Thermodynamics
1/55
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
Define the enthalpy change of formation (delta f H).
The enthalpy change when one mole of a compound is formed from it’s constituent elements in their standard states under standard conditions.
Define lattice enthalpy of formation (delta lattice H).
The enthalpy change when one mole of a solid ionic compound is formed from it’s ions in the gaseous state under standard conditions.
Define the lattice enthalpy of dissociation (delta lattice H) (opposite of lattice enthalpy of formation).
The enthalpy change when one mole of a solid ionic compound is dissociated into it’s gaseous ions under standard conditions.
Define enthalpy change of dissociation (delta diss H).
The enthalpy change when one mole of bonds of the same type of molecule in the gaseous state is broken.
Define the enthalpy change of first ionisation (delta IE1 H).
The enthalpy change when one mole of gaseous 1+ ions are made from one mole of it’s gaseous atoms.
Define enthalpy change of atomisation (delta at H).
The enthalpy change when one mole of gaseous atoms is made from an element in it’s standard state.
Define enthalpy change of second ionisation (delta IE2).
The enthalpy change when one mole of gaseous 2+ ions are made from one mole of gaseous 1+ ions.
Define first electron affinity (delta ea1 H).
The enthalpy change when one mole of gaseous 1- ions are made from one mole of gaseous atoms.
Define second electron affinity (delta ea2 H).
The enthalpy change when one mole of gaseous 2- ions are formed from one mole of gaseous 1- ions.
True or false? You can calculate lattice enthalpies directly from experiments.
False. YOU CANNOT CALCULATE LATTICE ENTHALPIES DIRECTLY FROM EXPERIMENTS.
What can Born-Haber cycles be used to calculate?
Lattice enthalpies.
What is a Born-Haber cycle?
A thermochemical cycle that includes all the enthalpy changes involved in the formation of a compound.
How can you use the blocked road analysis for working lattice enthalpy of formation when you have all the other values in the Born-Haber cycle?
1) Label the line after the starting point.
2) Label the bottom line the end point.
3) The direct line from the start point to the end point is blocked, so you must take another road to get to the end point.
4) Move from the start point backwards towards the endpoint.
5) If any arrows you draw are in the opposite direction to the ones indicating enthalpy change for each process, swap the + or - on the enthalpy change value for the other sign.
6) Add up all these values to get the lattice enthalpy of formation.
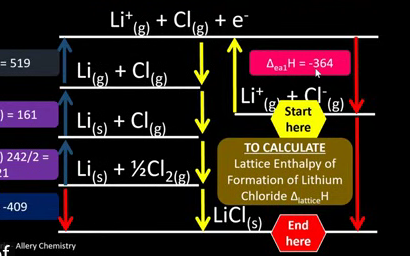
YOU CAN APPLY THIS METHOD TO ANY UNKNOWN VALUE IN A BORN-HABER CYCLE AS LONG AS YOU HAVE FIVE OTHER VALUES.
Is the formation of an ionic lattice normally exothermic or endothermic? Why?
The formation of an ionic lattice is normally exothermic. This is due to large amounts of energy being released.
Sketch and label a general diagram of a Born-Haber cycle.
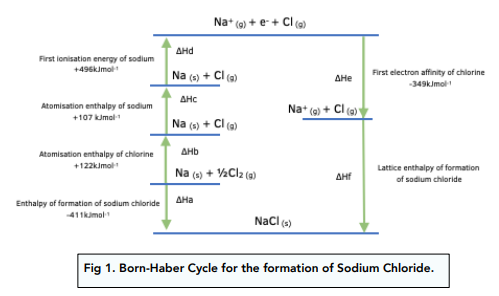
What does the direction of the arrow for a process in a Born-Haber cycle show about the enthalpy change for that process?
It shows whether the process is exothermic or endothermic (if the arrow goes up, the process is endothermic and if the arrow goes down, the process is exothermic).
True or false? All the values in a Born-Haber cycle should add up to 0.
True.
How does an extended Born-Haber cycle differ from a “normal” Born-Haber cycle?
The extended Born-Haber cycle has two electron affinities (first and second) whereas a “normal” Born-Haber cycle has one electron affinity (first).
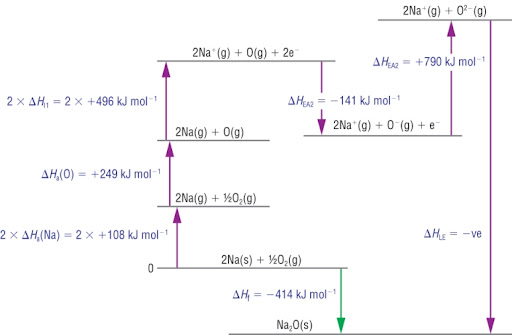
True or false? Second electron affinity is an exothermic process.
False. SECOND ELECTRON AFFINITY IS AN ENDOTHERMIC PROCESS.
Explain why the second electron affinity is and endothermic process.
Adding a negatively charged electron to an already negatively charged ion has lots of repulsion, energy must be put in to overcome that repulsion.
True or false? Theoretical and experimental enthalpy values are always identical.
False. THEORETICAL AND EXPERIMENTAL ENTHALPY VALUES ARE NOT USUALLY THE SAME.
Why are theoretical and experimental enthalpy values not always the same?
Theoretical lattice enthalpy values are calculated using the perfect ionic model while in reality ionic compounds don’t follow the perfect ionic model and actually have some covalent character.
What is the perfectly ionic model?
The ion is spherical, the charge is evenly distributed in the ion.
What is covalent character?
When one of the ions (usually the positive one) polarises the other ion (usually the negative one) causing the ions to not be spherical and have an uneven distribution of charge.
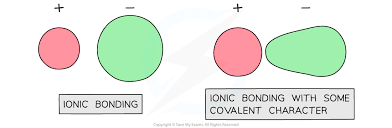
Which one of these drawings follows the perfect ionic model and which shows covalent character?
The first drawing follows the perfect ionic model while the second drawing shows covalent character.
True or false? The greater the polarisation the greater the covalent character.
True.
What does a greater difference between the theoretical lattice enthalpy and the experimental lattice enthalpy tell us about a substance?
The substance is less purely ionic.
How does covalent character change experimental values from theoretical values of lattice enthalpy?
Covalent character increases lattice enthalpy values.
What does a greater difference in theoretical and experimental lattice enthalpy values suggest about the covalent character of the substance?
Greater covalent character.
Define the enthalpy change of solution (delta solution H).
The enthalpy change when one mole of an ionic substance is dissolved in the minimum amount of solution to ensure no further enthalpy change is observed upon further dilution. (the enthalpy change when one mole of a solid ionic compound is just dissolved in water)
What two things (to do with bonding) must happen for a substance to dissolve? Label each thing as either endothermic or exothermic.
Bonds in the substance must break (endothermic).
New bonds between the solvent and the substance must be formed (exothermic).
Draw a diagram showing how bonds form between both water and cations and water and anions.
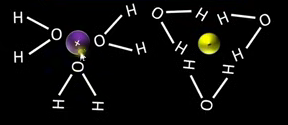
Why do ionic compounds dissolve well in polar solvents?
Ionic compounds are polar and polar substances dissolve well in polar solvents.
What is the name of the process in which water molecules surround an ion?
Hydration.
What must the strength of the new bonds formed be like in relation to those broken for hydration to take place?
The new bonds must be stronger/of equal strength to those broken in order for hydration to take place.
What two values do you need to calculate the enthalpy change of solution?
Lattice dissociation enthalpy.
Enthalpy of hydration.
Define enthalpy of hydration (delta hyd H).
The enthalpy change when one mole of aqueous ions is formed from one mole of gaseous ions.
Draw an enthalpy of solution cycle.
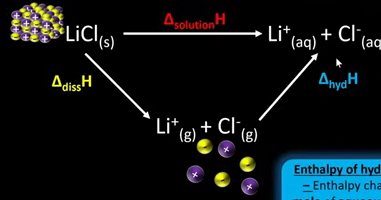
Like a Hess cycle.
True or false? Go with the arrow keep the sign the same, go against the arrow change the sign.
True.
What is entropy and what does it measure?
Entropy is the number of ways energy can be shared out between particles in a system. Entropy measures the disorder of a system.
The greater the entropy the … the disorder in a system.
Greater.
How does entropy change as a substance goes from a solid to a liquid to a gas?
Entropy increases (gases are more disordered than solids).
True or false? Disorder is less preferable in a system.
False. DISORDER IS PREFERRED IN A SYSTEM.
As the number of particles increases, entropy …
Increases.
Why does entropy increase when the number of particles in a system increases?
More particles means more ways that energy can be distributed in a system meaning it’s entropy increases.
True or false? A reaction can be feasible (spontaneous) even if it is enthalpically unfavourable. Explain your answer.
True. If the increase in entropy of a reaction is able to overcome the unfavourable changes in enthalpy then a reaction will be feasible.
Why are endothermic reactions not enthalpically favourable?
The products have a higher energy than the reactants.
Give an equation for calculating entropy change (delta S).

What are the units of entropy?
J K-1 mol-1
True or false? A reaction with a positive entropy change value is entropically favourable.
True.
What does Gibbs free energy (delta G) tell us about a reaction?
It tells us whether a reaction is feasible or not.
Give the Gibbs free energy equation.

Gibbs free energy = enthalpy change - (temperature x entropy change)
Enthalpy change = J mol-1
Temperature = K
Entropy change = J K-1 mol-1
Gibbs free energy = J mol-1
True or false? A reaction is feasible if it has 0 or positive Gibbs free energy value.
False. A REACTION IS FEASIBLE IF IT HAS A 0 OR NEGATIVE GIBBS FREE ENERGY VALUE.
Why might a feasible reaction not take place in real life?
The activation energy of the reaction is too high.
Does temperature affect the feasibility of a reaction?
Yes.
Give the equation for calculating at what temperature a reaction becomes just feasible.
