when J reacts with NaOH(aq). [2]
hydrolysis
acid-base / neutralisation
![<p>Phenol and benzene both react with nitric acid, as shown in the figure.</p><p>Explain why the reagents and conditions for these two reactions are different. <strong>[3]</strong></p>](https://assets.knowt.com/user-attachments/7ff91025-d2a2-460d-8fd2-781e8352ff8b.png)
Phenol and benzene both react with nitric acid, as shown in the figure.
Explain why the reagents and conditions for these two reactions are different. [3]
p-orbital on oxygen overlaps with ring / π system OR lone pair of e– on oxygen is delocalised into the ring [1]
electron density in ring increases [1]
attracts/polarises electrophile better [1]
![<p>Lidocaine is used as an anaesthetic. A synthesis of lidocaine is shown.</p><p>W can be formed by reacting HOCH<sub>2</sub>COOH with an excess of SOCl<sub>2</sub>, PCl<sub>5</sub>, or PCl<sub>3</sub>.</p><p>Write equations for these reactions. <strong>[3]</strong></p>](https://assets.knowt.com/user-attachments/41f54fb9-a727-490d-85e2-6b0a351e2857.png)
Lidocaine is used as an anaesthetic. A synthesis of lidocaine is shown.
W can be formed by reacting HOCH2COOH with an excess of SOCl2, PCl5, or PCl3.
Write equations for these reactions. [3]
SOCl2 by-products: SO2 + HCl
PCl5 by-products: POCl3 + HCl
PCl3 by-products: H3PO4
After W and X have reacted together, an excess of CH3COONa(aq) is added to the reaction mixture. Suggest why. [1]
to remove / neutralise excess H+ / acid produced
OR to react with any acidic by-products / HCl / SO2
OR to react with any unreacted W
![<p>The reaction of W with X, reaction 1, follows an addition–elimination mechanism.</p><p>Complete the mechanism for the reaction of W with X. </p><p>Include all relevant curly arrows, lone pairs of electrons, charges and partial charges. </p><p>Use Ar–NH<sub>2</sub> to represent X. <strong>[4]</strong></p>](https://assets.knowt.com/user-attachments/9970d42a-b493-4f11-a948-1b0ec9f476f7.png)
The reaction of W with X, reaction 1, follows an addition–elimination mechanism.
Complete the mechanism for the reaction of W with X.
Include all relevant curly arrows, lone pairs of electrons, charges and partial charges.
Use Ar–NH2 to represent X. [4]
Describe the mode of action of a heterogeneous catalyst in a reaction. [3]
M1 adsorption of reactants to the surface of the catalyst
M2 bonds in the reactants weaken (lowering the activation energy)
M3 reaction occurs and the products are desorbed
Manganese(VII) oxide, Mn2O7, can be made by treatment of KMnO4 with concentrated sulfuric acid (reaction 1).
Mn2O7 readily decomposes at room temperature to form manganese(IV) oxide and a colourless diatomic gas (reaction 2).
Construct equations for both the reactions described. [2]
Aqueous manganese(II) ions show similar chemical properties to aqueous copper(II) ions when reacted separately with NaOH(aq) and with concentrated HCl.
Write the ionic equation, and state the type of reaction, for the reaction of [Mn(H2O)6]2+ with NaOH(aq). [2]
Write the ionic equation, and state the type of reaction, for the reaction of [Mn(H2O)6]2+ with concentrated HCl. [2]
Define standard electrode potential, Eo. [1]
voltage / EMF / potential difference when a half-cell is connected to a (standard) hydrogen electrode under standard conditions
A salt bridge is used in an electrochemical cell.
State the function of the salt bridge. Explain your answer [1]
ions move (from the salt bridge) to maintain charge balance / complete the circuit
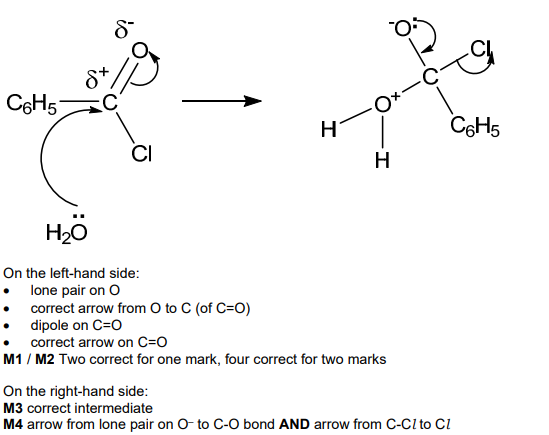
![<p>Benzoyl chloride is hydrolysed by water at room temperature to form benzoic acid. </p><p>Complete the diagram to show the mechanism for the reaction between C<sub>6</sub>H<sub>5</sub>COCl and H<sub>2</sub>O. </p><p>Include charges, dipoles, lone pairs of electrons and curly arrows as appropriate. <strong>[4]</strong></p>](https://assets.knowt.com/user-attachments/5f529328-e786-4a74-b7f4-cdfd31e494d5.png)
Benzoyl chloride is hydrolysed by water at room temperature to form benzoic acid.
Complete the diagram to show the mechanism for the reaction between C6H5COCl and H2O.
Include charges, dipoles, lone pairs of electrons and curly arrows as appropriate. [4]
Thin-layer and gas-liquid chromatography can be used to analyse mixtures of substances.
Each type of chromatography makes use of a stationary phase and a mobile phase.
stationary phase | mobile phase | |
thin-layer chromatography | ||
gas-liquid chromatography |
The solubility of the Group 2 sulfates decreases down the group.
Explain this trend. [3]

The solubility of the Group 2 hydroxides increases down the group.
Explain this trend. [3]
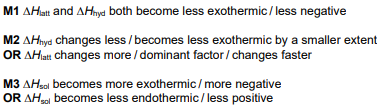
Describe the general trend in first electron affinities for Cl, Br and I. Explain your answer.

The thermal stability of Group 1 hydrogencarbonates increases down the group. Suggest an explanation for the trend in thermal stability of the Group 1 hydrogencarbonates. [2]
M1 ionic radius of M+ / cationic radius increases OR charge density of ion / M+ decreases down Group 1
M2 less distortion / polarisation of the anion / HCO3 – OR CO bond / C-O / C=O less weakened
![<p>Complete Table 5.1 to predict the substance liberated at each electrode during electrolysis of the indicated electrolyte with inert electrodes.<strong> [3]</strong></p>](https://assets.knowt.com/user-attachments/93e5939e-027d-46e0-8200-eea5538c4744.png)
Complete Table 5.1 to predict the substance liberated at each electrode during electrolysis of the indicated electrolyte with inert electrodes. [3]

Concentrated HNO3 reacts with concentrated H2SO4 to generate the electrophile NO2+ .
Write the equation(s) to show how this happens. [2]
Compare the relative ease of nitration of benzene, benzoic acid and phenol. Explain your reasoning; include reference to the structures of the three compounds in your answer. [4]

Compare the relative acidities of ethanol, ethanoic acid, chloroethanoic acid and phenol. Explain your reasoning. [4]

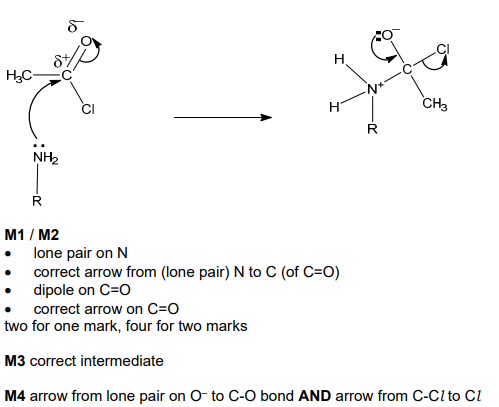
Compare the relative basicities of ammonia, cyclohexylamine and phenylamine. Explain your reasoning. [3]

![<p>Complete the mechanism of the reaction between cyclohexylamine and CH<sub>3</sub>COCl. </p><p>R–NH<sub>2</sub> is used to represent cyclohexylamine. Include all relevant lone pairs of electrons, curly arrows, charges and partial charges. <strong>[4]</strong></p>](https://assets.knowt.com/user-attachments/09fbb1f4-75a8-4230-8e5b-0326650da068.png)
Complete the mechanism of the reaction between cyclohexylamine and CH3COCl.
R–NH2 is used to represent cyclohexylamine. Include all relevant lone pairs of electrons, curly arrows, charges and partial charges. [4]
Explain the reasons why the lattice energy of MgCl2 is more exothermic than the lattice energy of KCl. [2]
Mg2+ is smaller (than K+ )
Mg2+ is greater charge (than K+ )
greater attraction between Mg2+ and Cl – /between the ions (in MgCl2) OR stronger ionic bonds (in MgCl2)
Describe the standard conditions used in a half-cell. [1]
ion concentration of 1 mol dm-3
298K
![<p>Equal volumes of 1.0mol dm<sup>–3</sup> Sn<sup>2+</sup>(aq) and 1.0mol dm<sup>–3</sup> Cl<sup>-</sup> (aq) are mixed. </p><p>Use relevant E<sup>o</sup> values to explain whether a reaction occurs between these two ions. <strong>[2]</strong></p>](https://assets.knowt.com/user-attachments/e4cc4e18-0934-4150-8887-4190d03466c2.png)
Equal volumes of 1.0mol dm–3 Sn2+(aq) and 1.0mol dm–3 Cl- (aq) are mixed.
Use relevant Eo values to explain whether a reaction occurs between these two ions. [2]
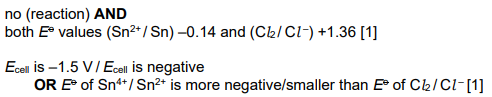
Equal volumes of 1.0mol dm–3 of Sn2+(aq) and acidified 1.0mol dm–3 VO2+(aq) are mixed.
Write an equation for the reaction that takes place in the resulting mixture. [2]
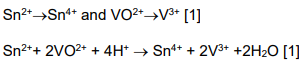
State what is meant by partition coefficient. [1]
ratio of concentration of the solute in two solvents at equilibrium [1]
The partition coefficient, Kpc, for butanoic acid between ethoxyethane and water is 3.50.
A solution of 2.00g of butanoic acid in 100cm3 ethoxyethane is added to water. This mixture is left until there is no further change in the concentration of butanoic acid in either solvent. The mass of butanoic acid dissolved in the ethoxyethane layer is now 1.62g.
Calculate the volume of water used. [2]
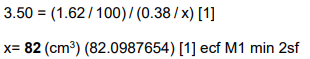
A is a pale green salt containing Fe2+ ions. A sample of 2.62g of A is dissolved in water and the solution is made up to exactly 100cm3 with water. 25.0cm3 samples of this solution are placed in conical flasks and titrated against 0.0100mol dm–3 acidified potassium manganate(VII).
The equation for the only reaction that occurs is shown.
5Fe2+ + MnO4– + 8H+ → 5Fe3+ + Mn2+ + 4H2O
The average titre value is 35.0cm3 of 0.0100mol dm–3 acidified potassium manganate(VII).
Describe the colour change that is seen in the conical flask at the end-point of this titration. [1]
Calculate the percentage by mass of iron in A. [Ar : Fe, 55.8] [2]
colourless / pale green to pink / purple
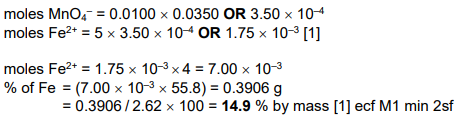
Define stability constant. [1]
the equilibrium (constant) for the formation of the complex ion in a solvent from its constituent ions or molecules
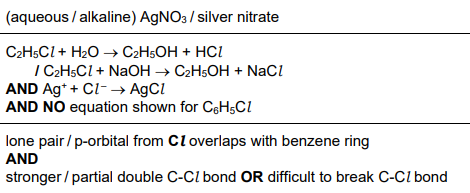
Chlorobenzene and chloroethane have different reactivities in nucleophilic substitution reactions.
Identify a suitable reagent to illustrate this difference in reactivity. The reagent chosen should give visibly different results with chlorobenzene and chloroethane. [1]
Write equations to describe any reactions that occur. [1]
Explain the difference in the reactivities of chlorobenzene and chloroethane in nucleophilic substitution reactions. [1]
![<p>Complete Table 9.1 to give the number of peaks in the carbon-13 NMR spectrum of each of the five isomers of C<sub>5</sub>H<sub>10</sub>O<sub>2</sub> that has an ester group. <strong>[2]</strong></p>](https://assets.knowt.com/user-attachments/2fbf2bad-47ab-4d36-8e5b-116cdf0683aa.png)
Complete Table 9.1 to give the number of peaks in the carbon-13 NMR spectrum of each of the five isomers of C5H10O2 that has an ester group. [2]
5 5 5 4 4
State the number of peaks that would be seen in the proton (1 H) NMR spectrum of methyl butanoate, CH3CH2CH2CO2CH3. Name all the splitting patterns seen in this spectrum. [2]
H2PO2- is a strong reducing agent. It can be used to reduce metal cations without the need for electrolysis.
HPO32- + 2H2O + 2e- ⇌ H2PO2- + 3OH- Eo= –1.57V
In an experiment, an alkaline HPO32-/H2PO2- half-cell is constructed with [H2PO2-] = 0.050 mol dm-3. All other ions are at their standard concentration.
Predict how the value of E of this half-cell differs from its Eo value. Explain your answer. [2]

The Cr3+/Cr half-cell has a standard electrode potential of –0.74V. An electrochemical cell consists of an alkaline HPO32-/H2PO2- half-cell and a Cr3+/Cr half-cell.
Calculate the standard cell potential, Eocell.
+0.83V
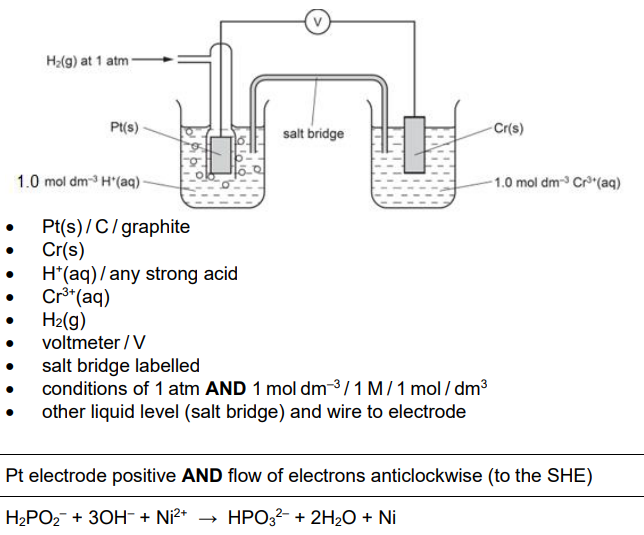
Complete the diagram in Fig. 2.1 to show how the standard electrode potential of the Cr3+/Cr half-cell can be measured relative to that of the standard hydrogen electrode.
Identify the chemicals, conditions and relevant pieces of apparatus. [3]
Label Fig. 2.1 to show: [1]
which is the positive electrode
the direction of electron flow in the external circuitH2PO2- reduces Ni2+ to Ni in alkaline conditions. Use equation 1 to construct the ionic equation for this reaction.
equation 1 HPO32- + 2H2O + 2e- ⇌ H2PO2- + 3OH- [1]
State and explain the relative ease of hydrolysis of acyl chlorides, alkyl chlorides and aryl chlorides. [3]

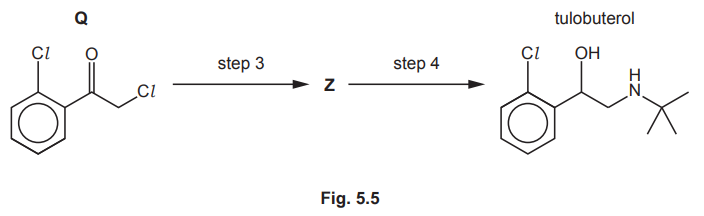
Tulobuterol is produced from Q as shown in Fig. 5.5.
Suggest reagents and conditions for steps 3 and 4.
Draw the structure of Z.
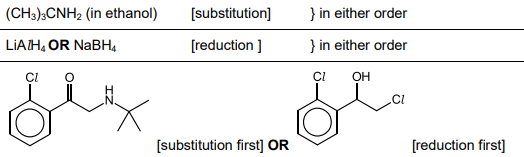
Define enSuggest one disadvantage of producing two enantiomers in this synthesis. Suggest a method of adapting the synthesis to produce a single enantiomer.