31. Amino acids, proteins and DNA
1/117
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
118 Terms
What are the 2 functional groups of amino acids?
Amino group (NH₂)
Carboxyl group (COOH)
How many naturally occurring amino acids are there in the body?
20
What type of amino acids are found in the body?
What does this mean about their structure?
⍺-amino acids (alpha) It means that NH₂ is always on the carbon next to COOH
Draw a general formula for a-amino acids
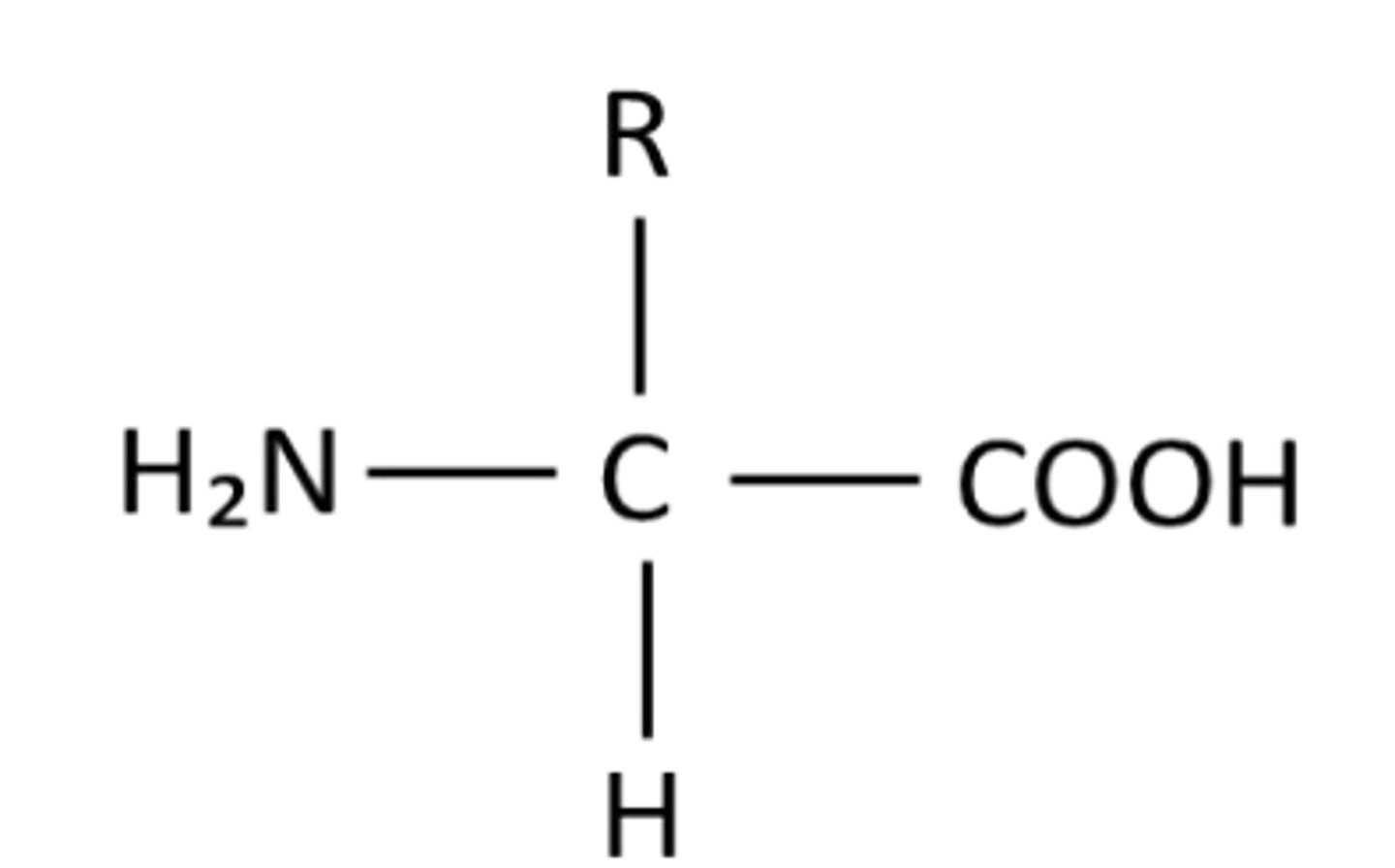
What does the R group affect ?
Name
Polarity
Size
Are a-amino acids chiral? Why?
● 20/21 amino acids (glycine is the exception, where R=H) are chiral.
● Carbon has 4 different substituents
● The body only ever uses 1 type of isomer and not the other.
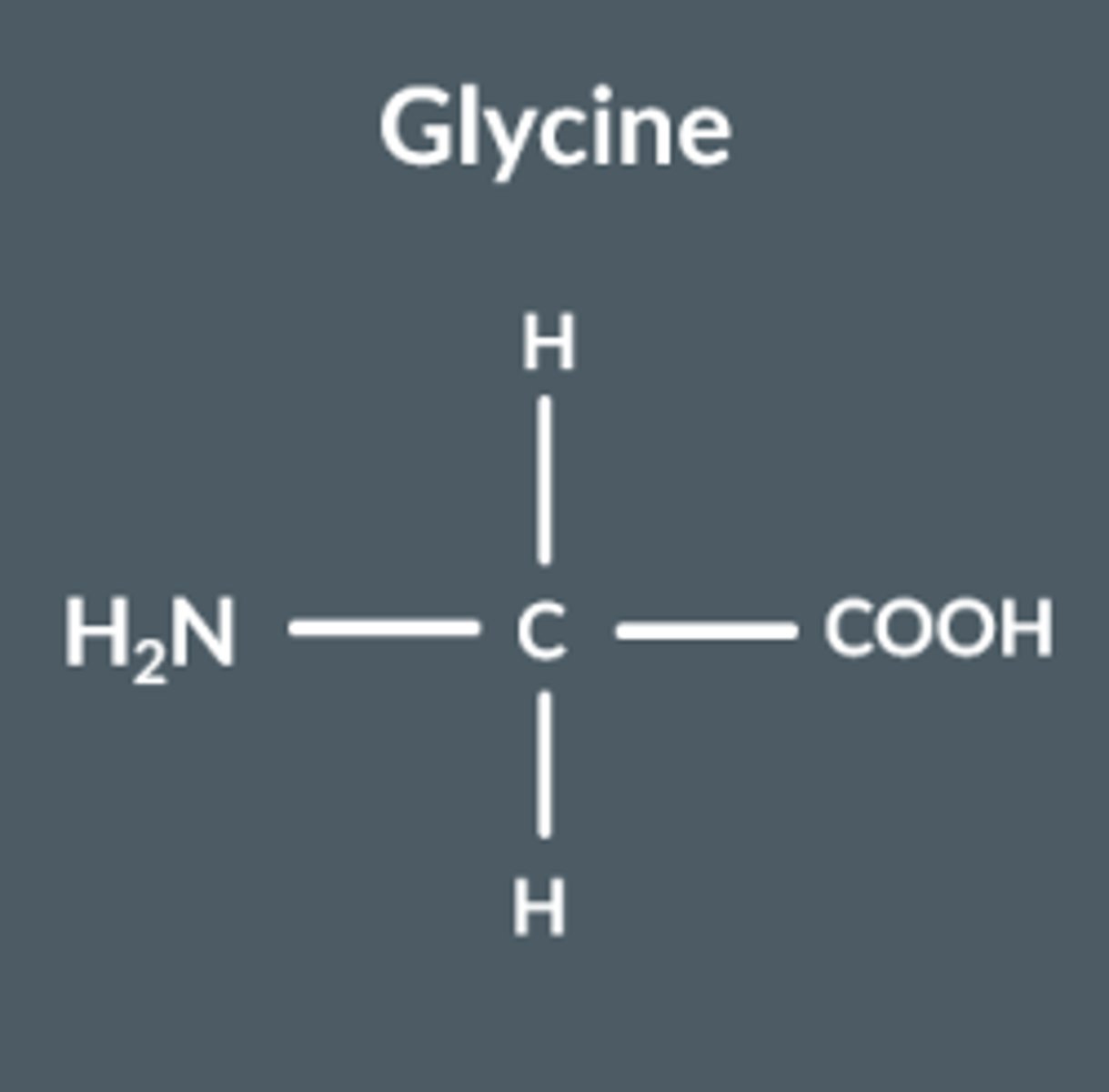
Glycine
Does NOT display optical isomerism, since it's bonded to 2 of the same group
Essential and nonessential amino acids
● Humans use 21 amino acids
● 12 amino acids can be created by your body, 9 cannot.
● All 21 are important to us, so it’s essential that we get the 9 that we can’t make from our diet instead.
● So, we call these 9 ‘essential amino acids’ and the other 12 ‘non-essential’ amino acids. (it's not that they're not important)
Melting point of amines
MUCH higher than similar sized molecules with similar functional groups
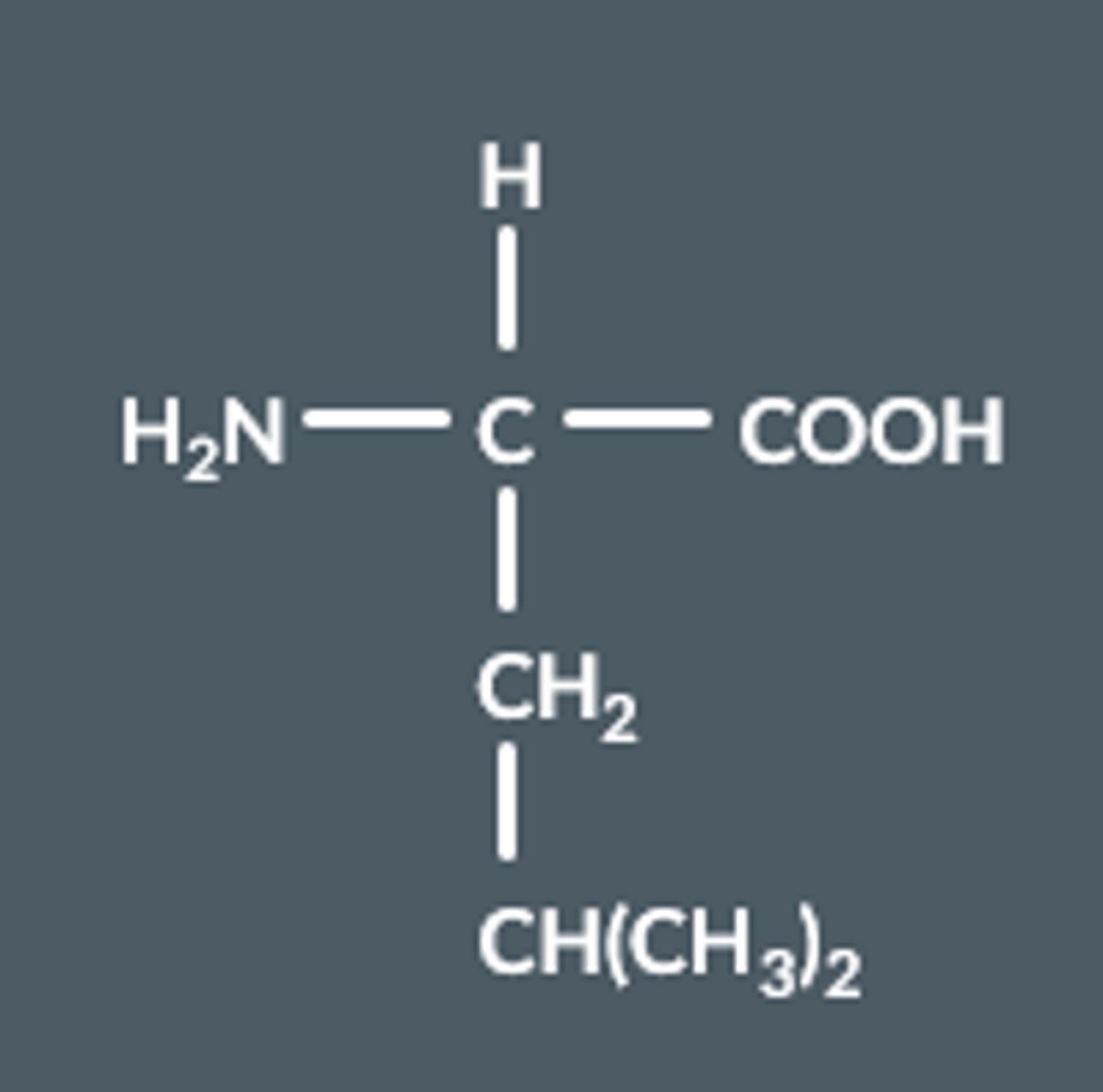
Name this amino acid
2-amino-4-methylpentanoic acid
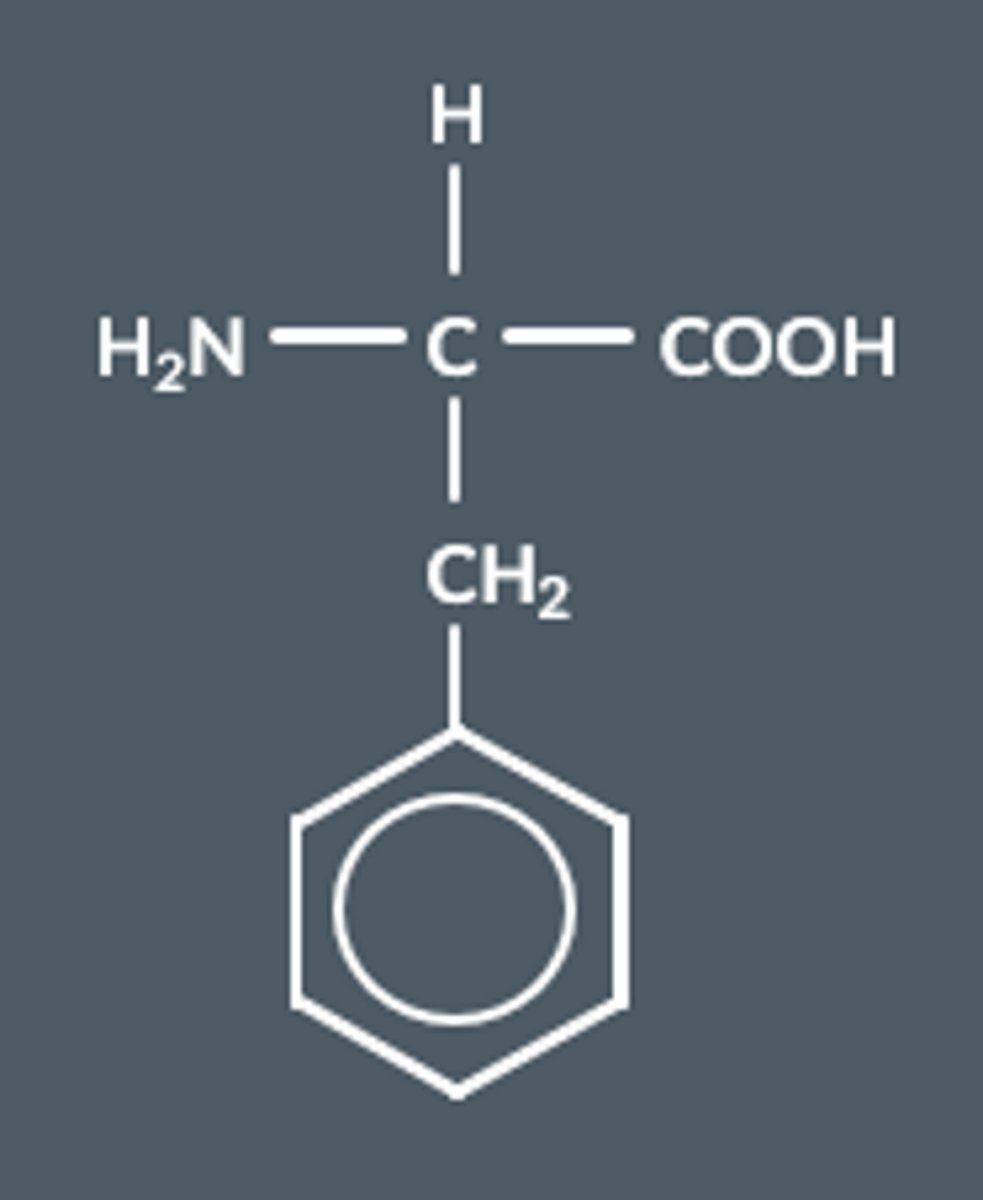
Name this amino acid
Which enantiomer do a-amino acids exist as in nature?
( - ) enantiomer
How can amino acids be synthesised industrially?
RCHO + NH₄CN → RCH(NH₂)CN via nucleophilic addition.
RCH(NH₂)CN + HCl + 2H₂O → RCH(NH₂)COOH + NH₄Cl (hydrolysis, HCl is dilute)
Need to reflux the reaction mixture
Is the product from amino acids being synthesised naturally optically active?
Why?
● No, a racemic mixture is formed as the CN⁻ ion can attack from above or below the planar C=O bond with equal likelihood.
● An equal amount of each enantiomer is formed, so no net effect on plane polarised light.
In what form do amino acids exist as solids? What consequences does this have?
Zwitterions (ionic lattice) - high melting and boiling points
What is a zwitterion?
Dipolar ions which have both a permanent positive and negative charge, but are neutral overall.
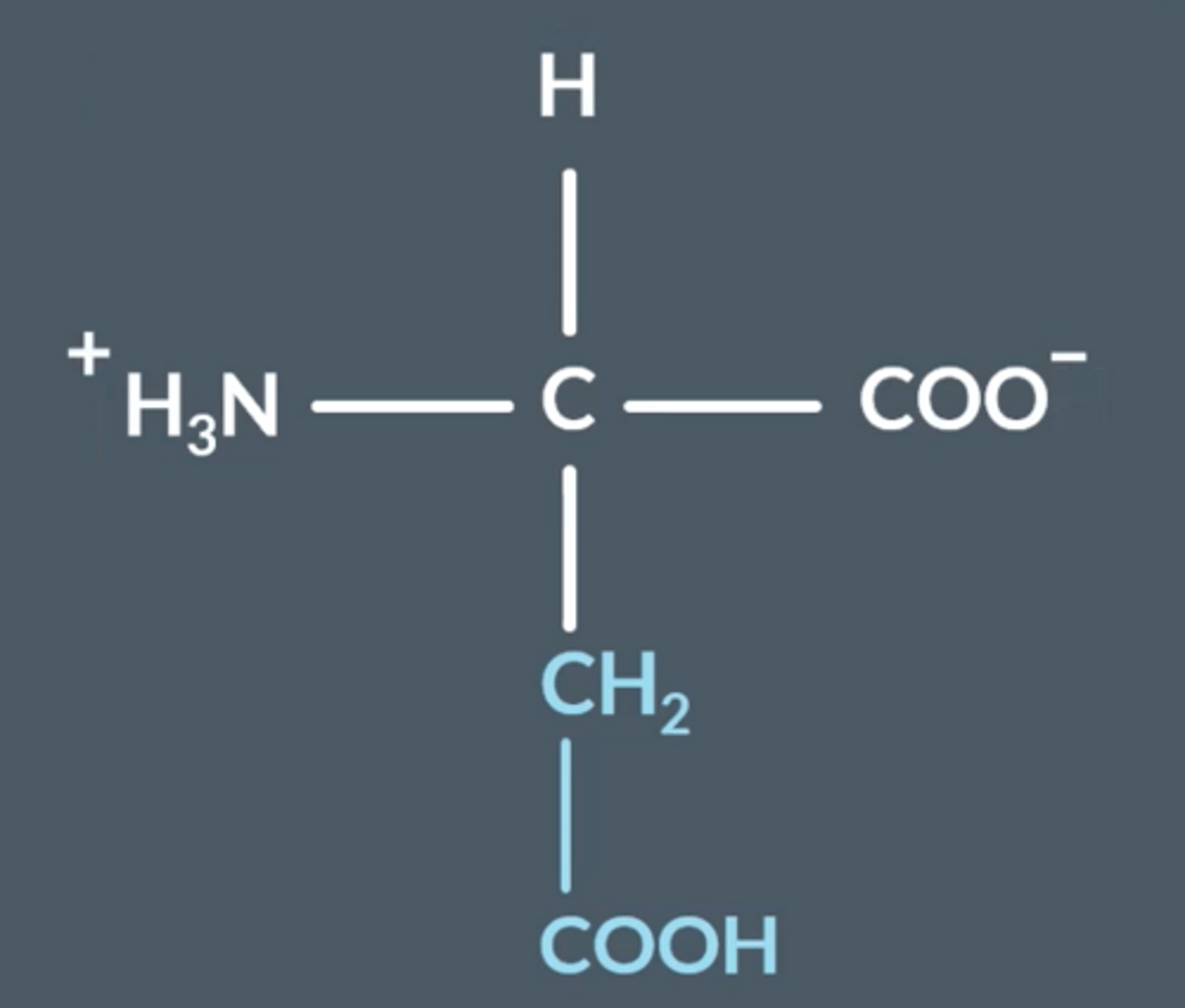
Isoelectric point
The pH at which the Zwitterion exist (pH 0)
What colour solids are most zwitterions at room temperature?
White solids
Behave like an ionic salt
Do zwitterions dissolve in water? Non-polar solvents? Why?
Yes, but NOT in non-polar solvents.
Due to ionic nature/polar bonds (can form ion-dipole forces between zwitterions and water molecules)
Describe the melting point of amino acids.
Much higher melting point than similar sized molecules due to strong electrostatic forces of attraction between zwitterions
Melt poorly in organic solvents
How do zwitterions occur in amino acids? Draw a general structure of one
COOH is deprotonated → COO⁻
NH₂ is protonated → NH₃+
How do amino acids appear in neutral conditions ?
The NH₂ group changes to NH₃
The COOH group changes to COO⁻
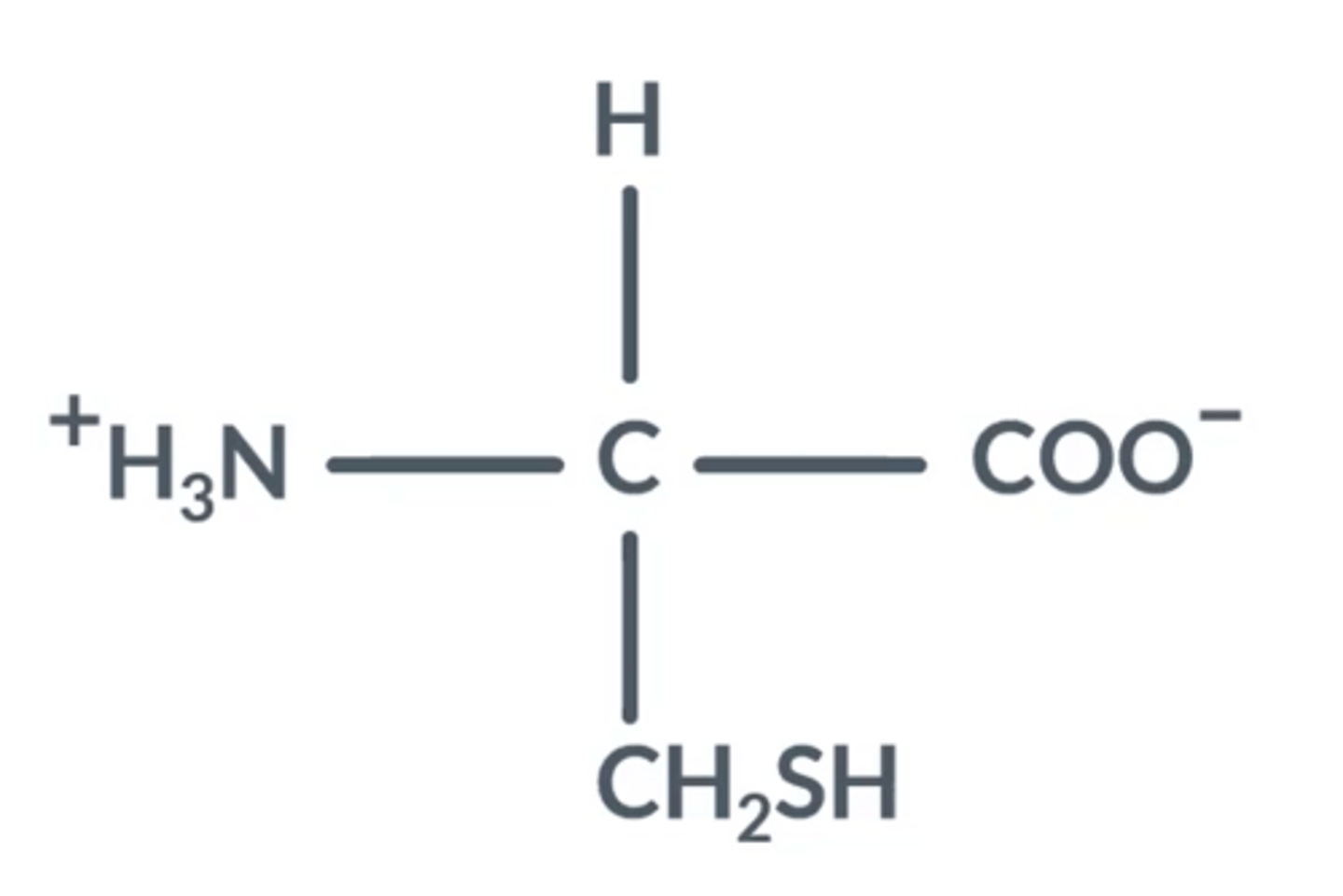
What happens to amino acids in acidic conditions?
Draw this.
NH₂ groups gain a proton, become NH₃⁺
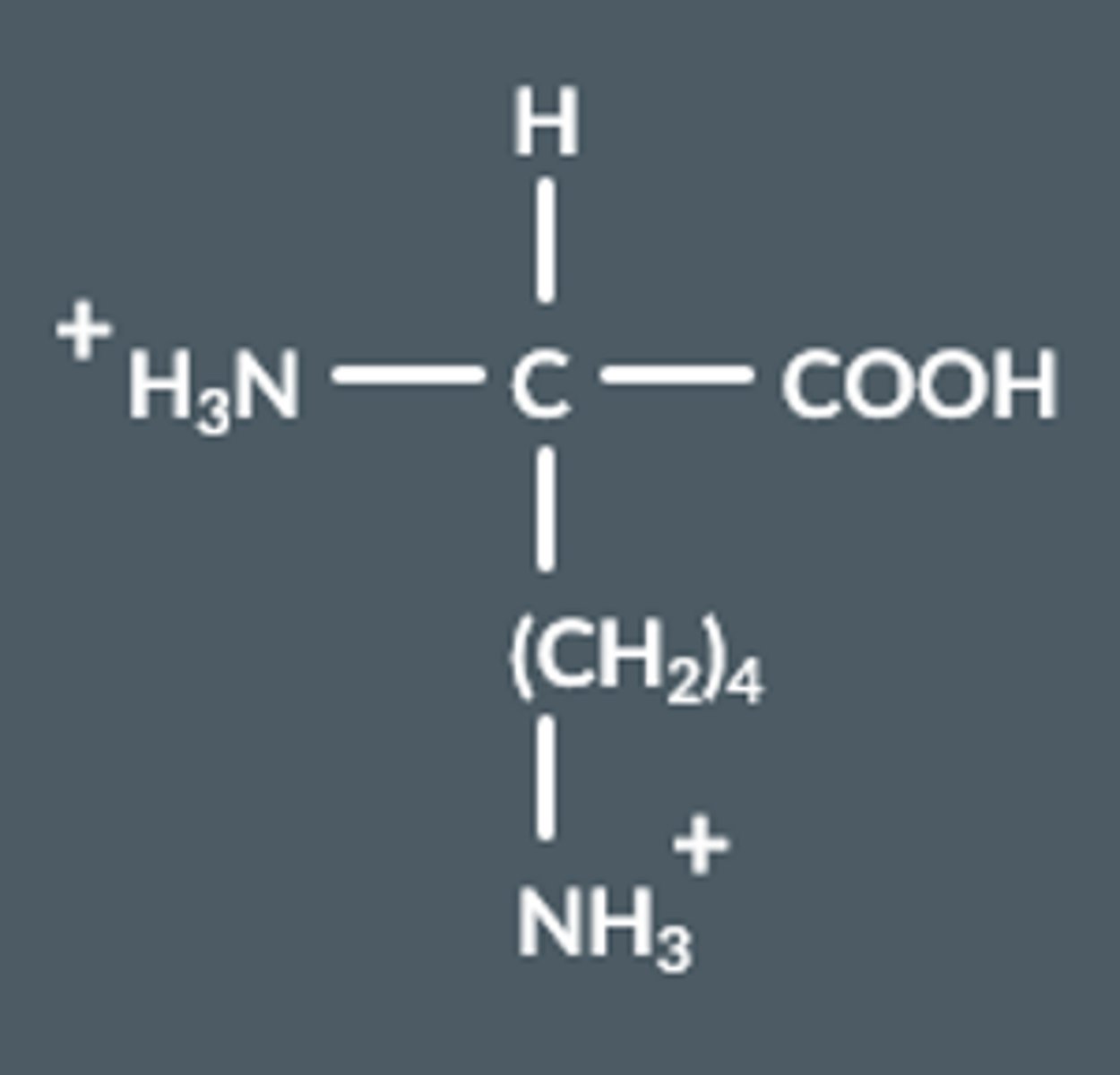
What happens to amino acids in alkaline conditions?
Draw this.
COOH groups lose a proton, becomes COO⁻
What is the peptide linkage?
CONH
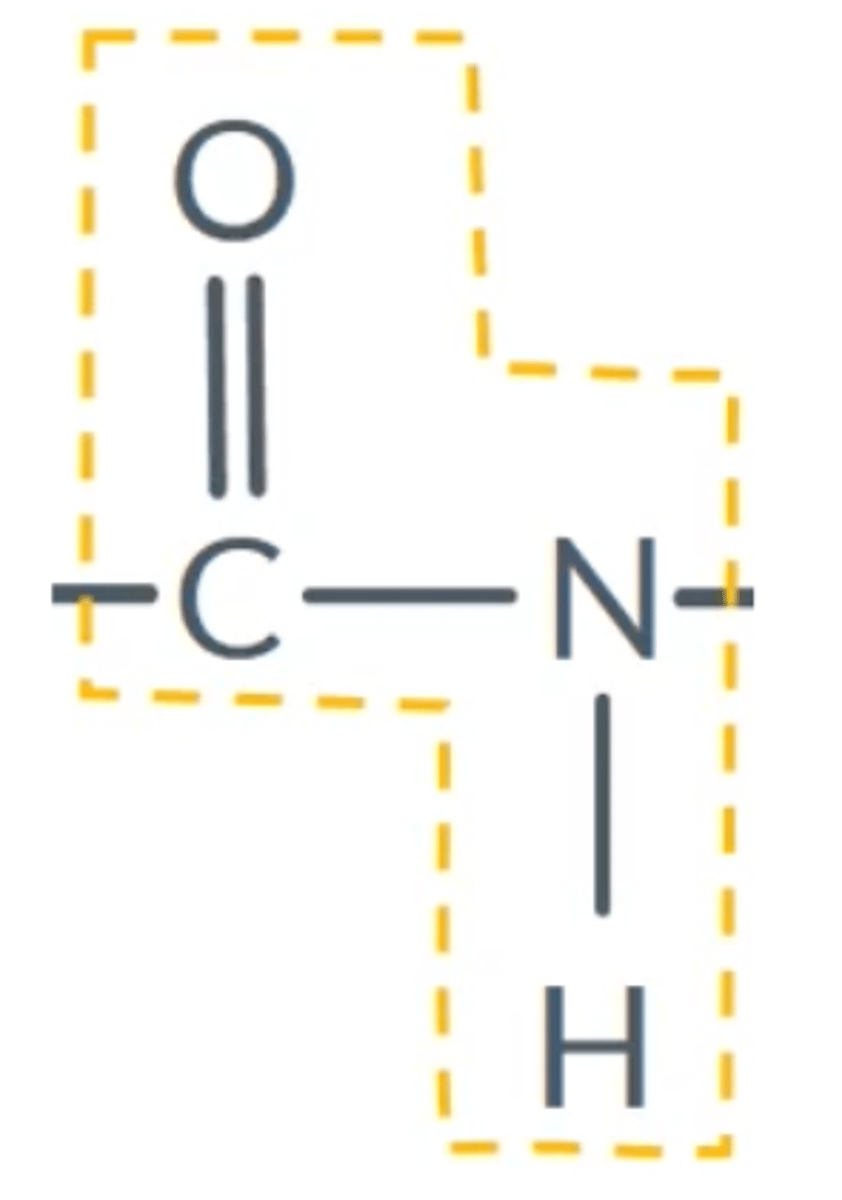
What is a dipeptide? Draw a general one for amino acids.
Two amino acids bonded together (a dimer)
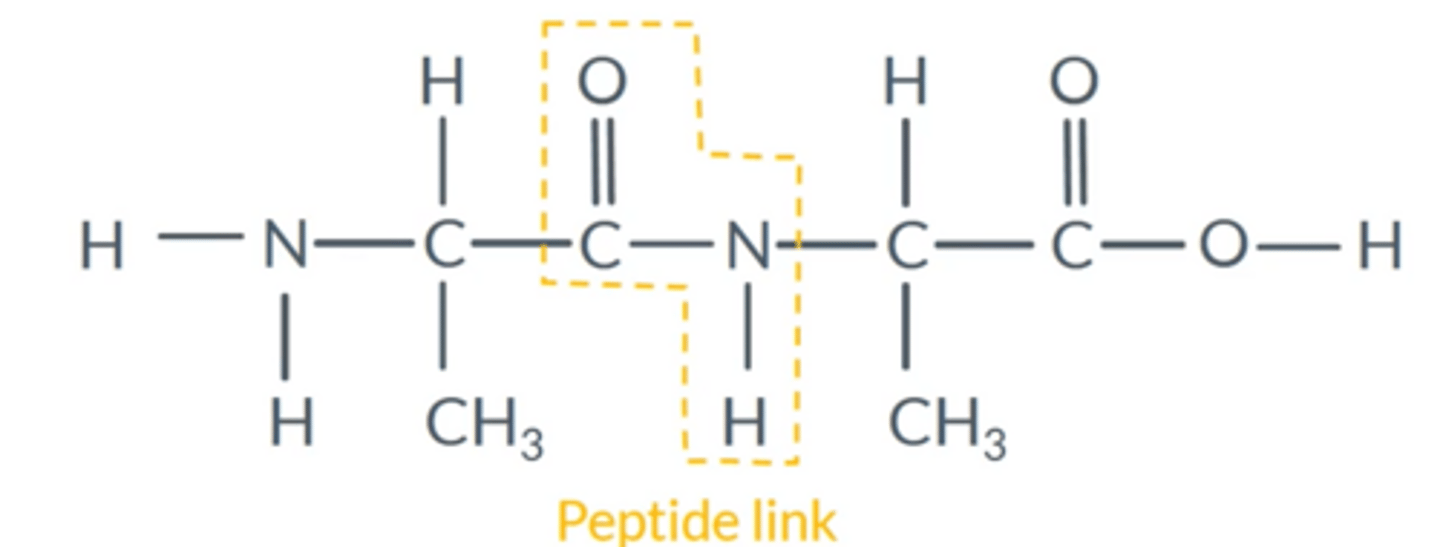
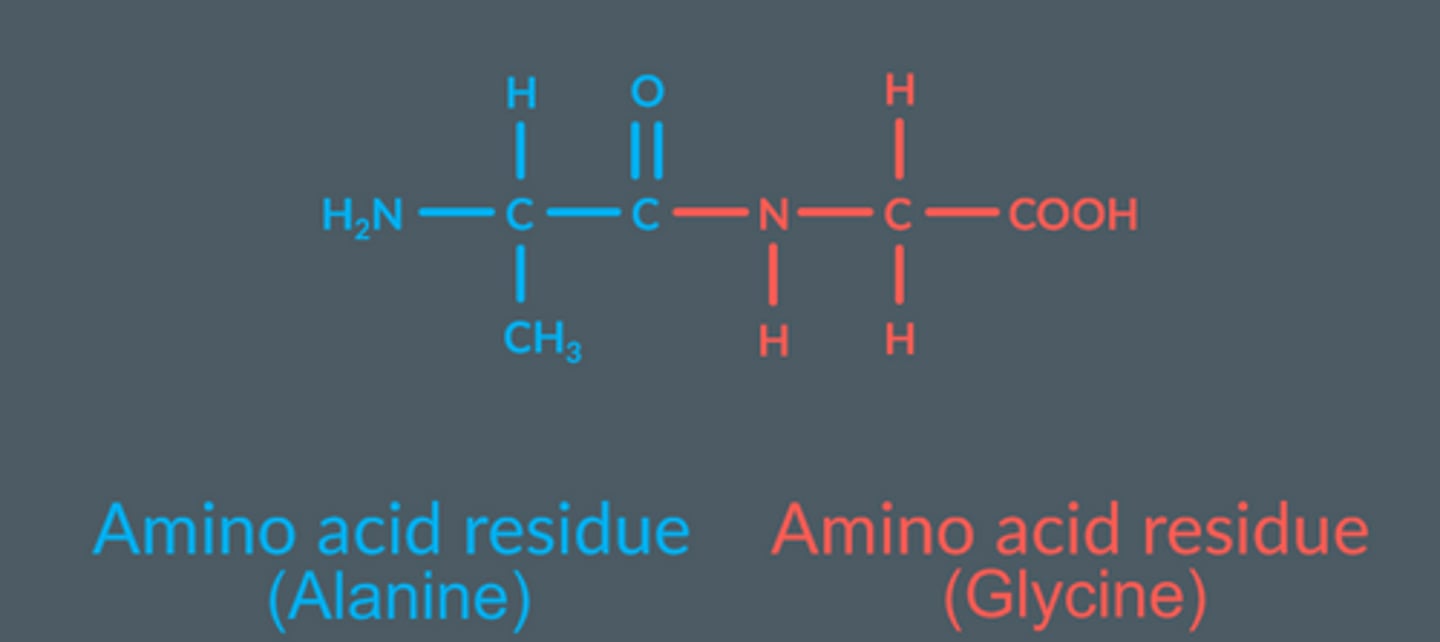
Amino acid residue
Leftover parts of an amino acid
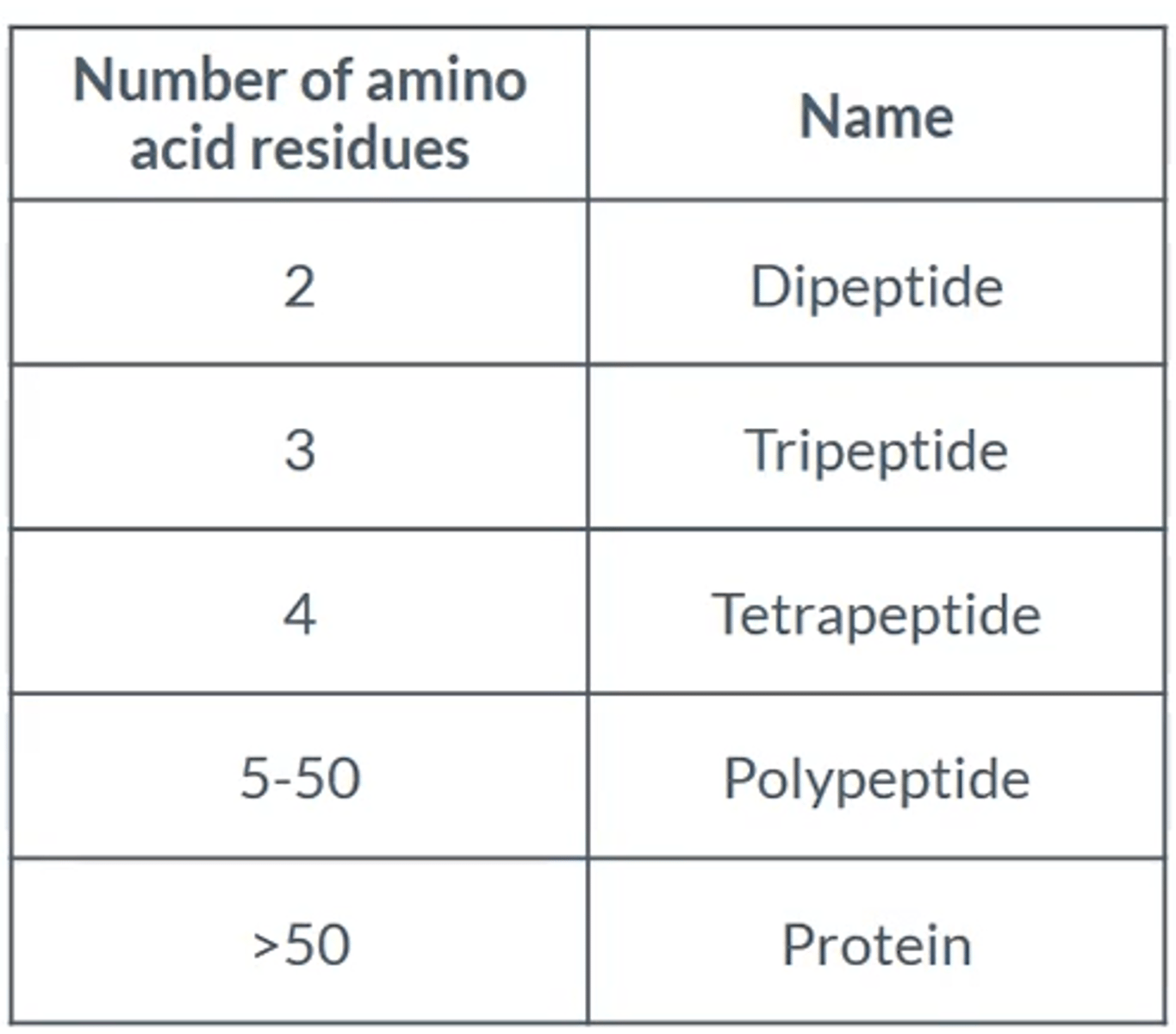
What name is given to chains of amino acids up to 50 amino acids?
Polypeptides
What name is given to chains of amino acids with more than 50?
Proteins
Give the number of amino acids and what the molecule is called
2 different amino acids reacting with each other...
● Create 2 different products, depending on how the reaction takes place.
● (depends on which molecule is on which side, aka the side the NH₂ and COOH are)
What are polypeptides and proteins found in?
Enzymes, Wool, Hair, Muscles
What is the process called by which polypeptides or proteins can be broken down into their constituent amino acids?
Hydrolysis
What conditions are needed for hydrolysis to occur?
6 mol dm⁻³ HCl
reflux for 24 hours
What is the primary structure of a protein? How is it bonded?
The sequence of amino acids along the protein chain. Bonded by covalent bonds
Shows the covalent bonding between amino acid residues and peptide links.
How is the primary structure represented?
Sequence of 3 letter abbreviations of the amino acids
Peptide bonds replaced by straight lines

How can the primary structure of a protein be broken up?
Hydrolysis
6 mol dm⁻³ HCl
reflux for 24 hours
What is the secondary structure of a protein?
The shape of the protein chain; how the primary structure folds on itself.
What are the two options for the secondary structure?
Alpha-helix shape or beta-pleated sheets
How is the secondary structure held together?
Hydrogen bonding, e.g. between C=O and N-H groups (between peptide links)
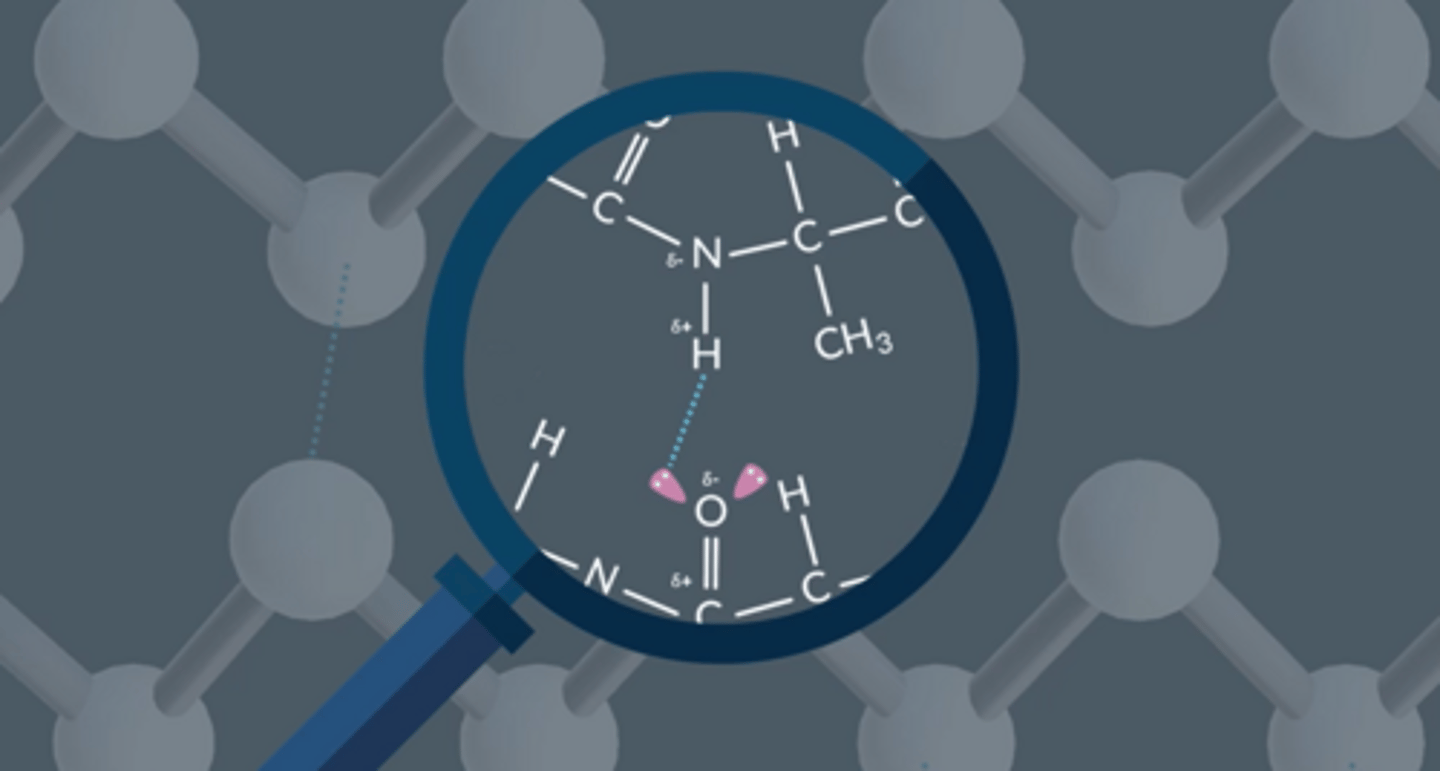
What is the difference between α - helix and β - pleated sheet?
In the α - helix the hydrogen bonds form in the same molecule whereas in the β-pleated sheet the hydrogen bonds form between adjacent protein molecules
Hydrogen bonding between proteins/polypeptides
Two protein molecules will experience hydrogen bonding between a hydrogen with a δ- nitrogen, and an oxygen with a δ+ carbon
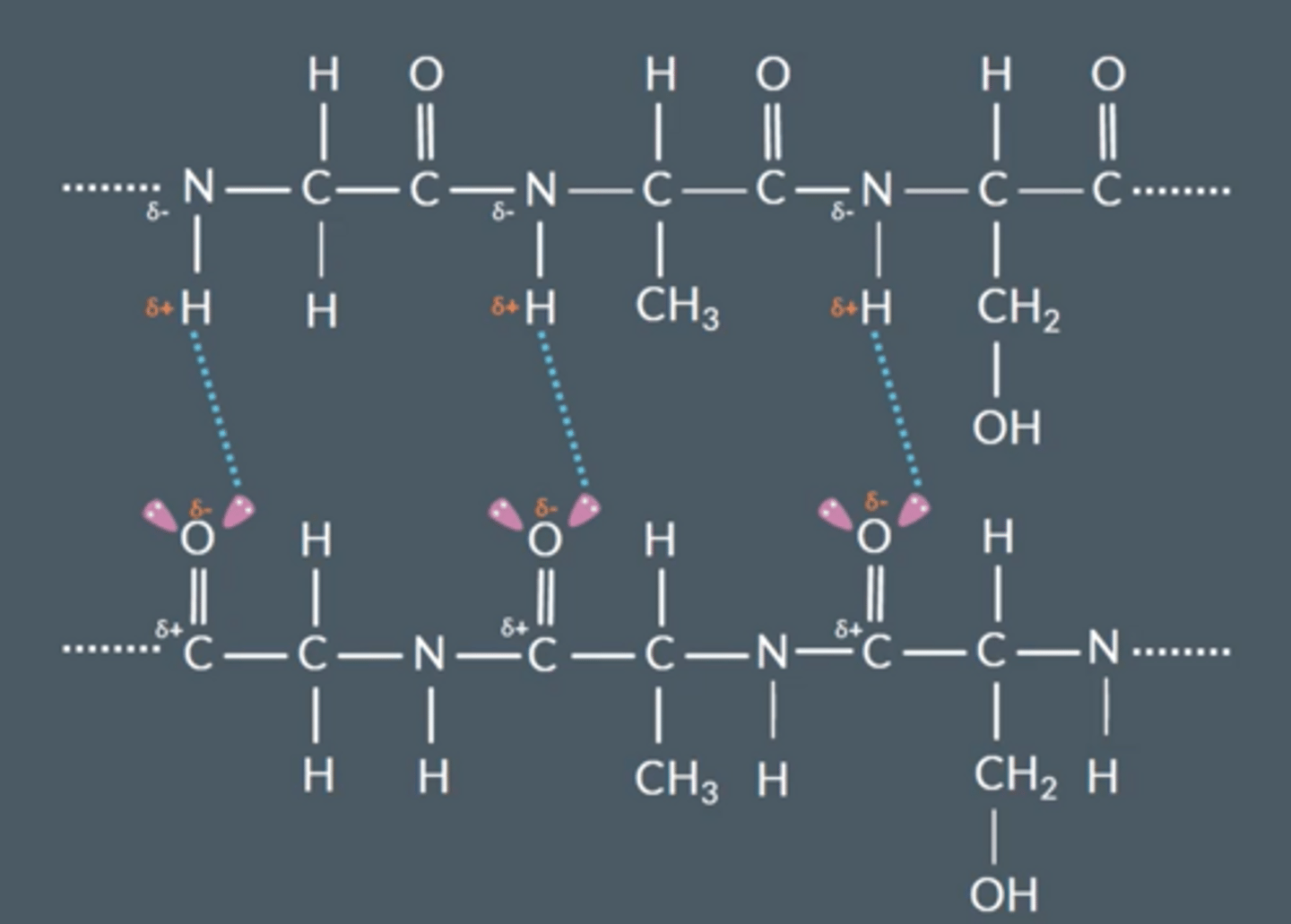
Explain how hydrogen bonds arise between chains
● Nitrogen and oxygen are very electronegative
● C=O and N-H bond is polar
● H bond between lone pair on oxygen and δ+ H on a different molecule
What is the tertiary shape of a protein?
Alpha-helix or beta-pleated sheet is folded into a complex 3D shape; this is the tertiary structure
How is the tertiary structure held together?
● Hydrogen bonding
● Ionic interactions between R groups (NH₃⁺ and COO⁻ groups)
● Sulfur-sulfur bonding (disulfide bridges from cysteine residues)
Van der Waals forces of attraction can also affect a protein's tertiary structure if side chains have large hydrocarbons.
Why is the tertiary structure important?
The shape of protein molecules is vital in their function - e.g. for enzymes
How can amino acids bond/be attracted to each other? (3 main ways)
● Hydrogen bonding
● Ionic interactions between groups on side chains
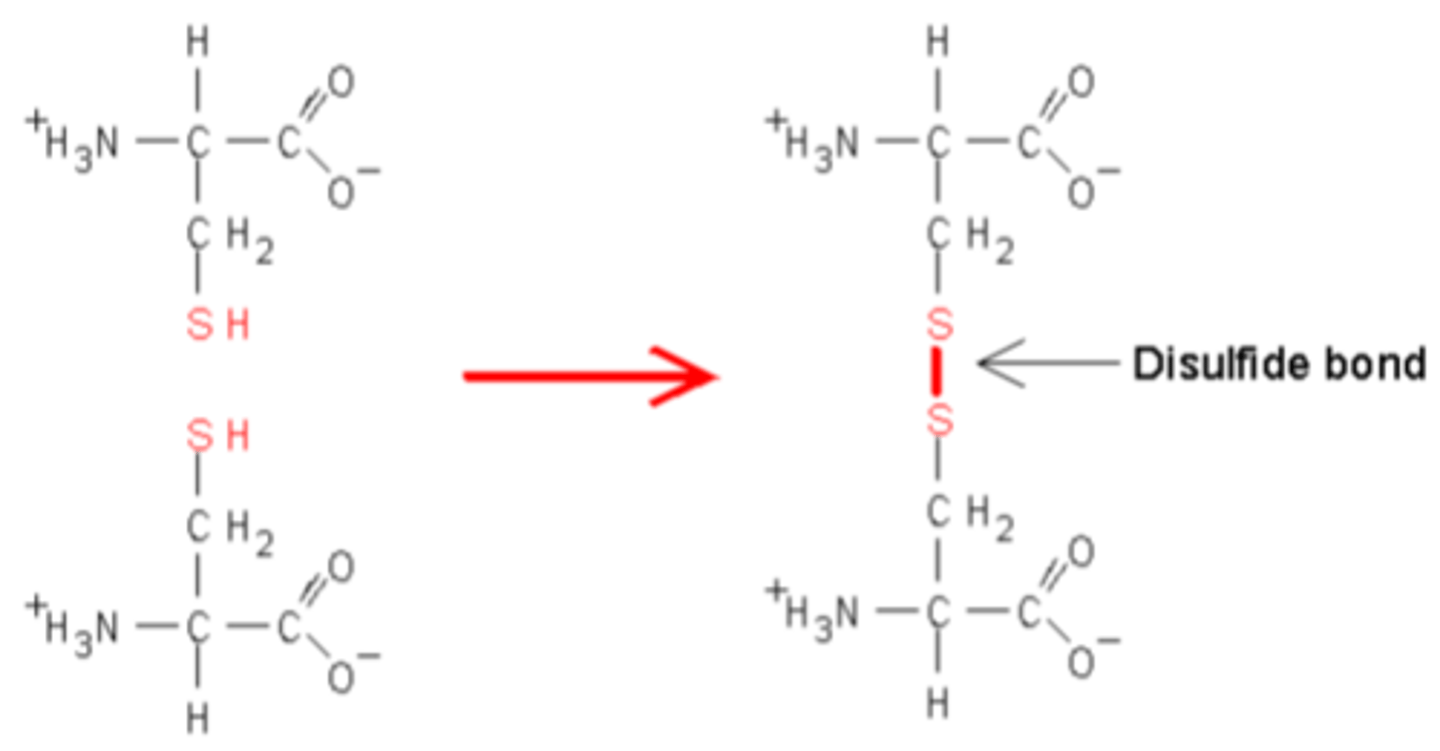
● Sulfur-sulfur bonds/disulfide bridges; 2 S atoms oxidised to form an S-S bond
Disulfide brudge
● The sulfur-sulfur bonds that hold tertiary structures together
● They keep the protein structure stable by losing 2 hydrogen atoms
What is wool? How is it held together?
Protein fibre with secondary alpha-helix structure; held together by hydrogen bonds
What does wool's structure and bonding mean for wool's properties?
● Can be stretched, H bonds extend.
● Release it and it returns to its original shape
● Wash too hot and H bonds permanently break so garment loses its shape.
Which technique is used to determine the primary structure of a protein?
● The protein is hydrolysed (see conditions above)
● The amino acids are separated using TLC.
● Because amino acids are colourless, ninhydrin is used and the amino acids turn purple OR UV light, Rf values are calculated and compared against data base to identify the amino acids
What is a TLC plate made of?
● Plastic sheet coated with silica, SiO₂.
● This is the stationary phase. (The solvent is the mobile phase)
Describe how you would carry out Thin Layer Chromatography
● Spot the samples onto a pencil line a few cm above the base of the TLC plate.
● Place this in a beaker or tank, with solvent level below the pencil line. Ensure there is a lid on the beaker to keep the inside saturated with solvent vapour.
● Wait until the solvent front is almost at the top of the TLC plate; then remove from the beaker and analyse.
Why does TLC separate amino acids (or other molecules)?
● Solvent carries amino acids up the TLC plate.
● The rate of movement depends on the balance between that amino acid's affinity for the solvent (solubility in it) and affinity for the stationary phase (attraction to the silicon with hydrogen bonding).
Why do amino acids have different Rf values?
Amino acids have different Rf values because they have different polarities
These will affect the retention of amino acids in the stationary phase and the solubility in the moving phase.
State why the amino acids separate on the TLC plate.
Difference in the balance between solubility in solvent/mobile phase and attraction to/retention on stationary phase
ALLOW difference between (relative) affinity/attraction for solvent and stationary phase
ALLOW absorption/adsorption for retention on stationary phase
What do you often have to do to enable the amino acids to be seen on the chromatogram?
● Spray with ninhydrin (amino acids are colourless, ninhydrin turns their spots purple)
● Or shine UV light on them
How do you calculate an Rf value?
Distance moved by that substance / distance moved by the solvent front
How can Rf values verify which amino acid is which?
● Compare the experimental Rf values to known/accepted values in the same solvent.
● Or run pure amino acids in the same solvent and compare results to identify amino acids
What is 2D TLC?
● Uses a square TLC plate.
● Spot the amino acids in one corner, then run TLC in first solvent.
● Flip the plate through 90° and repeat TLC in a second, different solvent.
What are the benefits of 2D TLC (2 main ones)?
● Separates the spots more - it is extremely unlikely that 2 amino acids will have identical Rf values in 2 solvents.
● Gives you 2 Rf values for each amino acids; you can be more confident in verifying the identity of the amino acids when comparing to known values, as 2 Rf values can be verified
How do you find the primary structure of a protein?
● Reflux for 24 hours with 6 mol dm⁻³ HCl (Hydrolysis)
● Carry out TLC to find the number and type of amino acids present.
How do you find the secondary and/or tertiary structure of a protein?
Various techniques, e.g. X-Ray
Diffraction
What is an enzyme?
Protein based catalysts that speed up reactions in the body by factors of up to 10¹⁰ without being used up
How many reactions is each enzyme designed to catalyse?
One reaction - they are very specialised
What is the structure of an enzyme?
Globular protein with a crevice in it known as an active site (very particular shape), where a substrate molecule can bond to the amino acid side chains through a variety of
interactions including:
● Hydrogen bonding
● Van der waals forces
● Permanent dipole-dipole forces
● Ionic interactions
What allows enzyme-substrate complexes to form?
Intermolecular forces between R groups.
Describe the nature of the interactions between the enzyme and substrate.
The interactions need to be strong enough to hold the substrate for long enough for the enzyme catalysed reaction to occur, but weak enough for the product to be released.
How does its structure help the function of the enzyme? What is this hypothesis known as?
The reacting molecules fit precisely into the active site and are held at exactly the right orientation to react - the lock and key hypothesis
How else do enzymes increase the rate of reaction?
● Reacting molecules form temporary bonds (via intermolecular forces) to the enzyme.
● This weakens the bonds in the molecules, promotes electron movement and lowers EA
How do enzymes bind to substrates ?
● Through intermolecular forces.
● Only formed temporarily between the substrate and the enzyme's active site, which shapes itself around the substrate.
● This causes bonds to break and new bonds to form within the substrate.
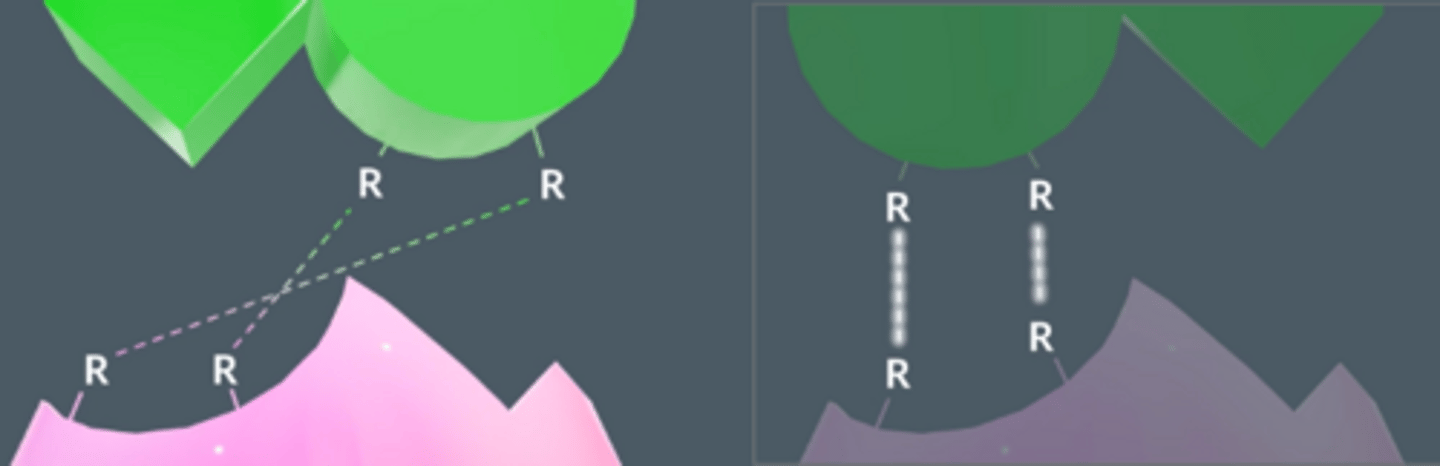
What can these intermolecular forces be?
Van der Waal forces
Hydrogen bonds
Ionic interactions
NOT disulfide bridges, these forces would be too strong and substrates wouldn't be able to leave enzymes
What does the stereospecificity of enzymes mean?
Active sites are so selective of the shape of substrates that only reactions involving one enantiomer are catalysed.
What does stereospecificity mean for most naturally occurring molecules?
Most naturally occurring molecules only occur as one enantiomer due to stereospecific enzymes
How are enzymes denatured?
Change in temperature or pH
How does temperature affect enzyme catalysis?
● The weaker bonds will break first - van der Waals attractions between side groups, and then hydrogen bonds.
● As soon as these bonds holding the tertiary structure together are broken, then the shape of the active site is likely to be lost; the enzyme is denatured
How does concentration of the substrate affect the enzyme catalysis?
● At very, very low concentrations, the rate is proportional to the substrate concentration.
● Increasing the concentration more has less and less effect - and eventually the rate reaches a maximum.
● There are not any enzyme molecules free to help the extra substrate molecules to react.
How does enzyme inhibition work?
● A molecule with a very similar shape and structure to the substrate is devised.
● Binds to and blocks the enzyme's active site (does not desorb easily).
● Substrate cannot adsorb to the active site, so reaction cannot be catalysed
An example of a drug that works through enzyme inhibition?
Penicillin
What are the benefits of modelling new molecules on computers?
Now we understand factors that affect the shapes of extremely complex proteins, we can:
● Model drugs that haven't even been synthesised,
● Predict their properties
● Design drugs that will treat a range of medical conditions
Give at least 2 reasons why designing enzyme inhibitors is really complex.
1. Enzymes are stereospecific,
2. Enzymes are made up of hundreds of amino acids,
3. Enzymes have a 3d shape,
4. Inhibitors might have enantiomers that interact differently with other enzymes.
May use computers to design inhibitors
What does DNA stand for?
Deoxyribonucleic acid
What does DNA do?
It is present in all cells and is a blueprint from which all organisms are made
What structure does DNA take?
A polymer with 4 monomers; they can be combined differently
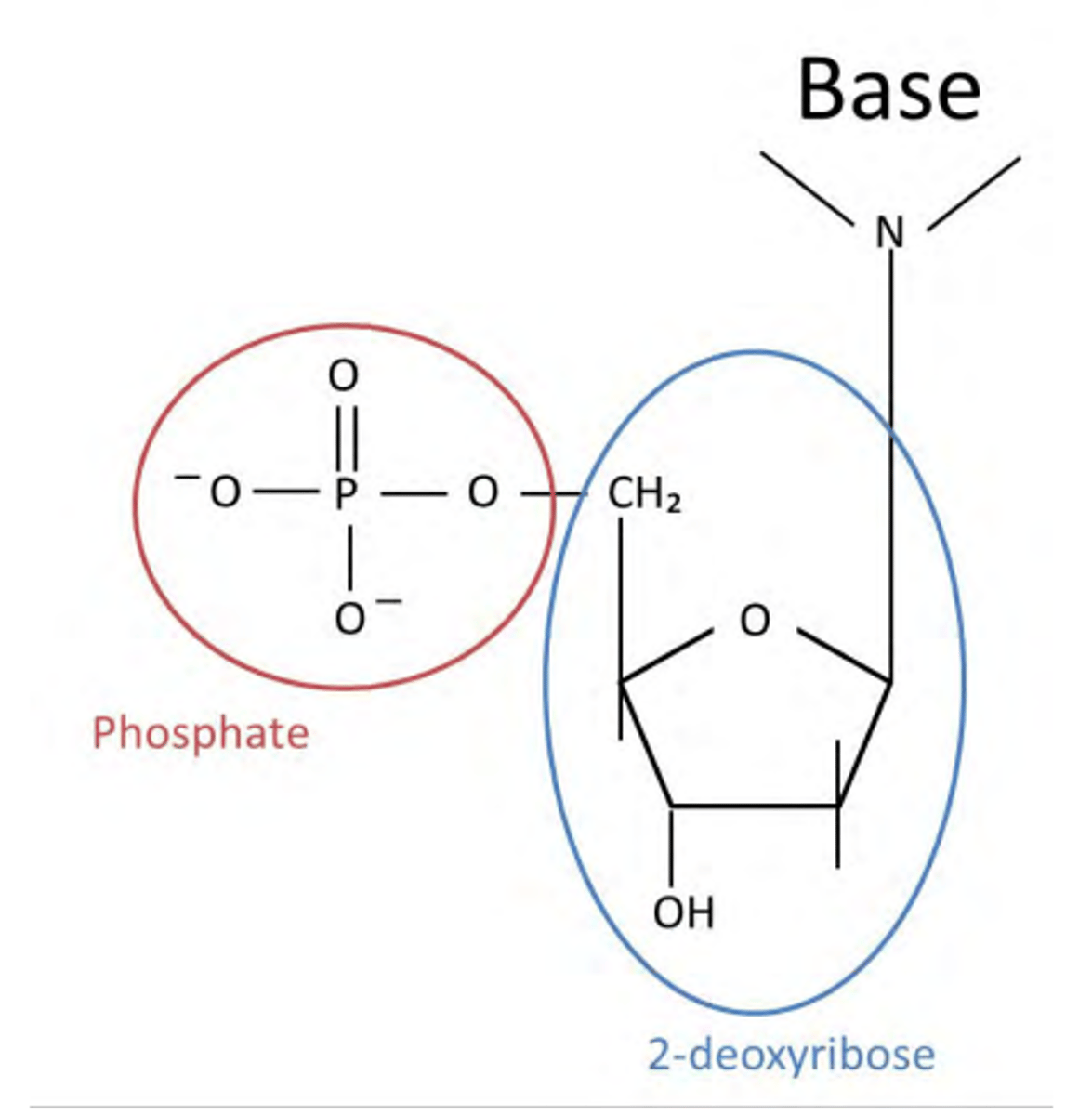
What constitutes a nucleotide?
A phosphate ion
a sugar (2-deoxyribose)
a base: ATCG
Draw a nucleotide.
What forms between bases of adjacent nucleotides?
Hydrogen bonding
Which bases pair up between nucleotides?
A + T
C + G
How does DNA polymerise?
OH on phosphate group and OH on Carbon 3 of 2-deoxyribose react to eliminate a molecule of H₂O, forming a larger molecule
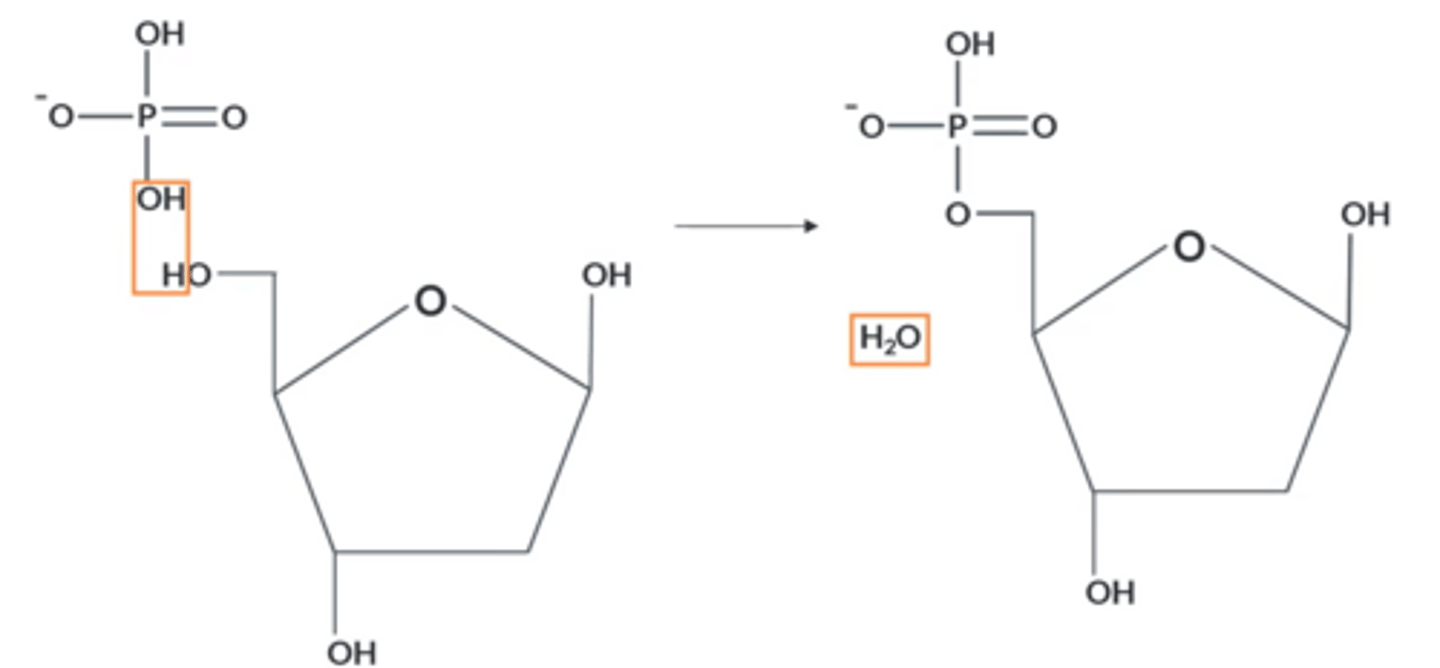
What kind of polymer does the polymerisation of DNA lead to?
● Nucleotides join together via condensation polymerisation:
● Phosphodiester (covalent) bonds between monomers.
● A molecule of H₂O is lost for each bond
● Forms a polynucleotide chain with a sugar-phosphate backbone.
What defines the properties of the DNA molecule?
The order of the bases
How can you distinguish between bases ?
Ring 2-oxygen or 1-CH₃
Ring 2-oxygen = Guanine, if not adenine
1-CH₃ = thymine, if not cytosine
Draw the larger molecule formed when adenine reacts with a sugar.
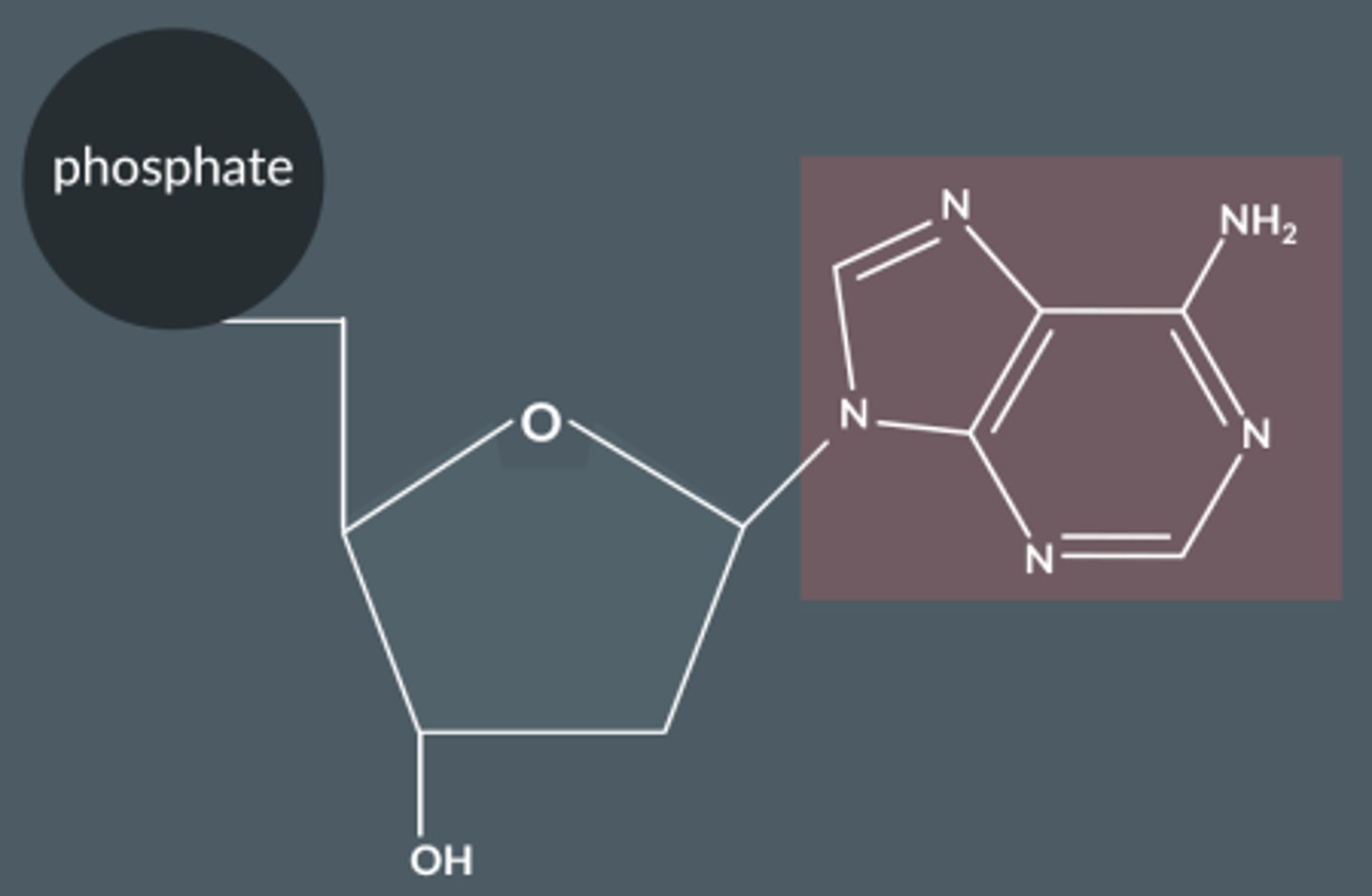
How do bases join with 2-deoxyribose ?
Bases react via their lowest NH group with the OH group on C1 in the sugar.
The reaction between the sugar and base forms a larger molecule with a C-N bond and water.
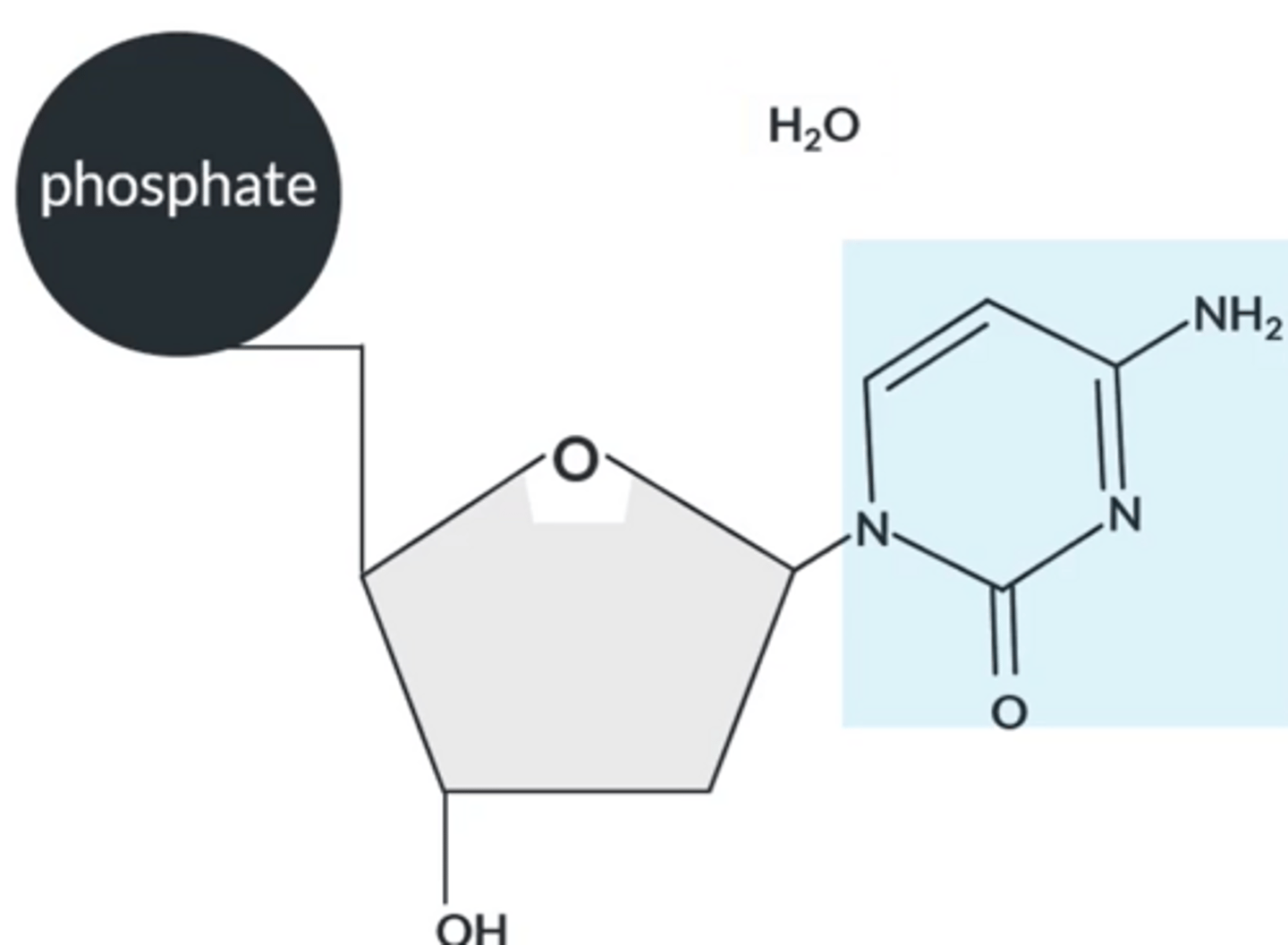
Nucleotide chain
● Nucleotides bond together to form a nucleotide chain
which are the macromolecules that form DNA.
● These macromolecules are held together by the phosphate-sugar backbone.
How many hydrogen bonds does each base form? Draw them.
Thymine and adenine form...
2 hydrogen bonds
Cytosine and guanine form...
3 hydrogen bonds
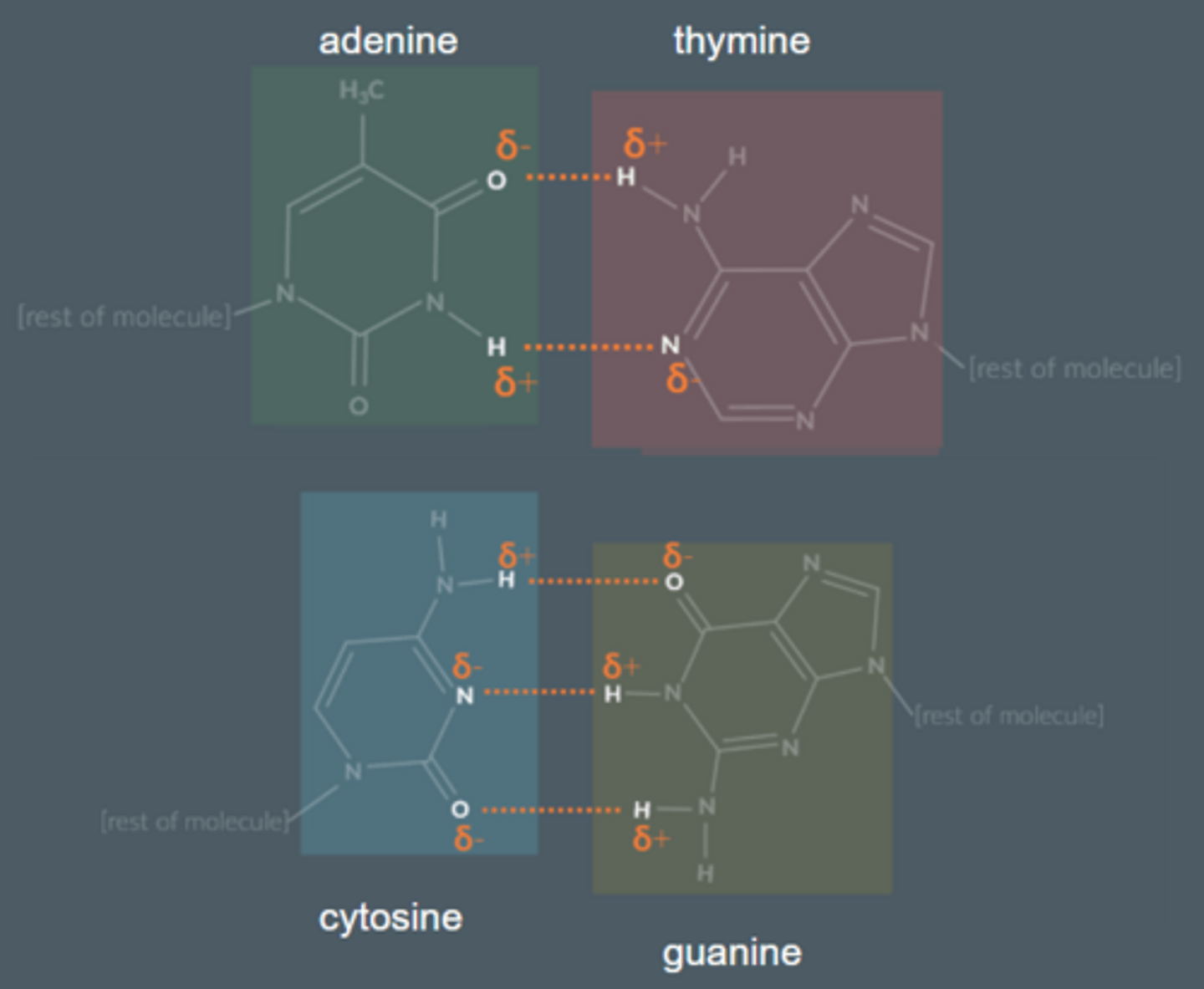
Explain how we know that adenine always bonds with thymine and that cytosine always bonds with guanine.
● 1:1 ratio between adenine and thymine and a 1:1 ratio between cytosine and guanine.
● The distance between two complementary DNA strands corresponds to around three carbon rings. This gives DNA a symmetrical helix shape. When bonded together, the base combinations of adenine and thymine or cytosine and guanine form three carbon rings.
● Adenine complements thymine fit perfectly by forming 2 hydrogen bonds. Guanine complements cytosine in the same way, by forming 3 hydrogen bonds.
What are the steps to draw interactions between complementary bases?
1. Identify the given base using the phrase "ring 2-oxygen
or 1-CH₃"
2. Identify the complementary base.
3. Label the partial charges on the atoms involved in the hydrogen bonds.
4. Rotate or flip the complementary base so that the
partial charges line up.
5. Draw on the base and use dashed lines for the hydrogen bond.
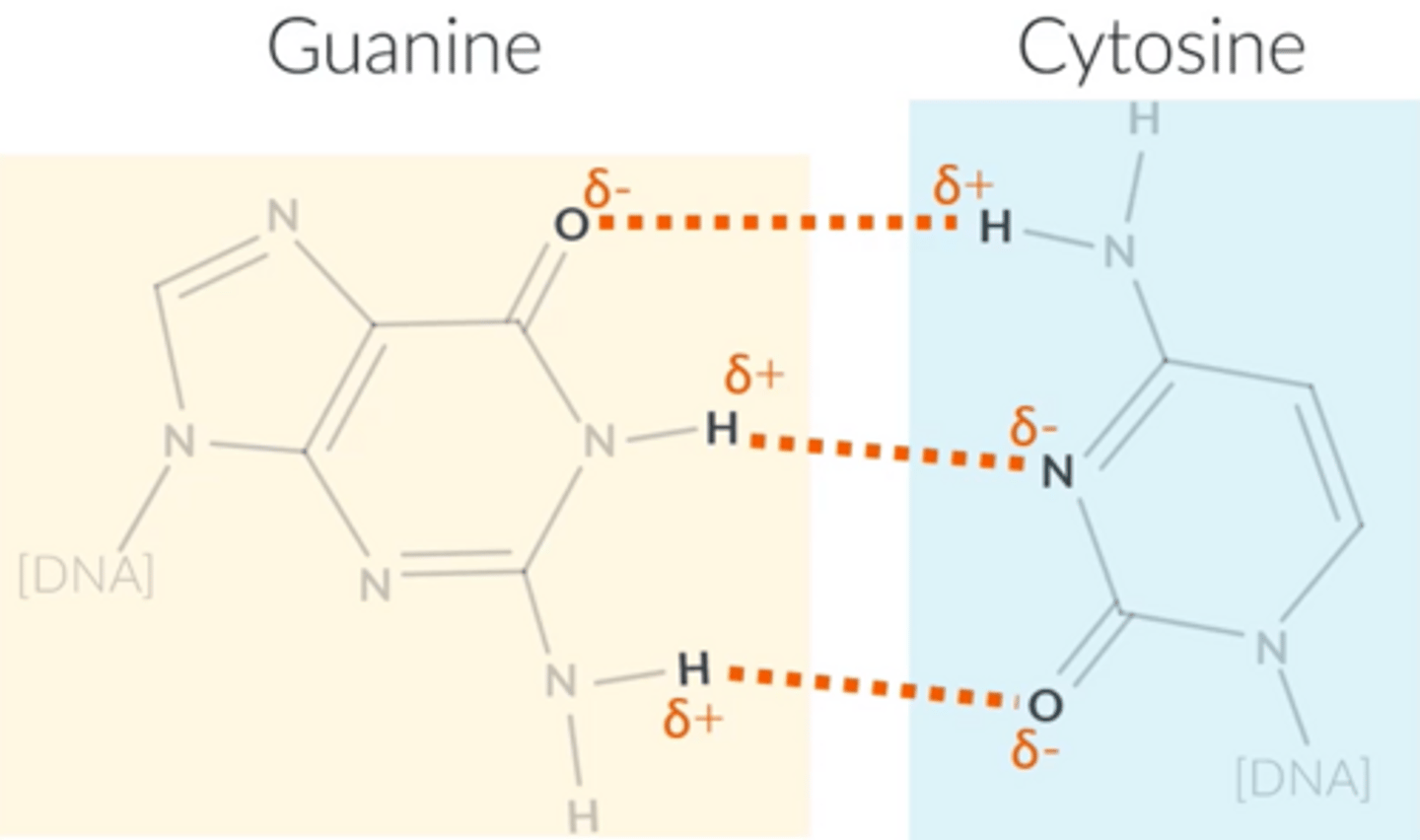
Why does DNA have a double helix shape?
● Exists as 2 strands; these 2 strands are held together by hydrogen bonding (C and G and A and T).
● The complementary DNA molecule has bases that hydrogen bond in the same order to those on another molecule → double helix shape is formed
Why is it important that DNA is exactly copied when cells divide?
Because it codes for proteins and makes all cells